Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 no.6 Pretoria jun. 2012
FORUM
HISTORY OF MEDICINE
Transplantation of the heart: an overview of 40 years' clinical and research experience at Groote Schuur Hospital and the University of Cape Town. Part II. Laboratory research experience
J Hassoulas
Professor Jannie Hassoulas is Associate Professor in the Medical School, University of Crete, Greece
Extensive experimental research on various aspects of heart transplantation was undertaken during the first 2 decades. An overview of this work is presented, and some still unpublished work has been included. Experimental laboratory investigation was an integral activity of the cardiac transplantation programme at the University of Cape Town over these years, and has remained so ever since. These studies provided invaluable fundamental information upon which future clinical work was based. It is therefore necessary to briefly mention and discuss this information, most of which has been published in detail by the various investigators concerned.
Heterotopic cardiac transplantation with a xenograft1,2
In an active cardiac surgery unit many occasions arise where temporary assistance of the left ventricle in cardiogenic shock after cardiopulmonary bypass is needed. Although counter-pulsation with an intra-aortic balloon pumping apparatus will provide excellent assistance in the majority of cases, not all patients benefit from it. Partial assistance of the circulation with a pump oxygenator, although superior to balloon counter-pulsation, is not very practical for technical reasons. An artificial heart for routine use was not yet available at the time. This option became a possibility only during the past decade. The use of a heterotopic cardiac transplant is therefore ideal in this situation, i.e. to provide temporary support to the circulation until the recipient's heart recovers.
Human donors, however, are not always available when required under these circumstances. High-risk procedures that may require such assistance can sometimes be postponed until a suitable donor is available. An alternative method is to use a xenograft heart temporarily for 3 - 4 days until the recipient's heart has recovered. This was done on two occasions when a human donor was not available and other methods of assisting the circulation had failed.
The first patient to receive a heterotopic cardiac transplant using a xenograft was a 25-year-old woman who underwent repeat aortic valve replacement together with an aortoventriculoplasty procedure to enlarge a small aortic root. On completion of the operation the patient could not be weaned from the pump oxygenator despite institution of all conventional supportive measures, including the intra-aortic balloon pump. Heterotopic heart transplantation was performed using the heart of a 30 kg baboon. After this, all other mechanical support was able to be discontinued. The recipient heart, however, fibrillated repeatedly in the postoperative period and eventually would not respond to electrical defibrillation. The donor heart was not large enough to support the circulation adequately under these conditions, although it maintained a moderate circulation alone for about 30 minutes. This heart also eventually fibrillated, about 5 hours postoperatively. At postmortem the recipient heart was found to have moderate but diffuse atherosclerosis of all major coronary arteries. The donor heart appeared to be normal macroscopically. On histological examination no signs of acute rejection were present, only a few contraction bands and mild interstitial oedema.
The second heterotopic xenograft heart transplant recipient was a 60-year-old man who, after aortic valve replacement for severe calcific aortic stenosis, also could not be weaned from the pump oxygenator. This time the donor heart was obtained from a blood group A rhesus-positive male chimpanzee. The operation was uneventful and the patient was returned to the intensive care unit with good circulation and fully conscious. High doses of immunosuppressive drugs were administered. During the following few days a steady and gradual deterioration in the function of both hearts was observed. Once the recipient heart had fibrillated, the donor heart could not support the circulation owing to the severe acute rejection it had undergone in this 4-day postoperative period. This rejection process was histologically documented at postmortem.
With the current development of mechanical circulatory support systems, this type of surgery is probably no longer necessary.
Orthotopic transplantation of a baboon heart after 20 - 24 hours' preservation3,4
Of patients accepted for cardiac transplantation, up to 50% die from progression of their cardiac disease while awaiting transplantation. In order to increase the availability of donor hearts, organs have to be procured from other centres in the country.
Short-term preservation of the donor heart, for up to 5 hours, has been successfully accomplished for many years, both locally and abroad. The method used for short-term preservation consists of initial cardioplegic arrest of the donor heart with a hyperkalaemic and hypertonic solution, together with topical cooling of the myocardium. After this the heart is harvested, placed in a sterile container containing cold saline, and then packed in ice for transportation. The clinical results following this procedure have been excellent. However, the cost is prohibitive as hire of a private aircraft is required. Also the time is too short for very long distances to be covered. A method of safe prolonged myocardial preservation would overcome these drawbacks, as there would be sufficient time to make use of commercial flights for transportation. The cost of obtaining donor hearts from distant centres would then be minimal. Our experimental efforts in this direction have proved that donor hearts can be successfully preserved for up to 24 hours.
Baboon hearts were rapidly excised after being flushed with 500 ml of cardioplegic solution at 4ºC and then immersed in cold -4ºC saline or cardioplegic solution for 2 minutes. The hearts were then perfused at a pressure of 8 - 10 cmH2O for 20 - 24 hours under refrigeration with a hyperosmolar clear fluid perfusate at 6 - 8ºC through which 95% oxygen and 5% carbon dioxide were continually bubbled to maintain the perfusate pH between 7.2 and 7.4. Myocardial temperature remained at approximately 6 - 8ºC. The hearts were then orthotopically transplanted into recipient baboons matched for size and AB blood group the next day. Two groups (A and B) were studied, differing significantly only in respect of the constitution of the cardioplegic solution and perfusate used. The cardioplegic agent used in group B contained a higher concentration of magnesium than that used in group A, and included the calcium antagonist verapamil. Perfusate B had higher osmolality than perfusate A, largely owing to the inclusion of sucrose. A preliminary group of 10 baboons in group A received no immunosuppression. Five of the remaining 6 immunosuppressed baboons in this group survived more than 48 hours to rejection or until sacrificed at 2 - 29 days. Cardiac catheterisation was performed in 6 surviving baboons (4 in group A, 2 in group B) between postoperative days 6 and 10 and showed good haemodynamic function. Histological examination of the hearts after death has shown only minor ischaemic changes in the hearts that functioned well.
Haemodynamic and metabolic effects of brain death on the donor heart
This subject was the topic of investigation in the research laboratory by a research team and was undertaken once the programme investigating prolonged preservation of the heart had been completed. Major haemodynamic changes definitely do occur after donor brain death, mainly owing to the diabetes insipidus caused by the brain oedema and elevated intracranial pressure. A consequence is loss of excessive amounts of fluids that in turn results in haemodynamic instability. To counteract this, large amounts of fluids are transfused to the donor together with inotropic agents and vasopressin. It follows that with these fluid changes, metabolic effects may occur as well.
Endocrine changes found to occur included catecholamine secretion and thyroid hormone depletion.5 In a further study,6 beneficial effects of hormonal therapy in donor management were shown. However, other studies in the literature, although they do document hormonal changes in the brain-dead animals or the donors, could not find any benefit in hormonal therapy.7,8 This topic remains very controversial.
Experimental heterotopic heart and single right lung transplantation (unpublished)
The transplantation of heart and lungs en bloc has the advantage of replacing these diseased organs in the recipient with healthy ones, thus deriving maximal physiological benefit from this procedure. Furthermore, all diseased recipient lung tissue, which could be a source of infection during the period of heavy immunosuppression, is removed. In certain conditions that would benefit from heart and lung transplantation however, such as Eisenmenger syndrome, idiopathic pulmonary hypertension and extreme pulmonary atresia, the retention of one relatively 'healthy' lung may be beneficial. Complete denervation of the whole respiratory tract is avoided, which may retain a more physiological respiratory pattern in the postoperative period. Consequently, this normal respiratory function will help prevent or diminish the magnitude of postoperative complications, including the lung 'reimplantation' response. With these considerations in mind we developed a method for the heterotopic transplantation of the heart together with the right lung en bloc after right pneumonectomy of the recipient.
Experimentation to prove the technical feasibility of this procedure was carried out in the animal laboratory. Briefly, the technique is as follows. Baboons weighing between 15 and 25 kg, matched for size and AB blood groups, were used. Two of the operations were performed through a right lateral thoracotomy through the 4th intercostal space, one with the aid of cardiopulmonary bypass and one without. A further 2 operations were performed through a median sternotomy; again, one was performed utilising cardiopulmonary bypass and one without.
Pneumonectomy of the recipient right lung was performed using standard techniques. The pericardial sac was opened above the right phrenic nerve and a piece of pericardium removed in the same fashion as for heterotopic heart transplantation. The donor heart and right lung were removed en bloc. After cardioplegic arrest of the heart and topical cooling of both donor organs, the pulmonary artery and veins were ligated and divided. The inferior vena cava was then ligated and divided. Next, the superior vena cava and ascending aorta were divided. The right bronchus was divided nearer the hilum than the carina to avoid possible ischaemia at the bronchial anastomosis site later on (Fig. 1). Further pericardial and hilar attachments of these organs were freed, taking control of possible bleeding sites that could occur after transplantation.
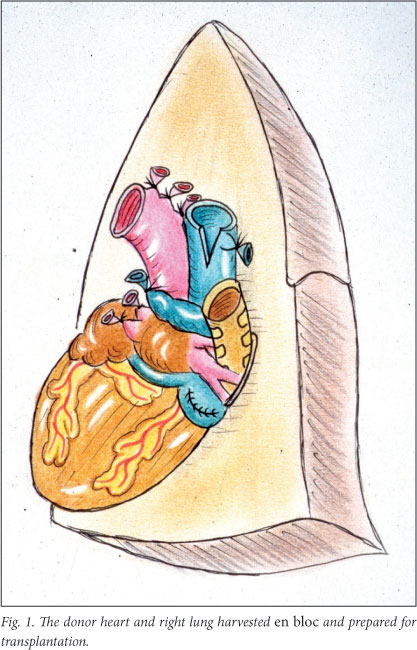
In this series of experiments great care was taken in the construction of the bronchial anastomosis. This contributed to the successful outcome in all 4 cases. The donor bronchus was telescoped into the recipient bronchus and the tissue of the suture line bolstered with vascularised tissue where possible.
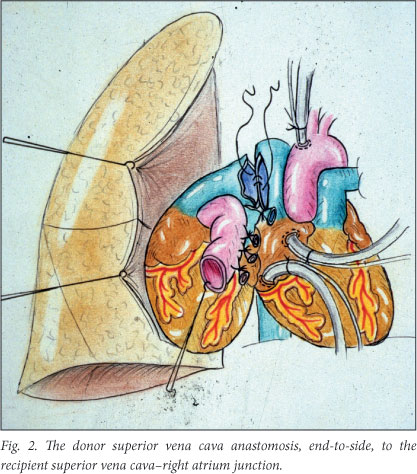
For completion of the operation (Fig. 2), the donor superior vena cava was anastomosed end-to-side to the recipient superior vena cava-right atrium junction, this anastomosis being made as wide as possible, and similarly the donor ascending aorta was anastomosed to the recipient ascending aorta in an end-to-side manner as well (Fig. 3). On releasing the superior vena cava and aortic side-clamps on completion of the anastomoses, a severe drop in blood pressure was experienced if the animal was not on cardiopulmonary bypass. In order to overcome this, prompt blood transfusion was necessary. Great care had to be taken to remove air from the donor left ventricle.

All animals lived for an average of 5 days postoperatively, and all died as a result of pulmonary oedema in the transplanted lung. At autopsy the bronchial anastomosis was found to be intact and open widely in all animals. No necrosis or tissue ulceration was observed. The cause of the pulmonary oedema in these experiments was thought to be a combination of 'reimplantation' response and early acute rejection. Although 'conventional' immunosuppression was given to these animals, no other efforts were made to distinguish between the two possible causes of this phenomenon and no added therapy was attempted. Our main concern was the technical feasibility of the operation.
Our conclusions were that the operation can be performed just as easily through a right lateral thoracotomy as through a median sternotomy, and that cardiopulmonary bypass can be instituted as easily from either approach. Cardiopulmonary bypass is not essential in the experimental situation, but may be necessary clinically when the recipient's left lung is diseased. To ascertain this, temporary clamping of the right lung before institution of cardiopulmonary bypass is necessary. The bronchial anastomosis should not be a cause for concern when performed in the manner described. Better immunosuppression for lung transplantation, as afforded by cyclosporin A alone or in combination with other more recent drugs, will be necessary in the clinical situation. Although unilateral lung transplants have been shown to afford only a modest improvement in the recipient's condition in the past, unilateral lung and heart transplantation does have a definite theoretical advantage in a select group of recipients.
References
1. Barnard CN, Wolpowitz A, Losman JG. Heterotopic cardiac transplantation with a xenograft for assistance of the heart in cardiogenic shock after cardiopulmonary bypass. S Afr Med J 1977;52:1035-1038. [ Links ]
2. Cooper DK, Novitsky D, Hassoulas J, Barnard CN. Heart transplantation: The South African experience. Heart Transplant 1982;2:78-84. [ Links ]
3. Wicomb W, Cooper DKC, Hassoulas J, Rose AG, Barnard CN. Orthotopic transplantation of the baboon heart after 20 to 24 hours preservation by continuous hypothermic perfusion with an oxygenated hyperosmolar solution. J Thorac Cardiovasc Surg 1982;83:133-140. [ Links ]
4. Hassoulas J, Barnard CN. Heterotopic cardiac transplantation: A seven year experience at Groote Schuur Hospital. S Afr Med J 1984;65:675-682. [ Links ]
5. Novitzky D, Cooper DKC, Morrell D, Isaacs S. Change from aerobic to anaerobic metabolism after brain death and reversal following triiodothyronine (T3) therapy. Transplantation 1988;45:32-36. [ Links ]
6. Novitzky D, Cooper DKC, Reichart B. Hemodynamic and metabolic responses to hormonal therapy in brain-dead potential organ donors. Transplantation 1987;43:852-854. [ Links ]
7. Macoviak JA, McDougall RI, Bayer MF, Brown M, Tazelaar H, Stinson EB. Significance of thyroid dysfunction in human cardiac allograft procurement. Transplantation 1987;43(6):824-826. [ Links ]
8. Hagl C, Szabo G, Sebening C, et al. Is the brain death related endocrine dysfunction an indication for hormonal substitution therapy in the early period? Eur J Med Res 1997;2(10):437-440. [ Links ]
Corresponding author: J Hassoulas (pariskal@yahoo.gr)














