Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 no.6 Pretoria Jun. 2012
RESEARCH
Phaco-emulsification versus manual small-incision cataract surgery in South Africa
Colin CookI; Henri CarraraII; Landon MyerIII
IFCS (Ophth) SA. Division of Ophthalmology, Groote Schuur Hospital and University of Cape Town
IIMPH. School of Public Health and Family Medicine, University of Cape Town
IIIPhD. School of Public Health and Family Medicine, University of Cape Town
ABSTRACT
OBJECTIVES: To compare the results of phaco-emulsification cataract surgery and manual small-incision cataract surgery.
METHODS: Consecutive patients aged >50 years undergoing surgery for age-related cataract were recruited into a randomised prospective clinical trial. Randomisation was done using opaque sequentially numbered envelopes opened by the surgeon immediately prior to surgery. The patients were seen after 1 day, 2 weeks, and 8 weeks.
OUTCOME MEASURES: The primary outcome measure was the uncorrected visual acuity at week 8. The secondary outcome measures were the uncorrected visual acuity on day 1, the best corrected visual acuity at week 8, the refraction at week 8, and the intra- and postoperative complications.
RESULTS: One hundred patients were recruited into each arm of the study. There was no difference in the incidence of intraocular complications (p=0.19). There was no difference in the day 1 visual acuities (p=0.28). However, both the uncorrected and the corrected week 8 visual acuities were better in the eyes that had phaco-emulsification (p=0.02 and p=0.03), and there was less astigmatism (p=0.001) at week 8 in the eyes that had phacoemulsification.
CONCLUSIONS: While manual small-incision surgery has been recommended as an acceptable alternative to phaco-emulsification in middle- and low-income countries, we have found that the results of phaco-emulsification are better. Where appropriate, consideration should be given to encouraging a transition to phaco-emulsification in our Vision 2020 programmes in Africa.
Cataract is the leading cause of blindness globally and in Africa, and the delivery of high-volume, high-quality, low-cost cataract surgery has been prioritised in the Vision 2020 programmes in Africa.1
While phaco-emulsification surgery has become the standard in high-income countries,2 financial constraints (capital costs for the purchase of equipment and running costs for the provision of consumables) preclude the routine use of phaco-emulsification in low-income countries.1 Manual small-incision surgery has many of the benefits of phaco-emulsification surgery, and is the standard in most Vision 2020 programmes in Africa.3
With the increasing availability of low-cost consumables for phaco-emulsification machines and low-cost foldable intra-ocular lenses, there is interest in introducing phaco-emulsification surgery in our Vision 2020 programmes. There remains a concern, however, that it may be unsuitable for some cataracts because of the advanced maturity and hardness of the lens nucleus.4
Three published randomised clinical trials have compared the results of phaco-emulsification versus manual small-incision surgery: in India, Gogate and others5 found that phaco-emulsification gave better results at six weeks post operatively; in Nepal, Ruit and others,6 and in India, Venkatesh and others,7 found that both techniques gave similar results, but that manual small-incision surgery is faster, less expensive, and less technology-dependent than phaco-emulsification. Thus manual small-incision surgery appeared more appropriate in low-income countries.6,7 There have been no reported randomised clinical trials comparing the 2 techniques in Africa. In a retrospective review of all cataract surgery done over a 12-month period at Groote Schuur Hospital (GSH), there was more postoperative astigmatism following manual small-incision surgery, the visual acuity at the first postoperative visit was better following phaco-emulsification surgery, but at the final postoperative visit there was no difference in either the uncorrected or the corrected visual acuity.
This prospective randomised clinical trial was conducted as a follow-up to this retrospective review to compare the outcomes of both techniques and to determine whether phaco-emulsification surgery should be promoted for Vision 2020 programmes in Africa.
Methods
Ethical approval was obtained from the Ethics Committee of the University of Cape Town, and the trial was registered with the South African Department of Health (DOH-27-0810-3226). Informed consent was obtained from each of the study participants. Consecutive patients aged over 50 years who were undergoing surgery for age-related cataract at GSH and who agreed to participate in the study were recruited. Patients with early cataract (visual acuity better than 6/36), and patients with co-existent glaucoma or corneal scar, were excluded. Assuming 1:1 randomisation, 90% power (á=0.05), and a precision error of 5% to detect a difference of 20% or more in the week 8 postoperative uncorrected visual acuity between the 2 groups, the required sample size was calculated to be 266. To account for loss to follow-up, the aim was to randomly assign 280 patients to either of the 2 surgical techniques. An interim analysis was conducted after 200 patients had been recruited, and no further patients were recruited.
Randomisation to the 2 arms of the study was done using opaque, sequentially numbered envelopes. The randomisation sequence allocation was generated by a research assistant who randomly selected and numbered sequential envelopes containing an instruction on the type of surgery to be done. These envelopes were kept in the operating room, and the next numbered envelope was opened by the surgeon immediately prior to surgery.
Patients were not informed about the method of surgery, and the ophthalmic assistants and nurses, who tested and recorded the postoperative visual acuities, were blinded to the surgery undertaken.
All patients underwent a routine pre-operative assessment, with measurement of visual acuity, dilated slit-lamp examination of the anterior segment, measurement of the intra-ocular pressure by Goldmann applanation tonometry, A-scan biometry, and fundus examination with either a 90-dioptre lens and indirect ophthalmoscope or B-scan ultrasound.
On the day of the operation, the pupil was dilated with topical tropicamide 1% and sub-tenon's anaesthesia administered approximately 10 minutes before surgery. The procedure was performed by any one of 5 consultants or 10 registrars, all of whom were competent in the selected technique. Standard surgical techniques were used.
For phaco-emulsification surgery, a temporal 3.0 mm clear corneal incision was made and a continuous curvilinear capsulorrhexis was created, using trypan blue if necessary, following which hydrodissection was performed. Phaco-emulsification was performed using an Infinity phaco-emulsification system (Alcon) with a phaco-chop technique. The remaining cortex was aspirated using the irrigation/aspiration tip. The capsule bag and anterior chamber were filled with hydroxypropyl methylcellulose 2%, and a 6.0 mm optic foldable acrylic intra-ocular lens (Tecsoft) was implanted in the capsule bag. The corneal incision was hydrated and was left unsutured in most cases. After aspiration of the visco-elastic, cefuroxime 1.0 mg in 0.1 ml was injected into the anterior chamber.
For manual small-incision surgery, a 7.0 - 8.0 mm wide and 4 mm long superior sclerocorneal tunnel was constructed, starting 2 mm from the 12 o'clock limbus and extending 2 mm into the cornea. Either a continuous curvilinear capsulorrhexis or an envelope capsulotomy was performed, with trypan blue if necessary. The nucleus was prolapsed from the capsular bag by hydrodissection, and it was then extracted by hydro-expression using a Simcoe cannula. The capsule bag and anterior chamber were filled with hydroxypropyl methylcellulose 2%, and a 6.0 mm optic single-piece rigid polymethylmethacrylate intra-ocular lens (Fred Hollows Foundation) was implanted in the capsule bag. If an envelope capsulotomy had been performed, the capsulectomy was completed. The scleral incision was sutured with one 10-0 nylon suture. After aspiration of the visco-elastic, cefuroxime 1.0 mg in 0.1 ml was injected into the anterior chamber.
Any intra-operative complications were documented. Routine postoperative care included a topical antibiotic-steroid combination drop 4 times daily for 8 weeks.
Patients were requested to return for review after 1 day, 2 weeks, and 8 weeks. At each visit, the visual acuity and objective refraction were recorded by an ophthalmic technician or nurse, and slit-lamp examination of the anterior segment and fundus was undertaken by the surgeon. At the week 8 visit, a subjective refraction was done and the corrected visual acuity recorded.
The primary outcome measure was the uncorrected visual acuity at week 8. The secondary outcome measures were the uncorrected visual acuity on day 1, the best corrected visual acuity at week 8, the refraction at week 8, and the intra- and postoperative complications.
Data were analysed using Stata (version 11.1) on an intention-to-treat basis. Proportions were compared using the chi-squared test and, where cell frequencies were <5, the exact test was employed. Means and standard deviations were reported for normally distributed variables and compared in the 2 groups using the t-test. Medians and interquartile ranges were reported for variables that were found not to be normally distributed, and comparisons between the 2 groups were made using the Wilcoxon rank sum test. Two-sided p-values were reported.
Results
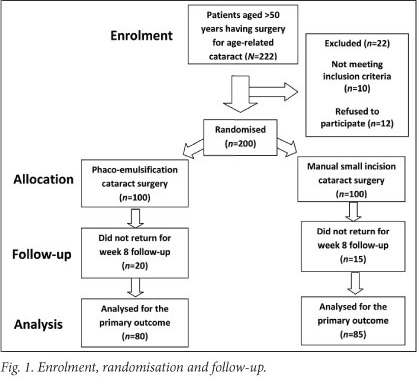
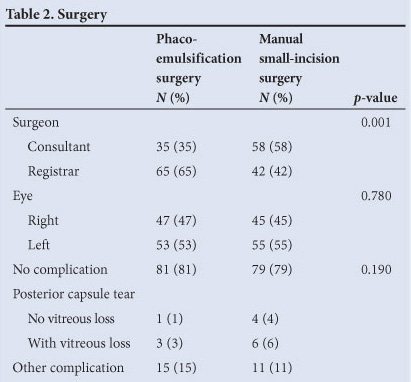
Two hundred subjects were recruited. Fig. 1 summarises the flow of the participants through each stage of the study. Thirty-five subjects (18%) were lost to follow-up at week 8. Table 1 shows the participant baseline demographic and clinical characteristics. There was no difference between the 2 groups. Table 2 shows the number of operations performed by consultants and by registrars, and the intra-operative complications. The registrars performed more phaco-emulsification procedures, and the consultants performed more manual small-incision operations. While there was a higher incidence of posterior capsule tear, both with and without vitreous loss, in the eyes having manual small-incision surgeries, this difference was not significant (p=0.34). Eight planned phaco-emulsification operations were converted to manual small-incision procedures because the nucleus was considered to be too hard for phaco-emulsification.
Day 1 complications and visual acuities. More eyes in the phaco-emulsification group had corneal oedema compared with the manual small-incision group on day 1 (35 v. 29), but this difference was not significant (p=0.36) and there was no difference in the visual acuities.
Week 8 visual acuities and refractions. Table 3 shows the visual acuities and refractions at week 8 following surgery. There was less astigmatism in the phaco-emulsification group, and both the uncorrected and the corrected visual acuities were better in the phaco-emulsification group.
Discussion
At week 8 post-surgery, there was less astigmatism and both the uncorrected and the corrected visual acuities were better in the phaco-emulsification group. The operations were done by a team of 5 consultants and 10 registrars, with competence in both surgical techniques, but with varying levels of expertise. Registrars did more of the phaco-emulsification procedures, and consultants more of the manual small-incision operations. Assuming that consultants had greater surgical expertise than residents, this could have introduced a bias favouring the outcome of the manual small-incision operations.
While there might have been an advantage to using an expertise design and limiting the surgeons to just one or two 'experts',8 our lack of an expertise design probably better reflects the reality in most African Vision 2020 programmes and may enhance the external validity of our findings. The earlier trials did use an expertise design but found no difference between the 2 procedures.6,7 The trial reported by Gogate et al.5 did not use an expertise design, and found phaco-emulsification to be better. Our findings, and those of Gogate and others, would seem to suggest that the results of phaco-emulsification are better.
Eight per cent of the planned phaco-emulsification operations were converted to manual small-incision operations because the lens nuclei were considered to be too hard for phaco-emulsification. The problems attendant on phaco-emulsification surgery in brunescent cataracts with hard nuclei are recognised.9 Venkatesh et al.7 found that phaco-emulsification and manual small-incision surgery gave comparable results in mature white cataracts, and in only 3/133 eyes randomised to phaco-emulsification surgery was conversion to manual small-incision surgery necessary.
It was obviously not possible to blind the surgeons to the surgery technique used, and they examined the eyes postoperatively. However, the patients, and the ophthalmic technicians and nurses who documented the postoperative visual acuities and refractions, were masked.
Eighteen per cent of our patients were lost to follow-up at 8 weeks (Table 3). (Our patients are indigent people living both within the Cape Town Metropole and in more distant rural areas, and this loss to follow-up is difficult to control.)
The earlier studies quoted5-7 emphasised the advantage of manual small-incision surgery because it is faster. In our African setting, with lower population densities and lower surgery volumes, reductions by a few minutes in individual surgery times are less critical.
We have found that the results of phaco-emulsification are better. Where appropriate, consideration should be given to encouraging the inclusion of phaco-emulsification in our African Vision 2020 programmes.
Acknowledgements
We wish to thank the consultants and registrars of the ophthalmology department at Groote Schuur Hospital.
References
1. Ellwein LB, Kupfer C. Strategic issues in preventing cataract blindness in developing countries. Bull World Health Org 1995;73(5):681-690. [ Links ]
2. Zaidi FH, Corbett MC, Burton BJ, Bloom PA. Raising the benchmark for the 21st century - the 1 000 cataract operations audit and survey. Br J Ophthalmol 2007;91(6):731-736. [ Links ]
3. Hennig A, Kumar J, Yorston D, Foster A. Sutureless cataract surgery with nucleus extraction - outcome of a prospective study in Nepal. Br J Ophthalmol 2003;87(3):266-270. [ Links ]
4. Singh R, Vasavada AR, Janaswamy G. Phacoemulsification of brunescent and black cataracts. J Cataract Refract Surg 2001;27(11):1762-1769. [ Links ]
5. Gogate PM, Kulkarni SR, Krishnaiah S, Deshpande RD, Joshi SA, Palimkar A. Safety and efficacy of phacoemulsification compared with manual small incision cataract surgery by a randomized controlled clinical trial - six week results. Ophthalmology 2005;112(5):869-874. [ Links ]
6. Ruit SA, Tabin G, Chang D, Bajracharya L, Kline DC, Richheimer W. A prospective randomised clinical trial of phacoemulsification vs manual sutureless small incision extracapsular cataract surgery in Nepal. Am J Ophthalmol 2007;143(1):32-38. [ Links ]
7. Venkatesh R, Tan C, Sengupta S, Ravindran R, Krishnan K, Chang D. Phacoemulsification versus manual small incision cataract surgery for white cataract. J Cataract Refract Surg 2010;36:1849-1854. [ Links ]
8. Devereaux PJ, Bhandari M, Clarke M, et al. Need for expertise based randomised controlled trials. BrMed J 2005;330:88. [ Links ]
9. Bourne R, Minassian D, Dart J, Rosen P, Kaushal S, Wingate N. Effect of cataract surgery on the corneal endothelium - modern phacoemulsification compared with extracapsular cataract surgery Ophthalmology 2004;111(4):679-685. [ Links ]
Accepted 31 January 2012.
Corresponding author: C Cook (colin.cook@uct.ac.za)














