Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.101 n.7 Pretoria Jul. 2011
ORIGINAL ARTICLES
Associations between asthma and bronchial hyperresponsiveness with allergy and atopy phenotypes in urban black South African teenagers
M E LevinI; R MuloiwaIII; C MotalaII
IMB ChB, FCPaed (SA), MMed (Paed), Dip Allerg (SA), PhD. Division of Allergy, Department of Paediatrics and Child Health, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town
IIIMB ChB, FCPaed (SA), FACAAI, FAAAAI. Division of Allergy, Department of Paediatrics and Child Health, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town
IIIMB ChB, DCH (SA), FCPaed (SA), MSc (LSHTM). Ambulatory and Emergency Paediatrics, Department of Paediatrics & Child Health, University of Cape Town and Red Cross War Memorial Children's Hospital
ABSTRACT
OBJECTIVES: To determine asthma and allergy phenotypes in unselected urban black teenagers and to associate bronchial hyperresponsiveness (BHR) with asthma, other atopic diseases and allergen sensitisation.
METHODS: This was a cross-sectional study of 211 urban highschool black children of Xhosa ethnicity. Modified ISAAC questionnaires regarding asthma, eczema and rhinitis were administered. BHR was assessed by methacholine challenge using hand-held nebulisers. Skinprick tests (SPTs) were performed for 8 aeroallergens and 4 food allergens.
RESULTS: Asthma was reported in 9%, and 16% demonstrated BHR. Rhinitis was reported in 48% and eczema in 19%. Asthma was strongly associated with BHR. Asthma was associated with eczema whereas BHR was associated with rhinitis. SPTs were positive in 34% of subjects, aeroallergens in 32%, and food allergens in 5%. The most common sensitivities were to house dust mites (HDM) and German cockroach. BHR was associated with sensitivity to any aeroallergen, cat, HDM, cockroach and bermuda grass. The number of positive SPTs was associated with asthma and BHR. With each level of SPT positivity, there was 40% increased prevalence of asthma and 70% increased prevalence of BHR. The rate of allergen sensitisation in subjects with BHR (72%) was much higher than those without BHR (28%); house dust mite sensitivity was 69% in subjects with BHR and 18% in those without.
CONCLUSIONS: These are the highest rates of allergen sensitisation in subjects with BHR documented in an African setting and the widest difference in sensitisation rates between subjects with and without BHR.
Studies in South Africa in the 1970s reported very low rates of asthma in indigenous blacks,1 especially in rural settings.2 The subjects also demonstrated low rates of allergen sensitisation. Later studies showed increasing prevalence rates of asthma in urban and rural black communities3,4 and narrowing of the urban-rural gradient.5 An increased allergen sensitisation rate was also noted in rural blacks,6 with different patterns of sensitisation in those with or without bronchial hyper-responsiveness (BHR).5
There are few data on BHR rates in urban black children, associations between asthma and BHR, and the relationship between BHR, allergen sensitisation and other atopic diseases. We aimed to determine asthma and allergy phenotypes in unselected urban black teenagers and to associate BHR with asthma, other atopic diseases and allergen sensitisation.
Methods
A cross-sectional study of 211 urban black children (mean age 17 years) of Xhosa ethnicity attending a high school a few kilometres from central Cape Town, was undertaken during May - September 2005. Questionnaires administered in the preferred language (English or Xhosa) were modified from the International Study of Asthma and Allergies in Childhood (ISAAC) regarding asthma, eczema and rhinitis. Prevalence of asthma symptoms was assessed by determining the occurrence of wheeze in the preceding 12-month period rather than physician-diagnosed asthma.
In 206 subjects, BHR was assessed by a novel method (based on the Yan Protocol7) using hand-blown DeVilbiss number 45 glass nebulisers. Saline diluent was administered initially and thereafter methacholine in doubling dosages from 0.0015 mg to 0.5 mg until a cumulative dose of 1.1675 mg was attained. BHR was defined as a fall in FEV1 >20% at any dose below the maximal dose administered. The administration of methacholine by handheld nebulisers to assess BHR used in this study was compared with dosimeter in an unpublished subset of this cohort. There was a correlation between the handheld method and the dosimeter (Spearman correlation co-efficient r=0.0854, p<0.001) but there was both systematic and proportional bias precluding the development of a cutoff point with the handheld method that accurately corresponded with previously defined measures of BHR. Therefore the handheld method can be used as a gross screening test to accurately differentiate people with no BHR from those with some BHR, but not to delineate population prevalences of BHR according to the American Thoracic Society definition.
Skinprick tests (SPTs) were performed to house dust mites (Dermatophagoides pteronyssinus and Dermatophagoides farinae), grass mix, Bermuda grass, mould mix, cat, dog, German cockroach, egg-white, milk, peanut and potato (ALK-Abello). SPTs were deemed positive if weal size was 3 mm or more larger than the negative control.
Ethical approval was received from the Ethics Committee of the Health Sciences Faculty of the University of Cape Town, the Provincial Department of Education and the school where the research was conducted. All subjects completed informed consent statements.
Statistical analysis
Data were analysed using STATA statistical package (StataCorp, Version 11). Proportions were depicted as percentages for categorical variables. Strength of association between variables was tested using a chi-square or Fisher's exact test. Variables showing strong association were further analysed to determine the degree of effect. Prevalence ratios were deemed an appropriate measure of effect for a cross-sectional study. A Poisson regression model with a robust error variance was used to estimate prevalence ratios controlling for gender and age. A significance level of p<0.05 was used for all analyses.
Results
Descriptive data
Females comprised 60% (127) of the study population (211). The median age was 18 years (range 15 - 24 years). The prevalence of self-reported asthma was 9% (20). Of the 206 subjects who had a methacholine challenge, 16% (32) demonstrated BHR. Symptoms of rhinitis were reported in 48% (101) and symptoms of eczema in 19% (41).
SPTs were positive in 34% (72). The most common sensitivities were to house dust mites (HDM) (26%) and German cockroach (17%). SPTs were positive for aeroallergens in 32% (67) of the subjects; 14% (29) were monosensitive to aeroallergens and 18% (38) had 2 or more positive skin prick tests for aeroallergens (Table I). SPTs were positive for food allergens in only 5% (11) - most commonly to egg white.
Association of asthma with BHR
Eight (40%) subjects, who reported symptoms of asthma had a positive methacholine challenge; having symptoms of asthma was strongly associated with a higher probability of BHR (p=0.005).
Subjects (N=8/32) with BHR (25%) had a much higher prevalence of asthma than those who did not exhibit any BHR (N=12/174, 7%). After controlling for age and gender, the risk of asthma in subjects with BHR was 4 times that of subjects without BHR (prevalence ratio (PR) 4.1, 95% confidence interval (CI) 1.6 - 10.1, p=0.003).
Associations of asthma and BHR with other atopic diseases
Subjects with a history of eczema had a significantly higher prevalence (50%) of self-reported asthma compared with those who did not have eczema (16%, PR 4.2, 95% CI 1.8 - 9.6, p=0.001). The prevalence of asthma in those who had rhinitis was slightly higher but not statistically significant. However, the presence of rhinitis was associated with a statistically borderline higher prevalence (69%) of a positive BHR test compared with those without rhinitis (44%). The prevalence of BHR was increased in subjects with eczema but this association was not statistically significant (Table II).
Association of asthma with SPT
Self-reported asthma showed a strong association with positive SPT to specific aeroallergens (Table III). The prevalence of positive SPT to aeroallergens was higher in subjects with asthma. This association was significantly strong for HDM, cockroach and cat. The association increased with the number of aeroallergen sensitivities. With each level of increase in SPT positivity, there was a 40% increased prevalence (overall trend) of asthma v. no asthma. This effect was statistically significant (Table IV).
Association of BHR with skin prick tests
There was a strong association between a positive methacholine (positive BHR) challenge and sensitivity to specific aeroallergens. This association was significantly strong for HDM, cockroach, cat and Bermuda grass (Table III).
With each level of increase in SPT positivity, there was a 70% increased prevalence (overall trend) of BHR v. no BHR. This effect was statistically significant (Table IV). Sensitivity to food allergens showed a weak association with both self-reported asthma and BHR, with prevalence ratios of 2.3 (95% CI 0.3 - 16.4) and 1.8 (95% CI 0.4 -7.2) respectively.
Discussion
The study population was situated in Cape Town; all were black children, mostly living in peri-urban shanty-towns to the east of the city and commuting to a school located a few kilometres from central Cape Town. The home environment and school were not near areas of major industrial pollution. The population was not highly parasitised, and their total IgE levels were related to atopy predominantly and secondarily to ascaris infection.8 The limited sample size could preclude generalisation of the findings to other populations.
Self-reported asthma
Early data on asthma diagnosis in South Africa is scanty, although under-diagnosis appeared to be common. From 1963 to 1967, there was an admission rate of 0.02% (of all hospitalisations) in Durban for black children compared with 0.79% for white children.1 At Baragwanath Hospital, in the black township of Soweto, 3.8% of paediatric hospital admissions during 1992 and 1993 were for asthma,9 which constitutes an almost 200-fold increase in paediatric admissions for asthma compared with the 1969 study.1
In 1998, the International Study of Asthma and Allergies in Childhood (ISAAC)10 reported a 12-month prevalence of selfreported asthma symptoms in all ethnic groups in Cape Town ranging from 7.5% to 17% (video questionnaire v. written questionnaire). The prevalence of 'asthma ever' was 13.3% in this group of children. The ISAAC study was repeated in Phase III in 2005.11 Polokwane participated in this phase of the study, with a documented prevalence of asthma symptoms of 18% using a written questionnaire - similar to the 20.7% in Cape Town. Although ethnicity was not recorded in the ISAAC study, it included all groups and attempted to take a cross-section of the populations of Cape Town and Polokwane by purposive sampling of schools in different areas.
In 2004 -2005, a questionnaire study12 of only black children in Polokwane found an 11.2% prevalence of wheeze, and 5.7% prevalence of severe wheeze in 6 - 7-year-old children.
In the present study, the prevalence of self-reported asthma was 9% - similar to the 2005 Polokwane study but much lower than the ISAAC data. This apparent disparity may be partly explained by the different age groups, ISAAC being conducted on 13 - 14-yearold children, and our study on grade 11 pupils (median age 18 years); the Polokwane study used younger children. An alternate explanation is persistence of a different (lower) prevalence rate of asthma among black urban children in South Africa, reflected in the lower prevalence rates in urban studies of only black children when compared with the ethnically diverse urban population studied in ISAAC.
BHR
BHR has been extensively studied in both urban and rural black children (Table V). In 1976, the seminal study of Van Niekerk et al.2 reported a prevalence of asthma (positive exercise challenge tests) of 3.1% in peri-urban black children v. 0.14% in rural black children. Because the subjects were all of the same ethnic origin, this 20-fold difference was ascribed to environmental influences.
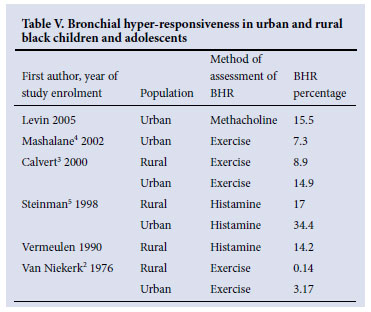
Using histamine challenge via hand-held nebulisers, Vermeulen found in 1990 a much higher prevalence of BHR (14.2%) among rural black children and adolescents (personal correspondence, unpublished data). Steinman's5 study of children in 1998 reported increased BHR (histamine challenge) in 17% of rural black children, in 34.4% of urban black children, and in 33% of urban white children. Calvert3 reported in 2000 a higher prevalence of exercise-induced bronchospasm in urban (14.9%) than in rural (8.9%) black children aged 8 - 13 years.
In 2002, a study in Thokoza (an exclusively black community in Gauteng) reported a 7.3% prevalence of exercise-induced bronchoconstriction (EIB) in 9 - 10-year-old children.4
Our prevalence of BHR (positive methacholine challenge) was lower than that reported by Steinman,5 similar to Calvert's study3 in black children that utilised exercise challenge, but higher than the study in Thokoza.4
Differences in the methods used to induce hyper-reactivity could account for some of the differences (histamine or methacholine challenge is likely to be more sensitive for asthma than exercise challenge, but less specific).
However, there is evidence that the prevalence of BHR and asthma has been increasing over time in the same ethnic group in urban and rural areas and that the urban-rural gradient is narrowing. This probably represents the effect of changes in both the urban and rural environments.
Association between BHR and asthma
Although there is a strong statistical association between BHR and asthma, there is limited overlap between both conditions. BHR is not a consistent feature in asthma,13,14 but the limited overlap (40%) in this study may call into question both diagnostic methods. The handheld methodology used to assess BHR limits it to a gross screening tool. In addition, there may be inaccuracies in the self-diagnosis of asthmatics by questionnaire in this population. Most studies find discrepancies between questionnaire-based self-diagnosis of asthma and other measures of asthma such as by video questionnaires,15-17 physician-diagnosed asthma and asthma control tests.18 Language differences and discordant definitions of medical terminology19 may lead to inaccuracies in self-diagnosed asthma and physician diagnosis of asthma in this setting.20
Association between BHR and asthma with other atopic diseases
The prevalence of symptoms of rhinitis (48%) is higher than that reported by the ethnically diverse 13 - 14-year-old children in both ISAAC I in 1995 (30.4%) and ISAAC III in 2002 (38.5%).21 The prevalence of symptoms of eczema (19%) is higher than that reported in ISAAC I (11.8%) and very similar to that in ISAAC III (19.4%).
In Wichmann's 2005 study12 of black children in Polokwane, the presence of eczema symptoms and rhinoconjunctivitis symptoms increased the likelihood of wheeze by 104% (odds ratio (OR) 2.04) and 226% (OR 3.26) respectively. Rhinoconjunctivitis was also a predictor of severe wheeze (OR 2.07).
In the present study, asthma was significantly associated with a higher prevalence of eczema (PR 4.2) whereas positive BHR was associated with a higher prevalence of rhinitis (PR 2.0).
Association between BHR and asthma with skin tests
Allergen sensitisation has been extensively studied in black children (Table VI). Van Niekerk et al. found no sensitisation to house dust mite (positive skin test) in rural black asthmatics, but a 65% sensitisation rate in urban black asthmatic children and teenagers. Potter et al.6 found no specific IgE to house dust mites in (unselected) rural black children. Vermeulen (written correspondence, unpublished data) found low sensitisation rates (positive SPT >2 mm) in rural unselected black children, which were not significantly different between those with BHR (6.2%) and those without (5.5%). SPTs were positive for HDM in 6.2% of both groups. However, there was a significant difference in allergen sensitisation by IgE testing in those with BHR (21.3%) compared with those without (14.4%) - predominantly owing to differences in grass and mould sensitisation.
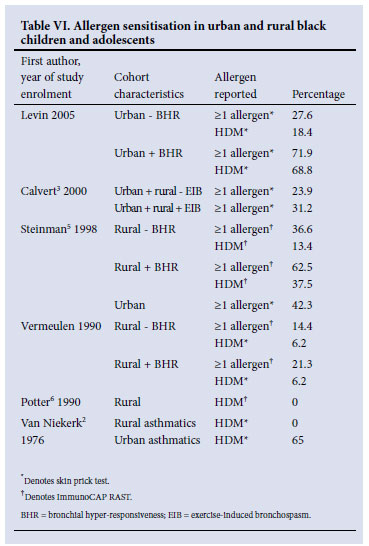
Of 455 black asthmatic children attending asthma clinics in Soweto22 (1991 - 1993), 69.8% had a positive skin test to one or more of a panel of 11 aeroallergens. Bermuda grass sensitivity (46.3%) was the most common allergen, followed by D. pteronyssinus (44.2%). However, the investigators used a cut-off of any reaction larger than the negative control as being a 'positive' skin test. High rates of other atopic diseases were also present, with 54.5% of children having a history of hay fever, 36.3% conjunctivitis, 18.7% eczema, and 15.5% food allergy.
Steinman et al.6 documented high levels of house dust mite sensitisation in rural and urban Xhosa children in 1998 and found no association between BHR and positive SPTs in urban black children (42.3%). However, in rural black children, allergen sensitivity (raised specific IgE) was found to be more common in those with BHR (62.5%) than those without BHR (36.6%). In addition, they found an association between BHR in rural black children and specific IgE to house dust mites (37.5% of those with BHR, 13.4% of those without) and grass pollen.
In Calvert's 2000 study,3 SPTs were positive in 31.2% of subjects with EIB compared with 23.9% of controls (p=0.02). The geometric mean weal size was significantly larger in those with EIB than those without EIB. Urban children were twice as likely to have a positive skin test result compared with rural children. The difference in EIB risk between urban and rural subjects was associated with atopy and affluence.18
This study showed a significant association between positive methacholine challenge and sensitivity to any aeroallergen (PR 1.7), cat (PR 7.2), HDM (PR 5.7), cockroach (PR 3.6), and Bermuda grass (PR 3.0). Allergen sensitivity (one or more positive skin prick tests) was far more common in those with BHR (72%) than those without BHR (28%). House dust mite and cockroach sensitivities were the dominant prevalent allergens, similar to inner city children in the USA.
The study confirms the increasing prevalence of allergic sensitisation in black teens in Cape Town and the difference in sensitisation rates between these and rural black subjects. It also demonstrates a gradient that appears to be increasing in allergic sensitisation rates between children with BHR and those without BHR. SPTs may be a useful investigation in assessing asthma and BHR in such subjects.
We gratefully acknowledge the assistance and contributions made by Jack Larsen (Phadia diagnostics) for skinprick test and Immuno CAP RAST reagents; Marilyn Koumbari (ACIC Ltd/Methapharm) for methacholine; Bartha Fenemore for performing total and specific IgE assays; and Anne Toerien for performing skin prick tests and methacholine challenges.
References
1. Wesley AG, Clyde JH, Wallace HL. Asthma in Durban children of three racial groups. S Afr Med J 1969;43(4):87-89. [ Links ]
2. Van Niekerk CH, Weinberg EG, De Shore SCV, Heese H, Van Schalkwyk DJ. Prevalence of asthma: A comparative study of urban and rural Xhosa children. Clin Allergy 1979:9:319-324. [ Links ]
3. Calvert J, Burney P. Effect of body mass on exercise induced bronchospasm and atopy in African children. J Allergy Clin Immunol 2005:116:773-779. [ Links ]
4. Mashalane MB, Stewart A, Feldman C, Becker P, de Charmoy S. Prevalence of exercise-induced bronchospasm in Thokoza schoolchildren. S Afr Med J 2006;96:67-70. [ Links ]
5. Steinman HA, Donson H, Kawalski M, Toerien A, Potter PC. Bronchial hyper-responsiveness and atopy in urban, peri-urban and rural South African children. Pediatr Allergy Immunol 2003;14(5):383-393. [ Links ]
6. Potter PC, Malherbe D, Lee S, Vermeulen J. Comparison of paediatric and adult Phadiotop screening assays in Xhosa and Caucasian Children. Clin Exp Allergy 1990:20(Suppl.):15. [ Links ]
7. Yan K, Salome C, Woolcock AJ. Rapid method for measurement of bronchial responsiveness. Thorax 1983;38(10):760-765. [ Links ]
8. Levin ME, Le Souëf PN, Motala C. Total IgE in urban Black South African teenagers: the influence of atopy and helminth infection. Pediatr Allergy Immunol 2008;19(5):449-454. [ Links ]
9. Luyt DK, Green RJ. Prevalence of asthma in Southern African children. Current Allergy and Clinical Immunology 1994;7:4-7. [ Links ]
10. ISAAC Steering Committee. Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis and atopic eczema: ISAAC. Lancet 1998;351:1225-1232. [ Links ]
11. Ait-Khaled N, Odhiambo J, Pearce N, et al. Prevalence of symptoms of asthma, rhinitis and eczema in 13-to 14-year old children in Africa: the International Study of Asthma and Allergies in Childhood Phase III. Allergy 2007;62:247-258. [ Links ]
12. Wichmann J, Wolvaardt JE, Maritz C, Voyi KV. Household conditions, eczema symptoms and rhinitis symptoms: relationship with wheeze and severe wheeze in children living in the Polokwane area, South Africa. Matern Child Health J 2009;13:107-118. [ Links ]
13. Tager IB, Weiss ST, Speizer FE. Occurrence of asthma, nonspecific bronchial hyperresponsiveness and atopy. Insights from cross-sectional epidemiologic studies. Chest 1987;91(6 Suppl):114S-119S. [ Links ]
14. Warner JO. Bronchial hyperresponsiveness, atopy, airway inflammation, and asthma. Pediatr Allergy Immunol 1998;9(2):56-60. [ Links ]
15. Crane J, Mallol J, Beasley R, Stewart A, Asher MI. Agreement between written and video questions for comparing asthma symptoms in ISAAC. Eur Respir J 2003;21:455-461. [ Links ]
16. Pizzichini MM, Rennie D, Senthilselvan A, Taylor B, Habbick BF, Sears MR. Limited agreement between written and video asthma symptom questionnaires. Pediatr Pulmonol 2000;30(4):307-312. [ Links ]
17. Pearce N, Aït-Khaled N, Beasley R, et al. Worldwide trends in the prevalence of asthma symptoms: phase III of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax 2007;62(9):758-766. [ Links ]
18. Khalili B, Boggs PB, Shi R, Bahna SL. Discrepancy between clinical asthma control assessment tools and fractional exhaled nitric oxide. Ann Allergy Asthma Immunol 2008;101(2):124-129. [ Links ]
19. Levin ME. Different use of medical terminology and culture-specific models of disease affecting communication between Xhosa-speaking patients and English-speaking doctors at a South African paediatric teaching hospital. S Afr Med J 2006;96(10):1080-1084. [ Links ]
20. Levin ME. Use of asthma terminology by Xhosa-speaking patients in South Africa - how it affects asthma-control questionnaires and questionnaire-based epidemiological studies. Current Allergy and Clinical Immunology 2007;20(2):74-78. [ Links ]
21. Zar HJ, Ehrlich RI, Workman L, Weinberg EG. The changing prevalence of asthma, allergic rhinitis and atopic eczema in African adolescents from 1995 to 2002. Pediatr Allergy Immunol 2007;18(7):560-565. [ Links ]
22. Luyt DK, Davis G, Dance M, Simmank K, Patel D. Clinical characteristics of black asthmatic children. S Afr Med J 1995;85:999-1001. [ Links ]
Accepted 7 March 2011.
Corresponding author: M Levin (michael.levin@uct.ac.za)














