Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 no.6 Pretoria Jun. 2012
RESEARCH
A randomised controlled trial of suture materials used for caesarean section skin closure: Do wound infection rates differ?
A ChunderI; J DevjeeII; S M KhedunIII, J MoodleyIV; T EsterhuizenV
IMB BS, Dip O&G (SA). Department of Obstetrics and Gynaecology, Addington Hospital, and Women's Health and HIV Research Group, University of KwaZulu-Natal, Durban
IIMB ChB, Dip O&G. Department of Obstetrics and Gynaecology, Addington Hospital, and Women's Health and HIV Research Group, University of KwaZulu-Natal, Durban
IIIMMed. Women's Health and HIV Research Group, Nelson R Mandela School of Medicine, University of KwaZulu-Natal S
IVMB ChB, FCOG, FRCOG, MD. Women's Health and HIV Research Group, Nelson R Mandela School of Medicine, University of KwaZulu-Natal S
VMSc. Women's Health and HIV Research Group, Nelson R Mandela School of Medicine, University of KwaZulu-Natal S
RESUMO
OBJECTIVE: The aim of this study was to determine wound complication rates following the use of suture materials and staples for skin closure at caesarean section (CS).
STUDY DESIGN: A randomised, controlled, prospective study was undertaken.
RESULTS: A total of 1 100 women was assigned randomly into 3 groups: polyglycolic acid (PGA) suture group (N=361), skin staple (SS) group (N=373) and nylon suture group (N=366). The overall wound infection rate was 7%. There was no difference in respect of number of patients, age, parity and gestation between the study groups. Those who had nylon sutures as opposed to PGA sutures were 9.5 times more likely to experience wound infection (p=0.055). Women who had SS were at 6.93 times higher risk of wound infection than those who had PGA sutures (p=0.014). Other factors influencing wound infection rates included: rupture of membranes >12 hours were 13.7 times (95% confidence interval (CI) 3.9 - 47.9, p<0.0001) more likely to have wound infection than those with rupture of membranes <12 hours. For every 1-minute increase of surgery duration, the risk of infection increased 1.094 times (95% CI 1.046 - 1.145; p<0.0001). HIV-infected women were 53.4% less likely to develop wound infection than their uninfected counterparts (odds ratio 0.466, 95% CI 0.238 - 0.913; p=0.026). As the time period of observation increased from baseline to day 3 and from day 3 to day 10, wound infection risk increased by 35 times (95% CI 8.155 - 150.868; p<0.001).
CONCLUSION: The use of SS for CS wound closure is associated with a significantly greater risk of wound infections. SS for wound closure at CS is not recommended for use in South African district hospitals.
A variety of suture materials and skin staples (SS) are used for skin closure after caesarean section (CS). Some of these suture materials have been associated with lower wound infection rates, reduced pain, improved cosmetic outcomes and cost-effectiveness.1 On the other hand, SS are easier to use and are associated with a three- to fourfold reduction in time for skin closure at similar rates of wound infection.2 However, they are more expensive than suture materials and it is reported that SS are more painful and result in a poorer cosmetic appearance.3
Most studies on suture materials and SS for skin closure following CS are limited to cosmetic aspects, patient satisfaction and postoperative pain relief,4-6 with conflicting outcomes. Furthermore, these studies were done in high-income countries. In middle- and low-income countries, such as South Africa, where infection rates associated with pregnancy are high and most CS are done for prolonged labour and/ or cephalopelvic disproportion, there is no standard material for skin wound closure at CS. We observed that currently polyglycolic acid (PGA), nylon sutures and SS are used for wound closure following CS, with surgeon preference guiding the choice.
A Cochrane review concluded that there were insufficient data to recommend any technique or materials for CS wound closure.7 We therefore planned to determine whether there is any difference in the frequency of wound complications with two suture materials (PGA and nylon) and SS used for skin closure at CS.
Methods
A randomised prospective clinical trial was conducted from August 2009 until May 2010 at a district hospital in South Africa, after institutional ethical permission had been granted. All women undergoing CS were given the opportunity to participate and were excluded if they declined.
A computer-generated randomisation sequence with sealed envelopes randomly assigned women to 3 groups (i) PGA sutures, ( ii) SS and ( iii) nylon sutures. Given that the incidence of wound complication ranges between 5% and 10% worldwide, 1 620 patients (540 in each group) were scheduled to be studied. An interim analysis was not planned but was done because patients in the SS group complained of severe discomfort at the time of removal of the SS and it was felt that it was unethical to proceed with the study. The interim analysis was therefore carried out following the recruitment of 1 100 patients.
The CS technique was standardised except for the materials used for wound closure, which were allocated according to the randomised sequence. All CS were carried out by medical officers or interns under supervision. All patients had a Pfannenstiel incision and all skin sutures were inserted subcutaneously: PGA (absorbable), straight needle size 2/0, continuous suture (Ethicon); nylon (non-absorbable) monofilament, curved needle size 2-0, interrupted sutures (Clinisut); and staples (interrupted placement) (Smith & Nephew)). Prior to CS, the surgeon opened the sealed opaque envelope next in sequential order to identify the closure material.
Intravenous prophylactic antibiotics were given following clamping of the umbilical cord after both emergency and elective CS. HIV-infected patients received a 3-day course of therapeutic antibiotics (gentamicin 400 mg daily and Augmentin 1.2 g 3 times a day). Patients who had premature or prolonged rupture of membranes were also given therapeutic antibiotics. Demographic and delivery data were obtained prospectively from the medical records. The body mass index (BMI) of patients was not calculated because this hospital did not routinely measure height.
The surgical wounds were assessed by medical officers on day 1 and day 3 after CS and on day 10 at an outpatient clinic to assess the presence of wound infection. Wound infection was classified as follows: clean, healed wound; superficial (serous or serosanguinous discharge with or without breakdown of skin; deep (breakdown of skin and exposure of sheath); wound dehiscence (breakdown of sheath but peritoneum intact) and fasciitis (spreading of deep wound infection to fascial tissue).8 Nylon sutures and SS were removed on day 10 postoperatively. There was no loss to follow-up of patients.
Statistical analysis
SPSS version 18 was used to analyse the data. A p-value <0.05 was considered statistically significant. Outcome was dichotomised into any unhealed wound (codes 1 - 3) v. no unhealed wounds (code 0), and generalised estimating equations were used to estimate the effect of the intervention on having any unhealed wounds while controlling for various confounders such as age, parity, HIV, etc. Odds ratios (ORs) and 95% confidence intervals (CIs) are reported.
Results
Of the 1 522 women who were eligible to participate, 422 were excluded because: (i) 177 declined participation, (ii) 13 initially booked for CS delivered vaginally, and (iii) 232 were eligible but were not randomised because of logistical problems. The remaining 1 100 were assigned randomly into 3 groups: 361 were in the PGA group, 373 in the SS group, and 366 in the nylon group (Fig. 1). There was no significant difference in the number of participants in each group.
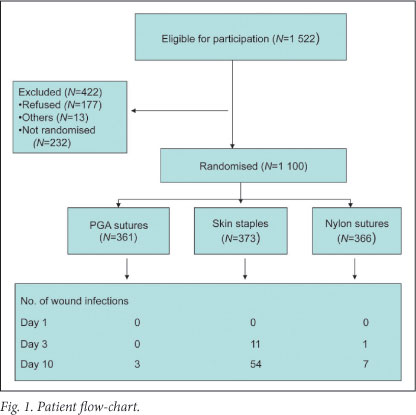
Table 1 lists the demographic and the clinical characteristics of participants. There was no difference in clinical characteristics between the study groups.
Wound complication rates
Wound complications occurred in 76 (7%) of the 1 100 patients: 1% (3/361) in group 1 (PGA sutures), 17% (65/373) in group 2 (SS), and 2% (8/366) in group 3 (nylon sutures).
Of wound complications, 64/76 (84%) occurred following discharge from hospital; 12 (16%) occurred during hospitalisation (11 in group 2 and 1 in group 3). All 12 wound complications that occurred in hospital were classified as superficial and were detected within 24 hours of CS.
Sixty-four (84%) wound complications were observed on day 10 at the follow-up visit. Of the 64 wound complications, 10 were classified as superficial, 34 as deep, and 20 involved wound breakdown. Two wound complications classified as deep and 12 classified as wound breakdown occurred in the SS group and the patients required hospitalisation, while the remainder were treated as outpatients. No patients with wound infections required surgical closure.
After controlling for confounders such as age, parity, number of vaginal examinations, HIV status and rupture of membranes >12 hours, patients who had nylon sutures v. those with PGA sutures were 9.5 times more likely to experience wound infection, but the difference was not statistically significant (p=0.055). Patients who had SS were at 6.93 times higher risk of wound infection (p=0.014) than those who had PGA sutures. However, besides the type of suture, other factors influenced wound infection. Women with rupture of membranes >12 hours were 13.7 times (95% CI 3.9 - 47.9; p<0.0001) more likely to have wound infection than those with rupture of membranes <12 hours. For every 1-minute increase in the duration of surgery, the risk of infection increased by 1.094 times (95% CI 1.046 - 1.145; p<0.0001). HIV-infected women were 53.4% less likely to develop wound infection than uninfected women (OR 0.466, 95% CI 0.238 - 0.913; p=0.026). As the time period of observation increased from baseline to day 3 and from day 3 to day 10, the risk of wound infection increased by 35 times (95% CI 8.155 - 150.868; p<0.001). The post-CS wound infection rate did not appear to be affected by patient weight, level of surgeon, whether the CS was an emergency or an elective procedure, or the number of vaginal examinations.
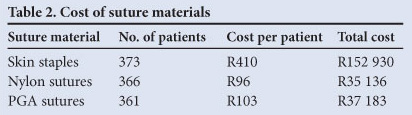
Cost of suture materials
Table 2 shows the cost (in rands) of the suture materials used in the study.
Discussion
The overall rate of wound complications following CS in this randomised clinical trial was 7% compared with rates from 0 to
25% in other studies.6,9-13 This variation probably results from several factors in the various studies.9-13 Firstly, studies with broad exclusion criteria have lower wound complication rates than studies with stringent exclusion criteria. The wound complication rate in our study was 7%, with the only exclusion criterion being declining to participate. Secondly, studies (including our study) in which the subcutaneous fat layer was sutured prior to skin closure, reported lower wound complication rates than those in which the subcutaneous fat layer had not been sutured.4,6,11
A limitation of our study was that we did not measure BMI and therefore did not compare wound infection rates in different BMI groupings. The mean weights in all 3 groups were similar.
A pilot study involving 416 patients randomised to groups (sutures and SS) showed that post- CS wound complications were significantly greater when SS were used;6 the incidence of wound infections in this study was obtained by telephonic interview, 2 - 4 weeks after surgery. Our larger sample size and actual detection of CS wound infections on patients' return to hospital showed an incidence of 7%. The high rate of wound complications observed with the use of SS could be explained by either the difficulty in SS placement in a population group in which the incidence of obesity is high, or allergic or inflammatory reactions associated with the metallic nature of the SS. However, we did not study this objectively. Reviews and meta-analysis indicate that SS used for skin closure after CS are associated with a high incidence of wound complications.13,14
Surgical site infection surveillance studies following CS show that most wound infections (637/758 (84%) and 148/241 (61.4%)) occur at home after discharge from hospital.15,16 In our study, 64 (84%) of the 76 wound infections occurred following discharge from hospital; this was despite the fact that our standard practice is to prescribe prophylactic antibiotics at the time of CS and patients are given information regarding wound care. Most of our patients are from low socio-economic backgrounds.
An earlier study demonstrated an association between maternal age, previous CS, anaemia, multiple vaginal examinations, prolonged labour and post-CS wound infection.17 Our study could not confirm this association.
Our study confirmed the role of rupture of membranes >12 hours as a predisposing factor to developing wound infection, as reported by Koigi-Kamau et at.10 A short operation time considerably reduces the risk of developing postoperative wound infection.14 In our study, the risk of wound infection increased for every 1-minute increase in the duration of surgery. Women who were HIV-infected were less likely to develop wound infection than those uninfected. This finding was surprising; it may be because, in our study, all HIV-infected patients received therapeutic antibiotics compared with their non-infected counterparts, who only received prophylactic antibiotics.
Although detailed cost analysis was not performed, there was a fourfold greater financial cost incurred when SS were used for skin closure. Had sutures been used, hospital savings of more than R117 000 would have been achieved - a significant saving for health systems in low- and middle-income countries. Comparing subcuticular sutures and SS for CS wound closure, Basha et al.6 found that sutures were four times cheaper than SS. We found no differences in wound infection rates and financial costs between PGA and nylon suture materials. However, we did not take into account other costs such as anaesthetic, surgical time and staffing.
We conclude that using SS for CS wound closure is associated with a significantly greater risk of infections, and recommend that SS not be used for CS skin closure in district hospitals.
References
1. Altman AD, Allen VM, McNeil SA, Dempster J. Pfannenstiel incision closure: a review of current skin closure techniques. J Obstet Gynaecol Can 2009;31(6):514-520. [ Links ]
2. The CORONIS Trial Collaborative Group. International study of caesarean section surgical techniques: a randomised fractional, factorial trial. BMC Pregnancy Childbirth 2007;7:24-58. [ Links ]
3. Alderdice F, McKenna D, Dornan J. Techniques and materials for skin closure in caesarean section. Cochrane Database Syst Rev 2003;2:CD003577. [ Links ]
4. Rousseau JA, Girard K, Tircot-Lemay L, Thomas N. A randomised study comparing skin closure in cesarean section: staples vs subcuticular sutures. Am J Obstet Gynecol 2009;200:265.e1-265.e4. [ Links ]
5. Cromi A, Ghezzi F, Gottardi A, et al. Cosmetic outcomes of various skin closure methods following cesarean delivery: a randomised trial. Am J Obstet Gynecol 2010;203(1):36e1-36e8. [ Links ]
6. Basha SL, Rochon ML, Quinones JN, Coassolo KM, Rust OA, Smulian JC. Randomized controlled trial of wound complication rates of subcuticular suture vs staples for skin closure at cesarean delivery. Am J Obstet Gynecol 2010;203(1):285.e1-8. [ Links ]
7. Anderson ER, Gates S. Techniques and materials for closure of the abdominal wall in caesarean section. Cochrane Database Syst Rev 2004;4:CD004663. [ Links ]
8. Olsen MA, Butler AM, Williers DM, Devkota P, Gross GA, Fraser VJ. Risk factors for surgical site infections after low transverse caesarean section. Infect Control Hosp Epidemiol 2008;29(6):477-484. [ Links ]
9. Gaertner I, Burkhardt T, Beinder E. Scar appearance of different skin and subcutaneous tissure closure techniques in caesarean section: a randomized study. Eur J Obstet Gynecol Reprod Biol 2008;138(1):29-33. [ Links ]
10. Koigi-Kamau R, Kabare LW, Wanyoike-Gichuhi J. Incidence of wound infection after caesarean delivery in a district hospital in central Kenya. East Afr Med J 2005;2(7):357-361. [ Links ]
11. Islam A, Ehsan A. Comparison of suture material and technique of closure of subcutaneous fat and skin in caesarean section. North Am J Med Sci 2011;3(2):85-88. [ Links ]
12. Ezechi CO, Edet A, Akinlade H, Gab-Okafor CV, Herbertson E. Incidence and risk factors for caesarean wound infection in Lagos Nigeria. BMC Research Notes 2009;2:186-190. [ Links ]
13. Tuuli MG, Rampersad RM, Carbone JF, Stamilio D, Macones GA, Odibo AO. Staples compared with subcuticular suture for skin closure after cesarean delivery: a systematic review and meta-analysis. Obstet Gynecol 2011;117(3):682-690. [ Links ]
14. Clay FS, Walsh CA, Walsh SR. Staples vs subcuticular sutures for skin closure at cesarean delivery: a metaanalysis of randomized controlled trials. Am J Obstet Gynecol 2011;204(5):378-383. [ Links ]
15. Petrosillo N, Drapeau CMJ, Nicastri E, Martini L, Ippolito G, Moro ML, ANIPIO. Surgical site infections in Italian Hospitals: a prospective multicentre study. BMC Infect Dis 2008;8:34-49. [ Links ]
16. Ward VP, Charlett A, Fagan J, Crawshaw SC. Enhanced surgical site infection surveillance following caesarean section: experience of a multicentre collaborative post-discharge system. J Hosp Infect 2008;70(2):166-173. [ Links ]
17. Litta P, Vita P, Konishi de Toffoli J, Onnis GL. Risk factors for complicating infections after cesarean section . Clin Exp Obstet Gynecol 1995;22(1):171-175. [ Links ]
Accepted 29 December 2011.
Corresponding author: J Moodley (tombe@ukzn.ac.za)














