Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 n.5 Pretoria May. 2012
RESEARCH
Clinical practice guidelines for management of neuropathic pain: expert panel recommendations for South Africa
S ChettyI; E BaalbergenII; A I BhigjeeIII; P KamermanIV; J OumaV; R RaathVI; M RaffVII; S SaldukerVIII
IDepartment of Anaesthesiology, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg. MB ChB, DCH (SA), DA (SA), FCA (SA), Cert Crit Care (SA)
IILife Vincent Pallotti Rehabilitation Unit, Pinelands, Cape Town.BSc, MB ChB
IIIDepartment of Neurology, Nelson R Mandela School of Medicine, Mayville, University of KwaZulu-Natal, Durban. MB ChB, MD, MMed(Neur), FCP (SA), FRCP (UK), FCN (SA)
IVBrain Function Research Group, School of Physiology, University of the Witwatersrand, Johannesburg.PhD
VDepartment of Neurosurgery, University of the Witwatersrand, Johannesburg. MB ChB, MMed (Neurosurgery), FCS (SA) Neurol
VIJacaranda Hospital, Muckleneuk, Pretoria. MB ChB, MMed (Anaes), FIPP (WIP)
VIIChristiaan Barnard Memorial Hospital, Cape Town. BSc, MB ChB, FCA (SA)
VIIISt Augustines Hospital, Durban. MB ChB, FCPsych (SA)
ABSTRACT
Neuropathic pain (NeuP) is challenging to diagnose and manage, despite ongoing improved understanding of the underlying mechanisms. Many patients do not respond satisfactorily to existing treatments. There are no published guidelines for diagnosis or management of NeuP in South Africa. A multidisciplinary expert panel critically reviewed available evidence to provide consensus recommendations for diagnosis and management of NeuP in South Africa. Following accurate diagnosis of NeuP, pregabalin, gabapentin, low-dose tricyclic antidepressants (e.g. amitriptyline) and serotonin norepinephrine reuptake inhibitors (duloxetine and venlafaxine) are all recommended as first-line options for the treatment of peripheral NeuP. If the response is insufficient after 2 - 4 weeks, the recommended next step is to switch to a different class, or combine different classes of agent. Opioids should be reserved for use later in the treatment pathway, if switching drugs and combination therapy fails. For central NeuP, pregabalin or amitriptyline are recommended as first-line agents. Companion treatments (cognitive behavioural therapy and physical therapy) should be administered as part of a multidisciplinary approach. Dorsal root entry zone rhizotomy (DREZ) is not recommended to treat NeuP. Given the large population of HIV/AIDS patients in South Africa, and the paucity of positive efficacy data for its management, research in the form of randomised controlled trials in painful HIV-associated sensory neuropathy (HIV-SN) must be prioritised in this country.
1. Introduction
Neuropathic pain (NeuP) is defined as pain that arises as a 'direct consequence of a lesion or disease affecting the somatosensory system'.1 Importantly, NeuP differs from nociceptive pain in respect of causes, mechanisms, symptomatology and different therapeutic approaches required for successful management.
The burden of NeuP for the patient is substantial. NeuP is associated with psychological distress, physical disability and reduced overall quality of life.2-5 A systematic review and meta-analysis by Doth et at.6 showed lower health-utility scores in patients with NeuP than the general population and in people with other chronic conditions like Parkinson's disease, heart failure, motor neurone disease, cancer, and stroke. Patients with peripheral NeuP are generally affected by difficulty in sleeping, lack of energy, drowsiness, and difficulty in concentrating.7 The problem is further compounded by the fact that globally, and in South Africa, NeuP is often underdiagnosed and inappropriately treated, exacerbating the burden of this already debilitating condition.
The costs of NeuP are considerable,3,8 with misdiagnosis, mistreatment, and mental and physical comorbidities such as depression and nerve damage contributing to the cost, in addition to usual diagnostic and treatment costs. Indeed, it has been reported that patients with NeuP have annual healthcare costs threefold higher than the costs for matched control populations.9
Reduced work ability of patients and carers, and medical expenses also contribute to the overall cost of NeuP.10 A survey in the USA revealed that almost 65% of working patients with painful diabetic neuropathy reported absence from work or decreased work productivity due to pain.11 Another study reported that the employment status was reduced, owing to pain, in 52% of patients with peripheral NeuP.7
In South Africa there are a number of specific challenges to evaluating and treating NeuP. Lack of education and awareness among physicians, including specialists, was noted as a problem in South Africa, leading to suboptimal identification, assessment and management of NeuP. For example, inappropriate use of non-steroidal anti-inflammatory drugs (NSAIDs) and opioids as first-line treatment is widespread, and inappropriate back surgery is common. Referrals to pain clinicians often come too late, and even in specialist centres a multidisciplinary approach is not always taken.
Patient access to care varies widely in South Africa, from rural to urban areas and across socioeconomic divides. But access to care does not guarantee access to the most appropriate drugs, as financial and supplychain constraints, and restricted formulary in the public sector and restricted reimbursement in the private sector limit access to appropriate medications.12 Along with access issues, lack of trained personnel is also a problem.13,14 Added to these challenges, which are not necessarily unique to South Africa, is the high rate of HIV in this country and the paucity of evidence for treating painful HIV-related neuropathy.15
To improve NeuP management in South Africa, regional guidelines for NeuP management, which take local settings into account, are vital. The consensus recommendations described here aim to help healthcare practitioners in South Africa become more aware of NeuP, better skilled at its diagnosis, and equipped to select appropriate treatment options for patients suffering from NeuP.
2. Methods
2.1 Expert panel
A panel with special expertise in diagnosis and management of NeuP met in Johannesburg, South Africa on 9 July 2011. The panel included specialists from the fields of psychiatry, neurology, neurosurgery, anaesthesiology, family medicine and basic science.
The panel collaborated with a French NeuP specialist to critically analyse available randomised controlled trials (RCTs) and evidence-based international and regional guidelines for the evaluation and treatment of NeuP. The objective of the meeting was to develop clear clinical practice guidelines to aid the diagnosis and medical management of NeuP in South Africa.
2.2 Evidence evaluation
Recommendations from recent international and regional guidelines were reviewed in addition to discussion of recent systematic reviews, meta-analyses, and peer-reviewed randomised, double-blind, placebo-controlled studies;15-30 a number of Cochrane reviews were also referred to.31-40 The validity, clinical relevance, and applicability of the evidence for peripheral and central NeuP in South Africa were discussed.
The main sources of evidence were the 2010 guidelines from the European Federation of Neurological Societies (EFNS)26 and recommendations from both the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain (IASP)27,41 and the French Pain Society,16 all based on systematic reviews of available evidence. A systematic review of evidence by Danish pain experts,17 consensus recommendations from the Canadian Pain Society19 and consensus recommendations from experts in Latin America,18 the Middle-East region (MER)21 and the Maghreb region22 were also consulted. Reference was also made to the American Academy of Neurology (AAN) guidelines for management of painful diabetic peripheral neuropathy (DPN);20 postherpetic neuralgia (PHN)24 and trigeminal neuralgia (TN)23 were also referred to.
It was decided against using number-needed-to-treat (NNT) as the sole measure of efficacy in making recommendations for South Africa, since NNT does not provide a complete picture of the quality of a study, particularly as the studies assessed vary widely in number of participants and quality of study design.
After considering the evidence, the panel achieved consensus on a number of recommendations that are supported by best scientific evidence. The recommendations include some agents that may not be indicated for use in NeuP. Similarly, some agents that are supported by best scientific evidence are not available in South Africa (e.g. the topical lidocaine patch), so are mentioned here but have been excluded from the final recommendations.
The levels of evidence stated in this review follow the levels attributed in the formal systematic reviews from which the data were sourced (refer to Appendix A).
2.3 Guideline development
The discussions and consensus statements were recorded at the meeting and written up as a full manuscript draft by a professional medical writer. The panel reviewed, edited, and provided comments on the outline and drafts of the manuscript until a final version was reached that was approved by all members.
3. Results
3.1 Epidemiology and burden of NeuP Estimating the prevalence of NeuP is notoriously difficult - a recent systematic review by Smith and Torrence42 found that estimates vary widely, confounded by underreporting and inconsistent definitions and diagnostic criteria. They suggest a prevalence of 6 - 8% in the general population. They estimate that approximately 20% of patients with diabetes and 8% of people who have had herpes zoster suffer from NeuP.
There are no published estimates of NeuP prevalence in South Africa. The prevalence of NeuP resulting from common aetiologies (see Table 1) is likely to be similar to other countries, but with a large additional component resulting from the high rate of HIV in this country.
Low back pain is a major contributor to NeuP prevalence globally, and there may be a neuropathic component in nearly 50% of black Africans with lower back pain.43 A similar rate of neuropathic pain (55%) was reported in adults with lower back pain in an outpatient setting in the Arabian Gulf region.44 PHN and DPN are also leading causes of NeuP, but data on the prevalence of these causes in South Africa are limited. The International Diabetes Federation (IDF) Diabetes Atlas estimates the prevalence of type II diabetes in the Africa region in 2010 to be 3.8%,45 which is below the global average but expected to rise disproportionately in the developing world in the coming decades.46 In diabetes patients attending outpatient clinics in the Middle East, 54% met the criteria for painful DPN.47 The reported occurrence of peripheral neuropathy in patients with diabetes varies widely in sub-Saharan African countries, from 4% in Zimbabwe to 69% in Nigeria,48 and was estimated at 28% among black African diabetes patients in a 1997 audit of public-sector diabetes care in South Africa.49 While not all diabetes-related neuropathy is painful, as many as 20% of diabetes patients could suffer from NeuP related to DPN,42 and this clearly represents a large, and growing, cause of NeuP, in South Africa.
According to the 2010 global report by the United Nations Program on HIV/AIDS (UNAIDS), 5.6 million people in South Africa are living with HIV.50 HIV-associated sensory neuropathy (HIV-SN), a frequent complication of both HIV and neurotoxic antiretroviral medications such as stavudine, is therefore a major concern in South Africa.
Prevalence of NeuP was reported to be 20.9% among South African AIDS patients who had not received prior antiretroviral treatment.51 The prevalence of symptomatic HIV-SN was 57% in 395 HIV-positive black South Africans exposed to stavudine, with 76% of affected individuals experiencing pain as their primary symptom.52 In 598 HIV-infected individuals in South Africa, the frequency of HIV-SN was 37% in individuals never exposed to antiretroviral drugs, increasing to 60% in individuals receiving antiretroviral therapy. In both groups of patients, the neuropathy was symptomatic in approximately 60% of individuals, with almost all these individuals reporting pain and/or paraesthesias.53
A recent study conducted in a South African hospital revealed that although 71% of the patients with HIV/AIDS had pain documented in their medical charts, only 34% of the patients reported adequate pain management.54 HIV-positive outpatients are no better off, with over 40% of ambulatory patients in pain not receiving any treatment, and of those patients who received treatment, less than 3% received drugs recommended for the treatment of NeuP, despite over a third of the patients having symptoms consistent with HIV-SN.55 These studies highlight that the neuropathic component of HIV-related pain is probably poorly recognised and undertreated in South Africa.
3.2 Pathophysiology of NeuP
NeuP, by definition, arises as a 'direct consequence of a lesion or disease affecting the somatosensory system'.1 While the detailed mechanisms that underlie NeuP are not fully understood, they are thought to operate at both central and peripheral levels (Fig. 1): (A) at the level of peripheral nerves, there is sensitisation, ectopic transmission and spontaneous discharges; (B) changes in central modulatory systems, predominantly in spinal neurones, lead to central sensitisation.
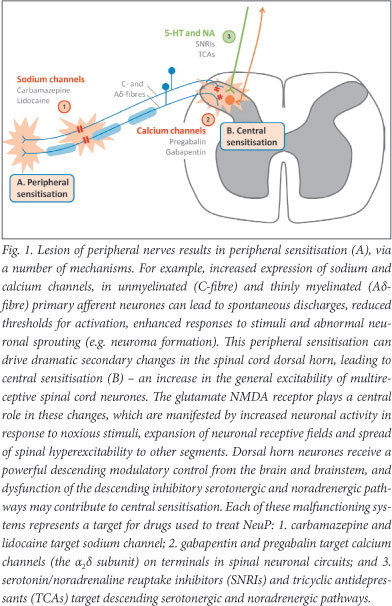
The relationship between these mechanisms and the resulting symptoms is not straightforward - one mechanism may give rise to more than one symptom and one individual symptom may result from multiple mechanisms.56
Knowledge of the possible mechanisms underlying NeuP is helpful in understanding and improving treatment of NeuP. An overview of the basic mechanisms and targets for disease is given in Fig. 1.
3.3 Aetiology of NeuP
Currently there is no universally accepted classification for NeuP types. However, four broad classes of diseases are recognised based on aetiology and anatomy (Table 1).
3.4 Clinical features of NeuP
Patients with NeuP experience symptoms arising in an area of altered sensation (numbness/loss of sensation and/or hyperexcitability) and exhibit a number of typical observable signs.57
The painful symptoms include both spontaneous pain (i.e. occurs with no apparent stimulation), which can be continuous or paroxysmal, and evoked pain. Terms commonly used to describe painful and unpleasant sensations (dysaesthesias) include burning, shooting, and electric shock-like pain. A number of altered, but not unpleasant, sensations (paraesthesias) - tingling, ants crawling, and pins and needles - are also common. Stimulus-evoked pain is described as allodynia if normally non-painful stimuli (e.g. light breeze, skin contact with clothing, temperature change) evoke pain, and as hyperalgesia when a normally painful stimulus (e.g. pinprick) evokes a heightened pain sensation.58
3.5 Diagnosis and evaluation of NeuP
NeuP is distinct from other chronic pain types that have an intact nociceptive system (nociceptive pain). For the differential diagnosis of NeuP it is helpful to analyse the exact quality of somatosensory abnormalities in the affected area as well in the areas adjacent to the sensory deficit.56 Clinical tools, such as questionnaires for screening and assessment, focus on the presence and quality of neuropathic pain, and can be used to alert a clinician to the likelihood of NeuP and the need for a careful examination. It is important to note that screening tools fail to identify about 10 - 20% of patients with clinician-diagnosed NeuP,59 and they should be used as a guide for further diagnostic evaluation and pain management but cannot replace clinical judgment.
3.5.1 Screening tools
In recent years, several standardised screening tools have been developed to aid the identification and classification of NeuP on the basis of patient-reported verbal descriptors of pain qualities.59 These include (among others) painDetect, ID-Pain, Leeds Assessment of Neuropathic Symptoms and Signs (LANSS), Neuropathic Pain Questionnaire (NPQ) and Douleur Neuropathique en 4 questions (DN4). Most of these questionnaires include questions about burning pain, paraesthesias, pain attacks, mechanical and thermal hypersensitivity, and numbness.60,61 They are attractive because of their ease of use by both professionals and patients, in clinic or via telephone or Internet, and because they provide immediate information.61
The painDetect questionnaire was developed and validated in Germany to identify NeuP components in back pain, whereas ID-Pain, DN4 and LANSS were developed to help differentiate nociceptive pain and NeuP.62,63
The DN4 scale is based on the patient's description, and physician examination, of sensory dysfunction - it has a sensitivity of 82.9% and specificity of 89.9%.64 The 10-item questionnaire includes 7 items related to symptoms and 3 related to clinical examination. A total score of 4 or higher suggests NeuP. The 7 sensory descriptors can be used as a self-report questionnaire with similar results. The DN4 has validated translations in 15 languages (in addition to its original French), and while it is not validated in South African languages, the DN4 questionnaire (Fig. 2) is recommended as it is short, quick and easy to follow in regular clinical practice.
3.5.2 Clinical assessment
A simple examination-based way to identify NeuP and differentiate from nociceptive pain is the '3L' approach: Listen, Locate and Look (Table 2).65
Listen to the verbal description of pain and any non-painful symptoms in the same area as the pain.
Locate the region of pain and document with a pain drawing, created either by the patient or by the physician. Any abnormal sensations may also be highlighted on the same illustration.
Look for sensory abnormalities and recognise the distribution pattern. A careful inspection of the painful body area should be carried out and any differences in colour, texture, temperature, etc. should be noted. A simple bedside examination of somatosensory functions is recommended, including touch, cold, warmth and pain sensibility (Table 3).59 The aim is to identify altered sensation in the painful area, and hence responses should be compared with a non-painful adjacent area.
Physicians need to consider a holistic approach to diagnose and treat the underlying condition and comorbid conditions. This will lead to improvement of patients' overall quality of life, physical functioning and sleep quality, along with a reduction of the psychological distress associated with NeuP conditions. Where the underlying pathology is understood, it is recommended that both symptomatic treatment (pain management) and treatment of the aetiology should be initiated. Where the underlying pathology is not clear, symptomatic treatment should be initiated while further testing is done to clarify the pathology.
3.5.3 Recommendations
- Apply screening tools and careful clinical examination and screening tools to help identify and evaluate NeuP.
- Use simple screening tools such as DN4 to help identify likely NeuP.
- Employ the 3L approach to differentiate NeuP from nociceptive pain: listen to the verbal description of pain, locate the region of pain and look for somatosensory deficits with the help of simple bedside tests.
3.6 Pharmacological treatments
Despite a reported 66% increase in published randomised, placebo-controlled trials (RCTs) for NeuP in the past 5 years,17 there are several gaps in the evidence for NeuP treatments. Although many types of peripheral and central NeuP occur in clinical practice, most RCTs have included patients with either PHN or painful DPN. Importantly, there are very few head-to-head trials comparing different treatments, making direct comparisons of efficacy and tolerability difficult or impossible. HIV neuropathy and chronic radiculopathy seem less responsive to drugs generally found useful in other NeuP conditions based on large-scale trials, particularly tricyclic antidepressants (TCAs), pregabalin, and gabapentin.15,66 Central NeuP is also difficult to treat, and while it appears to respond to the same drug treatments as peripheral NeuP, the response is generally less robust.66
3.6.1 Treatment recommendations by international guidelines
In the past few years, several national, regional and international guidelines, systematic reviews and expert panel recommendations have been published for the treatment of NeuP,16-19,21,22,26,27,41 and for specific aetiologies;20,23,24 these are summarised in Table 4a and 4b.
The first-line treatments recommended by most of the guidelines are TCAs, α2δ-ligands or gabapentinoids (pregabalin and gabapentin), and topical lidocaine (for localised NeuP), with selective serotonin/ noradrenaline reuptake inhibitors (SNRIs) sometimes included as first-line, sometimes second-line therapy. All guidelines recommend reserving tramadol and stronger opioid analgesics for second- or third-line treatment (Table 4a).
The EFNS26 and the French16 publications provide recommendations separately for specific NeuP aetiologies, while the others make general recommendations for peripheral (and central) NeuP.
3.6.2 Treatment framework
The initial approach to treatment of NeuP should include a thorough investigation and treatment of underlying pathology. The treatment choice should address the possible pain mechanisms as well as comorbid conditions (anxiety, depression, sleep disorders) associated with pain. Other considerations for treatment selection include potential for adverse effects, drug interactions, contraindications, risks of misuse and abuse, patients' response to prior therapy, and cost. Patient education is a vital aspect of NeuP management. It is important to clearly explain the mechanisms of NeuP as well as the goals of treatment to the patient in order to maximise treatment benefits and manage treatment expectations. The patient should be informed that the onset of analgesic effect will take time and reduction of pain is not achieved quickly, in most cases. Non-pharmacological methods of coping with pain should be discussed, including the importance of stress reduction and good sleep hygiene, and access to physical therapy and psychotherapy should be recommended or arranged.
3.6.3 Peripheral NeuP
Four classes of drugs have good evidence of efficacy in the treatment of non-localised NeuP: α2δ-ligands (pregabalin and gabapentin), TCAs (low-dose amitriptyline or other TCA), SNRIs (duloxetine and venlafaxine), and opioids (tramadol, methadone and morphine).
The efficacy and safety of these agents are briefly discussed below and also summarised in Table 5.
3.6.3.1 α2δ-ligands (pregabalin and gabapentin)
Pregabalin and gabapentin are recommended (grade A) as first-line therapy by IASP, EFNS, and French guidelines, based on high-quality evidence of efficacy established in multiple RCTs.16,26,27 The AAN guidelines for painful DPN recommend pregabalin (level A) because of the availability of strong evidence and gabapentin (level B evidence).20 A systematic review by Danish pain experts17 and several Cochrane reviews32,35,67 confirm the efficacy of these α2δ-ligands for the treatment of NeuP.
Although pregabalin and gabapentin appear to have similar efficacy, there are minor differences in the pharmacokinetic profile of these two drugs.27 Gabapentin pharmacokinetics are nonlinear (due to saturable absorption), and dosing requires careful titration. Treatment should be initiated at low dosages with gradual increases until pain relief, dose-limiting adverse effects, or a dose of 3 600 mg/day in 3 divided doses is/are reached. Pregabalin has linear pharmacokinetics and dosing is more straightforward. Dosing can start at 25 mg/day (at night), and be titrated slowly up to a maximum dose of 300 - 450 mg/day (in 2 divided doses). Because of its shorter titration period and potentially efficacious starting dosage, pregabalin may provide analgesia more quickly than gabapentin.27,68 Thus, pregabalin has pharmacokinetic advantages compared to gabapentin.
The IASP NeuPSIG guidelines16 acknowledge the additional efficacy of gabapentin and pregabalin in sleep disorders, and pregabalin in anxiety disorders associated with pain. Although gabapentin and pregabalin have few drug interactions, both can produce dose-dependent dizziness and sedation, which can be reduced by starting with lower dosages and titrating cautiously. It is also important to note that both these medications require dosage reduction in patients with renal insufficiency.69,70
3.6.3.2 SNRIs (duloxetine and venlafaxine)
SNRIs are considered a first-line treatment option by most of the international guidelines, including the NeuPSIG guidelines27 (grade A) and the EFNS guidelines26 (level A for DPN), thus highlighting the efficacy of SNRIs for management of NeuP. Although the French guidelines16 recommend SNRIs for second-line therapy because of the lack of marketing authorisation, duloxetine and venlafaxine have grade A recommendations for DPN and sensory polyneuropathy respectively. Danish pain experts17 state in their review that duloxetine and venlafaxine have a well-documented efficacy in painful polyneuropathy.
Although both duloxetine and venlafaxine have been studied in peripheral NeuP, especially in painful DPN, more evidence of efficacy is available for duloxetine.28,30,34,71 Venlafaxine has shown efficacy in painful polyneuropathies of different origins.31,72 Both duloxetine and venlafaxine are approved for the treatment of major depression disorder (MDD) and generalised anxiety disorder (GAD)73,74 and hence are the treatment of choice in NeuP patients with these co-morbid conditions. Nausea, the most frequent side-effect with duloxetine, occurs less frequently if treatment is initiated at 30 mg/day and titrated after one week to 60 mg/day.75 According to the IASP NeuPSIG guidelines,41 duloxetine 60 mg once daily appears to be as efficacious as 60 mg twice daily and is associated with fewer side-effects in painful DPN.
SNRIs in general and duloxetine in particular pose a minor to moderate hepatic risk; the use of duloxetine is contraindicated in patients with severe hepatic impairment.73 Elevated blood pressure and clinically significant electrocardiogram (ECG) changes are associated with patients treated with venlafaxine.74 Therefore, venlafaxine should be prescribed with caution in patients with cardiac disease and with regular BP monitoring. Venlafaxine should be tapered when treatment is being discontinued as a withdrawal syndrome has been described.76 Antidepressants are generally associated with increased risk of suicide; hence patients should be closely monitored (refer to Table 5 for additional considerations). An additional consideration, when using relatively high doses (120 mg duloxetine, 225 mg venlafaxine), is the risk of precipitating manic episodes in vulnerable individuals.
3.6.3.3 Low-dose TCAs (amitriptyline, imipramine, nortriptyline)
Published international guidelines including the EFNS26 (level A evidence for DPN and PHN), IASP NeupSIG27 (grade A), French guidelines (grade A scientific evidence in several aetiologies) as well as the systematic review by Danish experts17 have documented the efficacy of TCAs for treating a variety of types of NeuP. A Cochrane review34 that considered data from 17 studies validated the efficacy of TCAs in NeuP.
TCAs are an attractive option mainly because they are inexpensive and have a convenient once-daily dosing. Although TCAs are approved to treat MDD, the analgesic effect is independent of the antidepressant effect, and occurs at a lower dose.27 Therefore, low-dose TCAs are not the NeuP treatment of choice in patients with comorbid depression. Starting doses of amitriptyline should be low (10 - 25 mg/day), and titrated slowly until pain is adequately controlled or side-effects limit continued titration.
It is important to take into account the potential for drug interactions, especially when amitriptyline is co-administered with drugs that inhibit CYP2D6 enzyme. TCAs are associated with cardiac toxicity and hence amitriptyline is contraindicated in patients who have ischaemic heart disease or an increased risk of sudden cardiac death.77,78 The MER guidelines21 recommend a screening ECG before beginning treatment with TCAs in patients over 40 years of age. Amitriptyline should be avoided in elderly patients. Please refer to Table 5 for additional safety considerations.
3.6.3.4 Opioids (tramadol, morphine and methadone)
The IASP NeuPSIG guidelines27 reviewed several high-quality RCTs that showed the efficacy of opioid analgesics including tramadol in patients with different types of NeuP and recommend them as second-line agents (grade A), except in certain specific clinical situations in which first-line use could be considered. The EFNS guidelines26 recommend opioids as second- or third-line agents with level A evidence for DPN and PHN. A systematic review by Danish pain experts17 also acknowledged the consistent efficacy of opioids in NeuP.
Tramadol is a weak μ-opioid agonist that inhibits the reuptake of noradrenalin and serotonin. It has been shown to reduce pain in DPN and sensory polyneuropathies; although it may be less efficacious than strong u-agonists.79 The risk of abuse with tramadol appears considerably less compared with opioid analgesics.75 The EFNS guidelines26 cautions the use of tramadol in elderly patients because of risk of confusion and does not recommended tramadol with drugs acting on serotonin reuptake such as selective serotonin reuptake inhibitors (SSRIs). The French guidelines16 recommend tramadol for treatment of mixed pain (pain with nociceptive and neuropathic components) as it is effective in nociceptive pain.
Cochrane reviews have demonstrated the effectiveness of strong opioids (oxycodone, morphine, and methadone) in different types of NeuP, providing greater pain relief than placebo.38,80 In head-to-head comparisons, opioids provided at least as much analgesia as TCAs and gabapentin.81,82 Despite strong evidence of efficacy, most of the international guidelines reserve opioid analgesics as second- or third-line agents mainly because of risk of long-term side-effects and possible opioid misuse and addiction. The IASP NeuPSIG guidelines estimate that the frequency of these problems associated with opioid analgesics ranges widely from less than 5% to as much as 50%. Hence, prior to initiating opioids, clinicians should take into account the risk factors for abuse, which include active or previous substance abuse and family history of substance abuse.75
3.6.4 Recommendations for peripheral NeuP
The panel reviewed the evidence and constructed a treatment algorithm (Fig. 3) to aid step-wise management of non-localised NeuP.
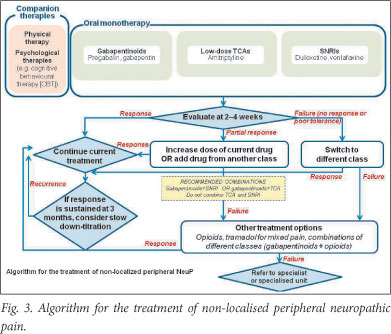
3.6.4.1 First-line treatment
Three classes of drugs are recommended for first-line monotherapy: μ2δ-ligands (pregabalin or gabapentin), TCAs (low-dose amitriptyline or other TCA) and SNRIs (duloxetine or venlafaxine). Pregabalin is the preferred first-line option because of its simple pharmacokinetics and good tolerability. The choice of drug also depends on additional factors summarised in Table 5.
Patients should be evaluated at 2 - 4 weeks after initiating therapy to determine response to treatment. If the response is good, the current treatment should be maintained, and if the response is sustained for 3 months, slow down-titration can be attempted. If symptoms return, treatment should be titrated back to an effective dose. If a partial response is seen at 2 - 4 weeks, consider increasing the dose of the current agent. If the response is poor, or the drug is not tolerated, move to second-line approaches.
3.6.4.2 Second-line therapy - combination
In case of partial response to first-line therapy, recommendations include either increasing the dose of the current drug or adding a drug from a different class. In case of complete failure to first-line therapy, the patient should be switched to a drug from a different class.
For combination treatment, pregabalin with either an SNRI or amitriptyline is recommended. It is important to note that although TCA and SNRI are different classes of antidepressant they target the same mechanism, so a combination of SNRI and TCA is not recommended.
Combination therapy may offer additional analgesic benefits and benefits on associated symptoms,83 but potential advantages must be weighed against the possibility of additive adverse effects, drug interactions, increased cost, and reduced adherence to a more complex treatment regimen.41
3.6.4.3 Third-line treatment
If the patient does not respond to combination therapy or the switch strategy, tramadol is recommended (especially in NeuP with a nociceptive component) followed by strong opioids (e.g. morphine, oxycodone, hydromorphone), or a combination of first-line options with opioids.
Evidence for these combinations is limited, but the combination of morphine and gabapentin seems to provide better pain relief than each drug given alone.82 In another study, a combination of gabapentin and an opioid was associated with significant pain relief and improved sleep, without an exacerbation of opioid-induced adverse events.84
3.6.4.4 Follow-up
The tools and scales used for diagnosis may be useful for clinical monitoring (though not all are validated for this use) to establish a baseline and assess the patient's response. Monitoring for potential drug interactions, adverse events, co-morbidities, need for dose titration, etc., should be part of the follow-up plan.
If a patient does not show a satisfactory therapeutic response, he/ she should be referred to a pain specialist centre.
3.6.5 Aetiology-based recommendations 3.6.5.1 Polyneuropathy
Painful DPN: The EFNS guidelines26 recommend the use of TCAs, gabapentin, pregabalin and SNRI (duloxetine, venlafaxine) as first-line treatment in painful polyneuropathy (notably related to diabetes), tramadol as second-line therapy and strong opioids as third-line agents.
Recommendations: The panel recommends use of pregabalin or gabapentin, low-dose amitriptyline (or other TCA), duloxetine or venlafaxine (SNRIs) for treatment of painful polyneuropathies, including painful DPN. If response to treatment is poor, patients should be switched to, or have added, a drug from a different class. Tramadol and opioids are recommended after failure of second-line or combination therapy.
Painful HIV-SN: A recent systematic review of pharmacological treatment of HIV-associated neuropathy15 identified only 3 agents with good evidence of efficacy (v. placebo): smoked cannabis (1 - 8% δ-9-tetrahydrocannabinol), high-dose topical capsaicin (8%), and recombinant human nerve growth factor (rhNGF). Lamotrigine had limited efficacy in one trial, demonstrating superiority over placebo in a secondary endpoint and only in patients exposed to neurotoxic ARVs.15 Drugs that are generally effective for peripheral neuropathic pain of other aetiologies (amitriptyline, pregabalin, and gabapentin) have been studied but with no evidence of efficacy, and there have been no RCTs of SNRIs in HIV-associated neuropathy.
Recommendations: Because of the lack of evidence for treatment of HIV-SN, the panel recommends following the framework outlined for other polyneuropathies and the step-wise management as illustrated in Fig 3. In addition, if the onset of the neuropathy is associated with starting antiretroviral therapy (even if it is a tenofovir-based regimen), then an alternative regimen should be considered, where possible.
3.6.5.2 Postherpetic neuralgia
Systematic reviews including a review by the AAN concur that gabapentin, pregabalin, TCAs, lidocaine patches and strong opioids have strong evidence of efficacy in PHN.24,29,34 Opioids have similar or slightly better efficacy compared with TCA but are associated with more frequent discontinuation because of side-effects.26,29 Because of the lack of RCTs, the efficacy of SNRIs duloxetine and venlafaxine for the treatment of PHN is not known.
The EFNS guidelines26 state that although topical lidocaine patches are effective for the treatment of PHN with brush-induced allodynia, the level of evidence is lower compared with systemic agents.85 Topical capsaicin has also reported modest benefits in patients with PHN.29
Recommendations: The panel recommends pregabalin, gabapentin or amitriptyline for first-line treatment of PHN, and to combine drugs from different classes as a second-line approach. Opioids (tramadol, then stronger opioids) should be reserved for third-line treatment.
As a topical lidocaine patch is not available in South Africa, the panel could not recommend its use despite strong supporting evidence. Topical capsaicin is also not available in South Africa, so it cannot be recommended. The panel suggests that the regulatory authorities in South Africa consider approval of these agents for use in neuropathic pain.
3.6.5.3 Trigeminal neuralgia (TN)
The AAN-EFNS guidelines for TN23 recommend carbamazepine (200 -1 200 mg/day) as the drug of choice in classic TN because of its robust treatment response; however, its efficacy may be compromised by poor tolerability and pharmacokinetic interactions.23,37 Oxcarbazepine has shown similar efficacy to carbamazepine for controlling pain in TN,23,26 but with fewer drug-drug interactions. The AAN-EFNS guidelines also comment on the lack of evidence for treatment of TN following failure of first-line therapy and acknowledge some evidence supporting add-on therapy with lamotrigine or a switch to baclofen, but recent Cochrane reviews conclude that there is insufficient evidence to recommend them in TN.36,39
Recommendations: The panel recommends the use of carbamazepine and oxcarbazepine for the treatment of TN.
3.6.6 Central NeuP (CP)
Relatively few RCTs have been conducted in patients with CP, but results and clinical experience suggest that such conditions may be relatively more refractory to treatment than peripheral NeuP.27
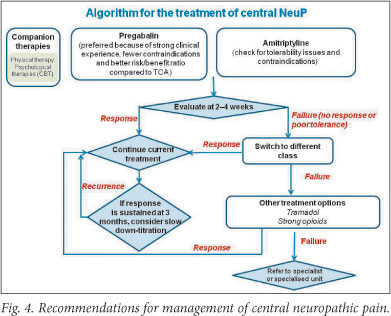
The EFNS guidelines,26 IASP NeuPSIG group recommendations,27 and a systematic review by Danish pain experts17 assessed the available data and agreed that the use of pregabalin, gabapentin, and TCAs (specifically amitriptyline) is best supported for CP states, specifically spinal cord injury (SCI) and poststroke pain. The EFNS guidelines26 recommend these three agents as first-line options for CP, with tramadol or stronger opioids as second-line. Cannabinoids are suggested in multiple sclerosis (MS) if other treatments fail,26,27 although poor availability and concerns about risk of abuse and precipitation of psychosis limit use. There is some mixed evidence for lamotrigine in SCI and post-stroke pain.26, 27
A systematic review of evidence by Danish pain experts did not include any RCTs with SNRIs in CP.17 A recent RCT which evaluated the effects of duloxetine on pain relief concluded that there is insufficient evidence for the efficacy of duloxetine in treatment of CP.86
Recommendations: Based on the scientific evidence and added benefit in treating comorbidities (depression, insomnia, anxiety), the panel recommends using pregabalin or amitriptyline for first-line treatment of CP (Fig. 4). As a result of the consistent clinical experience, fewer contraindications and better risk/benefit ratio compared with TCAs, the panel agrees that pregabalin should be the preferred option. Treatment trials should be approached as for peripheral NeuP; switching to other first-line agent or combining drugs if treatment fails. Tramadol should be considered next, followed by stronger opioids. As cannabinoids are not available in South Africa they cannot be recommended.
3.7 Non-pharmacological treatments 3.7.1 Companion treatments
A recent review of the evidence supporting the potential complementary role of psychosocial treatments of patients with chronic pain suggest that a combination of psychological, pharmacological and physical therapies, tailored to the needs of the individual patient, may be the best approach.87 Transcutaneous electrical nerve stimulation (TENS) is widely used for NeuP and nociceptive pain, and while it lacks robust efficacy data,88 it is recommended by EFNS Task Force89 as a preliminary or as an adjunct to analgesic therapy as it is inexpensive, non-invasive, safe, and can be self-administered. A review of non-pharmacological treatment approaches by Guastella et al.,90 indicate TENS in focal neuropathic pain when upstream stimulation is possible for a superficial sensitive nerve trunk. There are no good data supporting the use of acupuncture in NeuP.
Recommendation: The panel recommends the use of psychotherapy, particularly cognitive behavioural therapy, and TENS alongside appropriate physiotherapy and pharmacological treatment, for the management of NeuP. Comprehensive patient education can also help improve treatment outcomes.
3.7.2 Stimulatory treatments and surgical management (noninvasive and invasive)
Non-invasive electrical stimulation of the brain, using a variety of methods, has been studied in some chronic pain conditions with very limited evidence of efficacy.91
Spinal cord stimulation, via electrodes implanted into the spinal cord, has limited evidence of efficacy in failed back surgery syndrome and complex regional pain syndrome type I,90 the EFNS Task Force identified level B evidence of efficacy in several systematic reviews, as well as primary studies for spinal cord stimulation in these two conditions.89 Guastella et al.90 suggest the use of spinal cord stimulation in segmental mononeuropathies refractory to drug treatment.
Dorsal root entry zone lesioning (DREZotomy) involves destruct-tion of nociceptive fibres and the dorsal root entry zones in an aim to destroy the neurones that sustain the painful state. Guastella et al.90 suggest its use in refractory pain due to plexus avulsion.
Recommendations: The panel did not discuss these non-pharmacological treatment approaches extensively, but recommends spinal cord stimulation in cases of pain that cannot be managed by pharmacological and companion treatments. The panel does not recommend DREZotomy for management of any NeuP, because of limited evidence and risk of worsening of NeuP after this invasive procedure.
4. Discussion
The management of NeuP is challenging, and even when NeuP is diagnosed and treated according to the best evidence available, not all patients can achieve a satisfactory response. This article provides recommendations for the management of NeuP in South Africa, with the aim of raising awareness of NeuP and improving its diagnosis and treatment in this country. These recommendations apply published, international, evidence-based guidelines for NeuP management to the South African setting.
NeuP is widely underdiagnosed in South Africa, and the panel recommends the use of simple questionnaires, such as DN4, to identify NeuP. A raised awareness of common signs and symptoms of NeuP, and of the descriptors used by patients, will also help clinicians to better identify those patients who have neuropathic aspects to their pain.
For management of peripheral NeuP, the α2δ-ligands pregabalin and gabapentin, low-dose TCAs, and the SNRIs duloxetine and venlafaxine are recommended as first-line options. Pregabalin is the preferred option, based on tolerability and pharmacokinetics. Opioids should be reserved for later use, and only after switching to another monotherapy or combination therapy with multiple first-line agents fails.
For painful DPN, recommendations are as for peripheral NeuP in general; for PHN, first-line recommendations are pregabalin (preferred), gabapentin and low-dose amitriptyline; and for TN, oxcarbazepine (preferred) and carbamazepine. Some agents with good evidence, recommended in guidelines from other regions, are not available in South Africa. The panel requests that the South African regulatory authorities evaluate the evidence for the lidocaine patch and topical capsaicin in localised peripheral NeuP and consider approval of these agents in South Africa.
Based on current international recommendations, the committee cannot recommend specific therapy for the management of HIV-associated neuropathy. Currently these patients should be managed following the same recommendations used for the management of peripheral neuropathic pain.
Evidence in CP is less consistent than for peripheral NeuP, but first-line recommendations are pregabalin (preferred) and amitriptyline.
Companion therapies, such as cognitive-behavioural therapy (and other psychotherapy) and physical therapy are recommended to accompany pharmacological management. Invasive options like DREzotomy are not currently recommended.
The recommendations presented here have several limitations. Evidence is still lacking for the relative efficacy of agents for NeuP, as there are very few head-to-head trials. There are also limited data available for pain due to specific aetiologies other than painful DPN, PHN, and TN. In particular, the paucity of evidence for treatment of painful HIV-SN makes it impossible to provide an evidence-based recommendation for this problem that is so common in South Africa. This must be a priority area of future research. In addition, because there are few placebo-controlled RCTs in South African populations, the recommendations given here have to assume that results in other populations can be extrapolated to the various ethnic groups represented in South Africa.
Disclosures. The authors who contributed to this article have disclosed the following industry relationships: Dr Ahmed Bhigjee reports receiving honoraria from Janssen and Pfizer. Dr Sean Chetty reports receiving honoraria from Abbott, Adcock Ingram, Aspen, AstraZeneca, Dráger, Fresenius Kabi, GSK, Janssen, MSD, Nycomed, Pfizer, St. Jude Medical and Teleflex. Dr Eduard Baalbergen reports receiving honoraria for lectures. Prof Peter Kamerman reports receiving honoraria from MSD, Novartis and Pfizer. Dr John Ouma reports an honorarium from Pfizer. Dr Russell Raath reports receiving honoraria from Aspen, Adcock Ingram and Pfizer, consultancy fees from Earth Medical, clinical trial investigator meeting fees from Pfizer and St. Jude Medical, payment for board membership and development of educational presentations from Adcock Ingram. Dr Milton Raaf reports receiving honoraria from Boehringer Ingelheim, MSD, Johnson & Johnson, and Pfizer. Dr Shaquir Salduker reports receiving honoraria from AstraZeneca, Eli Lilly, Lundbeck, Novartis and Pfizer. All authors received honoraria and support to travel to the meeting at which these guidelines were discussed (funded by Pfizer).
Acknowledgements
The authors thank Prof Gérard Mick (Hôpital Neurologique, CHU de Lyon, France) for his invaluable contribution and expertise in the preparation of these consensus recommendations and the methodology, and for critical review and comments on the article content. Medical writing support in the development of this manuscript was provided by Choice Healthcare Solutions and funded by Pfizer Inc.
Society affiliations. The expert panel members represented a range of national societies, as follows: Sean Chetty, Pain South Africa (painsa); Eduard Baalbergen, South African Spinal Cord Association (SASCA); Ahmed Bhigjee, Neurological Association of South Africa (NASA); Peter Kamerman, painsa; John Ouma, South African Society of Neurosurgeons (SASN); Russell Raath, Pain Interventions and Regional Anaesthesia (PIRA); Milton Raff, South African Society of Anaesthesiologists (SASA); Shaquir Salduker, South African Society of Psychiatrists (SASOP).
Endorsements. These guidelines have been endorsed by painsa, NASA, PIRA, SASA, and SASCA.
References
1. Jensen TS, Baron R, Haanpaa M, et al. A new definition of neuropathic pain. Pain 2011;152:2204-2205. [ Links ]
2. Jensen MP, Chodroff MJ, Dworkin RH. The impact of neuropathic pain on health-related quality of life: review and implications. Neurology 2007;68:1178-1182. [ Links ]
3. Khenioui H, Cahagne V, Brissot R. Assessment of chronic pain as a disability in patients with spinal cord injuries. Ann Readapt Med Phys 2006;49:125-137. [ Links ]
4. Schmader KE. Epidemiology and impact on quality of life of postherpetic neuralgia and painful diabetic neuropathy. Clin J Pain 2002;18:350-354. [ Links ]
5. Gilron I, Watson CP, Cahill CM, Moulin DE. Neuropathic pain: a practical guide for the clinician. Can Med Assoc J 2006;175:265-275. [ Links ]
6. Doth AH, Hansson PT, Jensen MP, Taylor RS. The burden of neuropathic pain: a systematic review and meta-analysis of health utilities. Pain 2010;149:338-344. [ Links ]
7. Meyer-Rosberg K, Kvarnstrom A, Kinnman E, Gordh T, Nordfors LO, Kristofferson A. Peripheral neuropathic pain--a multidimensional burden for patients. Eur J Pain 2001;5:379-389. [ Links ]
8. Cruz-Almeida. Chronicity of pain associated with spinal cord injury: A longitudinal analysis. J Rehabil Res Dev 2005;42:585-594. [ Links ]
9. Berger A, Dukes EM, Oster G. Clinical characteristics and economic costs of patients with painful neuropathic disorders. J Pain 2004;5:143-149. [ Links ]
10. O'Connor AB. Neuropathic pain: quality-of-life impact, costs and cost effectiveness of therapy Pharmacoeconomics 2009;27:95-112. [ Links ]
11. Gore M, Brandenburg NA, Hoffman DL, Tai KS, Stacey B. Burden of illness in painful diabetic peripheral neuropathy: the patients' perspectives. J Pain 2006; 7: 892-900. [ Links ]
12. HumanRightsWatch. Global State of Pain Treatment Access to Medicines and Palliative Care, 2011. http://www.hrw.org/reports/2011/06/02/global-state-pain-treatment-0 (accessed 4 October 2011) [ Links ]
13. Harding R, Powell RA, Kiyange F, Downing J, Mwangi-Powell F. Provision of pain- and symptom-relieving drugs for HIV/AIDS in sub-Saharan Africa. J Pain Symptom Manage 2010;40:405-415. [ Links ]
14. Maree JE, Wright SC, Makua MR. The management of HIV- and AIDS-related pain in a primary health clinic in Tshwane, South Africa. Pain Management Nursing 2012 (in press). [ Links ]
15. Phillips TJ, Cherry CL, Cox S, Marshall SJ, Rice AS. Pharmacological treatment of painful HIV-associated sensory neuropathy: a systematic review and meta-analysis of randomised controlled trials. PLoS One 2010;5:e14433. [ Links ]
16. Martinez V, Attal N, Bouhassira D, Lantéri-Minet M. Chronic neuropathic pain: diagnosis, evaluation and treatment in outpatient services. Guidelines for clinical practice of the French Society for the Study and Treatment of Pain. Douleur et Analgesie 2010;23:51-66. [ Links ]
17. Finnerup NB, Sindrup SH, Jensen TS. The evidence for pharmacological treatment of neuropathic pain. Pain 2010;150:573-581. [ Links ]
18. Acevedo JC, Amaya A, Casasola Ode L, et al. Guidelines for the diagnosis and management of neuropathic pain: consensus of a group of Latin American experts. J Pain Palliat Care Pharmacother 2009;23:261-281. [ Links ]
19. Moulin DE, Clark AJ, Gilron I, et al. Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society. Pain Res Manage 2007;12:13-21. [ Links ]
20. Bril V, England JD, Franklin GM, et al. Evidence-based guideline: treatment of painful diabetic neuropathy -- report of the American Association of Neuromuscular and Electrodiagnostic Medicine, the American Academy of Neurology, and the American Academy of Physical Medicine & Rehabilitation. Muscle Nerve 2011;43:910-917. [ Links ]
21. Bohlega S, Alsaadi T, Amir A, et al Guidelines for the pharmacological treatment ofperipheral neuropathic pain: expert panel recommendations for the middle East region. J Int Med Res 2010;38:295-317. [ Links ]
22. Griene B, Bouajina E, Haddad M, et al Traitement médicamenteux des douleurs neuropathiques périphériques: recommandations d'un groupe d'experts pour le Maghreb francophone. Douleur et Analgesie 2011;24:112-119. [ Links ]
23. Gronseth G, Cruccu G, Alksne J, et al. Practice parameter: the diagnostic evaluation and treatment of trigeminal neuralgia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology and the European Federation of Neurological Societies. Neurology 2008;71:1183-1190. [ Links ]
24. Dubinsky RM, Kabbani H, El-Chami Z, Boutwell C, Ali H. Practice parameter: treatment of postherpetic neuralgia: an evidence-based report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2004;63:959-965. [ Links ]
25. Freeman R, Durso-Decruz E, Emir B. Efficacy, safety, and tolerability of pregabalin treatment for painful diabetic peripheral neuropathy: findings from seven randomized, controlled trials across a range of doses. Diabetes Care 2008;31:1448-1454. [ Links ]
26. Attal N, Cruccu G, Baron R, et al EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision. Eur J Neurol 2010;17:1113-e88. [ Links ]
27. Dworkin RH, O'Connor AB, Audette J, et al. Recommendations for the pharmacological management of neuropathic pain: an overview and literature update. Mayo Clin Proc 2010;85:S3-14. [ Links ]
28. Rowbotham MC, Goli V, Kunz NR, Lei D. Venlafaxine extended release in the treatment of painful diabetic neuropathy: a double-blind, placebo-controlled study. Pain 2004;110:697-706. [ Links ]
29. Hempenstall K, Nurmikko TJ, Johnson RW, A'Hern RP, Rice AS. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med 2005;2:e164. [ Links ]
30. Sultan A, Gaskell H, Derry S, Moore RA. Duloxetine for painful diabetic neuropathy and fibromyalgia pain: systematic review of randomised trials. BMC Neurol 2008;8:29. [ Links ]
31. Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy or chronic pain. Cochrane Database Syst Rev 2009:CD007115. [ Links ]
32. Moore RA, Straube S, Wiffen PJ, Derry S, McQuay HJ. Pregabalin for acute and chronic pain in adults. Cochrane Database Syst Rev 2009:CD007076. [ Links ]
33. Moore RA, Wiffen PJ, Derry S, McQuay HJ. Gabapentin for chronic neuropathic pain and fibromyalgia in adults. Cochrane Database Syst Rev 2011:CD007938. [ Links ]
34. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain: a Cochrane review. J Neurol Neurosurg Psychiatry 2010;81:1372-1373. [ Links ]
35. Wiffen P, Collins S, McQuay H, Carroll D, Jadad A, Moore A. Anticonvulsant drugs for acute and chronic pain. Cochrane Database Syst Rev 2005:CD001133. [ Links ]
36. Wiffen PJ, Derry S, Moore RA. Lamotrigine for acute and chronic pain. Cochrane Database Syst Rev 2011:CD006044. [ Links ]
37. Wiffen PJ, Derry S, Moore RA, McQuay HJ. Carbamazepine for acute and chronic pain in adults. Cochrane Database Syst Rev 2011:CD005451. [ Links ]
38. Eisenberg E, McNicol E, Carr DB. Opioids for neuropathic pain. Cochrane Database Syst Rev 2006;3:CD006146. [ Links ]
39. Yang M, Zhou M, He L, Chen N, Zakrzewska JM. Non-antiepileptic drugs for trigeminal neuralgia. Cochrane Database Syst Rev 2011:CD004029. [ Links ]
40. Duhmke RM, Cornblath DD, Hollingshead JR. Tramadol for neuropathic pain. Cochrane Database Syst Rev 2004:CD003726. [ Links ]
41. Dworkin RH, O'Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 2007;132:237-251. [ Links ]
42. Torrance BHSN. Epidemiology of neuropathic pain. Pain Management 2011;1:87-96. [ Links ]
43. Ouedraogo DD, Nonguierma V, Napon C, et al. Prevalence of neuropathic pain among black African patients suffering from common low back pain. Rheumatol Int 2011; Apr 28. (Epub ahead of print). [ Links ]
44. El Sissi W, Arnaout A, Chaarani MW, et al Prevalence of neuropathic pain among patients with chronic low-back pain in the Arabian Gulf Region assessed using the leeds assessment of neuropathic symptoms and signs pain scale. J Int Med Res 2010;38:2135-2145. [ Links ]
45. Atlas ID. Country Summary Table, 2010. http://atlas.idf-bxl.org/ru/node/250. (accessed 20 September 2011). [ Links ]
46. Rheeder P. Type 2 diabetes: the emerging epidemic. South African Family Practice 2006;48:20. [ Links ]
47. Jambart S, Ammache Z, Haddad F, et al. Prevalence of painful diabetic peripheral neuropathy among patients with diabetes mellitus in the Middle East region. J Int Med Res 2011;39:366-377. [ Links ]
48. Abbas Z, Archibald L. The diabetic foot in sub-Saharan Africa: A new management paradigm. Diabetic Foot Journal 2007;10:128-136. [ Links ]
49. Levitt NS, Bradshaw D, Zwarenstein MF, Bawa AA, Maphumolo S. Audit of public sector primary diabetes care in Cape Town, South Africa: high prevalence of complications, uncontrolled hyperglycaemia, and hypertension. Diabet Med 1997;14:1073-1077. [ Links ]
50. UNAIDS. UNAIDS report on the global AIDS epidemic 2010, http://www.unaids.org/globalreport/Global_report.htm (accessed 12 September 2011). [ Links ]
51. Hitchcock SA, Meyer HP, Gwyther E. Neuropathic pain in AIDS patients prior to antiretroviral therapy. S Afr Med J 2008;98:889-892. [ Links ]
52. Wadley AL, Cherry CL, Price P, Kamerman PR. HIV neuropathy risk factors and symptom characterization in stavudine-exposed South Africans. J Pain Symptom Manage 2011;41:700-706. [ Links ]
53. Maritz J, Benatar M, Dave JA, et al HIV neuropathy in South Africans: frequency, characteristics, and risk factors. Muscle Nerve 2010;41:599-606. [ Links ]
54. Narasimooloo C, Naidoo SS, Gaede BM. Adequacy of pain management in HIV-positive patients South African Family Practice 2011;53:71-76. [ Links ]
55. Mphahlele NR, Mitchell D, Kamerman PR. Pain in ambulatory HIV-positive South Africans. Eur J Pain 2012;16:447-458. [ Links ]
56. Baron R, Binder A, Wasner G. Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol 2010;9:807-819. [ Links ]
57. Freynhagen R, Bennett MI. Diagnosis and management of neuropathic pain. BMJ 2009;339:b3002. [ Links ]
58. IASP. 2011 update of 1994 Part III: Pain Terms, A Current List with Definitions and Notes on Usage. In: Merskey H, Bogduk N, eds. Classification of Chronic Pain: Descriptions of Chronic Pain Syndromes and Definitions of Pain Terms. 2nd ed. Seattle: IASP Press; 2011:209-214. [ Links ]
59. Haanpaa M, Attal N, Backonja M, et al. NeuPSIG guidelines on neuropathic pain assessment. Pain 2011;152:14-27. [ Links ]
60. Cruccu G, Sommer C, Anand P, et al. EFNS guidelines on neuropathic pain assessment: revised 2009. Eur J Neurol 2010;17:1010-1018. [ Links ]
61. Bennett MI, Attal N, Backonja MM, et al. Using screening tools to identify neuropathic pain. Pain 2007;127:199-203. [ Links ]
62. Freynhagen R, Baron R, Gockel U, Tolle TR. painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin 2006;22:1911-1920. [ Links ]
63. Portenoy R. Development and testing of a neuropathic pain screening questionnaire: ID Pain. Curr Med Res Opin 2006;22:1555-1565. [ Links ]
64. Bouhassira D, Attal N, Alchaar H, et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005;114:29-36. [ Links ]
65. Hansson P, Haanpaa M. Diagnostic work-up of neuropathic pain: computing, using questionnaires or examining the patient? Eur J Pain 2007;11:367-369. [ Links ]
66. Attal N. Recent developments in the pharmacological management of neuropathic pain. European Neurological Journal 2010;2:25-30. [ Links ]
67. Wiffen PJ, McQuay HJ, Edwards J, Moore RA. Gabapentin for acute and chronic pain. Cochrane Database of Systematic Reviews 2011:CD005452. [ Links ]
68. Stacey BR, Barrett JA, Whalen E, Phillips KF, Rowbotham MC. Pregabalin for postherpetic neuralgia: placebo-controlled trial of fixed and flexible dosing regimens on allodynia and time to onset of pain relief. J Pain 2008;9:1006-1017. [ Links ]
69. Pharmaplan (Pty) Ltd. Epleptin (gabapentin) South African Package Insert. October 4, 2005. [ Links ]
70. Pfizer Laboratories (Pty) Ltd. Lyrica (pregabalin) South African Package Insert. November 30, 2007. [ Links ]
71. Raskin J, Smith TR, Wong K, et al Duloxetine versus routine care in the long-term management of diabetic peripheral neuropathic pain. J Palliat Med 2006;9:29-40. [ Links ]
72. Sindrup SH, Bach FW, Madsen C, Gram LF, Jensen TS. Venlafaxine versus imipramine in painful polyneuropathy: a randomized, controlled trial. Neurology 2003;60:1284-1289. [ Links ]
73. Eli Lilly (S.A.) (Pty) Ltd. Cymbalta (duloxetine) South African Package Insert. February 7, 2006. [ Links ]
74. Wyeth South Africa (Pty) Ltd. Efexor (venlafaxin) South African Package Insert. June 24, 2005. [ Links ]
75. O'Connor AB, Dworkin RH. Treatment of neuropathic pain: an overview of recent guidelines. Am J Med 2009;122:S22-32. [ Links ]
76. Fava M, Mulroy R, Alpert J, Nierenberg AA, Rosenbaum JF. Emergence of adverse events following discontinuation of treatment with extended-release venlafaxine. Am J Psychiatry 1997;154:1760-1762. [ Links ]
77. Pfizer Laboratories (Pty) Ltd., Norline (Amitriptyline), South African Electronic Package Insert. http://home.intekom.com/pharm/quatrom/noriline.html (accessed 12 September 2011) [ Links ]
78. QUATROMED Ltd. Noriline (amitriptyline) South African Package Insert. June 10, 1992. [ Links ]
79. Finnerup NB, Otto M, McQuay HJ, Jensen TS, Sindrup SH. Algorithm for neuropathic pain treatment: an evidence based proposal. Pain 2005;118:289-305. [ Links ]
80. Wu CL, Agarwal S, Tella PK, et al. Morphine versus mexiletine for treatment of postamputation pain: a randomized, placebo-controlled, crossover trial. Anesthesiology 2008;109:289-296. [ Links ]
81. Raja SN, Haythornthwaite JA, Pappagallo M, et al. Opioids versus antidepressants in postherpetic neuralgia: a randomized, placebo-controlled trial. Neurology 2002;59:1015-1021. [ Links ]
82. Gilron I, Bailey JM, Tu D, Holden RR, Weaver DF, Houlden RL. Morphine, gabapentin, or their combination for neuropathic pain. N Engl J Med 2005;352:1324-1334. [ Links ]
83. Gilron I, Bailey JM, Tu D, Holden RR, Jackson AC, Houlden RL. Nortriptyline and gabapentin, alone and in combination for neuropathic pain: a double-blind, randomised controlled crossover trial. Lancet 2009;374:1252-1261. [ Links ]
84. Hanna M, O'Brien C, Wilson MC. Prolonged-release oxycodone enhances the effects of existing gabapentin therapy in painful diabetic neuropathy patients. Eur J Pain 2008;12:804-813. [ Links ]
85. Khaliq W, Alam S, Puri N. Topical lidocaine for the treatment ofpostherpetic neuralgia. Cochrane Database Syst Rev 2007:CD004846. [ Links ]
86. Vranken JH, Hollmann MW, van der Vegt MH, et al. Duloxetine in patients with central neuropathic pain caused by spinal cord injury or stroke: a randomized, double-blind, placebo-controlled trial. Pain 2011;152:267-273. [ Links ]
87. Turk DC, Audette J, Levy RM, Mackey SC, Stanos S. Assessment and treatment of psychosocial comorbidities in patients with neuropathic pain. Mayo Clin Proc 2010;85:S42-50. [ Links ]
88. Johnson MI, Bjordal JM. Transcutaneous electrical nerve stimulation for the management of painful conditions: focus on neuropathic pain. Expert Rev Neurother 2011;11:735-753. [ Links ]
89. Cruccu G, Aziz TZ, Garcia-Larrea L, et al. EFNS guidelines on neurostimulation therapy for neuropathic pain. Eur J Neurol 2007;14:952-970. [ Links ]
90. Guastella V, Mick G, Laurent B. [Non pharmacologic treatment of neuropathic pain]. Presse Med 2008;37:354-347. [ Links ]
91. O'Connell NE, Wand BM, Marston L, Spencer S, Desouza LH. Non-invasive brain stimulation techniques for chronic pain. Cochrane Database Syst Rev 2010:CD008208. [ Links ]
92. Attal N, Cruccu G, Haanpaa M, et al EFNS guidelines on pharmacological treatment of neuropathic pain. [ Links ]
![]() Corresponding author:
Corresponding author:
S Chetty
seanchetty@gmail.com
Accepted 25 November 2011.
Appendix A. Evidence classification scheme, and levels of recommendation used by Attal et al.92
Class I: An adequately powered prospective, randomised, controlled clinical trial with masked outcome assessment in a representative population or an adequately powered systematic review of prospective randomised controlled clinical trials with masked outcome assessment in representative populations. The following are required:
(a) randomisation concealment
(b) primary outcome(s) is/are clearly defined
(c) exclusion/inclusion criteria are clearly defined
(d) adequate accounting for dropouts and crossovers with numbers sufficiently low to have minimal potential for bias
(e) relevant baseline characteristics are presented and substantially equivalent among treatment groups or there is appropriate statistical adjustment for differences.
Class II: Prospective matched-group cohort study in a representative population with masked outcome assessment that meets (a) - (e) above or a randomised, controlled trial in a representative population that lacks one criterion (a) - (e).
Class III: All other controlled trials (including well-defined natural history controls or patients serving as own controls) in a representative population, where outcome assessment is independent of patient treatment
Class IV: Evidence from uncontrolled studies, case series, case reports, or expert opinion
Rating of recommendations
[X] Level A rating (established as effective, ineffective, or harmful) requires at least one convincing class I study or at least two consistent, convincing class II studies
[X] Level B rating (probably effective, ineffective, or harmful) requires at least one convincing class II study or overwhelming class III evidence
[X] Level C rating (possibly effective, ineffective, or harmful) rating requires at least two convincing class III studies














