Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.101 no.12 Pretoria dic. 2011
ORIGINAL ARTICLES
One-year post-primary antibody persistence and booster immune response to a DTaP-IPV//PRP~T vaccine (Pentaxim) given at 18 - 19 months of age in South African children primed at 6, 10 and 14 weeks of age with the same vaccine
Shabir Ahmed MadhiI; Clare CutlandII; Stephanie JonesIII; Michelle GroomeIV; Esteban OrtizV
IMD. Department of Science and Technology/National Research Foundation: Vaccine Preventable Diseases and Medical Research Council: Respiratory and Meningeal Pathogens Research Unit, University of the Witwatersrand, Johannesburg
IIMD. Department of Science and Technology/National Research Foundation: Vaccine Preventable Diseases and Medical Research Council: Respiratory and Meningeal Pathogens Research Unit, University of the Witwatersrand, Johannesburg
IIIMD. Department of Science and Technology/National Research Foundation: Vaccine Preventable Diseases and Medical Research Council: Respiratory and Meningeal Pathogens Research Unit, University of the Witwatersrand, Johannesburg
IVMD. Department of Science and Technology/National Research Foundation: Vaccine Preventable Diseases and Medical Research Council: Respiratory and Meningeal Pathogens Research Unit, University of the Witwatersrand, Johannesburg
VMD. Sanofi Pasteur, Lyon, France
ABSTRACT
OBJECTIVE: To assess the immunogenicity and safety of a pentavalent diphtheria, tetanus, acellular pertussis, inactivated poliovirus, Hib polysaccharide-conjugate vaccine booster.
DESIGN, SETTING AND PARTICIPANTS: A DTaP-IPV//PRP~T vaccine (Pentaxim, a Sanofi Pasteur AcXim family vaccine) was given to 182 healthy children in South Africa at 18 - 19 months of age following priming with the same vaccine plus a monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age.
OUTCOME MEASURES: Seroprotection (SP) and seroconversion (SC) rates, geometric mean titres (GMTs) and concentrations (GMCs) were assessed before, and 1 month after, the booster dose. Safety was assessed using parental reports.
RESULTS: One month after primary vaccination, at least 94.3% of participants were seroprotected against tetanus (>0.01 IU/ml), diphtheria (>0.01 IU/ml), poliovirus (>8 1/dil) and Haemophilus influenzae type b (Hib) infection (>0.15 µg/ml). Before the booster dose, the SP rates ranged from 65.7% to 100%. One month after the booster dose, SP rates were 97.7% for Hib (anti-PRP titre >1.0 µg/ml), 100.0% for diphtheria (>0.1 IU/ml) and 100% for tetanus (>0.1 IU/ml) and poliovirus types 1, 2, 3 (>8 1/dil). At least 95.7% of participants had fourfold post-booster increases in anti-pertussis antibody titres. GMTs increased from 11.21 to 465.51 EU/ml and from 12.89 to 520.35 EU/ml for anti-PT and anti-FHA respectively. Anti-PRP GMT increased from 0.35 to 47.01 µg/ml. The DTaPIPV// PRP~T vaccine booster was well tolerated, with fever >39.0ºC in only 1.7% of participants.
CONCLUSIONS: Antibody persistence following priming was satisfactory. The pentavalent DTaP-IPV//PRP~T vaccine booster was highly immunogenic and well tolerated.
Continuing high vaccination coverage has resulted in dramatic decreases in the global incidence of many childhood diseases. The Expanded Program on Immunization (EPI) originally included vaccines for tuberculosis, diphtheria, tetanus, pertussis, poliomyelitis and measles, but others including hepatitis B (HB) and Haemophilus influenzae type b (Hib) have since been added. In South Africa, HB and Hib conjugate vaccines were added to the national schedule in 1995 and 1999, respectively; conjugated pneumococcal and rotavirus vaccines were introduced in 2008 and inactivated poliovirus vaccine (IPV) was added in 2009. In response to increasing childhood vaccinations, combination vaccines, typically including acellular pertussis (aP), IPV, and others such as Hib and/or HB have been licensed over the last 10 years, and are now used in national immunisation programmes worldwide.1
Combined vaccines facilitate high coverage and reduce administration costs.2 The aP components included in many combined vaccines consist of purified Bordetella pertussis antigens that are better tolerated than the traditional whole-cell pertussis (wP) vaccines, and have satisfactory immunogenicity and protection.3 Based on their protective efficacy and improved safety and reactogenicity, the World Health Organization (WHO) recommends aP vaccines for primary vaccination, and a booster dose preferably during the second year of life.4
Sanofi Pasteur has developed a DTaP-IPV liquid combination vaccine used to reconstitute a lyophilised tetanus protein-conjugated Hib capsular polysaccharide vaccine (PRP~T) at the time of injection. The PRP~T vaccine is licensed as ActHib in South Africa and worldwide. The IPV vaccine is also licensed worldwide as Imovax Polio, and both are WHO pre-qualified.5 This DTaP-IPV//PRP~T combination vaccine, first licensed in 1997 as Pentaxim or Pentavac, is now licensed in over 100 countries. It was added to the EPI schedule (6, 10 and 14 weeks of age) in South Africa in April 2009. Its safety and immunogenicity have been assessed in numerous clinical studies.6 The approved indication is for a three-dose primary immunisation and a booster vaccination during the second year of life.
According to the WHO, all available aP-containing vaccines have demonstrated high levels of effectiveness in preventing pertussis, irrespective of antigen content.4 The effectiveness of the aP vaccine used in this study has been confirmed by national pertussis surveillance in Sweden7 and surveillance data are available from Austria and France.8,9
We reported the immunogenicity and safety of the DTaP-IPV// PRP~T combined vaccine given as a primary vaccination at 6, 10 and 14 weeks of age in South Africa.10 The present study assessed the immunogenicity and safety of the same vaccine given as a booster to the same group of children at 18 - 19 months of age.
Methods
Study design and participants
This Phase III, open clinical trial (clinicaltrials.gov ID NCT00254969) was conducted at the Chris Hani Baragwanath Hospital, Johannesburg, South Africa. The protocol was approved by the University of the Witwatersrand Human Research Ethics Committee (HREC) and the Medicines Control Council (MCC) and conducted in accordance with guidelines of the Declaration of Helsinki, the International Conference on Harmonization (ICH) and Good Practice in the Conduct of Clinical Trials in Human Participants in South Africa. Participants' parents or legal guardians gave written informed consent.
Healthy full-term (>37 weeks) infants of birthweight >2 500 g had previously been sequentially screened and enrolled during the first 24 hours of life to receive a primary series vaccination with the DTaP-IPV//PRP~T vaccine, plus a concomitant monovalent HB vaccine, at 6, 10 and 14 weeks of age. Infants whose mothers were known to be seropositive for HIV were excluded from the study before the primary series vaccination. Participants who had completed the primary series vaccination were eligible to receive a booster dose of the same vaccine at 18 - 19 months of age.
Vaccines
The composition of each 0.5 ml dose of the combined DTaP-IPV// PRP~T vaccine (Pentaxim, Sanofi Pasteur, France, batch number A2053) has been described elsewhere.6 The PRP~T was supplied as a freeze-dried powder and reconstituted with the DTaP-IPV vaccine liquid suspension at the time of injection. The vaccine was administered intramuscularly into the anterolateral aspect of the right upper thigh.
Safety and reactogenicity
The investigator monitored participants for immediate adverse reactions in the 30 minutes following each vaccine injection. Parents or legal representatives recorded time of onset, duration, and severity of solicited injection site (tenderness, redness, swelling) and systemic reactions (axillary temperature >37.4ºC, vomiting, abnormal crying, drowsiness, loss of appetite, irritability) for the following 7 days. Unsolicited adverse reactions were recorded from each vaccination until the following study visit. Serious adverse events (SAEs) were recorded throughout the study period. The investigators evaluated all AEs.
Serology
Blood samples were collected for antibody measurement just before and 1 month after the booster vaccination. Serological analyses were performed at Sanofi Pasteur's Global Clinical Immunology laboratory in the USA following completion of the booster vaccination. The methodologies are described in the primary series article.10
The pre-defined serological correlates of seroprotection (SP) were: anti-diphtheria and anti-tetanus >0.01 (proxy of short-term protection) and >0.1 IU/ml (proxy of long-term protection), antipoliovirus >8 reciprocal dilution (1/dil), anti-PRP >0.15 (proxy of short-term protection) and >1.0 µg/ml (proxy of long-term protection), and anti-hepatitis B surface antigen (HBsAg) >10 mIU/ ml. Seroconversion (SC) to anti-pertussis antigens was defined as a > fourfold increase in anti-PT or anti-FHA titre (EU/ml) from pre- to post-booster vaccination, since a correlation between antibody level and protection against pertussis is not currently well established.11
Statistical methods
The objectives were to determine the SP rates for diphtheria, tetanus, poliovirus and PRP before and 1 month after the booster, the SC rates for PT and FHA 1 month after the booster vaccination, and to describe the safety of the booster vaccination. The statistical analysis was descriptive; no hypothesis was tested. For the evaluation of immunogenicity, SP and SC rates were calculated with their 95% confidence intervals (CIs) by the exact binomial Clopper-Pearson method.12 Geometric mean antibody concentrations (GMCs - for diphtheria, tetanus, PT, FHA, and PRP) and titres (GMTs - for polio 1, 2, and 3) were calculated with 95% CIs using the normal approximation method.13 Pre- to post-booster GMC and GMT ratios (GMRs) were also calculated with 95% CIs. Reverse cumulative distribution curves (RCDCs) were constructed for each antibody response. For the evaluation of reactogenicity, the number and percentage of participants with solicited symptoms after the booster were calculated. Participants who received the booster dose and had at least one antibody titration were included in the immunogenicity analysis. All participants who received the booster were included in the safety analysis.
Results
Participant disposition
Of 207 participants who completed the primary vaccination series, 182 (87.9%) received the booster injection. A total of 179 participants completed the booster phase. Three participants withdrew because of non-compliance with the protocol, voluntary withdrawal, and loss to follow-up. The percentage of males (52.8%) was slightly higher than females (47.2%). The mean age at booster administration was 18.3 (standard deviation 0.4) months.
Immunogenicity
Tables I and II present the SP/SC rates, GMC/GMT for each vaccine antigen, with 95% CIs. At 1 month after primary vaccination (18 weeks of age), 94.3 - 100% of participants had been seroprotected against diphtheria and tetanus (titre >0.01 IU/ml), polio (titre >8 1/dil)), and Hib (anti-PRP >0.15 µg/ml); 4-fold increases in anti-PT and FHA antibody titres were observed in 98.2% and 83.4% of participants, respectively. Just before the booster dose at 18 - 19 months of age, at least 97.2% of the children still had protective levels of antibodies to tetanus, diphtheria, and poliovirus; 99.4% had anti-HBsAg antibody titres >10 mIU/ml. Seroprotective anti-PRP antibody concentrations were observed in 65.7% of participants. Following booster vaccination, SP rates for diphtheria and tetanus (>0.1 IU/ml) and poliovirus (>8 1/dil) were 100%; anti-PRP titres >1.0 µg/ml were observed in 97.7% of participants, and at least 95.7% of participants had seroconverted (>400% increase in antibody titre) against the PT and FHA antigens.
For anti-PT and anti-FHA, before the booster vaccination 86.0% of participants had anti-PT and 85.8% had anti-FHA antibody concentrations >5 EU/ml. GMCs increased strongly following booster administration, from 11.2 to 465.5 EU/ml for anti-PT and from 12.8 to 520.3 EU/ml for anti-FHA. Anti-PRP GMC increased from 0.35 to 47.0 mg/ml. Similar strong increases were observed for polio, diphtheria and tetanus (Table II).
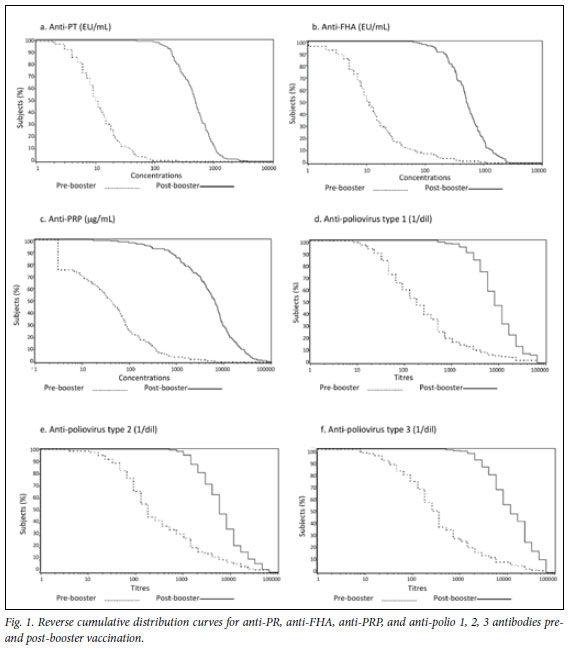
Geometric mean ratios were 39.4 for anti-PT and 40.7 for anti-FHA (Table II), and were also high for all other antigens. Reverse cumulative distribution curves showed large, linear increases in antibody titres from pre- to post-booster vaccination (presented for anti-PT, anti-FHA, anti-PRP, and anti-polioviruses in Fig. 1).
Reactogenicity and safety
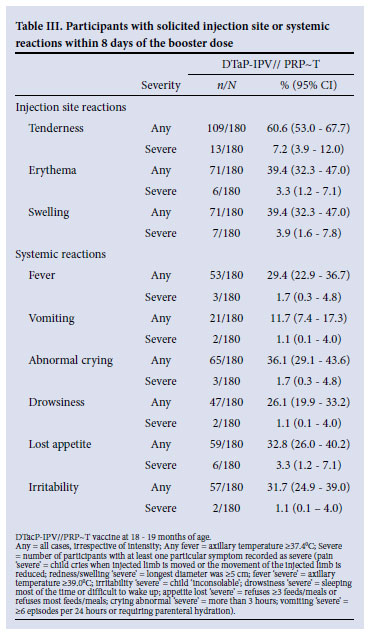
Most solicited reactions (Table III) occurred within 3 days of vaccination and resolved within the 8-day reporting period. The most frequent reported injection site reaction was tenderness (60.6%). Severe injection site reactions were reported by 3.3% (erythema) to 7.2% (tenderness) of participants. Abnormal crying, appetite loss and irritability were the most frequently reported solicited systemic reactions (31.7 - 36.1% of participants) followed by fever, drowsiness and vomiting (29.4 - 11.7%). Severe systemic reactions were reported by fewer than 2% of participants, except for lost appetite (3.3%).
A total of 64 (35.6%) participants experienced at least one unsolicited event within 8 days of the booster vaccination. Most were systemic events, upper respiratory tract infections in 23 participants and cough in 14 participants. Only one participant experienced an unsolicited mild injection site adverse event that was considered related to vaccination (injection site haemorrhage) that started on the day of injection, and resolved spontaneously in 3 days.
One SAE was reported: an episode of pulmonary tuberculosis 19 days after the booster injection that was considered by the investigator to be unrelated to vaccination. No hypotonic hyporesponsive episodes or seizures were reported. There was no case of litigation for any solicited reaction, unsolicited event, or SAE.
Discussion
This study evaluated the immunogenicity, reactogenicity and safety of a DTaP-IPV//PRP~T combination vaccine booster at 18 - 19 months of age. All participants had completed a primary series vaccination with the same vaccine at 6, 10 and 14 weeks of age concomitant with a monovalent HB vaccine.10 This combination vaccine has been evaluated in several other clinical studies.6 To obtain long-lasting immunity, booster doses are required, and our observed strong booster responses indicate long-lasting protection.6
Decreasing antibody levels between the primary series and booster administration has been previously observed with other DTaP- or DTwP-combined vaccines.6 In our study, immediately before the booster, at least 97.2% of children had protective levels of antibodies to tetanus, diphtheria, the 3 poliovirus types, and Hib (PRP). Anti-PRP antibody concentrations were also observed in the majority of children, although SP rates were lower than for other antigens. The strong booster response indicated satisfactory priming and a strong immune memory response and demonstrated that long-term protection can be expected. The persistence of a very high HB SP rate of 99.4% provides additional evidence for the compatibility of the monovalent HB vaccine that was co-administered with the pentavalent vaccine primary series.10
The anti-PRP concentration decreased in the interval between primary and booster vaccination. However, the immune memory and booster response induced by Hib conjugate vaccine were comparable to previous studies in children primed and then boosted in the second year of life with combinations containing the same DTaP and PRP~T vaccine antigens used in the study vaccine.6,14 The high vaccine effectiveness (VE) of DTaP-PRP~T combinations against Hib invasive disease has been demonstrated in Germany in an extensive, 5-year postmarketing surveillance study. The VE was 96.7% for the full 3-dose primary series, and 98.5% for those receiving a booster dose.15 Additional surveillance data from Sweden show high VE against invasive Hib disease, with incidences of 0.5 per 100 000 in 1997 and 0.16 per 100 000 in 2008.16
The high VE of Hib-conjugate vaccines has also been shown in South Africa17 and a number of other African countries that follow EPI schedules. In Kenya, Ghana, Uganda and Malawi there were significant decreases in the number of cases of invasive Hib disease in children under the age of 5 years, but especially in children under 1 year of age, following the introduction of the Hib vaccine at 6, 10 and 14 weeks of age.6 In The Gambia, which follows a similar 2-, 3- and 4-month schedule, no cases of Hib meningitis were detected 5 years after the introduction of Hib conjugate vaccine.18
Resurgence of invasive Hib disease among young children has occurred in countries where only a 3-dose primary series of Hib vaccine was administered, leading to the addition of a booster dose in the second year of life in various countries including South Africa.6
The waning of serum anti-pertussis antibodies following primary vaccination and the strong booster responses seen here are well documented.6,19 Today, most countries recommend pertussis booster doses in the second year of life, the timing of which differs between countries.3 The WHO recommends a pertussis booster dose for children, preferably during the second year of life, which is expected to ensure protection against pertussis for >6 years.4 National surveillance in Sweden following more than a decade of routine use demonstrated effective control of pertussis infection by DTaP-containing vaccines, including the study vaccine.6,7 These Swedish data show that vaccination at 3, 5 and 12 months of age resulted in a marked decrease in pertussis incidence compared with no vaccination, and that protection has remained high for 5 - 7 years after the third (booster) dose, at which time an additional booster dose is recommended. Indeed, the importance of the first booster has been shown by outbreaks of pertussis in children in countries when only primary vaccination has been given routinely.20 Although the schedule followed in South Africa is different, we believe that the available surveillance data are applicable, as high immunogenicity has been demonstrated in a range of vaccination schedules that have included a booster during the second year of life.6
Immunisation schedules for IPV vary widely among countries but generally include 2 or 3 doses in the first year of life and at least one booster dose 6 - 12 months after the last dose of the primary series. The persistence of anti-polio types 1, 2 and 3 antibody and strong IPV booster response observed in our study provide additional support for IPV administration in a 6-, 10- and 14-week EPI schedule with a booster at 18 - 19 months of age. According to the WHO, the possibility of exposure to either wild or vaccine-derived poliovirus is a risk in all parts of the world.21 Booster doses of IPV should be continued either until long-term antibody persistence has been demonstrated without additional doses after the second year of life, or until better worldwide control is achieved.22 Inclusion of IPV in a DTaP combination vaccine, as in South Africa, assures vaccination coverage as high as that for pertussis primary vaccination.
As expected for childhood combination vaccines, the incidence of common, solicited adverse reactions tended to be slightly higher for the booster dose compared with the primary vaccination.6 The overall reactogenicity of the study vaccine booster was satisfactory.
Booster vaccinations during the second year of life are recommended in many countries, including South Africa, to increase the duration of protection and reduce the burden of childhood infectious diseases. This study confirms that pre-booster antibody titres were satisfactory following an accelerated EPI priming schedule. The Pentaxim booster at 18 -19 months of age was well tolerated and induced high antibody responses to all the vaccine antigens, indicative of long-term protection against each of the five diseases.
Acknowledgements. The authors thank the participating clinicians and the infants and their parents who took part in this study. We would like to acknowledge Clement Weinberger (Le Stylo Communications) and Andrew Lane for assistance with manuscript preparation, Roy Fernando for data management, Valérie Bosch-Castells for the statistical analysis, and Catherine Bravo for study management. Andrew Lane, Roy Fernando, Valérie Bosch-Castells, and Catherine Bravo are employees of Sanofi Pasteur.
Clinicaltrials.gov ID NCT00254969.
The study was conducted with the financial support of Sanofi Pasteur, Lyon, France, and presented at the 26th International Pediatric Association Congress of Pediatrics, Johannesburg, 4 - 9 August 2010.
Conflict of interest. Esteban Ortiz is an employee of Sanofi Pasteur. Shabir Madhi, Clare Cutland, Stephanie Jones and Michelle Groome have acted as investigators for clinical trials sponsored by Sanofi Pasteur but have not received any direct payment from Sanofi Pasteur in this regard.
References
1. World Health Organization. WHO Vaccine Preventable Diseases Monitoring System: Country Profile Selection Center. http://apps.who.int/immunization_monitoring/en/globalsummary/countryprofileselect.cfm (accessed 17 August 2011). [ Links ]
2. Pichichero ME. New combination vaccines. Pediatr Clin North Am 2000;47(2):407-426. [ Links ]
3. Edwards K, Decker M. Pertussis vaccines. In: Plotkin SA, Orenstein WA, Offit PA, eds. Vaccines. 5th ed. Philadelphia: Saunders Elsevier, 2008:467-517. [ Links ]
4. World Health Organization. Pertussis position paper. Weekly Epidemiol Rec 2010;85(40):385-400. [ Links ]
5. United Nations pre-qualified vaccines: WHO list of vaccines for purchase by UN agencies. http://www.who.int/immunization_standards/vaccine_quality/PQ_vaccine_list_en/en/index.html (accessed 17 August 2011). [ Links ]
6. Plotkin S, Liese J, Madhi SA, Ortiz E. A DTaPIPV//PRP~T vaccine (PentaximTM): a review of 16 years' clinical experience. Expert Rev Vaccines 2011;10(7):981-1005. [ Links ]
7. Swedish Institute for Infectious Disease Control. Pertussis surveillance in Sweden. Appendix 2 to eleven-year report: person-time of follow-up and incidence calculations for Sanofi Pasteur vaccine specific cohorts. http://www.smittskyddsinstitutet.se/upload/Publikationer/11-y-report-app202-GSK.pdf (accessed 17 August 2011). [ Links ]
8. Bonmarin I, Levy-Bruhl D, Baron S, Guiso N, Njamkepo E, Caro V. Pertussis surveillance in French hospitals: results from a 10 year period. Euro Surveill 2007;12(1):34-38. [ Links ]
9. Rendi-Wagner P, Paulke-Korinek M, Stanek G, Khanakah G, Kollaritsch H. Impact of a pertussis booster vaccination program in adolescents and adults on the epidemiology of pertussis in Austria. Pediatr Infect Dis J 2007;26(9):806-810. [ Links ]
10. Madhi SA, Cutland C, Jones S, Groome, M, Ortiz E. Immunogenicity and safety of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (PentaximTM) and monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age in infants in South Africa. S Afr Med J 2011;101:126-131. [ Links ]
11. Plotkin SA. Correlates of protection induced by vaccination. Clin Vaccine Immunol 2010;17(7):1055-1065. [ Links ]
12 . Newcombe RG. Interval estimation for the difference between independent proportions: comparison of eleven methods. Stat Med 1998;17(8):873-890. [ Links ]
13. Newcombe RG. Two-sided confidence intervals for the single proportion: comparison of seven methods. Stat Med 1998;17(8):857-872. [ Links ]
14. Eskola J, Ward J, Dagan R, Goldblatt D, Zepp F, Siegrist CA. Combined vaccination of Haemophilus influenzae type b conjugate and diphtheria-tetanus-pertussis containing acellular pertussis. Lancet 1999;354(9195):2063-2068. [ Links ]
15. Kalies H, Verstraeten T, Grote V, et al. Four and one-half-year follow-up of the effectiveness of diphtheria-tetanus toxoids-acellular pertussis/Haemophilus influenzae type b and diphtheria-tetanus toxoids-acellular pertussis-inactivated poliovirus/H. influenzae type b combination vaccines in Germany. Pediatr Infect Dis J 2004;23(10):944-950. [ Links ]
16. Hallander HO, Lepp T, Ljungman M, Netterlid E, Andersson M. Do we need a booster of Hib vaccine after primary vaccination? A study on anti-Hib seroprevalence in Sweden 5 and 15 years after the introduction of universal Hib vaccination related to notifications of invasive disease. APMIS 2010;118(11):878-887. [ Links ]
17. Von Gottberg A, de Gouveia L, Madhi SA, et al. Impact of conjugate Haemophilus influenzae type b (Hib) vaccine introduction in South Africa. Bull World Health Organ 2006;84(10):811-818. [ Links ]
18. Adegbola RA, Secka O, Lahai G, et al. Elimination of Haemophilus influenzae type b (Hib) disease from The Gambia after the introduction of routine immunisation with a Hib conjugate vaccine: a prospective study. Lancet 2005;366(9480):144-150. [ Links ]
19. Grimprel E, Begue P, Anjak I, Njamkepo E, Francois P, Guiso N. Long-term human serum antibody responses after immunization with whole-cell pertussis vaccine in France. Clin Diagn Lab Immunol 1996;3(1):93-97. [ Links ]
20. Barret AS, Ryan A, Breslin A, et al. Pertussis outbreak in northwest Ireland, January - June 2010. Euro Surveill 2010;15(35):1-5. [ Links ]
21. World Health Organization. Polio vaccines and polio immunization in the pre-eradication era: WHO position paper. Wkly Epidemiol Rec 2010;85(23):213-228. [ Links ]
22. Rennels MB. Need for polio boosters after age two years. Vaccine 2009;27(2):179-180. [ Links ]
Accepted 23 August 2011.
Corresponding author: E Ortiz (esteban.ortiz@sanofipasteur.com)














