Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 n.10 Pretoria Oct. 2012
RESEARCH
Determinants of mortality in Nigerian children with severe anaemia
Samuel AdegokeI; Adenike AyansanwoII; Isaac OluwayemiIII; John OkeniyiIV
IMB ChB, MPH, FWACP; Department of Paediatrics and Child Health, Obafemi Awolowo University, Ile-Ife, Nigeria
IIBSc, MB ChB, FWACP; Department of Paediatrics and Child Health, Obafemi Awolowo University, Ile-Ife, Nigeria
IIIMB BS, FMC Path; Department of Haematology, University Teaching Hospital, Ado-Ekiti, Nigeria
IVMB BS, FWACP; Department of Paediatrics, University Teaching Hospital, Ado-Ekiti, Nigeria
ABSTRACT
BACKGROUND: Severe anaemia (haemoglobin concentration <50 g/l) is a major cause of paediatric hospital admissions and deaths in the tropics.
OBJECTIVES: To examine the pattern and predictors of mortality among severely anaemic children.
METHODS: A prospective cross-sectional study was conducted among children with severe anaemia at the Children's Emergency Room of the University Teaching Hospital, Ado-Ekiti, Nigeria. Sociodemographic characteristics, clinical features, laboratory findings and co-morbidities of the survivors and those who died were compared by logistic regression analysis.
RESULTS: Of the 1 735 children admitted, 311 (17.9%) had severe anaemia, with a case-fatality rate of 9.3%. The presence of respiratory distress (95% confidence interval (CI) 2.1 - 3.6, p=0.031); acidosis (95% CI 1.8 - 2.7, p=0.010); coma (95% CI 0.1 - 0.3, p=0.001); hypotension (95% CI 2.0 - 4.2, p=0.020); and bacteraemia (95% CI 3.1 - 3.9; p=0.008) were the significant independent predictors of death with regression analysis.
CONCLUSION: Early recognition with prompt and appropriate anticipatory intervention is essential to reduce mortality from severe anaemia.
Severe anaemia is a major cause of paediatric hospital admissions and deaths in many African countries, with a hospital prevalence of 10 - 30% and case fatality rates between 4 and 10%.1,2 Anaemia, in which blood oxygen carrying capacity is reduced, is characterised by a low haemoglobin concentration.3 The World Health Organization (WHO) defined severe anaemia in patients with malaria as a haemoglobin concentration less than 50 g/l.3Anaemia becomes life-threatening when affected individuals develop complications such as heart failure, metabolic acidosis and hypoxic encephalopathy.4
Severe falciparum malaria remains the leading cause of paediatric anaemia in sub-Saharan Africa.1,2,4 Anaemia may develop in children already debilitated by other conditions such as nutritional deficiencies, bacteraemia, human immunodeficiency viral (HIV) infections, haemoglobinopathy, glucose-6-phosphate dehydrogenase (G6PD) deficiency, hookworm infestation and leukaemia. In most cases, these factors tend to co-exist.1
Severe anaemia-related deaths continue to occur in many centres in the tropics, even where blood transfusion services are readily available.1,2 To limit this mortality, we studied children admitted with severe anaemia in a newly established tertiary health centre in south-western Nigeria to determine the mortality rate and its predictive factors. Identifying these factors may facilitate prompt use of adjunct therapies including blood transfusion in high-risk children.
Methods
This was a prospective cross-sectional study of consecutive children admitted with severe anaemia at the Children's Emergency Room (CHER) of the University Teaching Hospital, Ado-Ekiti, Ekiti state, Nigeria, from January to December 2010. The hospital was established about 50 years ago as a secondary health institution, and was upgraded about three years ago to a tertiary health institution to serve the growing health needs of a sizeable part of south-western Nigeria.
Severely anaemic children were those with venous haematocrit of <15% (haemoglobin concentration <50 g/l).3 All patients required emergency blood transfusion. Informed parental consent was obtained in all cases. Permission for the study was granted by the Ethics and Research Committee of the hospital.
Biodata (age, gender and socio-economic status) and other information, e.g. presenting symptoms (fever, pallor, jaundice and passage of dark-brown urine and their respective duration) and relevant clinical examination findings, were recorded. The socio-economic status was based on the highest parental level of education and occupation.5
Duration of illness was the interval between its onset and presentation in our unit and was considered prolonged when over 72 hours. Adequacy of antimalarial treatment before presentation was taken as WHO Artemisinin Combination Therapy (ACT) for the appropriate duration.
Nutritional status was assessed using the National Center for Health Statistics and WHO (NCHS/WHO) definition of wasting, underweight and stunting.6 Those with weight-for-height below 2 standard deviations (SD) were categorised as wasted, weight-forage below 2 SD as underweight, and height/length-for-age below 2 SD as stunted. The level of consciousness was assessed using the Blantyre Coma Scale (a modification of the Glasgow Coma Scale). Blood pressure (BP) was measured with the age-appropriate Accuson mercurial sphygmomanometer by standard techniques. Using the Paediatric Task Force guidelines on BP control, children with systolic and/or diastolic BP below the 5th percentile for age, gender and height were categorised as hypotensive.7
Oxygen saturations (SaO2) were measured using a pulse oximeter (Nell core, N-200, USA) with an appropriately sized paediatric probe attached to the finger or toenail bed. The reading of the oximeter was recorded after stabilisation of the reading for 1 minute by a paediatric nurse not involved in the clinical examination. Hypoxaemia was defined as SaO2<90%.8
Venous blood samples were taken on admission for the haematocrit concentration, white blood cell count (total and differential count), platelet count, thick and thin blood film for malaria parasites, serum glucose, bicarbonate and creatinine estimations, blood culture, haemoglobin electrophoresis and glucose-6-phosphate dehydrogenase (G6PD) assay. Haematocrit levels and complete blood counts were determined by standard manual methods.9 Malaria was defined as the presence of Plasmodium falciparum asexual parasites in the blood. All the patients had standard bacterial cultures and sensitivity tests.9
The survivors and the dead were compared in terms of sociodemographic characteristics (age, gender, socio-economic status), clinical symptoms and signs including duration of illness before presentation and adequacy of therapy before presentation.
Laboratory findings, e.g. haematocrit and complete blood count, and the presence of co-morbidities such as acidosis (serum bicarbonate <20 mmol/l), hypoglycaemia (serum glucose <2.2 mmol/l), bacteraemia, haemoglobinopathy and HIV infection, were also compared.
The data were analysed using SPSS version 17. Tests of association between the variables and the outcome were performed with chi-squared and Student's t-tests for categorical and continuous variables respectively. Variables associated with statistically significant mortality were further analysed with logistic regression modelling to identify the independent predictive factors of mortality in children with severe anaemia. Results of logistic regression were presented with odds ratios (ORs) and 95% confidence intervals (CIs). Statistical significance was established when p-values were <0.05 and the CI excluded unity.
Results
During the study, 1 735 children were admitted; 311 had severe anaemia, a prevalence of 17.9%; 199 (64.0%) were males (male-to-female ratio 1.8:1). Ages ranged from 2 to 161 months with the mean age 30.9 (SD 16.7) months. Of the 311 children, 56 (18.0%) were infants under 1 year of age, 218 (70.1%) were preschool children (>1 - 5 years), 29 (9.3%) were school-age (>5 - 10 years), 8 (2.6%) were adolescents (>10 years), 260 (83.6%) were of low socio-economic status (III - V) and 51 (16.4%) were of high socio-economic status (I and II).
Malaria, the leading cause of severe anaemia, occurred in 282 (90.7%), either alone or in combination with other diseases: 81 (26.0%) had bacteraemia, 40 (12.9%) had sickle-cell anaemia and were in haemolytic crisis, 20 (6.4%) had significant bleeding from injuries, 16 (5.1%) were G6PD-deficient and 3 (1.0%) had chronic kidney disease.
Of the 311 children with severe anaemia, 29 died during the study period (case-fatality rate 9.3%), representing 33.0% of the 88 deaths in CHER during the period. The remaining 282 (90.7%) survived and were discharged. Of the 29 children who died, 8 (24.1%) died before blood transfusion was given.
The mean age of the survivors was 36.2 (SD 30.5) months (range 2 -161 months). The ages of those who died ranged from 2 to 72 months with a mean of 18.8 (SD 8.1) months. Children with severe anaemia who died were significantly younger than survivors (t=2.44, p=0.014).
Among survivors, the duration of illness before presentation ranged from 1 to 13 days with a mean of 4.5 (SD 2.5) days; among those who died, illness duration ranged from 2 to 21 days, with a mean of 6.7 (SD 6.0) days. The average duration of illness before presentation was significantly longer among the severely anaemic children who died than among the survivors (t=3.07, p=0.002).
The mean interval between presentation and blood transfusion for those who survived was 3.1 (SD 3.6) hours and 2.5 (SD 3.8) hours for those who died. However, there was no statistically significant difference in the interval between presentation and transfusion among the two groups of children (t=0.69, p=0.492).
Table I compares the sociodemographic characteristics, and the clinical and laboratory findings, at presentation of the survivors and those who died. Respiratory distress, tachypnoea, heart failure, hypotension, coma and severe wasting were significantly related to poor outcome (p<0.005).
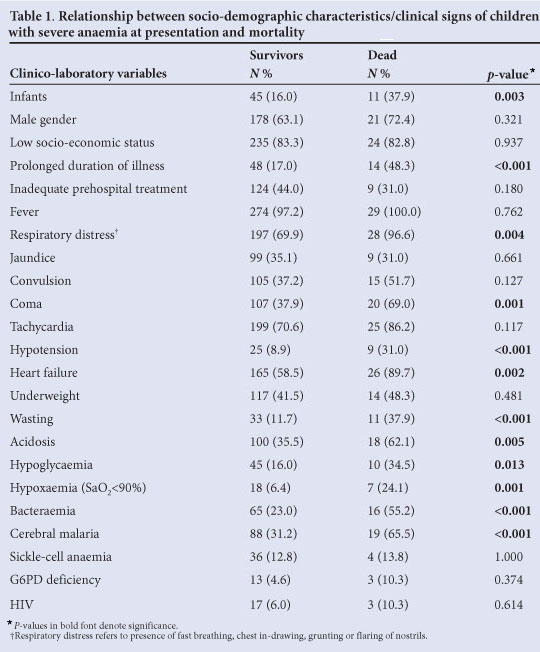
The haematocrit ranged from 6% to 19% among survivors and from 6% to 17% among those who died, with an overall mean of 12.9% (SD 1.1%). The mean haematocrit of those who died was 11.7% (SD 2.6%) and did not differ significantly from that of survivors (13.9% (SD 3.8%) (t=0.18, p=0.855)).
Of the 282 children with malaria, 123 (43.6%) had high-density parasitaemia (>10 000/μl), including 109 (38.7%) of the 282 survivors and 14 (48.3%) of the 29 who died. Although a higher proportion of those who died had high-density parasitaemia, the difference was not statistically significant (χ2=1.019, p=0.313). Similarly, the proportion of children with leucopenia or thrombocytopenia who died did not significantly differ from those who survived (p=0.152 and 0.217 respectively).
Of the 27 children with elevated serum creatinine levels (>110 μmol/l), 19 (70.4%) died (p=0.001). The mean serum creatinine level of the children who died was 119.6 μmol/l (SD 23.1) - significantly higher than that of survivors at 92.5 μπω!/! (SD 19.7) (p<0.001). The presence of acidosis, hypoglycaemia, hypoxaemia, bacteraemia and cerebral malaria was significantly related to death (p<0.05) (Table 1).
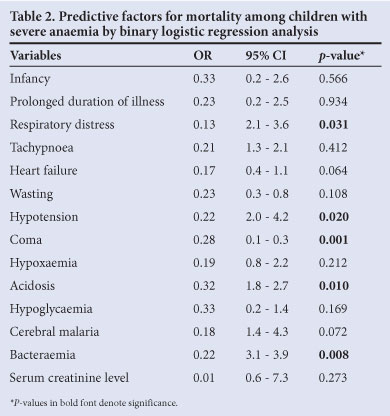
Predictive factors for mortality among children with severe anaemia were analysed by binary logistic regression of all the variables initially found to be significantly related to death using Pearsons's chi-squared test of association. Table 2 shows that the presence of respiratory distress (95% CI 2.1 - 3.6, p=0.031), acidosis (95% CI 1.8 - 2.7, p=0.010), coma (95% CI 0.1 - 0.3, p=0.001), hypotension (95% CI 2.0 - 4.2, p=0.020) and bacteraemia (95% CI 3.1 - 3.9, p=0.008) were significant independent predictors of death in children with severe anaemia.
Discussion
Childhood anaemia remains a serious health problem in most of sub-Saharan Africa.10 In this study, severe anaemia accounted for 17.9% of paediatric emergencies, which is within the 10 - 30% hospital prevalence in some African studies.1,2 However, in areas where delayed presentation to the hospital is common, hospital incidence of severe anaemia was higher.11 In this study, severe anaemia was more prevalent among preschool children and infant males from the low socio-economic status. This is probably because the same group of children are more susceptible to malaria, which accounted for more than 90% of cases of severe anaemia in our unit, either singly or in combination with other diseases.
Our in-hospital case fatality rate of 9.3% is also similar to those in previous studies.1,2 Mortality remains high, despite availability of blood transfusion services in our hospital. Sadly, one out of every four severely anaemic children died before blood transfusion was given.
In this study, severely anaemic children who died were significantly younger than survivors (mean age 18.8 (SD 8.1) v. 36.2 (SD 30.5) months). Younger children, particularly infants and preschool children, tend to develop severe anaemia more rapidly during haemolytic processes because of their low iron stores and poor immune response to P. falciparum infection.12
Prolonged duration of illness (>72 hours) before presentation was significantly related to poor outcome among our patients. Although it was not found to be an independent predictor of death with regression analysis, longer duration of illness or repeated attacks of untreated illness before presentation are likely determinants of anaemia severity.
Our finding of respiratory distress as a predictor of adverse outcome among children with severe anaemia agrees with previous studies.1,13 Respiratory distress (presence of fast breathing, chest in-drawing, grunting and flaring of nostrils) increases case fatality rates of severely anaemic children 10 times.13 This may result from lower oxygen concentration and metabolic acidosis commonly seen in such children. In this study, hypoxaemia (SaO2<90%) and metabolic acidosis were also significantly related to death. Features such as tachypnoea, tachycardia, hepatomegaly, gallop rhythm, lethargy and grunting, which are characteristic of heart failure in anaemic children, are thought to arise from lactic acidosis.4,13
Consistent with previous studies,12,13 high-density parasitaemia was neither significantly related nor a predictor of death from severe anaemia. A study on the clinical predictors of severe malaria anaemia in a holo-endemic P. falciparum transmission area found that children with uncomplicated malaria had more peripheral parasitaemia, geomean parasitaemia and proportion of high-density parasitaemia than those with severe anaemia.13 Our findings therefore support the hypothesis that immune response to P. falciparum rather than the cumulative parasite burden may be the primary factor responsible for the development and promotion of severe malaria anaemia in holo-endemic regions.13
Of our patients, 26% had bacteraemia and 55.2% of those who died had bacteraemia. Hence, in Africa, where bacterial infections such as staphylococcal and Salmonella infections are prevalent,14 the use of antibiotics for severely anaemic children may be justified, especially where blood culture facilities are not readily available. Our study found that hypotension predicted death among the children with severe anaemia, consistent with Maitland et al.14 who concluded that, although hypovolaemia and resultant hypotension are infrequently seen in children with severe malaria, impaired tissue perfusion shares features with sepsis syndrome and significantly contributed to deaths.
Children with severe anaemia may present with coma owing to metabolic dysfunction (lactic acidosis and/or hypoxia) which depresses the level of consciousness.15 In our study, more patients with coma died, which indicates that aggressive resuscitation measures are needed for children with coma and severe anaemia.
Our study shows that mortality risk in children with severe anaemia depends on clinical characteristics such as respiratory distress, coma, hypotension and presence of co-morbidities, especially acidosis and bacteraemia. In Africa, where severe anaemia is prevalent and causes considerable childhood morbidity and mortality, early identification of these factors, anticipatory supportive care and timely resuscitative measures in addition to blood transfusion are essential to reducing deaths from severe anaemia.
References
1. Phiri KS, Calis JCJ, Faragher B, et al. Long term outcome of severe anaemia in Malawian children. PLoS ONE 2008;3(8):e2903.[http://dx.doi.org/10.1371/journaLpone.0002903] [ Links ].
2. Lackritz EM, Campbell CC, Ruebush TK II, et al. Effect of blood transfusion on survival among children in a Kenyan hospital. Lancet 1992;340:524-528. [ Links ]
3. World Health Organization. Guidelines for the Treatment of Malaria. (WHO/HTM/MAL/2006.1108). Geneva: WHO, 2006:1-41. [ Links ]
4. Oyedeji OA, Oluwayemi IO, Oyedeji AT, Okeniyi JA, Fadero FF. Heart failure in Nigerian children. Cardiology 2010;5(3-4):18-22. [ Links ]
5. Oyedeji GA. Socioeconomic and cultural background of hospitalized children in Ilesa. Nig J Paediatr 1985;13:11-17. [ Links ]
6. Blössner M, de Onis M. Malnutrition: quantifying the health impact at national and local levels. (WHO Environmental Burden of Disease Series, No. 12). Geneva: WHO, 2005. [ Links ]
7. National High Blood Pressure Education Program. Working Group on High Blood Pressure in Children and Adolescents. The Fourth Report on the Diagnosis, Evaluation, and Treatment of High Blood Pressure in Children and Adolescents. Pediatrics 2004;114(suppl):555-576. [ Links ]
8. Basnet S, Adhikari R, Gurung C. Hypoxemia in children with pneumonia and its clinical predictors. Indian J Pediatr 2006;73:777-781. [ Links ]
9. Bain BJ. Basic Haematological Techniques. In: Davies JV, Lewis SM, eds. Practical Haematology. 8th ed. London: Churchill Livingstone, 1995:37-66. [ Links ]
10. English M, Ahmed M, Ngando C, Berkley J, Ross A. Blood transfusion for severe anaemia in children in a Kenyan hospital. Lancetm2002;359:494-495. [ Links ]
11. Adediran IA, Adejuyigbe EA, Oninla SO. Haematological profiles and malaria parasitaemia in Nigerian children requiring emergency blood transfusion. Niger J Med 2003;12(3):130-133. [ Links ]
12. Obonyo CO, Vulule J, Akhwale WS, Grobbee DE. Inhospital morbidity and mortality due to severe anaemia in western Kenya. Am J Trop Med Hyg 2007;77(6):23-28. [ Links ]
13. Novelli EM, Hittner JB, Davenport GC, et al. Clinical predictors of severe malaria anaemia in a holoendemic Plasmodium falciparum transmission area. Br J Haematol 2010;149(5):711-721.[http://dx.doi.org/10.1111/j.1365-2141.2010.08147.x] [ Links ].
14. Maitland K, Levin M, English M, et al. Severe Plasmodium falciparum malaria in Kenyan children: evidence for hypovolaemia. Q J Med 2003;96(6):427-434. [ Links ]
15. Idro R. Severe anaemia in childhood cerebral malaria is associated with profound coma. Afr Hlth Sci 2003;3(1):15-18. [ Links ]
Accepted 9 February 2012.
 Corresponding author:
Corresponding author:
SA Adegoke
(adegoke2samade@yahoo.com)














