Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 n.4 Pretoria Apr. 2012
SCIENTIFIC LETTER
The value of blood culture audits at peripheral hospitals
Chris R KenyonI; Geoff FattiIII; Neshaad SchruederII; Kim BonorchisIV; Graeme MeintjesV
IMB ChB, MPH, MA, FCP (SA). G F Jooste Hospital and Department of Medicine, Faculty of Health Sciences, University of Cape Town
IIMB ChB, FCP (SA). G F Jooste Hospital and Department of Medicine, Faculty of Health Sciences, University of Cape Town
IIIMB ChB, MPH. Kheth'Impilo, Cape Town
IVMB ChB, FC Path (SA) Micro. Greenpoint Laboratory, National Health Laboratory Service (NHLS), Cape Town
VMB ChB, FCP (SA), MRCP. G F Jooste Hospital, Institute of Infectious Diseases and Molecular Medicine, and Department of Medicine, Faculty of Health Sciences, University of Cape Town, and Department of Medicine, Imperial College London, UK
ABSTRACT
Knowledge of local antibiotic sensitivities is crucial to creating appropriate empiric antibiotic guidelines. The new National Health Laboratory Service (NHLS) Data Warehouse allows clinicians to access collated spreadsheets of culture isolates and antimicrobial susceptibility patterns for their facilities. We used this service to study the trends in blood culture (BC) results at GF Jooste Hospital from 2005 to 2010. We investigated the BC contamination rate and changes in the antibiotic sensitivity profiles of selected organisms, and estimated the proportion of infections that were hospital-acquired. Over 3000 BCs were performed per year in this period. A very high contamination rate was observed (7 - 9%) in 2005 -2007, with a gratifying reduction by 2010. Ceftriaxone resistance increased from 16% to 62% in Klebsiella pneumoniae (p<0.0001), and from 33% to 100% in Enterobacter spp. (p=0.053).
To the Editor: The auditing of health service provision has well-established benefits, but there is little published on the practical monitoring and handling of changes to microbial threats in individual hospitals. There is considerable concern about the rise of multidrug-resistant bacteria, and the roles of healthcare facilities - in particular, the lack of adequate infection control procedures - and inappropriate broad-spectrum antibiotic use in the genesis and spread of this problem. This concern is great in South African hospitals with a high proportion of immunocompromised patients at increased risk for severe and resistant bacterial infections.1-3 Appropriate empiric antibiotic guidelines should be based on accurate and up-to-date knowledge of local antibiotic sensitivities. We questioned how many hospitals collect, analyse and act on data regarding the prevalence and likely provenance of antibiotic resistance in key organisms. The National Health Laboratory Service (NHLS) Data Warehouse was used to study the trends in blood culture (BC) results at GF Jooste Hospital from 2005 to 2010. We investigated the BC contamination rate and changes in the antibiotic sensitivity of selected organisms, and estimated the proportion of hospital-acquired infections.
Methods
The major indications for BCs at GF Jooste are patients who present with pneumonia, unexplained fever - particularly in the immunocompromised - and in patients with suspected nosocomial sepsis. All specimens were processed at the NHLS Greenpoint laboratory. Isolation, identification and sensitivity testing was performed according to standard methods and the Clinical and Laboratory Standards Institute (CLSI) guidelines.4 Sensitivity testing was performed manually or with the Vitek 2 system. Changes in resistance trends over time were assessed with chi-squared tests using Stata 10. The study was approved by the Research Ethics Committee of the University of Cape Town.
Results
Over 3000 BCs were performed per year; 66% were performed in casualty and 18% in the 2 medical wards. The remainder were performed in the 2 surgical wards, high care unit and outpatients departments (Table 1). Although the number of BCs performed per year changed marginally between 2005 and 2010, there was a statistically significant decline in the number of BCs with positive growth (from 872 to 421, p<0.0000) and a decline in BCs positive for a coagulase-negative Staphylococcus (CNS) (323 to 145, p<0.0000). In 2005 - 2007, an alarming 7 - 9% of all BCs and 40% of all positive-growth BCs were positive for a CNS. About 97% of these were likely skin contaminants due to poor aseptic technique.4 The contamination rate was therefore 3 times higher than the internationally accepted rate of 3%.5 A statistically significant and gratifying reduction to 4.6% was observed in 2010.
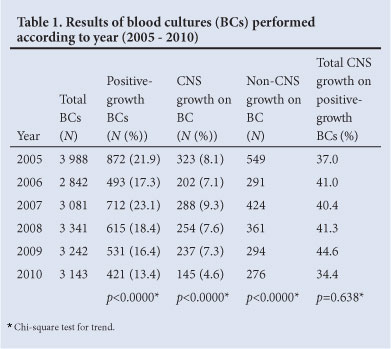
Over the 5-year period, there was also a decline in positive-growth BCs (22 - 13%), including a decline in the numbers of CNS and non-CNS organisms. This might have been attributed to a decline in admissions due to changes to the hospital referral area and a simultaneous increase in the rate of BCs performed per admission. The initial high BC positivity rate might have been, in part, related to excess workload and attributable to too few BCs being performed.
The decline in the contamination rate could also have been related to a reduced workload, but was more likely the result of a campaign focused on proper BC technique education and improved infection control with an increased supply of chlorhexidine-alcohol cleaning solution.
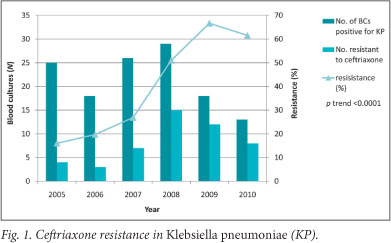
Klebsiella pneumoniae (KP) and the Enterobacter species demonstrated the most concerning increase in resistance (Fig. 1). The chi-squared test for trend revealed a highly significant increase in ceftriaxone resistance in KP between 2005 and 2010 (16 - 62%, p<0.0001) and a borderline increase in Enterobacter spp. (33 - 100%, p=0.053). Ceftriaxone resistance in E. coli remained relatively low (5 - 13%, p=0.089). The monitoring of ceftriaxone resistance is particularly important as it can reflect the acquisition of extended spectrum beta-lactamases (ESBLs). It is likely that at least 5 of the 9 ESBL-producing KP infections in 2009 were hospital-acquired, as the corresponding BCs first came up positive 9 - 21 days after admission. In 2009, 3 and 6 of the 9 ESBL-producing KP infections were intermediately and fully sensitive to amikacin, respectively.
There was no increase in cloxacillin resistance in Staphylococcus aureus, but at least 3 of the 7 MRSA BC isolates from 2009 were considered to be hospital-acquired (the time from admission to first positive BC was 11 - 37 days). Most MRSAs remained sensitive to clindamycin. Overall clindamycin resistance was low (7% average over 6 years); this is considerably lower than in neighbouring tertiary hospitals.6 Organisms from the 8-bedded high and intensive care unit revealed no MRSA isolates; all ESBL-producing organisms were sensitive to amikacin.
Discussion
From this simple and fast audit we concluded that: (i) the contamination rate, though reduced, was unacceptably high; (ii) there was a rising rate of ESBL-producing KP infection, most of which was nosocomially acquired and some of which was likely to have been driven by the widespread use of ceftriaxone; and (iii) the high proportion of hospital-acquired MRSA and ESBL-producing KP suggested a failure of basic infection control.
We presented these findings to each hospital department to motivate behavioural change in 3 domains. Firstly, with emphasis on the casualty department where most BCs were performed, we encouraged doctors to comply with our simple and clear guidelines printed on the BC trays. Secondly, we motivated for improved antibiotic stewardship. Thirdly, we reiterated the under-practised key points of infection control that facilitated the spread of resistant organisms, especially hand-washing before and after patient contact. Furthermore, bottlenecks in the supply of sufficient quantities of chlorhexidine to various wards were identified and addressed.
References
1. Pepper DJ, Rebe K, Morroni C, Wilkinson RJ, Meintjes G. Clinical deterioration during antitubercular treatment at a district hospital in South Africa: the importance of drug resistance and AIDS defining illnesses. PLoS One 2009;4(2):e4520. [http://dx.doi.org/10.1371/journal.pone.0004520] [ Links ]
2. Popovich KJ, Weinstein RA, Aroutcheva A, Rice T, Hota B. Community-associated methicillin-resistant Staphylococcus aureus and HIV: intersecting epidemics. Clin Infect Dis 2010;50(7):979-987. [http://dx.doi.org/10.1086/651076] [ Links ]
3. Cotton MF, Wasserman E, Smit J, Whitelaw A, Zar HJ. High incidence of antimicrobial resistant organisms including extended spectrum beta-lactamase producing Enterobacteriaceae and methicillin-resistant Staphylococcus aureus in nasopharyngeal and blood isolates of HIV-infected children from Cape Town, South Africa. BMC Infec Dis 2008;8:40. [http://dx.doi.org/10.1186/1471-2334-8-40] [ Links ]
4. Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial susceptibility testing; seventeenth informational supplement. CLSI document M100-S17. Wayne, Pennsylvania: CLSI, 2007. [ Links ]
5. Towns ML, Jarvis WR, Hsueh P. Guidelines on blood cultures. J Microbiol Immunol Infect 2010;43(4):347-349. [http://dx.doi.org/10.1016/S1684-1182(10)60054-0] [ Links ]
6. Bamford C, Badenhorst L, Duse AG, et al. Antimicrobial susceptibility patterns of selected invasive pathogens from public sector hospitals in South Africa, 2007. South Afr J Epidemiol Infect 2009;24(2):28-30. [ Links ]
Accepted 24 February 2012
Corresponding author: C R Kenyon (chriskenyon0@gmail.com)














