Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 n.1 Pretoria Jan. 2012
ORIGINAL ARTICLES
South african hypertension guideline 2011
Y K SeedatI; B L RaynerII
IMD, PhD, FRCP (London), FCP (SA), Hypertension Guideline Working Group, Southern African Hypertension Society, and Nelson Mandela School of Medicine, Faculty of Health Sciences, University of KwaZulu-Natal, Durban
IIMB ChB, FCP (SA), MMed, Hypertension Clinic, Department of Medicine, Faculty of Health Sciences, University of Cape Town, and Groote Schuur Hospital, Cape Town
ABSTRACT
OUTCOME: Extensive data from randomised controlled trials have shown the benefit of treating hypertension. The target blood pressure (BP) for antihypertensive management is systolic <140 mmHg and diastolic <90 mmHg with minimal or no drug sideeffects; however, stricter BP control is required for patients with end-organ damage, co-existing risk factors and co-morbidity, e.g. diabetes mellitus. The reduction of BP in the elderly and in those with severe hypertension should be achieved gradually over 1 month. Co-existent risk factors should also be controlled.
BENEFITS: Benefits of management include reduced risks of stroke, cardiac failure, chronic kidney disease and coronary heart disease.
RECOMMENDATIONS: The correct BP measurement procedure is described, and evaluation of cardiovascular risk factors and recommendations for antihypertensive therapy are stipulated. The total cardiovascular disease risk profile should be determined for all patients to inform management strategies. Lifestyle modification and patient education are cornerstones in the management of every patient. Major indications, precautions and contraindications to each recommended antihypertensive drug are listed. Combination therapy should be considered ab initio if the BP is >20/10 mmHg above goal. First-line drug therapy for uncomplicated hypertension includes low-dose thiazide-like diuretics, calcium channel blockers (CCBs) or angiotensin-converting enzyme inhibitors (ACEIs) (or ARBs - angiotensin II receptor blockers). If the target BP is not obtained, a second antihypertensive should be added from the aforementioned list. If the target BP is still not met, the third remaining antihypertensive agent should be used. In black patients either thiazide-like diuretics or CCBs can be used initially, because response rates are better than with ACE-Is or β-blockers. In treating resistant hypertension, a centrally acting drug, vasodilator, α-blocker, spironolactone or β-blocker should be added. This guideline includes management of specific situations, i.e. hypertensive emergency and urgency, severe hypertension with target organ damage, hypertension in diabetes mellitus, resistant hypertension, fixed drug combinations, new trials in hypertension, and interactions of antihypertensive agents with other drugs.
VALIDITY: The guideline was developed by the Southern African Hypertension Society.
1. Introduction
This is the 5th hypertension guideline published by the Southern African Hypertension Society (SAHS). It is an important step towards implementing a national standard to improve the quality of care for persons living with hypertension. The realistic objectives described can be applied widely and aim to diminish the impact of hypertension and related cardiovascular disease (CVD) risk in South Africa. Previous versions of the SAHS guideline emphasised improved diagnosis and treatment, tighter control and risk factor stratification.1-4Other guidelines support the same trends and the movement to evidence-based strategies.5-10 The SAHS continuously reviews its guideline, given the changing nature of the evidence.
Using criteria of BP >160/95 mmHg or persons aged >18 years receiving antihypertensive medication, the 1998 South African Demographic and Health Survey estimated that there were 3.3 million people with hypertension in the country.11 Acceptance of the international definition of hypertension (BP >140/90 mmHg; used in this guideline) adds a further 2.7 million people to this hypertensive population.11 CVD was previously ranked as the second highest cause-of-death category in South Africa.12 This has major cost implications for a developing country and requires a national strategy for prevention and management.
Hypertension is a global health burden affecting developed and developing countries, including South Africa.13 The high prevalence of hypertension worldwide contributes to the present and anticipated pandemic of CVD, which is of particular concern in developing countries.13 The control of hypertension, together with the curbing of other major risk factors such as cigarette smoking, dyslipidaemia and diabetes mellitus, constitutes the ideal approach to the primary prevention of atherosclerotic disease, and remains a major challenge for the community. The trend towards comprehensive cardiovascular (CV) risk factor management is the internationally accepted model of care.14
Hypertension is a major and costly contributor to CVD: it accounted for R4 - 5 billion in direct and indirect expenditure in 1991,15 and was previously shown to constitute 7.5% of the direct total healthcare spend in South Africa.16 This guideline has adopted an evidence-based approach to the estimation of CVD risk, intended to allow the treatment of patients at highest risk and those who can benefit most from lifestyle and drug interventions at the lowest cost, given the country's limited resources.17
2. Objective and methodology
The objective of this guideline is to promote evidence-based, accessible, and comprehensive management of hypertension by healthcare professionals in the public and private sectors in South Africa. It should act as a resource document to inform hypertensive patients of the national approach to hypertension care. For development of this guideline, hypertension and CVD treatment and prevention guidelines were reviewed, as well as hypertension trials reporting clinical end-points, including individuals with important co-morbidities, such as diabetes mellitus and chronic kidney disease (CKD).
3. Measurement of BP
Blood pressure (BP) measurement is a vital clinical sign, but can be poorly performed by healthcare professionals. Published recommendations of the European Hypertension Society and the American Heart Association are endorsed by the Hypertension Working Group.5,18
3.1 Generic measurement principles
These recommendations are generic and apply equally to all validated devices, especially in clinic and self BP monitoring (SBPM); considerations include arm position, posture of the patient, cuff size and the number of readings that should be taken. BP should be recorded using an approved device with the patient seated for at least 5 minutes before measurement (with the back supported, and arm bared and resting on a surface at heart-level). Patients should not have smoked, or ingested caffeine-containing beverages or food in the preceding 30 minutes. In persons aged 60+ years, those with diabetes mellitus and others at risk (Table III), the BP should also be recorded after the patient has stood for 1 minute, to document postural hypotension.
An appropriate size cuff should be used: a standard cuff (12 cm) for a normal arm and a larger cuff (15 cm) for an arm with a midupper circumference >33 cm; the bladder within the cuff should encircle 80% of the arm. If an undersized cuff is used, the BP can be overestimated (under-cuffing); the BP can equally be underestimated (over-cuffing) if the cuff and bladder are too large.
Systolic blood pressure (SBP) and diastolic blood pressure (DBP) should be recorded. BP should be measured in both arms at the initial consultation; if there is a discrepancy, it should subsequently be taken in the arm with the higher BP. SBP should be estimated first by palpation to avoid missing the auscultatory gap. SBP is measured at the first appearance of sound (Phase I) and DBP is measured at the disappearance of sound (Phase V). Measurement at Phase V is also recommended in pregnancy. In cardiac arrhythmia (e.g. atrial fibrillation) the highest Phase I and lowest Phase V are recorded as the SBP and DBP, respectively. Circumstances when both Phase IV (muffling) and Phase V should be recorded include aortic regurgitation, pregnancy and severe anaemia.
The BP recorded should be the average of 2 readings taken 1 minute apart. If the first 2 readings differ by >5 mmHg, additional readings should be taken. When the initial SBP is between 140 and 160 mmHg, or the DBP is between 90 and 100 mmHg, repeat measurements should be performed on 3 separate occasions within a period of 2 months, to determine whether a diagnosis of hypertension is valid. All measurements should preferably be taken at the same time of the day and in the same arm.
The elderly may present considerably variable BP measurements. It may be advisable to check the standing BP in hot weather - particularly in diabetics, the elderly, and those who have symptoms of postural hypotension.19The most common form of hypertension is isolated systolic hypertension (ISH), attributed to the stiffening of the large arteries that occurs with ageing.
The BP measurement device and its attachments (tubing, cuff, and valve) must be serviced and calibrated at least once each year.
3.2 Mercury sphygmomanometer
There is increasing criticism of the use of mercury sphygmomanometers: mercury is inert and does not degrade,20but is not toxic to patients or operators, provided that the device is intact; however, mercury becomes a major environmental hazard when it is discarded. There are international moves to replace mercury sphygmomanometers with battery-operated digital devices; however, in South Africa and other developing countries, there is concern about the availability of accurate devices, and the safe disposal of lead-containing batteries. If a mercury sphygmomanometer needs replacement, an inexpensive solar-powered validated oscillometric device should be considered.21
3.3 Self- and ambulatory BP monitoring
SBPM and ambulatory blood pressure monitoring (ABPM) is recommended in selected circumstances and for selected target groups:18
• In suspected 'white-coat' (higher readings in the office compared with readings outside) or masked hypertension (normal readings in the office and higher readings outside)
• To guide antihypertensive medication, especially in high-risk groups (e.g. the elderly and diabetics)
• In refractory hypertension
• To improve compliance with treatment (SBPM only).
Masked hypertension should be suspected if target organ damage (TOD) progresses or remains unresolved, despite BP control in the clinic. All devices used for SBPM and ABPM should be validated properly with the following independent websites: http://www.dableducational.com and http://afssaps.sante.fr.
For SBPM, only upper-arm devices are recommended, but are unsuitable in patients with sustained arrhythmias. These devices should not be used for BP measurement during exercise, and they are not as specific as ABPM devices for the diagnosis of whitecoat hypertension. Ideally, a reliable mean BP value should be established by calculating the average of 2 early morning and 2 late afternoon readings taken over 5 days. Patients must discuss any proposed change in medication with their healthcare professional. The advantages of SBPM are: improved assessment of drug effects; detection of causal relationships between adverse events and BP response; and improved compliance. The disadvantages include an increase in anxiety and the risk of self-medication.
ABPM provides the most accurate method to diagnose hypertension, assess BP control and predict outcome.22 In patients with a raised clinic BP, ABPM was shown to reduce misdiagnosis and save costs.23 Additional costs of ABPM were counter-balanced by the costs saved by better-targeted treatment. ABPM can also assess nocturnal BP control and BP variability, which are important predictors of adverse outcome; however, this is limited by access to equipment - particularly in the public sector - and the impracticalities of regular 24-hour monitoring (Table I).

4. Cardiovascular disease risk stratification
The principle of assessing and managing multiple major risk factors for CVD is unanimously accepted; however, there is much debate on which method of risk stratification to use.
4.1 Rationale for cardiovascular risk assessment
The rationale for assessing CVD risk is that certain risk factors confer a great possibility of morbidity and mortality. It is assumed that identification of individuals at highest risk allows scarce resources to be focussed on managing individuals with the greatest potential to benefit from treatment. Despite the availability of many CVD risk factor systems and charts, all have shortcomings, particularly in developing countries such as South Africa. Any consensus must address topics such as the weighting of BP readings and other risk factors in relation to ethnic group, resources, and the development of associated clinical conditions (ACC) and TOD. The SAHS remains committed to the format of the CVD risk assessment (Table II) until there is national consensus on a different model by all stakeholders (professionals, providers, government and healthcare funders) that is supported by adequate local data. The CVD risk assessment described in this guideline was developed by the European Hypertension Society and the European Society of Cardiology.7 Absolute risk and the continuous risk associated with BP is used in many other guidelines.6,8,9 This pragmatic risk assessment model is adaptable for use in many settings, including those constrained by limited resources and low budgets.
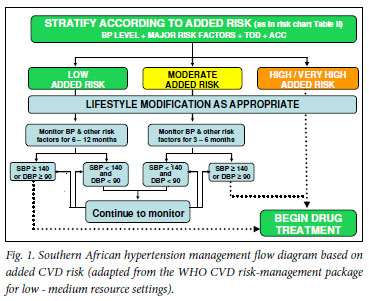
There is consensus on the necessity of immediate drug treatment for those with known ACC and/or TOD and/or a SBP >180 mmHg or DBP >110 mmHg (Fig. 1).7 In the absence of ACC, TOD and very high levels of BP, the exact BP level at which to initiate treatment has changed over time, and remains the subject of debate.
4.2 Risk factors, TOD and ACC
Table III lists the major risk factors, ACC and TOD. Modifiable risk factors (e.g. smoking and dyslipidaemia) should be the target of lifestyle intervention and treatment as appropriate. In addition to controlling hypertension, TOD and ACC must be managed appropriately and, if necessary, the patient must be referred to a higher level of care.
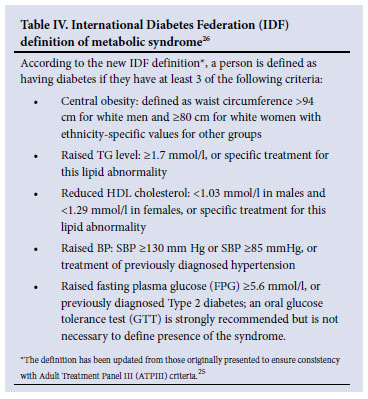
Consensus is lacking in treatment decisions on the importance of newer risk factors such as obesity and the metabolic syndrome, e.g. obesity may be measured using the body mass index (BMI), abdominal circumference or waist-to-hip ratio.24The metabolic syndrome has not been listed as a risk factor as in the JNC 7 report, despite evidence of its link to CV risk and future diabetes.5 The syndrome represents a combination of underlying and major risk factors; however, there is debate as to which criteria to adopt and whether the clustering of risk factors represents a greater CV risk over and above the individual components used in the Framingham risk calculation. However, where the metabolic syndrome is present, a SBP in >140 mmHg and or DBP >85 mmHg requires treatment (weight loss and exercise followed by drug therapy, after an appropriate trial of lifestyle modification). Table IV shows the new definition of the metabolic syndrome,25 which has recently harmonised with other major international bodies.26
4.3 Routine baseline investigations
Table V lists recommended routine basic investigations. Apart from measurements of overweight and obesity, the tests are performed annually, unless abnormal results are obtained. Tests with abnormal results must be repeated as clinically indicated.
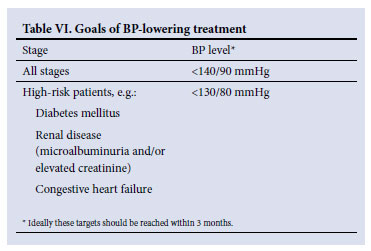
5. Goals of BP-lowering treatment
The goals of BP-lowering treatment vary according to number of major risk factors, TOD and/or ACC (Table VI). These goals should be added to the recommended goals for waist/abdominal circumference, blood sugar and lipid levels in patients with the metabolic syndrome (section 8.3).
6. Sustainable hypertension management and scarce resources
The Hypertension Working Group has given considerable time, thought and debate to the concerns about the economic sustainability of lifelong drug therapy. Affordability is important in South Africa's private and public sectors. This becomes more relevant given the proposals for the holistic management of hypertension, its complications and disease associations (section 8.3).
SAHS strongly reiterates the importance of lifestyle modification at all stages of hypertension. If resources are limited, drug treatment may be delayed if the added CV risk is low or moderate (Table II and Fig. 1). Studies emphasise the cost-effectiveness of lifestyle and drug management in reducing CVD risk in developed and less-developed regions of the world, and the importance of tight BP control in African-Americans.
Currently, the price of antihypertensive and other drugs fluctuates considerably irrespective of market sector. Where possible, generic equivalents and combinations are encouraged and the cheapest generic in a class should be considered, provided that it is a true equivalent. The patient should not be changed frequently from one generic to another in the same class, solely because of lower price. SAHS is concerned that guidelines overemphasise the use of limited resources (when to introduce medication, use of generics, and inadequate level of BP control) and may lead to poorly managed hypertension with the undesirable consequences of heart failure, stroke and chronic renal failure. Best practice recommendations should be stated clearly and compromises, based on limited resources, should be made deliberately and transparently.
7. Management
A diagnosis of hypertension may be made if repeat BP measurements, performed on 3 separate occasions within a period of 2 months, indicate a SBP >140 mmHg and/or a DBP > 90 mmHg. Where circumstances permit, ABPM should be considered, particularly in the absence of TOD. This section does not apply if the SBP is >180 mmHg or the DBP is >110 mmHg: refer to section 9 on severe (stage III) hypertension.
Lifestyle information should be given to all patients when BP measurement is performed. In the case of elevated BP, a programme of lifestyle modification should be implemented immediately (Fig. 1 and section 8 - further clarified by using Table II).
7.1 Lifestyle modification
A healthy lifestyle remains the cornerstone of managing hypertension regardless of BP level: in addition to decreasing BP, it enhances antihypertensive drug efficiency and decreases total CV risk. The Department of Health's strategy for a healthy lifestyle is supported - measures to improve BP include (Table VII):
• Achieving and maintaining ideal weight (BMI of 18.5 -24.9) - refer to the 2 local guidelines for the prevention and management of obesity.
•Limiting total sodium intake to < 2 400 mg/day (<1 teaspoon of salt). High sodium levels are found not only in table salt, but also in packet soups, stock cubes, gravies, processed cheese, many breakfast cereals, breads, salty snacks and tinned foods. Reducing the intake of such foods is crucial. The removal of the salt cellar from the table and a gradual reduction in added salt in food preparation should be recommended. Patients must be informed that food may taste bland initially and that taste adaptation to reduced sodium intake occurs with time; the use of lemon juice, herbs and spices as alternative seasoning should be encouraged. Salt content is listed as sodium in food labels:
• 'Sodium free': <5 mg per 100 g serving
• 'Very low sodium': <40 mg per 100 g serving
• 'Low sodium': <120 mg per 100 g serving
• Salt content can be calculated by multiplying sodium content by 2.5.
• Limiting alcohol intake to 2 standard drinks per day for men and 1 standard drink per day for women and small men. A standard drink (approximately 10 g of ethanol) is equivalent to 25 ml of liqueur/spirits, 125 ml of wine, 340 ml of beer, or 60 ml of sherry.
• Following the nutrition guidelines published by the WHO, which emphasise: a diet low in total fat with high intake of fruit and vegetables (5 portions per day), regular low-fat dairy products, high intake of high-fibre wholegrain foods, fish rather than red meat, products low in saturated fat, low salt, and sparing use of sugar and sugar-containing foods.8,14,17 Beverages with high caffeine levels should be avoided, but modest use (1 - 2 cups of coffee per day) will not increase BP.
• Regular moderate intensity exercise for at least 30 minutes on most - preferably all - days of the week, e.g. brisk walking at 40-60% of peak. Exercise bouts can be continuous or accumulated in shorter periods throughout the day. The benefit of exercise is dose-responsive: early adaptations from a sedentary lifestyle to becoming moderately active have the greatest effect. Patients with uncontrolled hypertension should only embark on exercise training after evaluation and initiation of therapy.
• Avoiding the use of all tobacco products, including snuff. Nicotine replacement therapy should be used for a patient with hypertension, while under medical supervision.
7.2 Drug therapy
In use of these recommendations for treatment, it is essential that the patient's added CV risk is assessed according to Table II, thereby informing the decision to implement drug therapy according to the decision flow chart (Fig. 1). Drug therapy is commenced in the following cases:
• Low added risk: SBP remains >140 mmHg or DBP remains >90 mmHg despite a period of lifestyle modification and observation (6 - 12 months)
• Moderate added risk: SBP remains > 140 mmHg or DBP remains >90 mmHg despite a period of lifestyle modification and observation (3 - 6 months)
• High or very high added risk.
The following must be considered prior to the selection of an antihypertensive agent: the cost of the drug class, patient-related factors such as the presence of major risk factors, conditions favouring use, contraindications, ACC and TOD (Tables III and VIII). In otherwise uncomplicated essential hypertension, there are 3 important antihypertensive agents: diuretics (thiazide and thiazide-like), angiotensin-converting enzyme inhibitors (ACE-Is) and calcium channel blockers (CCBs). A thiazide-like diuretic is advisable when consideration for the cost of therapy is relevant. Studies have led to reconsideration of the drugs of choice for the management of uncomplicated hypertension. The most cost-effective antihypertensive drugs are a thiazide-like diuretic (or a CCB in black patients).10 The combination of a thiazide diuretic with a β-blocker is discouraged, especially where there is abdominal obesity combined with hypertension; both classes of drugs have adverse metabolic consequences and increase the risk of new diabetes.
In uncomplicated essential hypertension, therapy should be initiated with a diuretic, ACE-I (or ARB - angiotensin II receptor blocker), or CCB. Combination therapy should be considered ab initio if BP is > 20/10 above goal, with either an ACE-I (or ARB)/diuretic, ACE-I/CCB or CCB/diuretic combination. A thiazide-like diuretic with a CCB is the preferable combination for black hypertensive patients. In general, fixed drug combinations (FDCs) are preferred. If control is not reached with monotherapy, combination therapy should be instituted with another drug from the first-line classes.
The choice of diuretic should be a low-dose hydrochlorothiazide (12.5 - 25 mg) or a thiazide-like diuretic-like indapamide (1.25 - 2.5 mg daily). Chlorthalidone (15 - 30 mg daily) is more potent compared with hydrochlorothiazide; it produces a sustained 24-hour BP-lowering response, and may replace hydrochlorothiazide as a hypotensive agent.10,27-29 Chlorthalidone is currently not available in South Africa, except as a fixed combination with atenolol. Loop diuretics such as furosemide should not be used because of their short duration of hypotensive activity (about 6 hours), unless there is evidence of chronic kidney disease (CKD) with an estimated glomerular filtration rate (GFR) <45 ml/min.
Table VIII lists the clinical considerations and possible contraindications of the major antihypertensive drug groups. Issues relating to drug adherence are critical and must be considered in each patient (section 14). Ensuring that each patient understands the importance of adherence to the treatment regimen and returns drug containers and unused drugs should be reinforced frequently. Patient empowerment and single daily dose regimens improve compliance. Fixed-dose combinations should be used where appropriate. Continued monitoring and management of drug side-effects is essential.
Hypertension can seldom be managed in isolation from other related chronic illnesses. Lifestyle modification, drug therapy and the targets of management should be broadened to include measures of other risk factors and co-morbidities, e.g. obesity, blood sugar, lipids and BP control (Table IX).
The metabolic syndrome is of increasing prevalence. The clinical criteria of the syndrome have been debated and may include obesity, dyslipidaemia and Type 2 diabetes mellitus in addition to hypertension. This guideline recognises that insulin levels or insulin/glucose ratios have no place in the diagnosis (Table IV). The syndrome is not a clearly defined entity with an evident aetiology or underlying mechanism; therefore, its management should reflect the accepted strategies to reduce CV risk, and each component of the syndrome should be managed if present (e.g. increased abdominal girth and BP is decreased with a combination of intensive diet and exercise).
Table VII reflects the current South African norms for the Dietary Approaches to Stop Hypertension (DASH).30A low-sodium diet will lower BP and also have a favourable effect on weight, lipids and glycaemic control. Evidence is mounting that so-called normal lipid levels may be inappropriately high in hypertensive patients, and are certainly so in patients with hypertensive complications, e.g. stroke and myocardial infarction (MI). Lipid-lowering therapy is increasingly becoming a part of standard drug therapy in both young and old patients with vascular disease. In some cases, the use of the biguanide and/or metformin may be required, in addition to exercise, to prevent the progression to frank Type 2 diabetes mellitus in hypertensive patients with central obesity.
Questions are frequently asked regarding the indications and contraindications for the use of aspirin, hormone replacement therapy and antioxidants. Low-dose aspirin should be used for secondary prevention of a transient ischaemic attack (TIA), stroke and MI, only once the BP is well-controlled. The use of hormone replacement therapy, antioxidants, homoeopathic or complementary drugs is of no benefit in hypertensive patients.
7.2.1 Consensus statement on the use of ACE-Is and ARBs
The use of ARBs and ACE-Is results in up to 95% and 75% blockade, respectively, of the renin-angiotensin system. No difference in this outcome was found in patients with diabetes and microalbuminuria (N=50)31 or in patients post myocardial infarction, with heart failure and/or impaired left ventricle (LV) dysfunction.32 The ARB losartan produced greater regression of left ventricular hypertrophy (LVH), a 14% reduction in the primary end-point of CV morbidity and death, and a 25% reduction in stroke, compared with a β-blocker.33 ACE-Is have been shown to prevent the progression of microalbuminuria from normoalbuminuria, and reduce established microalbuminuria in Type 2 diabetes.34 ARBs have also been shown to reduce microalbuminuria, and delay the progression of established diabetic nephropathy.35ACE-Is, in combination with indapamide36or an ARB, have proven effective in the secondary prevention of stroke.37Furthermore, the effect of the ARB, telmisartan, has been shown to be equivalent to that of the ACE-I, ramipril, in patients at high CV risk. Combination therapy, however, had no added advantage, and caused increased side-effects and adverse renal outcomes.38 Hence, there appears to be little difference between ACE-Is and ARBs; choice of therapy should be determined by cost and tolerability.31
7.2.2 Compelling indications for a specific drug class
Table VIII outlines the compelling indications (high-risk conditions) for certain classes of antihypertensive drugs, based on randomised
Table VIII. Indications and contraindications for the major classes of antihypertensive drugs*
Contraindications Class Conditions favouring use Compelling Possible controlled trials. These indications apply equally to patients from any ethnic group; however, the prevalence of other illnesses such as diabetes in certain groups such as the Asians must be considered (section 11.1).
7.2.3 Direct renin inhibitor
Since the last guideline publication, the direct renin inhibitor, aliskiren, has been registered for the treatment of hypertension in the USA and Europe. This drug provides antihypertensive efficacy similar to other classes of drugs and has a similar placebo-like profile to ARBs. It can be combined effectively with other major classes of antihypertensive drugs, especially diuretics and CCBs. In the absence of major outcome studies, the place of direct renin inhibitors is not established and it is uncertain whether aliskiren will have additional benefits over ACE-Is and ARBs. However, benefits of the combination of aliskiren with ACE-Is and ARBs have been shown in patients with diabetic nephropathy and heart failure, respectively.39,40 Long-term outcome studies - such as the ALTITUDE study in patients with Type 2 diabetic nephropathy41- are awaited before recommendations on the use of direct renin inhibitors can be made.
7.2.4 New trials
Seven recent trials, involving 83 112 patients with hypertension and other co-morbidities, have been published and have influenced this guideline: TRANSCEND,42 ON TARGET,38 ACCOMPLISH,43HYVET,44 PROFESS,45 AVOID,39 ADVANCE,46 and ACCORD.47
Prior to the HYVET study, the treatment of hypertension in very elderly patients was not proven to be beneficial; the study showed unequivocally that initiation of treatment with indapamide, with or without perindopril vs. placebo, resulted in significant reductions in mortality and CV end-points.44
The ACCOMPLISH study demonstrated that combination treatment with amlodipine plus benazepril vs. hydrochlorothiazide plus benazepril resulted in a 20% reduction in the primary endpoint.43 This influenced the recent ISHIB guidelines regarding combination therapy, where a CCB plus RAAS inhibitor combination was preferred, except in the event of volume overload.48 In the latter case, a RAAS inhibitor plus diuretic combination was preferred.
Two recent studies in Type 2 diabetes showed contrasting results. In the ADVANCE study, the addition of perindopril plus indapamide vs. placebo resulted in significant reductions in both macro-and microvascular complications, with no lower threshold for BP.46 In contrast, in the ACCORD study, intensive (<120/80 mmHg) vs. standard care did not result in a reduction in the primary end-point of CV morbidity and mortality, but did reduce the secondary endpoint of stroke.48 These contrasting results have created a dilemma regarding the target BP in diabetics.
The PROFESS45and TRANSCEND42studies showed no reduction in the primary end-point in patients with a previous stroke or those at high CV risk and intolerant to ACE-Is, despite significant lowering of the BP. This has prompted a re-evaluation of lower BP targets for patients at higher CV risk. Studies of a larger scale and longer timeframe may be required to demonstrate unequivocal benefit.
7.2.5 Combination treatment in hypertension
The pathophysiology of hypertension is multifactorial. Data show that the reductions in BP produced by monotherapy are too small to achieve recommended BP targets. The use of combination therapy is recommended, especially if the BP > 20/10 mmHg above goal and there are co-morbidities to consider such as diabetes, ISH, CKD or overt CVD. There is a revival of combination therapy owing to poor BP control in different countries and the need to achieve lower target BP levels.
Combination therapy may include free or fixed combination of a standard or lower dose. BP reduction with a low-dose combination of agents is comparable to that achieved with full-dose single agents, with a lower incidence of side-effects. FDC can be defined as 2 or more drugs in a single formulation, with independent modes of action, the combination of which is synergistic or complementary in effect. Free combinations can be defined as 2 or more drugs in separate formulations, usually taken at the same time. The requirement for a rational fixed-dose antihypertensive combination is that each component should be safe, efficacious, and have predictable pharmacokinetic and pharmacodynamic profiles, with a high troughpeak ratio and no excessive hypotensive episodes.
The benefits of combination therapy include an enhanced antihypertensive effect, better response rates and outcomes, and fewer adverse effects. Effective drug combinations include diuretics with β-blockers, diuretics with long-acting CCBs, ACE-Is or ARBs, and β- and α-blockers. Black patients respond best to diuretics or CCBs combined with other antihypertensive agents. Drug synergism exists with ACE-Is or ARBs combined with either CCBs or diuretics. Synergism is defined as a co-operative action resulting in a total effect that is greater than the sum of the effects taken individually; this may relate to BP and TOD.
7.3 Fixed drug combinations in hypertension and hyperlipidaemia
In many developing countries, the primary problem to be addressed with FDC therapy is not incomplete treatment, but rather the lack of treatment in patients unknown to the healthcare system. In this setting, the benefits of FDCs would be derived primarily from simplifying the process of patient identification and treatment provision.
There is limited empirical evidence to support or refute the main purported advantage of FDC, namely increased patient adherence.49Additional presumed advantages include: (i) a simpler dosage schedule which improves compliance and treatment outcomes; (ii) a reduction in inadvertent medication errors; (iii) reduced drug shortages, i.e. simplification of drug handling lowers the risk of being 'out of stock'; (iv) simplified drug procurement, management and handling; (v) lower production, packing and shipping costs; and (vi) reduced side-effects, if one of the combined drugs serves this purpose.50 The presumed disadvantages of FDCs include: (i) a greater expense than separate tablets, although not invariably so; (ii) cases where FDC therapy must be discontinued and replaced by separate tablets if a patient is allergic or has side-effects (this also applies with single-dose formulations); and (iii) irrational combinations may result from incompatible pharmacokinetics, i.e. different elimination half-lives of individual components.50
The pharmaceutical industry, academic and public health sectors advocate the expansion of secondary CVD prevention. A proposed FDC for established ischaemic heart disease is aspirin 75 mg, simvastatin 10 mg, lisinopril 10 mg and atenolol 25 mg daily, and for established ischaemic cerebrovascular disease a combination of aspirin 75 mg, lisinopril 10 mg, simvastatin 10 mg and hydrochlorothiazide 12.5 mg daily51. All of these drugs are available in generic formulation.
The polypill holds promise for the treatment of chronic disease.51-53Although the value of such a pill remains to be 'clearly demonstrated rather than simply assumed', a 3 - 4 drug polypill has been supported by the World Health Organization53,54and a global trial began in 2007. Only 20% of chronic diseases occur in high-income countries; 80% occur in low-and middle-income countries, with equal numbers of deaths in men and women.52High-cost physician models of care for chronic diseases are unsuitable in the latter. It has been shown that 20% of patients with coronary heart disease (CHD) in such countries were not receiving any aspirin, and patients requiring β-blockers - which are low-cost and widely available - were not receiving them.53 This has been one reason to introduce the polypill, and it will hopefully improve compliance; however, Reddy feels that the 'preventive polypill has much promise but insufficient evidence'.54
7.4 Antihypertensive drug interactions
A drug interaction is the quantitative modification of drug effect by the simultaneous or successive administration of a different drug. Antihypertensive agents have many drug interactions, some of which are highly significant; morbidity or mortality can result if steps are not taken to minimise this risk. Patients with hypertension frequently take multiple medications and are therefore at increased risk of drug interactions. There is also significant potential for drug interactions in the elderly, as they often receive larger number of drugs and show renal impairment in the excretion of several therapeutic agents.
Drug interactions may occur via pharmacokinetic (i.e. absorption, distribution, metabolism, and elimination) or pharmacodynamic means. The most frequent interactions related to clinical problems are pharmacokinetic; most stem from metabolism via the cytochrome P450 system or presystemic clearance by means of P-glycoprotein. Polymorphisms of the cytochrome P450 enzymes may explain the individual differences or the appearance of drug interactions. Certain drugs may impair the renal excretion of other agents, usually at the tubular level, e.g. drug plasma level and toxicity increases have been demonstrated with concomitant use of verapamil, amiodarone or quinidine.55 Of the statins, simvastatin is particularly prone to drug interactions, partly because it is extensively metabolised by the cytochrome CYP3A4 enzyme system.55 The SEARCH trial indicated that much of the increase in risk for myopathy noted in the high-dose simvastatin group was due to the concomitant use of medications such as amiodarone, diltiazem and amlodipine.56Judicious concomitant use of other medications with simvastatin will reduce the risk of myopathy (Table X).
There may be pharmacodynamic interactions between similarly acting drugs which may lead to additive or potentiation effects, e.g. the combination of intravenous verapamil and a β-blocker may cause additive impairment of a cardiac atrioventricular (AV) block. A combination of 2 or more antihypertensive agents may be expected to cause an additive BP-lowering effect. The antidepressant effects of all drugs that suppress the activity of the central nervous system enhance the side-effects of centrally acting antihypertensives (reserpine, α-methyldopa, guanfacine and clonidine). Recently, attention has been paid to the interaction of antihypertensives and non-steroidal anti-inflammatory drugs (NSAIDs).55 The ONTARGET study showed that telmisartan was as effective as ramipril, with marginally increased tolerability. The combination of the two was not superior to ramipril and had increased side-effects; the data suggest that the combination could precipitate acute renal failure and aggravate the onset of renal insufficiency.38 A summary of drug interactions in hypertension is shown in Table X.55
8. Management of severe hypertension
Severe hypertension (stage 3 DBP >110 mmHg and/or SBP >180 mmHg) may be classified into one of 3 categories to determine the urgency of treatment, namely asymptomatic severe hypertension, hypertensive urgency and hypertensive emergency. Patients should be managed or referred to the appropriate level of care in accordance with local protocols. Sustained severe hypertension requires immediate drug therapy and lifestyle modification.
8.1 Asymptomatic severe hypertension
These patients are asymptomatic but have severe hypertension with or without evidence of progressive TOD or ACC. The patient must be kept in the care setting and BP measurement must be repeated after 1 hour of rest. If the second measurement remains elevated at the same level, oral therapy should be started with a combination of 2 drugs, including a low-dose thiazide-like diuretic. The second drug is usually a dihydropyridine CCB. Follow-up should occur within a week, with escalation of treatment as required. Early referral is advised if BP is not controlled within 2 - 4 weeks.
8.2 Hypertensive urgencies and emergencies57
Despite advances in chronic hypertension management, hypertensive emergencies and urgencies remain serious complications. Factors for this include poor compliance with antihypertensive management, failure to institute effective antihypertensive therapy, failure to refer patients with resistant hypertension timeously, and failure to recognise important secondary causes.
Hypertensive emergencies and urgencies also occur in hypertension in pregnancy and in the preoperative period. Many classes of intravenous antihypertensive drugs are available to treat hypertensive emergencies, and specific agents may have an advantage in a given clinical situation. Orally active agents are used to treat hypertensive urgencies and include ACE-Is, CCBs, diuretics, α-β-blockers and β-blockers. Most patients respond well to drug therapy, but problems may arise if BP is normalised rapidly.
Hypertensive emergencies and urgencies are uncommon, but are likely to be encountered by all clinicians because of the high prevalence of chronic hypertension. All doctors must be familiar with treatment. Information is available from well-conducted studies on the outcomes of various antihypertensive drugs and BP-lowering strategies; therefore, any recommendation is based on case studies, clinical reports, comparative studies and expert opinion.
8.2.1 Hypertensive urgency58
Hypertensive urgency is symptomatic, usually with severe headache, shortness of breath and oedema. There are no immediate lifethreatening neurological, renal, eye or cardiac complications as in the case of hypertensive emergency (section 8.2.2). Ideally, all patients with hypertensive urgency should be treated in hospital. Treatment should be commenced with 2 oral agents with an aim to lower the DBP to 100 mmHg, slowly over 48 - 72 hours. Lowering of the BP can be achieved with the use of: (i) long-acting CCBs; (ii) ACE-Is used initially in very low doses and avoided if there is severe hyponatraemia (serum sodium <130 mmol/l indicates hyper-reninaemia and BP may fall dramatically with ACE-Is); (iii) β-blockers; and (iv) diuretics (which may potentiate the effects of the other classes of drugs). Furosemide should be used if there is renal insufficiency or evidence of pulmonary congestion.
8.2.2 Hypertensive emergency
A hypertensive emergency exists when acute elevation of BP is associated with acute and ongoing organ damage to the kidneys, brain, heart, eyes (grade 3 or 4 retinopathy) or vascular system. Such patients require rapid lowering of the BP to safe levels, within minutes or a few hours. Once a genuine hypertensive emergency is identified, immediate hospitalisation is essential, with monitoring in a modernfacility intensive care unit (ICU) with experienced staff. Standard care includes intravenous antihypertensive therapy tailored to the specific type of emergency (except in stroke patients - see 'Summary: management of acute stroke'). The potential threat of harm from overzealous lowering of BP exists together with the need for careful and structured BP reduction.
The definition of a hypertensive emergency does not explicitly include absolute BP levels, although most affected adults have a SBP >220 mmHg and/or DBP >130 mmHg. Hypertensive emergencies may also occur at modest BP elevations, e.g. in previously normotensive women during pregnancy (eclampsia) or in acute glomerulonephritis (especially in children). Hypertensive emergencies are uncommon, with an estimated occurrence of less than 1 - 2% of the hypertensive population; however, they are more common in black and older patients. Most patients know that they are hypertensive, and are receiving treatment.
Hypertensive emergencies and urgencies may be seen in the immediate postoperative period following vascular surgery. The initiating factors of hypertensive emergencies are poorly understood, but a rapid rise in BP associated with increased vascular resistance is suspected as the initial derangement. Smoking - possibly via associated endothelial injury - is suspected to be a risk factor: smokers have 5 times the risk of developing malignant hypertension. Hypertensive emergencies in patients with thrombotic (ischaemic) stroke and intracerebral haemorrhage should be managed according to the guideline of the Neurological Association of South Africa.59
Common clinical hypertensive emergencies are described in sections 8.2.2.1 - 8.2.2.4 below.
8.2.2.1 Acute cerebrovascular syndromes
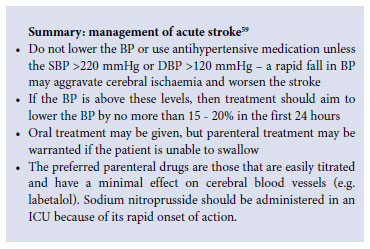
Severe hypertension is common in the case of acute stroke; it is debatable whether or not it should be treated, and if so, to which immediate target BP. In this setting, cerebral auto-regulation is impaired, and rapid BP reduction may result in an ischaemic stroke extension. The American Heart Association recommends the treatment of hypertension in the case of intracerebral bleeding when BP >180/105 mmHg, and the maintenance of mean arterial pressure (DBP plus one third of pulse pressure) above 130 mmHg. In the case of ischaemic stroke, BP should be observed for at least 1 - 2 hours to assess whether it will lessen spontaneously. Only a persistently elevated DBP >120 mmHg or SBP > 220 mmHg should be treated, with caution and an initial 20% reduction in mean arterial pressure.
Hypertensive encephalopathy is a cerebrovascular hypertensive emergency characterised by diffuse cerebral dysfunction with headache, nausea, vomiting, disturbed consciousness and - rarely - seizures. The condition is frequently accompanied by retinal findings of malignant hypertension and acute renal dysfunction. Computed tomography (CT) imaging usually appears normal with the condition, but may show diffuse cerebral oedema or posterior leuko-encephalopathy. The gradual lowering of BP generally leads to a fairly rapid improvement in symptoms. Sodium nitroprusside or labetalol are recommended for treatment. In patients failing to improve within 6 - 12 hours of BP reduction, an aggressive additional evaluation should be prompted for an alternative cause of the encephalopathy.
8.2.2.2 Acute cardiac syndromes
Severe hypertension in the setting of acute myocardial infarction, unstable angina, or pulmonary oedema should be treated aggressively and concurrently with all other indicated interventions. BP treatment reduces the ischaemic load on the left ventricle. Classic studies have shown that treatment with intravenous nitroglycerine is ideal in this setting, because it reduces myocardial oxygen consumption and increases blood flow beyond a stenosis. Sodium nitroprusside is also suitable and can be used alone or in combination with nitroglycerine.
8.2.2.3 Postoperative hypertension60
Postoperative hypertension is frequent (20 - 75%) and tends to be more prevalent in patients with poor pre-operative BP control, autonomic disorders, or a history of acute alcohol or cocaine use. Reversible causes of the hypertension may include pain, hypoxia, a full bladder, hyper- and hypovolaemia, persistent vomiting and anxiety. Patients with true emergencies should preferably be treated by an appropriate specialist, with admission of the patient to the ICU for parenteral drug therapy (Table XI) and monitoring. The BP should not be lowered by >25% within 30 - 120 minutes, with a goal of 160/100 mmHg within the following 2 - 6 hours. This may be achieved with the use of intravenous or oral drugs.

8.2.2.4 Resistant hypertension
Refractory or resistant hypertension remains >140/90 mmHg despite the use of 3 antihypertensive drugs in a rational full-dose combination with a diuretic component. An appropriate therapeutic plan must include lifestyle modification measures. In older patients with ISH, refractory hypertension is diagnosed when triple therapy (as above) has failed to control the BP below 160/90 mmHg. Table XII lists the causes of refractory hypertension in South Africa that must be considered in management of this condition.
The most common cause of resistant hypertension in South Africa is probably non-compliance (adherence) with lifestyle and medication; this includes the unavailability of medication and other drug-related causes. Unsuspected causes of secondary hypertension are less common, but bilateral renal disease and bilateral adrenal hyperplasia should be considered. These are suspected in patients with a reduced GFR and abnormal dipstick test results, or a positive ratio of plasma aldosterone to plasma renin. Once lifestyle and adherence to therapy have been managed satisfactorily, the addition of a fourth-line drug should be considered. Resistant hypertension should, where possible, be managed by specialist physicians. Fourthline therapy drugs are listed below and users should be conversant with their pharmacology:
direct vasodilators: hydralazine and minoxidil
centrally acting drugs: methyldopa and moxonidine
α-blockers: doxazosin
β-blockers: many cardio-selective agents are available
aldosterone antagonist: spironolactone61 and eplerenone.
9. Special considerations for hypertension in certain populations
9.1 Hypertension in blacks and Asians
Black persons are prone to complications such as stroke, heart failure and renal failure, while CHD - although emerging in frequency - is less common than in whites and Asians.62 Asians have a higher prevalence of diabetes mellitus and the metabolic syndrome compared with other racial groups.63
Compared with white patients, black patients respond poorly to antihypertensive monotherapy with ACE-Is and β-blockers, but do respond well to these agents in combination therapy with diuretics. Overall, CCBs show the most consistent response in black hypertensive patients compared with other classes of drugs used as monotherapy.64,65
9.2 Hypertension in children and adolescents66,67
Hypertension in children is an important issue beyond the scope of this guideline. Measurement of BP should be a routine part of paediatric examination, and the use of appropriate cuff size is essential. Hypertension in children is defined by a SBP and DBP greater than the 95th percentile according to age, sex and height (Table XIII). Hypertension is seldom primary in childhood; a detailed investigation should be made for an underlying secondary cause. Referral to a specialist for evaluation and treatment is essential.
In adolescents, hypertension is increasingly linked to obesity. Globally, poor diet and a lack of exercise in children is causing an epidemic of obesity, with an early onset of hypertension and Type 2 diabetes. Early recognition of this hypertension will be an important motivation for children and their parents to institute important lifestyle changes.
9.3 Hypertension in pregnancy
Hypertensive disease in pregnancy is the leading cause of direct maternal deaths in South Africa. Pre-eclampsia is a multi-organ disease unique to pregnancy, clinically evident by the presence of hypertension and proteinuria. In severe form, pre-eclampsia is the most common cause of maternal and perinatal morbidity and mortality.
9.3.1 Treatment
Antihypertensive treatment should be instituted when SBP >160 mmHg or DBP >110 mmHg. In the presence of other markers of potentially severe disease (e.g. thrombocytopenia, oliguria and/ or abnormal liver function), treatment should be initiated at lower degrees of hypertension. Diuretics and atenolol should generally be avoided, and ACE-Is and ARBs are contraindicated entirely. Suitable antihypertensive drugs to be used in pregnancy are:
• methyldopa (500 mg 6-hourly)
• nifedipine XL (30 - 60 mg daily)
• apresoline (25 - 50 mg 8-hourly)
• labetalol (100 - 200 mg twice per day, titrated to 600 mg if needed).
9.3.2 Hypertensive emergencies (impending eclampsia, eclampsia)
Very high BP should be lowered with an infusion of labetalol: 5 mg/ml at a rate of 4 ml/h via a syringe pump. The infusion rate should be doubled every 30 minutes to a maximum of 32 ml until the DBP has fallen and stabilised at an acceptable level (95 - 100 mmHg).
Labetalol can also be used as an intermittent bolus infusion - 50 mg (10 ml of labetalol 5 mg/ml) administered over at a period of at least 1 minute. This treatment should take effect within 5 minutes and should be repeated until the DBP is between 95 and 100 mmHg - to a maximum dose of 200 mg and provided that the pulse rate remains > 60 bpm. Nifedipine is an alternative drug administered via a 10 mg oral tablet (not a slow-release tablet); it should not be given sublingually, chewed, bitten or used buccally.
9.4 Hypertension in persons living with HIV/AIDS
Prolonged highly active antiretroviral therapy (HAART) is associated with a higher prevalence of systolic hypertension.68Individuals receiving HAART may be at increased risk of developing hypertension-related conditions, underscoring the importance of monitoring their BP. When antiretroviral drugs are used, the doses of CCBs are invariably influenced, especially at the start, termination or change of therapy. Frequent BP and dose checks are advised. Two of the 3 major classes of antiretroviral drugs - protease inhibitors (PIs) and non-nucleoside reverse transcriptase inhibitors (NNRTIs) - are involved in many drug interactions by inhibition or induction of the key hepatic enzyme system, cytochrome P450. CCBs are the major class of antihypertensives affected by such drug interactions.68-70
The first-line antiretroviral regimen is currently based on NNRTIs (efavirenz or nevirapine) which are enzyme inducers, thereby promoting the metabolism of all currently available CCBs - via a poorly understood mechanism - and potentially reducing their antihypertensive effect. If the first-line regimen fails, therapy is changed to a PI regimen, which decreases the rate of CCB metabolism to increase CCB blood levels with the risk of hypotension. Particular care must be taken in patients receiving first-line antiretroviral therapy where the BP is controlled with CCBs. If the antiretroviral therapy is switched to include a PI, a dramatic fall in BP may result; it is therefore better to avoid CCBs with concurrent antiretroviral therapy.70The metabolism of numerous β-blockers may be inhibited by PIs - a theoretical interaction of uncertain significance. It would be prudent to initiate a low-dose β-blocker treatment in patients receiving PIs.
9.5 Control of hypertension in diabetes
Diabetes is prevalent in South African Indians (15.8%)63 and whites (3.5%),71 and is becoming more common in blacks (4.8 - 6%).72 The illness has become the most common cause of end-stage renal disease (ESRD) because it is increasing in prevalence (particularly Type 2). Moreover, diabetic patients live longer at present, and are receiving treatment in ESRD programmes from which they were previously excluded.73 The onset and course of the disease can be ameliorated if measures are instituted early.
ESRD is the most common complication of Type 1 diabetes. A higher proportion of individuals with Type 2 diabetes are found to have microalbuminuria and overt nephropathy shortly after diagnosis because the disease had been present for many years prior. A correlation exists between the degree of albuminuria and CVD. The guideline was abbreviated and therefore does not meet the detailed information required for the prevention and treatment of diabetes with hypertension in the SA population.73
The earliest clinical evidence of nephropathy - i.e. incipient nephropathy - is microalbuminuria (3 - 30 mg/mmol on spot urine). Microalbuminuria rarely occurs with a short duration of Type 1 diabetes; therefore, screening is necessary after 5 years of disease course. Because of the difficulty in precise dating of the onset of Type 2 diabetes, screening should begin at diagnosis.73 The Diabetes Control and Complications Trial74 and the United Kingdom Prospective Study75have both shown that intensive therapy of blood sugar can significantly reduce the development of microalbuminuria and overt nephropathy in diabetic patients.
Hypertension in diabetes shows: (i) more ISH; (ii) more nondippers; (iii) abnormal BP regulation (variable with exercise and posture); (iv) a widened pulse pressure; (v) a correlation between SBP, heart failure and CV disease in Type 2 diabetes; and (vi) impaired sodium handling by the kidneys. In patients with Type 2 diabetes, hypertension is present in about one-third of patients at the time of diagnosis. Hypertension in diabetes is aggressive and progressive; it progresses rapidly to renal failure unless aggressively treated. The ACE-I, captopril, relieves albuminuria and prevents the progression of renal disease in Type 1 diabetes.76 Studies have shown that ACE-Is reduce the progression of microalbuminuria in Type 2 diabetes,77 and prevent the onset of microalbuminuria.
The combination of a third-generation dihydropyridine CCB (manidipine) and an ACE-I (delapril) failed to slow GFR but safely ameliorated CVD, retinopathy and neuropathy in the DEMAND study, involving 380 hypertensive Type 2 diabetic patients with albuminuria <200 mg/min. Notably, treatment was well tolerated, and the trial was double-blind, placebo-controlled and randomised to a 3-year follow-up.78Because of the high proportion of patients who progress from microalbuminuria to overt nephropathy in ESRD, the use of ACE-Is or ARBs is recommended for all patients with microalbuminuria or advanced stages of nephropathy.76, 77 The decision to use either an ACE-I or ARB in Type 2 diabetes should be left to the physician, with consideration of the affordability of therapy.
Patients with diabetic nephropathy are at very high CV risk, and there should be effective control of common CV risk. Patients should stop smoking, receive salicylates 80 - 150 mg daily (provided that the BP is controlled), and statins (and/or fibrates if indicated). In all stages of nephropathy, a BP <130/80 mmHg and blood HbA1c level<7% should be targeted. Albumin excretion should be monitored annually (albumin/creatinine ratio on spot urine). Treatment with an ACE-I or ARB is the preferred initial choice, combined with other antihypertensive drugs.
In more advanced nephropathy (CKD stages 4 and 5) the following should be observed: (i) glycaemic control must be modified due to risks of hypoglycaemia and metabolic acidosis related to metformin; (ii) BP <130/80 mmHg should be targeted, and furosemide twice daily should be considered; (iii) ACE-Is or ARBs should be continued (but with extreme caution in CKD stage 5), with awareness of the risks of hyperkalaemia and acute decline in renal function; and (iv) malnutrition should be avoided, but protein and phosphate restriction can be considered. Dialysis and renal transplantation should be considered in suitable cases with end-stage disease.
10. Primordial prevention
The main objective is to avoid or decrease the social, economic and cultural determinants that contribute to the development of hypertension. Primordial prevention relies on health policies that create a congenial environment in which healthy behaviour and population-wide education programmes are encouraged. In turn, policies rely on many factors, including political commitment, advocacy by health professionals, and involvement of community leaders and the mass media. Strategies are intended to prevent the acquisition or enhancement of CVD risk factors, particularly lifestyle and diet changes in black patients brought about by rapid urbanisation.80 The approach should be non-pharmacological, population-based and lifestyle-linked. Development of cost-effective methods for diagnosis and cost-saving measures for all risk factors of CVD is needed.
11. Prevention of hypertension
Prevention of hypertension is reliant upon the adoption of strict lifestyle measures. The prevalence of hypertension and CVD is increasing rapidly in Sub-Saharan Africa.80In a study of hypertension in Tanzania, under 20% of hypertensive subjects were aware of their diagnosis, approximately 10% reported receiving treatment, and less than 1% had controlled BP (<140/90 mmHg).81 Similarly, the treatment status for South African black males showed that 20% were aware of their hypertension, 14% were receiving treatment and only 7% had controlled BP. In females, 47% were aware of their hypertension, 29% were receiving treatment and only 15% had controlled BP.11
12. Patient education
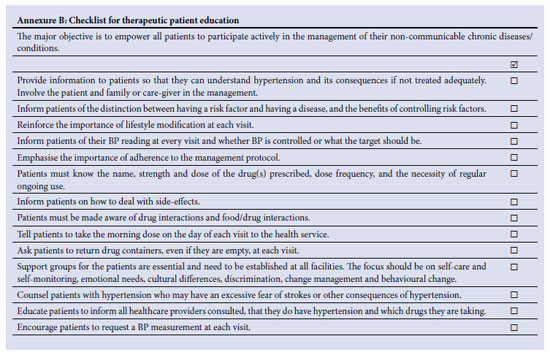
Hypertensive patients have the right to be informed about the status and progress of their condition. The main objective of patient education is to empower individuals to participate actively and ensure the quality of the management of their hypertension. Effective, honest and open two-way communication between the care provider and the patient is critical to the management of chronic life-long conditions. Acquisition of communication and counselling skills by health professionals is essential - preferably in the language of the target population. A checklist to guide the content of hypertensive patient education is provided in Annexure B. Poor adherence to therapy is the central cause of uncontrolled BP; obstacles to adherence are shown in Table XIV.
13. Ongoing management of the patient with hypertension
| • Dose titration or stepwise increase should be carried out after 2 months if the BP remains uncontrolled and adherence is a factor | |
| • Once a stable target BP has been achieved, follow-up BP measurement should be performed every 3 - 6 months | |
| • Drug dose should be reduced if the patient presents with symptoms of postural hypotension (i.e. dizziness or SBP fall >20 mmHg on standing) | |
| • Refer the patient from primary care to higher level care in the following: |
| • Young patients (18 - 30 years) | |
| • Pregnancy | |
| • Resistant hypertension (uncontrolled BP despite treatment with 3 drugs) | |
| • Treatment characteristics Patient and illness characteristics | |
| • Any patient with severe TOD and/or severe ACC (most patients with high added risk or very high added risk) | |
| • Hypertensive urgency or emergency. |
| • Most patients with low or moderate added risk can be managed at primary care level (general practitioner or clinic nurse) and assessed every 6 months. Patients with high or very high added risk with numerous risk factors should be managed by physicians or medical subspecialists (cardiologists, nephrologists and endocrinologists) and healthcare professionals with a special interest in hypertension; these patients may need frequent visits until the BP is controlled. |
14. Strategic implications for implementing this guideline
This section was developed by the Department of Health (DoH) and has been included as Annexure C. The SAHS endorses the strategy, which is mainly for policy-makers or those who administer healthcare facilities. Implementation of the guideline is an active process requiring more than dissemination and education; it requires the full collaboration and co-operation of policy makers, administrators and funders. The DoH and SAHS are committed to the full implementation of this guideline.
15. Disclaimer
This national clinical guideline is for educational and reference purposes only. It is not intended to be a substitute for the advice of the appropriate healthcare professional or for independent research and judgment. The SAHS accepts no responsibility or liability arising from any information contained within the guideline, or any error of omission from the protocol or from the use of any information contained within it.
References
1. Southern African Hypertension Society. Guidelines for the management of hypertension at primary health care level. S Afr Med J 1995;85:1321-1325. [ Links ]
2. Southern African Hypertension Society. Hypertension Clinical Guideline 2000. S Afr Med J 2001;91:163-172. [ Links ]
3. Milne PJ, Pinkney-Atkinson VJ. Southern African Society Hypertension Guideline Working Groups 2000 and 2003. S Afr Med J 2004;94:209-216. [ Links ]
4. Seedat YK, Croasdale MA, Milne FJ, et al. South African Hypertension Guideline 2006. S Afr Med J 2006;96:337-362. [ Links ]
5. Chobanian AV, Bakris GL, Black HR, et al. Seventh Report of the Joint National Conference on Prevention, Detection, Evaluation and Treatment of High Blood Pressure: The JNC 7 Report. JAMA 2003;289:2560-2572. [ Links ]
6. Harkam DG, Khan NA, Hemmelgarn BR, et al. The 2010 Canadian Hypertension Education Program recommendation for the management of hypertension: Part 2 - therapy. Can J Cardiol 2010;26:249-258. [ Links ]
7. Mancia G, Laurent S, Agahiti-Rosei, et al. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension task force document. J Hypertens 2009;27:2121-2158. [ Links ]
8. Whitworth JA. 2003 World Health Organization (WHO) / International Society of Hypertension (ISH) statement on management of hypertension. J Hypertens 2003;21:1983-1992. [ Links ]
9. Williams B, Poulter WB, Brown MJU, et al. Guidelines for management of hypertension: report of the fourth working party of the British Hypertension Society. J Hum Hypertens 2004;18:139-185. [ Links ]
10. National Clinical Guideline Centre. Hypertension: The clinical management of primary hypertension in adults. London: National Institute for Health and Clinical Excellence. http://www.nice.org.uk/nicemedia/live/12167/54727/54727.pdf (accessed 20 October 2011). [ Links ]
11. Steyn K, Gaziano T, Bradshaw D, et al. Hypertension in South African adults: results from the Demographic and Health Report 1998. J Hypertens 2001;19:1717-1725. [ Links ]
12. Bradshaw D, Groenewald P, Laubscher R, et al. Initial burden of disease estimates for South Africa, 2000. S Afr Med J 2003;93:682-688. [ Links ]
13. Kearney PM, Whelton M, Reynolds, et al. Global burden of hypertension: analysis of worldwide data. Lancet 2005;365:217-223. [ Links ]
14. World Health Organization. Integrated management of cardiovascular risk. Geneva: WHO, 2002. http://whqlibdoc.who.int/publications/9241562242.pdf (accessed 20 October 2011). [ Links ]
15. Pestana JAX, Steyn K, Leiman A, Hartzenberg GM. The direct and indirect costs of cardiovascular disease in South Africa. S Afr Med J 1996;86:679-684. [ Links ]
16. Dougherty J, McIntyre D, Bloom G. Value for money in South African health care: findings of a review of health expenditure and finance. Cent Afr J Med 1996;42:21-24. [ Links ]
17. World Health Organization. CVD - Risk Management Package for Low and Medium-Resource Settings. Geneva: WHO, 2002. http://www.who.int/cardiovascular_diseases/publications/cvd_package2002/en/index.html (accessed 20 October 2011). [ Links ]
18. O'Brien E, Asmar R, Beilin L, et al. Practice guidelines of the European Society of Hypertension for conventional, ambulatory and home blood pressure measurement. J Hypertens 2005;23:697-701. [ Links ]
19. Rosenthal T. Seasonal variations in blood pressure. Am J Geriatr Cardiol 2004;13:267-272. [ Links ]
20. O'Brien T. Replacing the mercury sphygmomanometer: requires clinicians to demand better automated devices. BMJ 2000;320:815-816. [ Links ]
21. Parati G, Kilama MO, Faini A, et al. Blood pressure measurement device. A new solar-powered blood pressure measuring device for low-resource settings. Hypertension 2010;56:1047-1056. [ Links ]
22. Bur A, Herkner H, Vlcek M, et al. Classification of BP levels by ambulatory BP in hypertension. Hypertension 2002;40:817-822. [ Links ]
23. Lovibond K, Jowett S, Barton P, et al. Cost-effectiveness of options for the diagnosis of high blood pressure in primary care: a modelling study. Lancet 2011;378:1219-1230. [ Links ]
24. Eckel RH, Alberti KG, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet 2010;375:181-183. [ Links ]
25. International Diabetes Federation (IDF). Consensus worldwide definition of the metabolic syndrome. Worldwide definition for use in clinical practice. Brussels: IDF. http://www.idf.org (accessed 20 October 2011). [ Links ]
26. Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009;120:1640-1645. [ Links ]
27. Kaplan NM. Editorial. The choice or thiazide diuretics. Why chlorthalidone may replace hydrochlorothiazide. Hypertension 2009;54:951-953. [ Links ]
28. Flack JM, Sica DA, Nesbitt S. Chlorthalidone versus hydrochlorothiazide as the preferred diuretic. Is there a verdict yet? Hypertension 2011;57:665-666. [ Links ]
29. Dorsch MP, Gillespie BW, Erickson ST, et al. Chlorthalidone reduced cardiovascular events compared with hydrochlorothiazide. A retrospective cohort analysis. Hypertension 2011;57:689-694. [ Links ]
30. Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med 1997;336:1117-1124. [ Links ]
31. Barnett AH, Bain SC, Bouter P, et al. Angiotensin receptor blockade versus converting - enzyme inhibition in Type 2 diabetes and nephropathy. N Engl J Med. 2004;351:1952-1961. [ Links ]
32. Pfeffer MA, McMurray JJ, Velazquez AJ, et al. Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med 2003;349(20):1893-1906. [ Links ]
33. Dahlöf B, Devereux RB, Kjeldsen SE, et al. Cardiovascular morbidity and mortality in the Losartan Intervention for Endpoint reduction in hypertension study (LIFE): a randomized trial against atenolol. Lancet 2002;359:995-1003. [ Links ]
34. Ruggenenti P, Fassi A, Ilieva AP, et al. Preventing microalbuminuria in type 2 diabetes. N Engl J Med 2004;351:1941-1951. [ Links ]
35. Parving HH, Lehnert H, Brochner-Mortensen J, et al. The effect of irbesartan in the development of diabetic nephropathy in patients with diabetic nephropathy. N Engl J Med 2001;345:870-878. [ Links ]
36. PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood- pressure lowering regimen among 6105 individuals with previous stroke or transient stroke. Lancet 2001; 358: 1033-1104. [ Links ]
37. Schrader J, Lüders S, Kulschewski A, et al. Morbidity and mortality after stroke eprosartan compared compared with nitrendipine for secondary prevention: principal results of a prospective randomized controlled study (MOSES). Stroke 2005;36(6):1218-1226. [ Links ]
38. Yusuf S, Teo KK, Pogue J, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med 2008; 358:1547-1559. [ Links ]
39. Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenburg NK. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 2008;358:2433-2446. [ Links ]
40. McMurray JJV, Pitt B, Latini R, et al. Effects of the oral direct renin inhibitor aliskiren in patients with symptomatic heart failure. Circ Heart Fail 2008;1:17-24. [ Links ]
41. Parving HH, Brenner BM, McMurray JJ, et al. Aliskiren trial in Type 2 diabetes using cardio-renal endpoints (ALTITUDE): rationale and study design. Nephrol Dial Transplant 2009;24:1661-1671. [ Links ]
42. Yusuf S, Teo K, Anderson C, et al. Effects of the angiotensin blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomized controlled trial. Lancet 2008;372(9644):1174-1183. [ Links ]
43. Jamerson K, Weber MA, Bakris GL, et al. Benazepril plus amlodopine or hydrochlothazide for hypertension in high-risk patients. N Engl J Med 2008;359:2417-2428. [ Links ]
44. Beckett NS, Peters R, Fletcher AE, et al. HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med 2008;358:2417-2428. [ Links ]
45. Yusuf S, Diener HC, Sacco RL, et al. Telmisartan to prevent recurrent strokes and cardiovascular events. N Engl J Med 2008;359:1225-1237. [ Links ]
46. Patel A, MacMahon S, Chalmers J, et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 2007;370:829-840. [ Links ]
47. Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood pressure control in type 2 diabetes mellitus. N Engl J Med 2010; 362:1575-1585. [ Links ]
48. Flack JM, Sica DA, Bakris G, et al. Management of high blood pressure in blacks: an update of high blood pressure in blacks consensus statement. Hypertension 2010;56:780-800. [ Links ]
49. Connor J, Rafter N, Rodgers A. Do fixed combination pills or unit-of-use packaging improve adherence? A systematic review. Bull World Health Organ 2004;82:935-939. [ Links ]
50. Kaplan W, Laing R. Fixed drug combinations as an innovative delivery mechanism. In: Priority Medicines for Europe and the World. A Public Health Approach to Innovation. Geneva: World Health Organization, 2004. [ Links ]
51. Wald NJ, Law MR. A strategy to reduce cardiovascular disease by more than 80%. BMJ 2003;326:1419. [ Links ]
52. Neal B, Chapman N, Patel A. Managing the global burden of cardiovascular disease. Eur Heart J 2002;4(F):F2-F6. [ Links ]
53. World Health Organization. The World Health Report 2002: Reducing Risks, Promoting Healthy Life. Geneva: World Health Organization, 2002. [ Links ]
54. Reddy KS. The preventive polypill-much promise, insufficient evidence. N Engl J Med 2007;356:212. [ Links ]
55. van Zwieten PA. Interactions between antihypertensive agents and other drugs. European Society of Hypertension Scientific Newsletter 2003;4(17):1-2. [ Links ]
56. Egan A, Colman E. Weighing the benefits of high dose simvastatin against the risk of myopathy. N Engl J Med 2011;365:285-287. [ Links ]
57. Herbert CJ, Vidt DG. Hypertensive crisis. Prim Care 2008;35:478-487. [ Links ]
58. Handler J. Hypertensive urgency. J Clin Hypertens 206;8:61-64. [ Links ]
59. South African Medical Association, Neurological Association of South Africa Stroke Working Group. Stroke therapy clinical guideline. S Afr Med J 2000;90:276-306. [ Links ]
60. Lindenauer PK, Pekow P, Wang K, et al. Perioperative beta blocker therapy and mortality after major noncardiac surgery. N Engl J Med 2007;2005:839-845. [ Links ]
61. Chapman N, Dobson J, Wilson S, et al. Effect of spironalactone on blood pressure in subjects with resistant hypertension. Hypertension 2007;49:839-845. [ Links ]
62. Opie LH, Seedat YK. Hypertension in sub-Saharan African populations. Circulation 2005;112:35623568. [ Links ]
63. Seedat YK. Diabetes mellitus in South African Indians. In: Mohan V, Rao GHD, eds. Type 2 Diabetes South Asians: Epidemiology, Risk Factors and Prevention. New Delhi: Jaypee Brothers, 2007;40-44. [ Links ]
64. Seedat YK. Varying responses to hypotensive agents in difference racial groups, black versus white differences. J Hypertens 1989;7:S15-S18. [ Links ]
65. Brewster LM, van Montfrans GA. Kleijnen systematic review: antihypertensive drug therapy in black patients. Ann Intern Med 2004;141:614-627. [ Links ]
66. Sinaiko AR. Hypertension in children. N Engl J Med 1996;335:1968-1973. [ Links ]
67. National Heart, Lung and Blood Institute. Task force on blood pressure control in children. Bethesda, Maryland: National Heart, Lung and Blood Institute, 1987;79:1-25. [ Links ]
68. Seaberg EC, Munoz A, Lu M, et al. Association between highly active anti-retroviral therapy in a large cohort of men followed from 1984 to 2003. AIDS 2005;19:953-960. [ Links ]
69. Glesby MJ, Aberg JA, Kendall MA, et al. Pharmacokinetic interactions between indinavir plus ritonavir and calcium channel blockers. Clin Pharmacol Ther 2005;78:143-153. [ Links ]
70. De Maat M, Ekhart GC, Huitema ADR, et al. Drug interactions between antiretroviral drugs and comedicated agents. Clin Pharmacokinet 2003;44:223-282. [ Links ]
71. Seedat YK, Mayet FGH, Gouws E. Risk factors for coronary heart disease in the white community of Durban. S Afr Med J 1994;84:257-262. [ Links ]
72. Mollentze WF, Moore AJ, Steyn AF, et al. Coronary heart disease risk factors in a rural and urban Orange Free State black population. S Afr Med J 1995;85:90-96. [ Links ]
73. Molitch ME, DeFronzo RA, Franz MJ, et al. Diabetic nephropathy. Diabetes Care 2003;26:S94-99. [ Links ]
74. Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Eng J Med 1993;329:977-998. [ Links ]
75. UK Prospective Diabetes Study Group. Intensive blood glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications with type 2 diabetes. Lancet 1998;352:837-853. [ Links ]
76. Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. The effect of angiotensin-converting enzyme inhibition on diabetic nephropathy. N Engl J Med 1993;329:1456-1462. [ Links ]
77. Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin- receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001;345:851-860. [ Links ]
78. Ruggenenti P, Lauria G, Iliev IP, et al. Effects of mandipine and delapril in hypertensive patients with type 2 diabetes mellitus: the delapril and mandipine for nephroprotection in diabetes (DEMAND) randomized clinical trial. Hypertension 2011;58:776-783. [ Links ]
79. Morar N, Seedat YK, Naidoo DV, Desai DK. Ambulatory blood pressure and risk factors for coronary heart disease in black and Indian medical students. J Cardiovasc Risk 1998;5:313-318. [ Links ]
80. Opie LH, Schall R. Old antihypertensives and new diabetes. J Hypertens 2004;22:1453-1458. [ Links ]
81. Edwards R, Unwin N, Mugusi F, et al. Hypertension prevalence and care in an urban and rural area of Tanzania. J Hypertens 2000;18:145-152. [ Links ]
Accepted 24 October 2011.
Corresponding author: Y K Seedat ((seedaty1@gmail.co)