Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.100 n.5 Pretoria May. 2010
EDITORIALS
A Pan-African clinical trials registry for the specific needs of triallists on the continent
Clinical trials provide the best evidence for which health care interventions work, which do not, and which may be harmful.1 Ideally we aim to base our clinical practice on the results from well-conducted trials. For us to be able to do so, all trial reports must be available in the public domain and accurately reflect the methods and the results of clinical trials.
Trial reports may not be available publicly due to publication bias - the tendency for positive and significant trial results to be published preferentially and for negative or neutral trial results to be refused, or withheld from, publication.2 Prospective trial registration arose out of the need to reduce the effects of publication bias and to encourage greater public disclosure, particularly in industry-led trials.3 Prospective registration encourages triallists to record the aim, objectives, outcomes and planned analysis of their trial on a clinical trial register before enrolling the first patient. After successful registration the trial is allocated a unique identification number.
Clinical trial registration has become an important part of the clinical trial process since the Ministerial Summit on Health Research called on the World Health Organization (WHO) in 2004 to establish 'a network of international clinical trial registers to ensure a single point of access and the unambiguous identification of trials'. In 2005 this call was endorsed by the 58th World Health Assembly and supported by the International Committee of Medical Journal Editors (ICMJE), who updated their statement so that only trials registered on WHO-endorsed primary registers would be published in participating journals (www.who.int/ictrp/). A WHO primary registry must comprise the WHO's 20-item minimum dataset in which trial registrants publicly disclose specific trial information. This standardised information is then regularly uploaded on to the WHO International Clinical Trials Portal (www.who.int/trialsearch/), a search engine that enables searching for trials according to specific characteristics such as by intervention or location.
In South Africa, the necessity to register trials is legislated through Section 11(r) in the National Health Act of 2004 (www.ufs.ac.za). The South African National Clinical Trials Registry (SANCTR) (www.sanctr.gov.za) is administered by the National Department of Health and is named as the venue through which to fulfil this obligation in the South African Good Clinical Practice Guidelines, 2nd edition (2006), in section 1.5.2 (www.doh.gov.za/nhrec/norms/gcp.pdf). Currently, the SANCTR does not fulfil the ICJME mandate for clinical trials registration because it is not a WHO-endorsed primary registry. We would advise South African triallists to also register their trial on another WHO-recognised primary registry to ensure that they may be considered for publication in ICJME journals.
To provide opportunities for local registration, the South African Cochrane Centre (SACC), based at the Medical Research Council, established the Pan-African Clinical Trials Registry (PACTR) in 2007. The PACTR (www.pactr.org) is funded by the European and Developing Countries Clinical Trial Partnership and provides a platform for prospective registration of all clinical trials in Africa. On 25 September 2009, the PACTR was officially launched as a WHO-endorsed primary register in Abuja, Nigeria, the only such register in Africa.
Trial work evolving in Africa
African triallists face challenges to registration such as limited, unreliable and costly Internet access. The PACTR seeks to provide feasible ways of overcoming this by allowing triallists to register by postal mail or facsimile in addition to online and via e-mail. Since the PACTR was first established in 2007, 42 applications for registration have been received with 6 of these sent by e-mail to be completed by the Project Manager. We also provide dedicated telephonic and e-mail support to registrants.
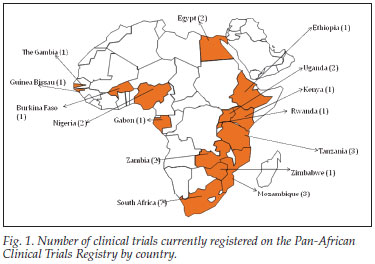
The number of trial applications has increased progressively over time. In 2008 there were only 12 applications, whereas 2009 saw 26 new applications, with most registrations following immediately after the official announcement of WHO primary register status in Abuja. As word of the registry spreads, representation among African nations is increasing on the PACTR. PACTR data reveal 10 single-centre trials taking place, in South Africa (4), Egypt (2), Kenya (1), Tanzania (1) The Gambia (1) and Uganda (1); 7 multi-centre trials have sites in 12 African countries, including Burkina Faso, Ethiopia, Gabon, Guinea Bissau, Mozambique, Nigeria, Rwanda, South Africa, Tanzania, Uganda, Zambia and Zimbabwe, and 1 non-African country, India (Fig. 1). Registered trials cover research on a range of diseases: HIV/AIDS (7), tuberculosis (4), co-morbid TB and HIV (3), malaria (1), birthing (1), hyperkalaemia (1) and prostate surgery (1).
Conclusion
Clinical trial registration does not replace the need for legislation and should not function as an ethics watch-group, but a registry can promote and encourage compliance with regulatory and legal requirements. If African countries participate actively in the PACTR, their prospectively registered trials will be traceable and accessible in the future. As more trials are registered, the PACTR will provide a comprehensive, searchable, free repository of African trials, ensuring that African trial activity is adequately represented globally.
Registration and information on registered trials is free of charge and easy to access. To learn more about PACTR or to register a trial, please visit our website www.pactr.org.
The project is funded by the European and Developing Countries Clinical Trials Partnership.
Amber Abrams
Pan-African Clinical Trials Registry Project Manager
South African Cochrane Centre
Medical Research Council, Cape Town
Nandi Siegfried
Co-director and Senior Specialist Scientist
South African Cochrane Centre
Medical Research Council, Cape Town
1. Chinnock P, Siegfried N, Clarke M. Is evidence-based medicine relevant to the developing world? PLoS Med 2005; 2(5): e107. [ Links ]
2. Dickersin K, Min Y-I, Meinert CL. Factors influencing publication of research results. JAMA 1992; 267: 374-378. [ Links ]
3. Askie LG, Simes J. Prospective registration of clinical trials. Aust J Physiother 2006; 52: 237-239. [ Links ]
Corresponding author: A Abrams (amber.abrams@mrc.ac.za)














