Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.100 n.5 Pretoria May. 2010
GUIDELINE
South African guideline for the diagnosis, management and prevention of acute viral bronchiolitis in children
Robin J Green; Heather J Zar; Prakash M Jeena; Shabir A Madhi; Humphrey Lewis
ABSTRACT
ENDORSEMENT: South African Thoracic Society, South African Society of Paediatric Infectious Diseases, United South African Neonatal Association.
OBJECTIVE: To develop and publish a guideline for doctors managing acute viral bronchiolitis, because this condition is extremely common in South Africa, it is responsible for significant morbidity in the population, and subsequently a great deal of patient and parental distress, and the disease is costly, since many children are unnecessarily subjected to investigations and treatment strategies that are of no proven benefit. The main aims of the guideline are to promote an improved standard of treatment based on understanding of the disease and its management, and to encourage costeffective and appropriate management.
EVIDENCE: A detailed literature review was conducted and summarised into this document by a selected working group of paediatricians from around the country.
RECOMMENDATIONS: These include the appropriate diagnostic and management strategies for acute viral bronchiolitis.
Acute bronchiolitis is defined as viral-induced inflammation of bronchioles. The clinical manifestations occur as a consequence of airway inflammation and air trapping.
Bronchiolitis most commonly occurs as an acute illness in children younger than 2 years of age. The most frequent cause is rhinovirus or respiratory syncytial virus (RSV) infection, other respiratory viruses being less common (Table I). The illness is generally self-limiting. Patients often feed poorly and have mild upper respiratory tract infection (URTI) signs, low-grade fever, hyperinflation of the chest and wheezing. Symptoms of severe illness include tachypnoea and lower chest wall retractions.1 Bronchiolitis must be differentiated from aspiration of a foreign body, in which URTI signs are lacking.

The most reliable clinical feature of bronchiolitis is hyperinflation of the chest, evident by loss of cardiac dullness on percussion, an upper border of the liver pushed down to below the 6th intercostal space, and the presence of a 'Hoover sign' (subcostal recession, which occurs when a flattened diaphragm pulls laterally against the lower chest wall). Diffuse expiratory wheezing is common.
1. Causative organisms
The most common respiratory viruses associated with bronchiolitis are listed in Table I.
Many studies now show rhinovirus to be a cause of bronchiolitis, particularly mild bronchiolitis. Rhinovirus infection is identified in older children more frequently than RSV-associated bronchiolitis (median ages 13 v. 5 months, respectively) and is often associated with atopic disease (atopic dermatitis and blood eosinophilia).2 Oxygen saturation is generally not as low in children with rhinovirus-associated bronchiolitis as in children with RSV-associated bronchiolitis.2 Rhinovirus appears to be a perennial isolate in South African children.
RSV is the commonest cause of moderate or severe bronchiolitis. Two RSV subtypes occur, which are genotypically characterised by the nucleotide sequence of the virus glycoprotein (G-protein).3 The existence of distinct lineages within the subgroups has been demonstrated on both antigenic and nucleotide levels.4 South African subgroup A and B isolates cluster into four and five genotypes, respectively. Different genotypes co-circulate in every season. There is some evidence that group A RSV is associated with more severe disease.5
2. Seasonality of RSV infection
There are several factors responsible for an outbreak of RSV infection. These include geographical locations (latitude and altitude) and climatic factors (temperature, barometric pressure, relative humidity, vapour tension, hours of light, precipitation, dewpoint).
In most temperate regions, such as the USA and Europe, RSV outbreaks last an average of 3 - 4 months with a peak incidence during winter, although the exact timing of the onset of the outbreak is uncertain. In tropical regions such as The Gambia and Singapore, RSV outbreaks do not have distinct seasonality but often occur during the hottest rainy season. In South Africa, the seasonality of RSV outbreaks varies between the different regions and from year to year. In Cape Town, in one year, a biennial peak was seen, the first in the rainy season and the second between autumn and winter. In Gauteng the epidemic begins in late February and ends in August, while along the KwaZulu-Natal coast the pattern follows that of the tropics with a peak between February and March, during the rainy season. RSV may be identified throughout the year in HIV-infected children because of prolonged shedding of the virus (up to 100 days postinfection), compared with shedding of 5 - 7 days in HIV-uninfected children.6
3. Clinical manifestations and diagnosis
Bronchiolitis may be diagnosed on the basis of clinical signs and symptoms. The clinical pattern of wheezing and hyperinflation in a young child is diagnostic. The illness may have a seasonal tendency coinciding with the peak in RSV circulation. Chest radiographs and blood testing are generally unhelpful. Measurement of peripheral arterial oxygen saturation is useful to indicate the need for supplemental oxygen. A saturation less than 92% at sea level and 90% inland indicates that the child requires hospital admission for oxygen. Routine measurement of C-reactive protein does not aid in management.7
3.1 Features of severe disease
Bronchiolitis is usually mild but may cause more severe morbidity, including the need for hospitalisation, especially in children who have underlying risk factors for severe disease (Table II). In the USA, rates of hospitalisation of infants with bronchiolitis increased substantially during 1980 - 1996.8 However, it is an uncommon cause of mortality.9

Human metapneumovirus (hMPV) may produce severe bronchiolitis requiring intensive care unit (ICU) admission.10 Between 1994 and 1996, the average observed incidence of hospital admissions of children younger than 1 year living in Banjul, The Gambia, with acute lower respiratory tract infections (ALRTIs) was 5.27 per 100 child-years; the rate for RSV-associated severe respiratory illness was 0.87 per 100 child-years, and that for RSV-associated hypoxaemia 0.11 per 100 child-years.11 In The Gambia in 1994 - 1996, RSV accounted for 19% of ALRTI hospitalisations and had a mortality rate of 0 - 0.5%.11 In the ICU, RSV infection was associated with a mortality rate of 3 - 5%.11 Mortality from RSV LRTI in industrialising countries is higher than in industrialised countries. In part this may be because a disproportionately greater percentage of RSV LRTIs in industrialising countries are associated with pneumonia and concurrent bacterial co-infection rather than bronchiolitis.
Severe disease is present if a child is hypoxic, has severe respiratory distress (tachypnoea, nasal flaring or lower chest retractions), is unable to feed or play because of shortness of breath, or has apnoea. Bronchiolitis with bronchopneumonia or hypoxia may require admission or mechanical ventilation.
3.2 Bacterial co-infection - risk and antibiotic use
Uncomplicated bronchiolitis in immunocompetent children is usually a self-limiting illness, and secondary bacterial infection is uncommon. However, immunosuppressed children or those presenting with severe illness may have higher rates of bacterial co-infection.12-14 Risk factors for bacteraemia among children with RSV bronchiolitis include severe presentation, ICU admission, nosocomial acquisition of RSV or cyanotic congenital heart disease.12 Among children with RSV bronchiolitis admitted to an ICU, especially those who required mechanical ventilation, the risk of bacteraemia was almost 4-fold that in children who did not require ICU admission.12 Further support for the role of bacterial-viral coinfection in children who develop severe respiratory disease is provided by comparing rates of hospitalisation for severe viral pneumonia in children immunised with pneumococcal conjugate vaccine with those in unimmunised children. Among immunised children, the number of hospitalisations for viral pneumonia was reduced by approximately 30% compared with unimmunised children.15 HIV-infected children have a higher risk of bacteraemia than HIV-uninfected children.16 Moreover, bacterial-viral co-infection is more common in HIV-infected children hospitalised with LRTI than in immunocompetent children.17
There are very few randomised trials of the use of antibiotics in children with bronchiolitis.18,19 A Cochrane review found no evidence for the use of antibiotics in bronchiolitis, as antibiotics did not impact on the primary outcome (time to resolution of signs or symptoms) or on secondary outcomes (hospital admission, duration of hospitalisation, need for re-admission, complications or radiological findings).16 However, only 1 study18 of reasonable quality was identified that could be included in this review. In this study, infants were randomised either to ampicillin or placebo. Outcomes in 2 additional studies of poorer methodological quality have also shown no evidence for use of antibiotics in bronchiolitis.16,19
The available evidence suggests that antibiotics should not be used in uncomplicated bronchiolitis. For children with more severe disease, requiring ICU admission or with risk factors for severe disease, antibiotics should be considered.
4. Management
Management should include:
• treating the symptoms
• treating the underlying cause of the disease (infection and inflammation)
• preventing complications.
The following supportive treatments have been used for management of bronchiolitis (Table III):
• humidified oxygen
• bronchodilator therapy
• nebulised hypertonic (3%) saline
• anti-inflammatory therapy (corticosteroids - oral and nebulised)
• in-hospital use of antiviral treatments, e.g. ribavirin in ventilated children
• montelukast.
Humidified low-flow oxygen (0.5 - 3 litres/minute) applied by nasal prongs is effective for hypoxic children. Nasal prongs give a maximum inspired oxygen of 28 - 35% except in small infants, when higher oxygen concentrations may be obtained. Headbox oxygen is an alternative that is well tolerated by young infants. It requires no humidification, but high flow and a mixing device are needed to ensure that the correct oxygen concentration is delivered. However, there is wastage of oxygen and the delivered oxygen concentration (FiO2) is unpredictable. Facemask oxygen delivers between 28% and 65% oxygen at a flow rate of 6 - 10 l/min. In severely hypoxic infants who are not ventilated, oxygen should be administered using a polymask, which enables FiO2 concentrations of 60 - 80% to be achieved.
Oxygen should be weaned when the child improves clinically and with resolution of hypoxia.
Rapid short-acting bronchodilator therapy has shown modest benefits in the treatment of bronchiolitis.20 Bronchodilators may produce short-term improvements in clinical signs, but have not been shown to affect any important clinical outcome, such as obviating the need for ventilation or reducing inpatient stay.21 A Cochrane review (22 clinical trials with 1 428 infants) found that almost 60% of children treated with bronchodilators had a modest improvement in clinical score, but there was no impact on oxygenation, need for hospitalisation or duration of hospitalisation.21 For studies of adrenaline use, only 1 (of 5) demonstrated modest benefit compared with placebo and 1 (of 7) modest benefit compared with salbutamol.22 Furthermore, these improvements must be considered against the cost of the medications, and the need to handle and therefore distress the infant. Ipratropium bromide is ineffective in the treatment of bronchiolitis.23
In some children with severe disease a trial of a bronchodilator (adrenaline or β2-agonist by spacer with facemask (preferably) or by nebulisation) is acceptable.
However, if there is a lack of response (improvement in oxygen saturation or reduction in signs of respiratory distress) following three consecutive doses of bronchodilator, this form of therapy should cease. In children who respond, ongoing use may be appropriate until there is resolution of signs of severity. The optimal frequency, dosage and duration of bronchodilator therapy in these children have not been established.
Hypertonic saline given as nebulised 3% or 5% saline may significantly reduce the length of hospital stay and improve the clinical severity score in infants with acute viral bronchiolitis.24 However, data on dose and frequency are inconsistent. Some studies use 2 ml, while most use 4 ml delivered with or without bronchodilator agents.24 Dosing frequency ranges from 2- to 6-hourly.
Oral corticosteroids have little or no effect on the clinical outcome of bronchiolitis and are therefore not routinely recommended.25-27 A recent study reported that prednisolone (2 mg/kg per day in 3 divided doses for 3 days) may have some benefit in rhinovirus-induced bronchiolitis.28 Prednisone decreases the rate of relapses (22% v. 57%) (odds ratio (OR) 19.06, confidence interval (CI) 2.52 - 144.03).28 However, rhinovirus bronchiolitis is usually mild. In contrast, prednisone did not appear to reduce the severity or duration of illness in children with RSV-associated bronchiolitis. Studies have also shown no evidence of benefit for injectable systemic steroids.29,30
Two randomised controlled trials showed no evidence of benefit for inhaled corticosteroids (ICS) in acute bronchiolitis.31,32 Routine use of systemic or inhaled steroids in the management of bronchiolitis is therefore not indicated.
Furthermore, there is no evidence that use of ICS reduces wheezing after bronchiolitis.33,34 The use of ICS during or after bronchiolitis is therefore not indicated.
Montelukast has no effect on the clinical course of bronchiolitis or its outcome. A study of montelukast (4 mg daily until discharge) found no improvement in the clinical course of the disease. Montelukast also had no significant effect on the T-helper 2/T-helper 1 cytokine ratio when given in the early acute phase.35 In a study of post-bronchiolitis wheeze, montelukast did not improve respiratory symptoms of post-RSV bronchiolitis in children.36
Similarly, ribavirin has not been shown to have any consistent effect in the management of bronchiolitis.37
The use of chest physiotherapy has not been shown to change the course of bronchiolitis or its outcome. Chest physiotherapy using vibration and percussion techniques does not reduce length of hospital stay or oxygen requirements or improve the severity clinical score in infants with acute bronchiolitis.38
4.1 Indications for hospitalisation
The majority of infants with bronchiolitis do not require hospitalisation, but certain children are at risk of severe disease or require oxygen39-41 (Table IV).
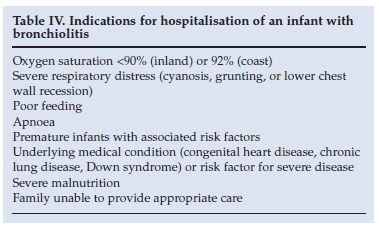
There is no evidence that clinical scoring systems perform better than clinical judgement in deciding who needs admission to hospital.42 Infants with bronchiolitis may deteriorate for 2 - 3 days after the onset of symptoms before symptoms and signs start to resolve.
4.2. Indications for ICU admission
The following are indications for ICU admission:
• failure to maintain an oxygen saturation of >90% on an inspired oxygen concentration of >70% (i.e. on a polymask), or if the arterial partial pressure of oxygen to fractional inspired oxygen (PaO2/FiO2) ratio is <100 (normal 350) if arterial blood gas measurements are available
• apnoea
• hypercarbia with resulting acidaemia (pH <7.25)
• exhaustion, which may be difficult to judge but should be considered if a high respiratory rate or severe chest wall indrawing persists.
4.3 Management of severe disease
Infants with bronchiolitis who require paediatric ICU admission may have co-infection with bacteria, and antibiotics should be considered. In addition, ventilation should be with minimum positive end-expiratory pressure (PEEP), low tidal auto-PEEP occurs.
4.4 Infection control in hospital
RSV is spread predominantly by droplet spread and direct inoculation. Hospital-acquired outbreaks of bronchiolitis occur and must be prevented. All reasonable barrier and isolation procedures must be adopted by hospital staff when caring for sick children. Careful hand-washing between patients is the most important infection control measure and the mainstay of prevention. Children hospitalised with RSV should be cohorted with other RSV-infected children or with patients who are not at risk for severe disease.
5. Prevention of RSV in high-risk children
Specific RSV monoclonal antibody, palivizumab, is available for children at risk of severe bronchiolitis. In a randomised, double-blind, placebo-controlled trial including 1 287 children with congenital heart disease (CHD) randomly assigned to receive 5-monthly intramuscular injections of 15 mg/kg palivizumab or placebo, palivizumab recipients had a 45% relative reduction in RSV hospitalisations (p=0.003), a 56% reduction in total days of RSV hospitalisation per 100 children (p=0.003) and a 73% reduction in total RSV hospital days requiring supplemental oxygen per 100 children (p=0.014).43 In the IMpact-RSV Study, significantly lower RSV hospitalisation rates were observed among premature infants who received palivizumab than among those who received placebo in each of the subgroups. The highest reduction in hospitalisation rate was seen in infants of gestational age 32 - 35 weeks, where RSV prophylaxis with palivizumab reduced hospitalisations by 80% (p=0.002).44 The efficacy of palivizumab was a 47% reduction in hospitalisations in infants with a gestational age <32 weeks.44 Palivizumab is therefore indicated in premature infants or young children with an underlying risk factor for severe disease (Table V). It should be remembered that in South Africa, because of resource constraints, these recommendations may need to be subject to cost-efficacy studies.

The recommended dose of palivizumab is 15 mg/kg body weight per dose, administered intramuscularly monthly. There are two dosing vials available - 15 mg and 30 mg. Since the seasonality of RSV in South Africa varies by province, it is recommended that prophylaxis be given for 5 months, beginning in December in KwaZulu-Natal and in February for the rest of the country.
5.1 Recurrent viral-induced wheeze
RSV-associated bronchiolitis is associated with an increased risk of recurrent wheezing episodes in as many as 43% of children.45 To date, there are no effective treatment strategies that reduce this risk. The reader is referred to the latest South African Childhood Asthma Working Group (SACAWG) asthma guideline for management of recurrent wheeze.46
6. Patient education
Education of parents and caregivers is a vital part of the successful management of bronchiolitis. Anxiety will compromise attempts to limit unnecessary medication use. The following principles regarding bronchiolitis should be explained to parents or caregivers:
• Generally the condition is not serious.
• Most cases do not require medication.
• Antibiotics are of no value.
• Parents must monitor respiratory symptoms and report concerns to their doctor.
• Symptoms may continue for weeks following the acute illness.
A useful patient education brochure may be found at: http://en.wikipedia.org/wiki/Bronchiolitis
Conflict of interest statements:
Robin J Green: Advisory Board: MSD, Wyeth, Abbott; Speakers
Bureau: MSD, Abbott, GSK, AstraZeneca
Heather J Zar: No conflict of interest
Prakash M Jeena: Speakers Bureau: MSD, Abbott
Shabir A Madhi: Advisory Board: Wyeth; Speakers Bureau: Abbott, Wyeth
Humphrey Lewis: Speakers Bureau: GSK
References
1. Wohl MEB. Bronchiolitis. In: Chernick V, Boat TF, Wilmot RW, Bush A, eds. Kendig's Disorders of the Respiratory Tract in Children. Philadelphia: Saunders, Elsevier, 2006: 423-432. [ Links ]
2. Matti K, Kotaniemi-Syrjänen A, Waris M, Vainionpää R, Reijonen TM. Rhinovirus-associated wheezing in infancy: comparison with respiratory syncytial virus bronchiolitis. Pediatr Infect Dis J 2004; 23: 995-999. [ Links ]
3. Madhi SA, Venter M, Alexandra R, et al. Respiratory syncytial virus associated illness in highrisk children and national characterisation of the circulating virus genotype in South Africa. J Clin Virol 2003; 27: 180-189. [ Links ]
4. Venter M, Collinson M, Schoub BD. Molecular epidemiological analysis of community circulating respiratory syncytial virus in rural South Africa: comparison of viruses and genotypes responsible for different disease manifestations. J Med Virol 2002; 68: 452-461. [ Links ]
5. Hall CB, Walsh EE, Schnabel KC, et al. Occurrence of groups A and B of respiratory syncytial virus over 15 years: Associated epidemiologic and clinical characteristics in hospitalized and ambulatory children. J Infect Dis 1990; 162: 1283-1290. [ Links ]
6. King JC, Burke AR, Clemens JD, et al. Respiratory syncytial virus illnesses in human immunodeficiency virus- and noninfected children. Pediatr Infect Dis J 1993; 12: 733-739. [ Links ]
7. Moodley T, Masekela R, Kitchin O, Risenga S, Green RJ. Acute viral bronchiolitis. Aetiology and treatment implications in a population that may be HIV co-infected. S Afr J Epidemiol Infect 2010 (in press). [ Links ]
8. Shay DK, Holman RC, Newman RD, Liu LL, Stout JW, Anderson LJ. Bronchiolitis-associated hospitalizations among US children, 1980 - 1996. JAMA 1999; 282: 1440-1446. [ Links ]
9. Shay DK, Holman RC, Roosevelt GE, Clarke MJ, Anderson LJ. Bronchiolitis-associated mortality and estimates of respiratory syncytial virus-associated deaths among US children, 1979-1997. J Infect Dis 2001; 183: 16-22. [ Links ]
10. Morrow BM, Hatherill M, Smuts HE, Yeats J, Pitcher R, Argent AC. Clinical course of hospitalised children infected with human metapneumovirus and respiratory syncytial virus. J Paediatr Child Health 2006; 42: 174-178. [ Links ]
11. Weber MW, Milligan P, Sanneh M, et al. An epidemiological study of RSV infection in the Gambia. Bull World Health Organ 2002; 80(7): 562-568. [ Links ]
12. Bloomfield P, Dalton D, Karleka A, Kesson A, Duncan G, Isaacs D. Bacteraemia and antibiotic use in respiratory syncytial virus infections. Arch Dis Child 2004; 89: 363-367. [ Links ]
13. Kyneber M, van Oud-Alblas H, van Vliet M, Uiterwaal C, Kimpen J, van Vught A. Concurrent bacterial infection and prolonged mechanical ventilation in infants with respiratory syncytial virus lower respiratory tract disease. Intensive Care Med 2005; 31: 680-685. [ Links ]
14. Thorburn K, Harigopal S, Reddy V, Taylor N, van Saene HKF. High incidence of pulmonary bacterial co-infection in children with severe respiratory syncytial virus (RSV) bronchiolitis. Thorax 2006; 61: 611-615. [ Links ]
15. Madhi SA, Klugman KP, The Vaccine Trialist Group. A role for Streptococcus pneumoniae in virus-associated pneumonia. Nature Med 2004; 10: 811-813. [ Links ]
16. Spurling GKP, Fonseka K, Doust J, Del Mar C. Antibiotics for bronchiolitis in children. Cochrane Library 2007; 1: CD005189. [ Links ]
17. Madhi SA, Venter M, Madhi A, Petersen K, Klugman KP. Differing manifestations of respiratory syncytial virus-associated severe lower respiratory tract infections in human immunodeficiency virus type 1-infected and uninfected children. Pediatr Infect Dis J 2001; 20: 164-170. [ Links ]
18. Field CMB, Connolly JH, Slattery CM, Turkington EE. Antibiotic treatment of epidemic bronchiolitis - a double-blind trial. BMJ 1966; 1: 83-85. [ Links ]
19. Friis B, Andersen P, Brenøe E, et al. Antibiotic treatment of pneumonia and bronchiolitis. A prospective randomised study. Arch Dis Child 1984; 59: 1038-1045. [ Links ]
20. Kellner JD, Ohlsson A, Gadomski AM, Wang EEL. Efficacy of bronchodilator therapy in bronchiolitis. A meta-analysis. Arch Pediatr Adolesc Med 1996; 150: 1166-1172. [ Links ]
21. Gadomski AM, Bhasale AL. Bronchodilators for bronchiolitis. Cochrane Database Syst Rev 2006; 3: CD001262. [ Links ]
22. Hartling L, Wiebe N, Russell K, Patel H, Klassen TP. Epinephrine for bronchiolitis. Cochrane Datbase Syst Rev 2004; 1: CD003123. [ Links ]
23. Henry RL, Milner AD, Stokes GM. Ineffectiveness of ipratropium bromide in acute bronchiolitis. Arch Dis Child 1983; 58: 925-926. [ Links ]
24. Zhang L, Mendoza-Sassi RA, Wainwright C, Klassen TP. Nebulized hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst Rev 2008; 4: CD006458. [ Links ]
25. Garrison MM, Christakis DA, Harvey E, Cummings P, Davis RL. Systemic corticosteroids in infant bronchiolitis: A meta-analysis. Pediatrics 2000; 105(4): 1-6. [ Links ]
26. Roosevelt G, Sheenan K, Grupp-Phelan J, et al. Dexamethasone in bronchiolitis: a controlled trial. Lancet 1996; 348: 292-295. [ Links ]
27. Kimpen JLL. Prevention and treatment of respiratory syncytial virus bronchiolitis and postbronchiolitic wheezing. Respir Res 2002; 3: Suppl 1, S40-S45. [ Links ]
28. Jartti T, Pasi L, Timo V, et al. Evaluation of the efficacy of prednisolone in early wheezing induced by rhinovirus or RSV. Pediatr Infect Dis J 2006; 25: 482-488. [ Links ]
29. Teereatakulpisarn J, Limwattananon C, Tanupattarachai S, Limwattananon S, Teereatakulpisarn S, Kosalaraksa P. Efficacy of dexamethasone injection for acute bronchiolitis in hospitalized children: a randomized, double-blind, placebo-controlled trial. Pediatr Pulmonol 2007; 42: 433-439. [ Links ]
30. Corneli HM, Zorc JJ, Mahajan P, et al. A multicentre, randomized, controlled trial of dexamthasone for bronchiolitis. N Engl J Med 2007; 357: 331-339. [ Links ]
31. Cade A, Brownlee KG, Conway SP, et al. Randomised placebo controlled trial of nebulised corticosteroids in acute respiratory syncitial viral bronchiolitis. Arch Dis Child 2000; 82: 126-130. [ Links ]
32. Wong JY, Moon S, Beardsmore C, O'Callaghan C, Simpson H. No objective benefits from steroids inhaled via a spacer in infants recovering from bronchiolitis. Eur Respir J 2000; 15: 388-394. [ Links ]
33. Fox GF, Everard ML, Marsh MJ, Milner AD. Randomised controlled trial of budesonide for the prevention of post-bronchiolitis wheezing. Arch Dis Child 1999; 80: 343-347. [ Links ]
34. Blom D, Ermers M, Bont L, van Aalderen WM, van Woensel JB. Inhaled corticosteroids during acute bronchiolitis in the prevention of post-bronchiolitic wheezing. Cochrane Database Syst Rev 2007; 1: CD004881. [ Links ]
35. Amirav I, Luder AS, Kruger N, et al. A double-blind, placebo-controlled, randomized trial of montelukast for acute bronchiolitis. Pediatrics 2008; 122: 1249-1255. [ Links ]
36. Bisgaard H, Flores-Nunez A, Goh A, et al. Study of montelukast for the treatment of respiratory symptoms of post-respiratory syncytial virus bronchiolitis in children. Am J Respir Crit Care Med 2006; 178: 854-860. [ Links ]
37. Law BJ, Wang EEL, MacDonald N, et al. Does ribavirin impact on the hospital course of children with respiratory syncytial virus (RSV) infection? An analysis using the Pediatric Investigators Collaborative Network on Infections in Canada (PICNIC) RSV Database. Pediatrics 1997; 99: e7. [ Links ]
38. Perrotta C, Ortiz Z, Roque M. Chest physiotherapy for acute bronchiolitis in paediatric patients between 0 and 24 months old. Cochrane Database Syst Rev 2005; 2: CD004873. [ Links ]
39. Holman RC, Shay DK, Curns AT, Lingappa JR, Anderson LJ. Risk factors for bronchiolitis associated deaths among infants in the United States. Pediatr Infect Dis J 2003; 22: 483-489. [ Links ]
40. Gavin R, Anderson B, Percival T. Management of severe bronchiolitis: indications for ventilator support. NZ Med J 1996; 109: 137-139. [ Links ]
41. Bush A, Thomson AH. Acute bronchiolitis. BMJ 2007; 335: 1037-1041. [ Links ]
42. Scottish Intercollegiate Guidelines Network (SIGN). Bronchiolitis in children. A national clinical guideline. 2006. www.sign.ac.uk (accessed 28 November 2009). [ Links ]
43. Feltes TF, Cabalka AK, Meissner HC, et al. Palivizumab prophylaxis reduces hospitalization due to respiratory syncytial virus in young children with hemodynamically significant congenital heart disease. J Pediatr 2003; 143: 532-540. [ Links ]
44. The IMpact-RSV Study Group. Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from the respiratory syncytial virus infection in high-risk infants. Pediatrics 1998; 102: 531-537. [ Links ]
45. Sigurs N, Gustafsson PM, Bjarnason R, et al. Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med 2005; 171: 137-141. [ Links ]
46. Motala C, Green RJ, Kling S, Manjra A, Potter PC, Zar HJ, for the South African Childhood Asthma Working Group (SACAWG). Guideline for the management of chronic asthma in children - 2009 update. S Afr Med J 2009; 99: 898-912. [ Links ]
Correspondence to: robin.green@up.ac.za














