Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 no.4 Pretoria Abr. 2012
ORIGINAL ARTICLES
Acceptability, feasibility and impact of routine screening to detect undiagnosed HIV infection in 17 - 24-month-old children in the western sub-district of Cape Town
Michael LevinI; Hlengani MathemaII; Kathryn StinsonIII; Karen JenningsIV
IIMB ChB, FCPaed (SA), MMed (Paed), Dip Allerg (SA), PhD. Division of Allergy, Department of Paediatrics and Child Health, University of Cape Town and Red Cross War Memorial Children's Hospital, and Kidzpositive Family Fund
IIBSocSc (Hons). Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, University of Cape Town
IIIMPH. Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, University of Cape Town
IVMB ChB, DCH. Health Directorate, City of Cape Town
ABSTRACT
OBJECTIVES: To explore the acceptability and feasibility of routine HIV screening in children at primary healthcare clinics and ascertain the prevalence of previously undiagnosed HIV infection in17- 24-month-old children accessing curative and routine services.
METHODS: A survey was conducted in 4 primary health clinics in the western sub-district of Cape Town. Rapid HIV screening of 17 - 24-monthold children was performed for consenting caregiver-child pairs. Data on demographics, child health and antenatal history were collected using questionnaires.
RESULTS: During recruitment, 358 children (72%) were tested for HIV infection. Most of the children (95.8%) were accompanied by a parent. The prevalence of reported HIV exposure among children was 21% (107/499). Of these, 3 had previously confirmed HIV infection; 1 was reportedly confirmed by a 6-week HIV test, and the other 2 probably contracted the virus via late postpartum transmission. The overall transmission rate was 3.5% (3/86) and the confirmed proportion of HIV-infected children was 0.8% (3/361). No previously unknown HIV infection was detected.
CONCLUSIONS: Programmes to prevent mother-to-child transmission are effective, but at-risk infants who test negative at 6 weeks should be monitored for subsequent seroconversion. Parents of HIV-exposed infants are more likely to permit (re)testing of their infants than those whose offspring are not at risk. Routine HIV testing of children is feasible and acceptable at primary level, but may require additional resources to achieve universal coverage. Routine screening at an earlier age may detect previously undiagnosed HIV infection.
Mother-to-child transmission (MTCT) contributes significantly to the incidence of HIV in sub-Saharan Africa and the rising number of children contracting HIV. In 2007 it was estimated that about 300 000 of South Africa's 5.5 million HIV-infected persons were children;1 this rose to 330 000 of 5.6 million in 2009.2
Prevention of mother-to-child transmission (PMTCT) strategies include the South African PMTCT programme that incorporates interventions antepartum, during labour and delivery, and postpartum.3,4 Despite comprehensive guidelines, service and patient-related challenges compromise the effectiveness of the PMTCT programme, leading to the identification of children with previously undiagnosed HIV infection at public health facilities. These challenges include suboptimal PMTCT programme coverage, late presentation for the first antenatal visit, maternal seroconversion after the first visit with insufficient repeat testing in late pregnancy, and suboptimal postpartum follow-up.5
The coverage of antiretroviral (ARV) prophylaxis and antiretroviral therapy (ART) initiation remains inadequate. The coverage of short-course zidovudine is poorly documented. The national coverage for single-dose nevirapine between 2007 and 2008 was 76%,6 and a study in Cape Town reported that treatment was initiated in only 51% of pregnant women eligible for ART.7
An opportunity for prevention is missed when undiagnosed HIV-positive pregnant women are not identified by services. Booking should optimally occur at 14 weeks' gestation and HIV status tested at the initial visit. Patients who refuse testing should be counselled to test at subsequent visits. Repeat HIV testing at 32 - 34 weeks for women who test negative in early pregnancy is national PMTCT policy.4 However, this is often poorly implemented. In Botswana, a study attributed 43% of all MTCT of HIV to seroconversion in late pregnancy and postpartum,8 highlighting the increased risk of unknown MTCT without re-testing.
Low coverage of postpartum follow-up services creates more opportunities for MTCT. National and Western Cape province PMTCT guidelines include repeat testing for HIV-exposed children who tested negative at 6 weeks.3,4,9,10 In the case of breastfed infants, this is performed 6 weeks after breast-feeding is ceased. National guidelines also provide for re-testing of all HIV-exposed children at 18 months of age, but Cape Town City Health data show that few repeat tests are done.5 Furthermore, the risk of MTCT increases if mothers contract HIV postpartum; therefore, health services may not identify children with HIV in good time.
We explored the acceptability and feasibility of routine HIV screening in children at primary healthcare clinics to ascertain the proportion of previously undiagnosed HIV-positive children aged 17 - 24 months accessing curative and routine child healthcare services.
Methods
This was an exploratory, facility-based cross-sectional study in the western sub-district of Cape Town. Primary healthcare facilities offering immunisation services (Du Noon, Albow Gardens, Vanguard and Langa) were non-randomly selected based on clinic size.
Fieldworkers recruited all patients aged 17-24 months as they arrived to access health services. Paediatric HIV clinics were excluded from the study, but patients on HIV registers seeking other curative or routine health services were eligible. No sample size calculations were performed due to the exploratory nature of the study. The target recruitment number of 500 patients, realised between September 2010 and February 2011, was based on Cape Town City Health statistics for the number of children accessing routine 18-month immunisation services over 3 months.
Pre-consent eligibility was determined and demographic data of all potential participants were collected from caregivers (biological parents or legal/usual guardians). A PMTCT questionnaire administered to all eligible consenting caregivers covered maternal HIV status, antenatal history, child HIV testing and infant feeding.
Healthcare providers (HCPs) provided the required consultation to children and their caregivers. To facilitate routine screening, HCPs implemented a streamlined approach to provider-initiated HIV counselling and testing (PICT).11 PICT was introduced prior to commencement of the study and all healthcare workers were trained accordingly. The Abbott Determine HIV antibody test was used to detect HIV infection. Known HIV-positive children were not screened. Caregivers could decline the testing of their children even in the case of prior consent.
Data were captured into a secure Microsoft Access database, validated and audited to ensure quality. Data analysis was performed using STATA Data Analysis and Statistical Software (release 11).
This was a non-anonymous study at primary healthcare facilities. Ethical approval was granted by the Health Sciences Research Ethics Committee at the University of Cape Town. Approval was also obtained from Cape Town City Health and the Western Cape Department of Health.
Results
During recruitment, 567 caregiver-child pairs were identified as potential participants (Fig. 1); 91% of caregivers were able to give legal consent but 3% declined, making the final number of participant pairs 499. A parent accompanied 95.8% (478/499) of children, including 97.2% (104/107) of HIV-exposed children and 95.4% (374/392) of non-exposed children.
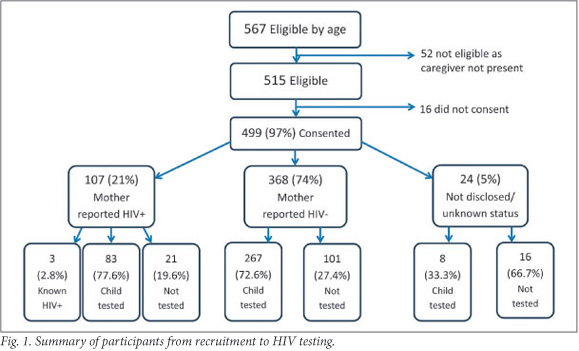
Ninety-nine per cent of pregnancies had been booked for delivery in an obstetric unit or hospital. The reported HIV prevalence of mothers was 21% (107/499). All except for 3 HIV infections were detected at early booking; these women tested negative at first booking, testing positive in late pregnancy, shortly post-partum and at recruitment, respectively. To their knowledge, 87% of all HIV-infected mothers had been on the PMTCT programme.
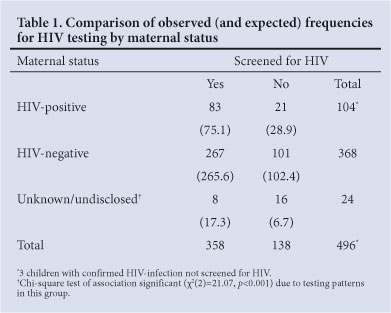
During recruitment, 72% of infants (358/499) were tested for HIV. The highest uptake of infant testing was in known HIV-positive mothers (83/107; 77.6%), followed by known HIV-negative mothers (267/368; 72.6%) (Fig. 1). The lowest uptake was in mothers of unknown or undisclosed status (8/24; 33.3%), which differed from the expected trend for random refusal to test (Table 1). An overall chi-squared test confirmed a significant association between the uptake of HIV testing and maternal HIV status (x2(2)=21.07, p<0.001). There was no significant difference in testing between HIV-positive and HIV-negative participants who disclosed maternal HIV status (x2=2.66, p=0.103). However, a significantly lower uptake of testing was found by those participants whose HIV status was unknown or undisclosed compared with known HIV-infected mothers (x2=21.40, p<0.001) and those known to be uninfected (x2=16.55, p<0.001).
No previously unknown HIV infection was detected. However, 87.9% (94/107) of known HIV-positive mothers (18.8% of total cohort) reported that their children were screened with HIV polymerase chain reaction (PCR) tests at 6 weeks. Of these, 1 child tested positive. In addition, 79 children had been tested for HIV in infancy (some were exposed to HIV but tested negative at 6 weeks). Two tested HIV-positive and were diagnosed with HIV infection in the 2 months preceding recruitment, because of intercurrent illness. One of these tested negative at 6 weeks. The other accessed PMTCT services in a neighbouring country, but it is uncertain whether the child had a 6-week HIV PCR test. The 6-week HIV transmission rate in this sample was 1.1% (1/94), the confirmed overall transmission rate was 3.5% (3/86), and the proportion of HIV-infected children (among those with confirmed HIV status) between 17 - 24 months of age was 0.8% (3/361). The HIV status of 28% (138) of the children remained unknown as they were not tested.
Ninety-two per cent (98/107) of HIV-exposed infants were reportedly never breastfed. The remaining 9 (8%) were exclusively breastfed (EBF) in the first 6 months. After this time, breast-feeding was ceased in favour of replacement feeding (formula and/or solid food) in 7 of the infants and 2 mothers reported mixed feeding. In the latter, the status of 1 mother was previously unknown; she tested HIV-positive on the day of recruitment, but her child tested negative.
The 2 children diagnosed with HIV in the 2 months preceding recruitment were EBF in the first 6 months and replacement-fed thereafter. One was reported to be HIV-negative at 6 weeks (PCR test), therefore infection may have been transmitted via breastfeeding.
Discussion
The percentage of HIV-exposed infants (21%) was in keeping with the antenatal HIV prevalence reflected by a survey in 2009.12 Overall, this was 18.2% (95% CI 17.0 - 19.3) and 17.0% (95% CI 14.1 - 20.0) in the Metro and western sub-districts, respectively. The higher result in our study may have been attributed to the smaller sample size, or the higher proportion of at-risk infants returning to follow-up clinics compared with those not at risk. It is more likely that our findings reflect the high prevalence of HIV in the low socioeconomic profile areas of Du Noon, Langa and Bonteheuwel. A comparable sub-district of Klipfontein has an HIV exposure rate of 24% (CI 21.0 - 27.1).
We found a low burden of HIV infection (0.8%) in 17 - 24-month-old children attending clinics for curative and routine visits. To our knowledge, this is the first published study of HIV burden in this age group in South Africa. This prevalence is lower than that estimated in the 2008 Human Science Research Council survey (1.1% among children aged 2 - 14 years in the Western Cape).13 While this could attest to effective PMTCT programme coverage, it is unclear whether the overall population is represented in these findings. The reported 6-week transmission rate of 1.1% was lower than the 2008/2009 provincial estimate of 4.5%.14 This could be attributed to differences in the study sites or because of participant selection bias.
Moreover, this raises questions about the patterns of access to health services among HIV-infected children. Up to 40% of HIV-exposed children may be lost to follow-up services by 18 months of age.15 The median survival time of untreated perinatally infected children is 1.1 years.16 HIV-related mortality rates are very high in the first year of life, especially when ART is not initiated early.17 Children in this age group with unknown HIV infection may present with advanced disease at secondary and tertiary healthcare facilities before 18 months of age. One of the 2 HIV-positive children diagnosed in the 2 months preceding recruitment in this study was diagnosed at a secondary level institution. Furthermore, HIV-infected children who survive hospital admission may be followed up and immunised in hospital-based services before referral to primary level services. Three of the 4 study clinics had a designated HIV clinic (excluded from recruitment) which may have provided routine health services including immunisations. Therefore, the low burden of HIV in this study does not account for HIV-infected children being sick, accessing health services elsewhere, or dying before their 18 month immunisation visit.
The overall transmission rate for participants was 3.5% (3 cases). This included a case of perinatal transmission (6-week HIV PCR test proved positive), and at least 1 case of postpartum transmission probably from breast-feeding (reported negative 6-week HIV PCR test). The age of the participant in the third case indicated that HIV contraction was also possibly via breast-feeding.
In this study, routine HIV testing of children was feasible and acceptable. Despite preconceptions of high rates of caregiver rotation at child health services, a high proportion of children accessed health services with a biological parent. This argues against routine HIV testing not being feasible due to legal issues when children are not accompanied by a guardian. The high rates of consent in this study reflect the acceptability of an HIV testing service to clients, as demonstrated in KwaZulu-Natal and Limpopo provinces.18 Our research also suggests that mothers are not opposed to PICT, which is part of the HIV component of the South African Integrated Management of Childhood Illness guidelines.
Managers at the research sites commented that the testing of children led some mothers to confirm their own status. The implementation of a more efficient approach to testing is valuable in integrating and streamlining a service with a sizeable impact on maternal and child health.11
Despite consenting to the study, only 72% of participants were tested for HIV. Reasons for declining testing were not determined. Researchers reported that some mothers retracted consent to avoid the trauma of additional procedures such as HIV testing on the same day as immunisations. This may indicate a need for additional resources to allow universal coverage.
This does not account for the discrepancy in consent to testing according to maternal HIV status. Known HIV-positive mothers were more likely to consent to the testing of their children (78%) than uninfected mothers (73%). The low rate of consent by mothers of unknown or undisclosed HIV status (33.3%) may indicate that known HIV-positive mothers perceive more benefit in re-testing their children. This could reflect a lack of knowledge of the possibility of late pregnancy and postnatal maternal acquisition of HIV. Mothers who have never been tested or refuse to acknowledge their results may be in denial about their HIV status or risk, thereby denying their children HIV screening and possible early detection.
Our results question whether routine screening of HIV in children should occur at all, and if so, whether at 18 months or at earlier routine healthcare visits. Two of the 3 confirmed cases of HIV were detected within 2 months of the study and could have been detected by earlier screening. Many children still present with undiagnosed HIV in the first year of life. Earlier diagnosis would lower HIV morbidity and mortality,17 especially in perinatal MTCT.16
It is important to consider the decline in access to services after the first year of life.15,19 Routine immunisation coverage in the Western Cape drops to just over one half by 18 months of age.19 Mothers do not wish to have invasive procedures (immunisations) and investigations (HIV test) performed on the same day; therefore, it would be preferable to link HIV testing to other routine health visits. Many mothers, however, only bring their children for immunisation visits. Furthermore, the Department of Health has called for the cessation of the free distribution of milk formula at health services, instead promoting EBF for the first 6 months of life - regardless of maternal HIV-status - together with ART provision to HIV-infected mothers and their infants.20 It may be advisable to recommend routine HIV tests after 7 months of age (e.g. at 9 months), but this does not take into account HIV-positive mothers who continue breast-feeding after 9 months of age whose children may require yet another follow-up test.
Since the additional cases detected arose in the HIV-exposed group, it is important to question whether there is any value in screening children of HIV-negative women. There may be more benefit to screening HIV-exposed children and previously HIV-negative women to detect seroconversion and test the infants accordingly.
The generalisability of the findings is hindered by the study's limitations. As an exploratory study, the sample size and study area were both small. These are unlikely factors in other programmes in the province and country. The probability of participant selection bias should be mentioned, including an apparent 'healthy child' effect at primary healthcare level. Furthermore, given the high rates of institutional booking of pregnancies and deliveries in this study, the caregiver-child pairs may have been more likely to access postnatal public health services than those who do not access antenatal care or deliver at health facilities.
Despite these limitations, lessons can be learned in terms of best practice, opportunities to be seized, and what these mean for the PMTCT programme and child health. The low burden of HIV demonstrates an effective PMTCT programme, but testing protocols must identify HIV infection not detected at 6 weeks, especially in breastfed children. Routine PICT is an acceptable and feasible service. The effect of routine HIV testing at 7 - 9 months of age should be assessed.
Funding acknowledgement. This research was funded by the US President's Emergency Plan For AIDS Relief (PEPFAR) through the United States Agency for International Development (USAID) under the terms of Award No. 674-A-00-08-00009-00 to Anova Health Institute, as well as the City of Cape Town.
USAID disclaimer. The opinions expressed herein are those of the authors and do not necessarily reflect the views of USAID or PEPFAR.
Acknowledgements. Staff at Du Noon, Albow Gardens and Langa clinics and Vanguard CHC for doing the HIV testing. Mrs Monica Sifanelo (City Health western sub-district manager) and Dr Kathy Grammer (MDHS sub-structure manager) and their management teams for supporting the project.
References
1. Department of Health. HIV and AIDS and STI strategic plan for South Africa, 2007 - 2011. Pretoria: National Department of Health, 2007. [ Links ]
2. Joint United Nations Programme on HIV/AIDS. Global report: UNAIDS report on the global AIDS epidemic 2010. UNAIDS, 2010. [ Links ]
3. Department of Health. Policy and guidelines for the implementation of the PMTCT programme. Pretoria: National Department of Health, 2008. [ Links ]
4. Department of Health. Clinical guidelines: PMTCT (Prevention of mother-to-child transmission. Pretoria: National Department of Health, 2010. [ Links ]
5. Cape Town City Health. Surveillance data (unpublished). 2008. [ Links ]
6. Day C, Barron P, Monticelli F, Sello, E, eds. The District Health Barometer 2007/08. Durban: Health Systems Trust, 2009. [ Links ]
7. Stinson K, Myer L, Boulle A. An evaluation of the approaches to the initiation of antiretroviral therapy during pregnancy among HIV-infected women in Cape Town. Cape Town: University of Cape Town, 2008. [ Links ]
8. Lu L, et al HIV incidence in pregnancy and the first post-partum year and implications for PMTCT programs, Francistown, Bostwana, 2008. 16th Conference on Retroviruses and Opportunistic Infections, Montreal, 2009. [ Links ]
9. Provincial Government of the Western Cape - Department of Health. Policy and guidelines for the prevention of mother-to-child transmission of HIV. Cape Town: Provincial Government of the Western Cape, 2009. [ Links ]
10. Provincial Government of the Western Cape - Department of Health. Circular No: H20/2011 - Re: PMTCT protocol - Implementation of revised PMTCT guidelines. [ Links ]
11. Futterman D, Stafford S, Michaels D, et al. ACTS (Advise, Consent, Test, Support). A new approach to routine HIV screening that is helping the Western Cape of South Africa maximize its clinical response to HIV/AIDS, 2010. http://www.actshivtest.org.za (accessed 22 February 2012) [ Links ]
12. HIV and Syphilis Prevalence in the Western Cape: Results of the 2009 HIV and syphilis antenatal provincial and sub-district surveys. Cape Town: Provincial Government Western Cape, Department of Health, 2009. [ Links ]
13. Human Sciences Research Council. South African national HIV prevalence, incidence, behaviour and communication survey, 2008. A turning tide among teenagers? Cape Town: Human Science Research Council Press, 2009. [ Links ]
14. Provincial Government of the Western Cape - Department of Health. Annual Report 2008/9. Cape Town: Provincial Government of the Western Cape, 2009. [ Links ]
15. Coetzee D, Hilderbrand K, Boulle A, Draper B, Abdullah F, Goemaere E. Effectiveness of the first district-wide programme for the prevention of mother-to-child transmission of HIV in South Africa. Bull World Health Organ 2005;83:489-494. [ Links ]
16. Violari A, Cotton MF, Gibb DM, et al. Early antiretroviral therapy and mortality among HIV-infected infants. N Engl J Med 2008;359(21):2233-2244. [ Links ]
17. Marston M, Becquet R, Zaba B, et al Net survival of perinatally and postnatally HIV-infected children: a pooled analysis of individual data from sub-Saharan Africa. Int J Epidemiol 2011;40(2):385-396. [http://dx.doi.org/10.1093/ije/dyq255] [ Links ]
18. Horwood C, Voce A, Vermaak K, Rollins, N, Qazi S. Routine checks for HIV in children attending primary healthcare facilities in South Africa: Attitudes of nurses and child caregivers. Soc Sci Med 2010;70:313-320. [http://dx.doi.org/10.1016/j.socscimed.2009.10.002] [ Links ]
19. Corrigal J, Coetzee D, Cameron N. Is the Western Cape at risk of an outbreak of preventable childhood diseases? Lessons from an evaluation of routine immunisation coverage. S Afr Med J 2008;98(1):41-45. [ Links ]
20. Department of Health, 2011 Media statement by Minister of Health Dr Aaron Motsoaledi, 23 August, 2011. http://www.doh.gov.za/show.php?id=3045 (accessed 22 February 2012). [ Links ]
Accepted 18 January 2012.
Corresponding author: ME Levin (michael.levin@uct.ac.za)














