Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 n.12 Pretoria Dec. 2009
GUIDELINE
Guideline for the management of chronic asthma in children - 2009 update
C Motala; R J Green; A I Manjra; P C Potter; H J Zar
South African Childhood Asthma Working Group (SACAWG)
ABSTRACT
OBJECTIVE: To revise the guideline for the diagnosis and management of chronic asthma in children in view of the following considerations: the existing South African Childhood Asthma Working Group (SACAWG) guideline was produced 10 years ago; diagnosis of asthma in young children remains a challenge; evidence-based treatment is the new paradigm; new treatment approaches to achieving and maintaining control; therapeutic roles of several medications have evolved; more studies and data on treatment in young children; new medications and formulations; a change of emphasis in assessing asthma control to guide treatment changes. The main aim of the guideline is to promote a better standard of treatment based on understanding of the pathophysiology and pharmacotherapy of asthma, and encouraging uniformity in asthma management.
EVIDENCE: A detailed literature review by a working group of clinicians from relevant disciplines. The strategies recommended are classified according to the evidence category in Appendix B, and denoted as Evidence A, B, C and D.
RECOMMENDATIONS: These include an appropriate diagnostic approach, environmental control measures, treatment options, definition of asthma control, and strategies to achieve control.
ENDORSEMENT: The guideline document was endorsed by the South African Thoracic Society (SATS), the National Asthma Education Programme (NAEP), the South African Paediatric Association (SAPA) and the South African Academy of Family Practice.
1. Background
The South African Childhood Asthma Working Group (SACAWG), a sub-committee of the Allergy Society of South Africa (ALLSA), first published its guideline for the management of chronic asthma in children and adolescents in 1992.1 The guideline was revised in 19942 and 2000.3 In the past decade, there have been significant advances in the management of asthma; these include treatment based on level of control, newer treatment options, new formulations of inhaled corticosteroids, and availability of low-cost spacers for delivering inhaled drugs.
The current revision was prompted by: (i) a need to expand the sections on diagnosis and treatment to include children <5 years old; (ii) the revised classification of asthma based on level of control; (iii) an ongoing need to emphasise the use of controllers (previously referred to as preventers) as the foundation of asthma treatment; and (iv) the positioning of newer treatment options for asthma (evidence-based approach), including combination treatment (inhaled corticosteroid (ICS) plus a long-acting β2-agonist (LABA)), leukotriene receptor antagonists, sublingual immunotherapy, omalizumab and macrolides.
The objectives of this guideline are to promote a better standard of treatment based on advances in the understanding of the pathophysiology and pharmacotherapy of asthma, and to encourage uniformity in the management of asthma.
2. Methodology
This 2009 asthma guideline update was undertaken after a meeting with a working group constituted by ALLSA. The working group is chaired by Professor Cas Motala.
The working group meetings were held in June 2008. Four task groups, each headed by a leader, were constituted: (i) Asthma diagnosis and assessment of level of control; (ii) Environmental control measures and immunotherapy; (iii) Pharmacotherapy: corticosteroids and steroid/LABA combinations; (iv) Pharmacotherapy: New treatments including leukotriene modifiers, anti-IgE and macrolides. The task groups were charged with reviewing the available scientific literature and assigning evidence levels according to the methodology utilized in current guideline documents: Categories A, B, C and D based on the strength of evidence levels (Appendix B). Subsequently, an editorial board, consisting of the chairperson and leaders of the task groups, was convened. The working group is aware of the trend toward application of GRADE4 technology and will consider utilising this methodology in the future.
The working group met again in July 2009 to discuss the draft guideline document. Thereafter, the editorial board was tasked to finalise the document for publication. This project was sponsored by ALLSA.
3. Disease profile
Asthma is the most common chronic childhood disease, affecting 10 - 20% of South African children.5 The burden of asthma affects the patients, their families, and general society in terms of lost work and school days, impaired quality of life, hospitalisations and mortality. Asthma typically begins in early life, and is often triggered by viral infections or environmental factors including allergens. Airway inflammation and airway hyper-responsiveness are the hallmarks of the disease. Characteristic symptoms include recurrent episodes of wheezing, breathlessness, chest tightness and coughing. In children <5 years, the clinical symptoms are variable and nonspecific. Furthermore, neither airflow limitation nor airway inflammation are possible to assess routinely in this age group. Diagnosing asthma in young children may therefore be difficult.
The goal of asthma treatment is to achieve control of the disease for prolonged periods with appropriate regard for the safety and cost of treatment required to achieve this. Defining satisfactory current clinical asthma control in children <5 years is problematic since it is almost exclusively dependent on reports from family/caregivers who may be unaware of or underreport the presence of asthma symptoms.
Recommendations in this guideline are based on currently available evidence. Health practitioners should strive to achieve the best possible therapy for each of their patients through motivation and education of parents and health care professionals, to ensure that all patients have access to appropriate medication.
4. Diagnosis
Asthma should be diagnosed in a child with chronic/recurrent wheeze with or without cough (owing to bronchoconstriction) triggered by multiple factors including viral infections, allergens, irritants (pollution), exercise and sudden emotional changes (e.g. crying, laughing) and which responds to an inhaled bronchodilator. Features supporting the diagnosis are a family or personal history of atopy, night cough, exercise-induced cough and/or wheeze and seasonal variation in symptoms. In children, allergy is often the main trigger that determines the severity of the disease. Early sensitisation, severe atopy and synergistic interaction between atopy and infections are risk factors for persistent asthma.6 Cough variant asthma is a rare form of asthma which presents with cough but no wheeze and no evidence of airway obstruction on spirometry.7,8
4.1 Children older than 5 years
A careful history and physical examination, together with objective evidence of reversible airflow obstruction after administration of a short-acting β2-agonist (SABA) (an increase in forced expiratory volume in 1 second (FEV1) >12% or in peak expiratory flow rate (PEF) >15% after 10 minutes), will in most instances confirm the diagnosis. Monitoring symptoms and PEF using a diary card is also useful for making the diagnosis; diurnal PEF variability >20% is highly suggestive of asthma. Conditions that need to be considered when the diagnosis of asthma is uncertain are listed in Table I.

4.2 Children 5 years and younger
The diagnosis of asthma in early childhood is challenging and has to be based largely on clinical judgement (assessment of symptoms and physical findings). Since the use of the label 'asthma' for wheezing in children has important clinical implications, it must be distinguished from other causes of persistent and recurrent wheeze (Table II).

Episodic wheezing and cough is very common even in children who do not have asthma, and particularly in those <3 years old. Three categories of wheezing have been described in children <5 years (retrospective epidemiological classification9): transient early wheezing, persistent early-onset wheezing, and late-onset wheezing. Despite this epidemiological classification, the diagnostic approach to these patients is poorly described, especially in low- and middle-income countries.
A simple clinical index (asthma predictive index) based on the presence of a wheeze before the age of 3, and the presence of 1 major risk factor (parental history of asthma or eczema) or 2 of 3 minor risk factors (eosinophilia, wheezing without colds, and allergic rhinitis), has been shown to predict the occurrence of asthma in later childhood in children in developed countries.10 While useful to diagnose asthma in large epidemiological studies in Europe and America (Evidence B), this finding has not been applied in the context of African children, who have less atopy.
A recent publication categorised wheeze in young children into 2 broad groups: episodic (viral) wheeze and multi-trigger wheeze.11 The group of children who have multi-trigger wheeze have symptoms precipitated by factors other than viral infections (such as allergens and exercise) and are likely to be asthmatic.
The diagnosis of preschool asthma should be based on a composite set of diagnostic criteria and, while no tests diagnose asthma with certainty in young children, the following may be considered as useful adjuncts in making a diagnostic decision:
4.2.1 Clinical features
The categories of symptoms and therapeutic responses listed in Table III are highly suggestive of asthma (based on current evidence and clinical experience).

4.2.2 Modified bronchodilator response test (MBR)12 (Evidence D)
In this test, a patient suspected of having asthma (usually a young child with persistent or recurrent wheezing) is given a bronchodilator by nebuliser or metered-dose inhaler (MDI) with a spacer, and symptoms/signs are evaluated 10 - 15 minutes later. Asthmatics usually demonstrate a significant clinical response to a bronchodilator, as measured by reduction in wheeze or respiratory rate or improvement in air entry. Some children with viral-induced wheeze may also respond, so the test forms part of an assessment for asthma and is not the sole criterion for diagnosis.
4.2.3 Lung function testing
Lung function testing is useful for aiding the diagnosis of asthma in children >5 years. In general, they do not have a diagnostic role in younger children because of their inability to perform reproducible expiratory manoeuvres. Lung function testing for infants13 and young children are only possible in specialised centres, and are mainly used for research purposes.
5. Goals and principles of management
5.1 Goals
The goal of asthma therapy is asthma control. Control implies that the asthmatic child is able to lead a normal and physically active life. The criteria for 'normal life' are to:
• be completely free from any symptoms, i.e. cough, wheeze and breathlessness
• attend school regularly and participate fully in all school activities, including sport
• sleep restfully, free from night-time cough and/or wheeze
• grow and develop normally
• minimise the number of attacks of acute asthma and avoid hospitalisation
• avoid or minimise medication-related side-effects.
5.2 Principles
A comprehensive therapeutic approach is required to meet the above objectives, which includes the following:
• early diagnosis and objective assessment of severity
• control of the environment to exclude cigarette smoke and reduce exposure to triggers such as viral infection and allergens
• optimal use of medications to limit side-effects and cost, using the most appropriate delivery system
• follow-up and regular re-evaluation (clinical evaluation and quality of life)
• patient and parent education.
6. Environmental control
Avoidance of allergens (based on history and allergy testing) and pollutants is recommended when there is a clear association between exposure and symptoms. Recommended avoidance measures are discussed below.
6.1 Indoors
6.1.1 Tobacco smoke/other pollutants
Smoking should not be allowed in the home, and active steps should be taken to inform parents of the problem, encouraging smokers to quit. Smoking during pregnancy should be strongly discouraged as the evidence indicates that in utero exposure to tobacco smoke is associated with a higher risk of developing wheezing early in life.
Other sources of biomass fuel exposure, such as open fires for cooking or heating, may trigger asthma or cause more severe asthma. Fluted chimneys and good ventilation in homes should be encouraged. Children should be kept away from open flames or fumes, e.g. a mother should avoid putting her baby on her back while cooking over open flames.
6.1.2 Indoor allergens14
A detailed history should identify which allergens are likely to be triggers of asthma symptoms. If there is uncertainty after taking a history, and the child has persistent asthma, then specific allergen testing by skin prick test or specific immunoglobulin E (IgE) is indicated. There is no evidence to support avoidance of allergens for primary prevention of asthma.
• House dust mites. Mattress, pillow and duvet covers with mite-impermeable characteristics are recommended, but mites will accumulate on mattress covers if these are not regularly washed15 (Evidence B). Bedding should be washed regularly at temperatures >60ºC. Rooms should be well ventilated. Where possible, carpets should be removed from the living areas and especially the bedrooms. Acaricides are ineffective. Other sources of house dust mites should also be considered, e.g. fluffy toys and feather pillows.
• Cockroaches. Cockroach allergy is widespread in South Africa. Cockroaches may be a cause of ongoing airway inflammation, and sensitivity to cockroaches is a risk factor for more severe asthma. Obsessive cleaning, bait stations and boric acid indoors can reduce cockroach numbers16 (Evidence B).
• Pets. Asthmatics known to be allergic to dogs or cats should avoid contact with them. Cat allergens are notoriously difficult to eliminate and may persist for several months after the cat has been removed from the home. Use of a dust cloth is three times more effective in reducing allergen levels than vacuuming with a brush attachment17 (Evidence B).
• Mould. Obvious sources of indoor mould in bathrooms, kitchens and damp areas of the home should be dealt with by proper plumbing, damp-proofing, mould-repellent paint and sodium hydroxide sprays. Products containing hypochlorite are effective in cleaning homes, achieving a low reduction in fungi, and improving quality of life - which was statistically significantly better than in a control group18 (Evidence B).
6.2 Outdoors
6.2.1 Outdoor allergens
Moulds are important sources of outdoor allergens, and sensitive children are advised to avoid exposure to mouldy places, e.g. farms, forests, compost heaps and parks, particularly in autumn, winter and spring. Grass pollens may be implicated in perennial asthma because of the particularly long grass season in South Africa.
7. Treatment
Pharmacotherapy is the cornerstone of asthma management, with appropriate medication and delivery devices to meet patients' needs and circumstances. When asthma is first diagnosed, it is convenient for initiation of treatment to classify it as mild intermittent or chronic persistent asthma that is mild, moderate or severe (Table IV).3 However, the severity of asthma is variable, does not predict response to treatment, and is of little value in patients already on treatment. Periodic assessment of asthma control (Table V)19 (www.ginasthma.com) and review of management are more relevant. Asthma severity assessment is used to initiate therapy, while asthma control is determined to monitor and adjust therapy.
7.1 Assessment of severity to initiate therapy
The method of assessment presented conforms to international assessment criteria. The following points should be noted:
• The assessment of severity is used to assign a child to a particular treatment group only as a starting point.
• The assessment of severity refers to a child's symptoms and lung function (PEF or FEV1) between acute episodes in a patient who is not receiving long-term therapy. Severity can also be measured, once asthma control is achieved, by the step of care (i.e. the amount of medication) required to maintain control.
• One or more features must be present to assign a grade of severity; a patient must be assigned to the most severe grade in which any feature occurs.
• In practice, about 70% of childhood asthmatics will fall into the mild persistent category, 25% into the moderate persistent group, and 5% into the severe persistent category.
7.2 Assessment of asthma control
Control refers to the degree to which the manifestations of asthma are minimised by therapeutic intervention and the goals of therapy are met. After therapy is initiated, the emphasis for clinical management changes to the assessment of asthma control (Table V). The level of asthma control will guide decisions either to maintain or to adjust therapy, i.e. step up if necessary, or step down if possible. Asthma control may be assessed clinically (symptoms, physical findings, reliever use), and by measurement of lung function and fractional exhaled nitric oxide (FENO) in certain situations.
7.2.1 Clinical assessment
Clinical assessment includes the frequency of daytime and nocturnal symptoms, the extent of limitation of activities, and the need for reliever treatment (Tables V and VI). Patient-centred questionnaires such as the Childhood Asthma Control Test (cACT) are very useful for identifying children who are uncontrolled.20 The cACT has been promoted for the assessment of asthma control in children >4 years old. The cACT has 7 questions regarding asthma symptoms present in the previous 4 weeks. The first 4 questions are completed by the child, while the last 3 are completed by the parent. A score of 20 or more is judged to be well-controlled asthma.

7.2.2 Spirometry
Spirometry (flow-volume loop) can only be performed reliably in children >5 years old. The most commonly used parameters derived from the volume/time plot are FEV1 and the forced vital capacity (FVC). The most commonly used parameters derived from the flow/volume plot are PEF and the maximum mid-expiratory flow rate (MMEF). FEV1 and PEF are utilised most frequently for assessing asthma control.
Spirometry should be performed:
• at the initial assessment
• after treatment is initiated and symptoms and PEF have stabilised
• during periods of progressive or prolonged loss of asthma control
• at least every 1 - 2 years, more frequently depending on response to therapy
Low FEV1 indicates current obstruction and is a useful measure of risk for exacerbations, although it must be emphasised that even children who have normal lung function experience exacerbations. PEF can be read directly off the flow-volume loop or it can be measured with a peak flow meter which can easily be used at home to monitor lung function. PEF has limitations in that it is effort-dependent; PEF changes do not always reflect changes in lung function, and are generally a late indicator of loss of asthma control. Among patients with mild intermittent or mild persistent asthma, PEF monitoring has no advantage over symptom monitoring.21 Short-term daily PEF monitoring, however, should be considered for patients who have moderate to severe persistent asthma or a history of severe exacerbations (it may be helpful for assessing the severity of obstruction and evaluating response to bronchodilator therapy) and for patients who poorly perceive airway obstruction or worsening asthma.
7.2.3 Exhaled nitric oxide (FENO)
FENO is not a routine test of asthma control. For most children, monitoring of exhaled NO offers no benefit over clinical monitoring. It may, however, have value in certain circumstances such as a child with difficult-to-control asthma. FENO values have been shown to increase before the onset of symptoms or loss of control and monitoring may, therefore, be used to predict loss of control.22 It may also be useful for assessing compliance with anti-inflammatory therapy.
7.3 Principles of medication
When selecting medication for an asthmatic patient, the following principles apply:
• regular anti-inflammatory medication is indicated for persistent asthma
• inhaled therapy is preferable
• drugs are classified as:
• relievers - short-acting bronchodilators with rapid onset of action that provide acute relief of symptoms
• controllers - drugs with anti-inflammatory and/or a sustained bronchodilator action.
7.4 Route of administration
Inhaled therapy is the cornerstone of asthma treatment for all children. Most children can be taught to use inhaled therapy effectively. Considerations in choosing an inhaler device include the efficacy of drug delivery, cost, safety, ease of use, convenience and efficacy in a specific age group.23,24 A metered dose inhaler (MDI) with spacer is preferable to nebulised therapy because of convenience, more effective lung deposition, fewer side-effects and lower cost.25,26
Spacers retain large drug particles that would be deposited in the oropharynx, so reducing oropharyngeal side-effects and systemic absorption and availability of inhaled drug. This consideration is especially important for ICS with poor first-pass metabolism such as beclomethasone dipropionate (BDP) and budesonide. Commercially produced spacers with well-characterised drug output characteristics, or a homemade spacer device made from a 500 ml plastic cold-drink bottle, are preferable.27 A child's inhalation technique should be observed, and corrected if necessary, at the health-care consultation.
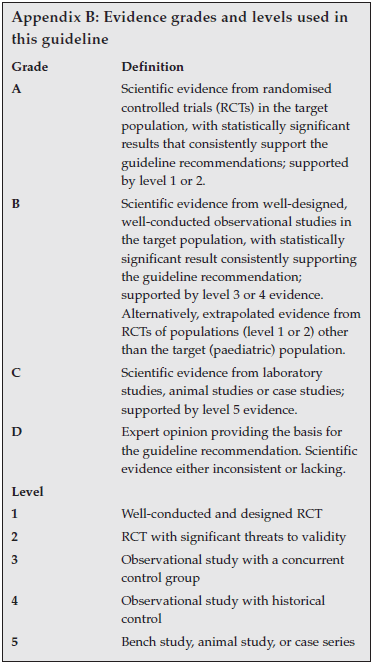
An MDI with a spacer is as effective as, or more effective than, nebulised treatment for of acute, severe asthma exacerbations.25,26 Nebulisers have imprecise dosing, are expensive, waste large amounts of drug into the surrounding air, are time-consuming to use and care for, and require maintenance. They are mainly reserved for children who cannot use other inhalation devices. In general, nebulisers should be discouraged for home use and rather utilised in settings where oxygen administration is necessary and available. A general inhaler strategy for children is given in Table VII.
7.5 Relievers
These include SABAs (Evidence A) and anticholinergics. Short-acting xanthines are not recommended in the maintenance treatment of asthma.
7.5.1 Short-acting β2-agonists
SABAs are generally used on an as-needed basis. Their use can be minimised by the optimal use of anti-inflammatory agents and controllers. SABA inhalers provide relief from acute symptoms of asthma.
7.5.2 Anticholinergics (ipratropium bromide)
These drugs work by inhibiting vagally mediated bronchoconstriction. They are less potent bronchodilators than inhaled β2-agonists and in general have a slower onset of action (30 - 60 minutes to maximum effect). They may be used in patients who cannot tolerate β2-agonists or as adjunctive bronchodilator treatment in patients who do not obtain adequate symptom relief during acute asthma. There is no evidence for the long-term use of ipratropium bromide in the management of asthma.
7.6 Controllers
Controller therapy is recommended for all patients with persistent asthma. ICSs are the most effective controller therapy for asthma (Evidence A). LABAs have prolonged bronchodilatory action, but have weak anti-inflammatory (controller) effects. Leukotriene receptor antagonists (LTRAs) also have anti-inflammatory effects via different pathways to ICSs. Sustained-release (SR) theophyllines have also been shown to have weak anti-inflammatory effects at doses lower than those required for bronchodilatation.
7.6.1 Inhaled corticosteroids (ICSs)
ICSs are the most effective medication for controlling symptoms, reducing the number of asthma-related hospitalisations, improving lung function, reducing the frequency of exacerbations, improving quality of life and reducing bronchial hyper-responsiveness19,28-34 (Evidence A).
Inhaled therapy via an MDI with spacer is preferable to nebulised therapy because of the convenience, more effective lung deposition, fewer side-effects and lower cost (Evidence A). For children >5 years old, a dry powder inhaler (DPI) or breath-actuated pressurised MDI may be alternatives to an MDI with spacer.
A number of different ICS preparations are available (Table VIII). Most ICSs should be given twice daily, but budesonide and ciclesonide are approved for use once daily in children with mild asthma. The most important determinant of appropriate dosing is the patient's response to therapy. ICS dose should be adjusted according to the level of control attained. Once control of asthma is achieved, the dose of medication should be carefully titrated to the minimum dose required to maintain control, thus reducing the potential for adverse effects.
Side-effects
Most studies evaluating the systemic effects of ICS suggest that clinically effective doses of ICS are safe and the potential risks are well balanced by the clinical benefits19,33,34 (Evidence A). However, studies using higher doses have been associated with detectable systemic effects on both growth and the hypothalamo-pituitary axis (HPA). Although there are fewer studies in children <5 years, the available data are similar to those from older children.19,34,35 Generally, low doses of ICS have not been associated with any clinically important adverse systemic effects in clinical trials, and long-term use is considered safe.19,34-36 Local side-effects, such as hoarseness and candidiasis, are rare in children when a spacer is used.
Children older than 5 years
Most children are controlled on low daily doses of ICS (100 - 200 g budesonide or equivalent (Table VIII). Some children require higher doses (400 µg/day) for control and for protection against exercise-induced symptoms. Clinical improvement occurs rapidly within 1 - 2 weeks, although maximum improvement may occur only after many weeks. Symptoms may recur after stopping ICS, with control deteriorating within weeks.
Children younger than 5 years
Several studies of ICS in young children with asthma have shown similar clinical effects as in older children, including increases in lung function and number of symptom-free days, reduction in symptoms, need for additional medication, caregiver burden, systemic glucocorticosteroid use, and exacerbations.30-32,34-36 A systematic review of randomised double-blind controlled trials of ICSs in preschool children with wheeze showed statistically significant improvements in symptoms, exacerbation rates, lung function and airway hyperresponsiveness.31 Of note is that exacerbations were reduced by almost 40%.31 However, the dose-response relationships have been less well studied. Use of ICSs for up to 2 years has not been shown to induce remission of asthma; symptoms usually return when treatment is stopped.30
The clinical benefits of ICS for episodic, viral-induced wheeze are controversial. Systematic reviews found that episodic high-dose ICSs provide some benefit, reducing the need for oral corticosteroids by 50%; however, no effect on hospitalisation rates or duration of symptoms was found. Maintenance treatment with 400 µg/day of budesonide equivalent does not reduce the number or severity of wheezing episodes in episodic, viral-induced wheeze.37,38
Intermittent use of ICSs in preschool children with episodic viral wheeze at the time of symptoms does not reduce the risk of persistent wheeze at the age of 6 years, and symptoms return when steroid therapy is discontinued.39 Therefore, the use of intermittent ICS for episodic wheeze cannot be recommended.
Children younger than 2 years
There is a paucity of data on the efficacy and safety of ICS in children <2 years old.34-36 In addition, wheezing caused by viral infections is very common in this age group but often resolves spontaneously or remits with age. In general, ICSs should only be used if symptoms are particularly troublesome and there is a clear response to treatment. As for older children, ICSs should be stepped down as control is achieved, so that the lowest ICS dose for control of symptoms is used.
7.6.2 Long-acting inhaled β2-agonists (LABAs) Children older than 5 years
A Cochrane review of 25 trials of 5 572 children reported that addition of LABAs did not significantly reduce the risk of asthma exacerbations requiring rescue systemic steroids, hospitalisation or duration of symptoms, but improved lung function compared with ongoing treatment with a similar dose of inhaled corticosteroids.40 There was no evidence of increased serious side-effects or withdrawals with the addition of LABAs.
The effect of LABAs added to ICSs (7 studies, 1 021 children) compared with a higher dose of ICS also reported no difference in the incidence of asthma exacerbations requiring oral steroids or in the rate of hospitalisation for asthma. However, lung function was significantly improved in children on LABAs.40 Studies have reported improvements in peak flow and lung function with the addition of a LABA.43-47 However, the effect on symptoms, need for rescue medication and frequency of exacerbations have been less consistent.41,42 The available paediatric studies suggest a modest improvement in lung function in children in whom LABAs are added to an ICS.40-47
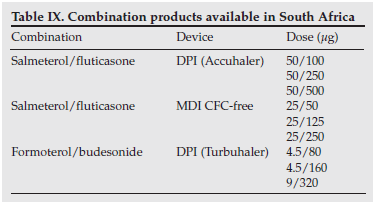
LABAs are therefore currently positioned for maintenance treatment of children with asthma only in combination with inhaled corticosteroids. Combination products (i.e. those containing an inhaled corticosteroid and a LABA in the same device) are preferable to administration via separate inhalers. Fixed combination inhalers ensure that the LABA is always accompanied by an ICS. Combination products available in South Africa are shown in Table IX.
Side-effects
LABAs are generally well tolerated in children. Side-effects are similar in type and frequency to those of SABAs, and include muscle tremor, headache and palpitations. An increased risk of severe asthma exacerbations and mortality has been reported, particularly when used as monotherapy; therefore, LABAs should never be used without an ICS.19,41,42
Children younger than 5 years
The effect of LABAs or combination products has not been adequately studied in children <5 years old. There are no published double-blind randomised placebo-controlled trials on the addition of LABAs to ICSs in this age group. LABAs are currently approved for use in South Africa in children >4 years old. Therefore, LABAs cannot be recommended at present for children <4 years old.
7.6.3 Leukotriene receptor antagonists/leukotriene modifiers
Leukotriene receptor antagonists (LTRAs) are an accepted treatment option for childhood asthma. They have a rapid onset of action (within 1 - 3 hours),48 are administered orally, treat asthma via a different pathway to other currently available medications (specific cysteinyl leukotriene receptor antagonists), and have been shown to be effective for aspirin-sensitive asthma.49
Side-effects
No significant safety concerns have been demonstrated for leukotriene modifiers in children. Headache and gastrointestinal upset are the most commonly encountered side-effects; skin rashes or flu-like symptoms are much less common.50,51
Children older than 5 years
LTRAs provide clinical benefit in children >5 years old at all levels of severity52-56 (Evidence B), but generally less than those of low-dose ICSs.57 Leukotriene modifiers provide partial protection against exercise-induced bronchoconstriction.56,57 As add-on treatment in children whose asthma is insufficiently controlled by low doses of inhaled glucocorticosteroids, leukotriene modifiers provide moderate clinical improvements, including a significant reduction in exacerbations52,58,59 (Evidence B).
Children younger than 5 years
LTRAs are safe and effective for treatment of asthma in young children, from as early as 6 months of age.60-66 LTRAs have been proposed as alternative first-line therapy to ICSs for episodic or mild persistent asthma67 (Evidence D), particularly in children who have difficulty in utilising inhalation treatment, with poor compliance, or where exercise-induced bronchospasm (EIB) is a dominant component of asthma. To date, no studies have evaluated the role of LTRAs as add-on therapy to inhaled steroids in children <5 years old.
Asthma and allergic rhinitis are frequently co-morbid conditions (80%); however, both are often treated as separate entities. With the concept of the 'united airway' disease in which asthma and allergic rhinitis are regarded as manifestations of a single disorder, treating one disease may affect the control of the other. In certain countries (but not in South Africa), LTRAs are approved for treatment of both allergic rhinitis and asthma.68-70
7.6.4 Theophylline
Theophylline has been used in the treatment of asthma mainly as a bronchodilator (10 - 20 mg/kg/day), though it may also have anti-inflammatory effects at lower doses (5 - 10 mg/kg/day).71-74 However, the anti-inflammatory effects of theophylline are small (less than that of low-dose ICSs) and side-effects are common.75 Theophylline may be used as alternative, adjunctive therapy with ICSs in children >5 years old (Evidence D).74 Monitoring of serum theophylline concentration is essential. Long-term treatment with theophylline is not generally recommended in young children because of its adverse effects.
Side-effects
The most common side-effects of theophylline are anorexia, nausea, vomiting and headache. Mild central nervous stimulation, palpitations, tachycardia, arrhythmias, abdominal pain, diarrhoea and, rarely, gastric bleeding may also occur. These side-effects are mainly seen at doses >10 mg/kg/day. The risk of adverse effects is reduced if treatment is initiated with daily doses around 5 mg/kg/day and then gradually increased to 10 mg/kg/day. Severe overdosing with theophylline can be fatal.
7.6.5 Oral corticosteroids
Oral corticosteroids such as prednisone may be considered in patients with poorly controlled severe asthma on optimal doses of ICS and additional controller therapy (Evidence D).19,34,76 Oral corticosteroids, while relatively inexpensive, are associated with serious systemic side-effects including growth suppression, obesity and adrenal suppression. Because of the side-effects of prolonged use of oral steroids, these agents should only be used for acute exacerbations, and then only for a maximum of 5 - 7 days at a dose of 1 - 2 mg/kg/dose of prednisone given once daily. It is not necessary to taper oral steroids if used as a short course. Maintenance treatment with daily or alternate-day oral steroids is indicated only in those patients not controlled with high-dose inhaled steroids and additional controller therapy. In children on oral steroids, increased dosage should be given during episodes of increased stress, e.g. surgery.
8. Other treatment options
8.1 Immunotherapy
The Expert Panel of the USA's Department of Health and Human Services76 recommends that allergen immunotherapy be considered for patients who have persistent asthma if there is clear evidence of a relationship between symptoms and exposure to an allergen to which the patient is sensitive (Evidence B).
A meta-analysis of 75 randomised placebo-controlled studies has confirmed the effectiveness of subcutaneous immunotherapy in asthma with a significant reduction in asthma symptoms and medication and with improvement in bronchial reactivity77 (Evidence A).
Immunotherapy is also effective for children when used sublingually78 (Evidence B). Sublingual immunotherapy is effective in paediatric patients with allergic rhinitis79 (Evidence A) and has an effect on inflammatory parameters and bronchial hyper-reactivity in asthmatic children sensitised to house dust mites80 (Evidence B). Although both subcutaneous and sublingual immunotherapy are effective in asthmatic children (monosensitive to house dust mite or pollen), the latter is safer (confirmed in post-marketing studies81) (Evidence B). Based on the above evidence, sublingual immunotherapy could be used as adjunctive treatment to pharmacotherapy in children >5 years old with rhinitis and mild to moderate asthma (FEV1 >80%), to enhance asthma control and, in some cases, effect a cure.
8.2 Omalizumab
Omalizumab (anti-IgE) is a monoclonal antibody that prevents binding of IgE to the high-affinity receptors on basophils and mast cells. Anti-IgE is used as adjunctive therapy for patients >12 years old with severe persistent asthma who have sensitivity to relevant inhalant allergens, e.g. house dust mite, cockroach, cat and dog. Isolated cases of anaphylaxis to anti-IgE have been reported in adults. Omalizumab is administered via subcutaneous injection every 2 - 4 weeks. The therapeutic index of this agent in children is still being defined. Clinicians who administer omalizumab should be prepared and equipped to identify and treat anaphylaxis that may occur. Anti-IgE should preferably be administered by a specialist. Mode of administration and cost could limit the use of this treatment, particularly in children.
8.3 Macrolides
Macrolide antibiotics have recognised anti-inflammatory properties in addition to their antimicrobial effects. Although some benefits have been reported in adults with chronic persistent asthma, a meta-analysis of 7 randomised controlled clinical trials involving both children and adult patients (N=416) with chronic asthma and treated with macrolides or placebo for >4 weeks, reported insufficient evidence to support their use in patients with chronic asthma.82
8.4 Antihistamines
Antihistamines have no proven benefit for asthma symptom control, but may be useful for control of symptoms of co-existing allergic diseases such as rhino-conjunctivitis, urticaria and atopic eczema.
8.5 Unnecessary therapy
The following are of no benefit in the treatment of childhood asthma: antibiotics, cough syrups, mucolytics, ionisers and breathing exercises. Complementary and alternative medications and interventions bear insufficient evidence to permit recommendations. Physiotherapy is indicated in children only where lobar collapse is documented.
9. Self-management plan for asthma
The responsibility for patient education rests with the doctor, and should be shared with trained health care professionals. Patients and parents must be encouraged to participate actively in their own management. In South Africa, the National Asthma Education Programme (NAEP) provides free access to patient educational materials as well as symptom/adherence diaries and a self-management action plan. These are available at www.asthma.co.za.
Patient education significantly improves asthma control and decreases hospitalisation rates (Evidence A).83 The influence of education is greater in moderate-severe, compared with mild-moderate, asthma. Intensive education is of greater benefit than limited education, and written plans are superior to oral instructions. Education given at follow-up visits is more effective than education during emergency visits.84 Culture-specific programmes for adults and children from minority groups with asthma are more effective than generic programmes in improving quality of life, asthma knowledge, asthma exacerbations and asthma control (Evidence B).85
Educational programmes should be considered a part of the routine care of all patients with asthma. Asthma self-management education should be integrated into all aspects of asthma care and reinforced at each follow-up visit. The elements of a self-management plan are summarised as follows:
• Avoid known allergens and nonspecific triggers, particularly tobacco smoke.
• Educate patients and parents about asthma and its treatment, e.g. difference between controller and reliever treatment, correct use of inhalers, use of a spacer where necessary, and correct use of peak flow meter where appropriate.
• Provide patients and parents with an asthma adherence and symptom diary as a tool for monitoring asthma control.
• Provide all patients with a written action plan that includes instructions for both daily management (long-term control medication, if appropriate, and environmental control measures) and actions to manage worsening asthma (what signs, symptoms and PEF measurements (if used) indicate worsening asthma; what medications to take in response; what signs and symptoms indicate the need for immediate medical care). Written action plans are particularly recommended for patients who have moderate or severe persistent asthma, or a history of severe exacerbations, or poorly controlled asthma.
• Arrange for a Medic-Alert badge for patients with severe steroid-dependent asthma, known drug hypersensitivity such as aspirin sensitivity, and brittle asthma.
10. Follow-up and review
Re-evaluate treatment within 2 - 6 weeks and adjust therapy as appropriate. The main objective of follow-up visits is to determine whether asthma is controlled or not (Table V). In addition, the following points should be assessed/monitored at each visit:
• inhaler technique
• adherence
• written action plan
• patient concerns
• side-effects of medication
• growth (weight, height).
11. Adjusting therapy based on control (Fig. 1)
If asthma is uncontrolled, treatment may be stepped up as follows:
• For children younger than 5 years: If asthma is not controlled on low-dose ICS, doubling the initial dose of ICS (medium-dose ICS) is the preferred controller option. Alternatively, consider adding a LTRA or LABA (LABA approved only in children >4 years old) to the ICS. If uncontrolled on medium-dose ICS and a LTRA (or LABA in children >4 years old), refer to a specialist for further evaluation and management.
• For children older than 5 years: Options for stepping up treatment include increasing the dose of ICS (medium dose) or adding a LABA or LTRA. If still uncontrolled, consider further increasing the ICS dosage or adding another controller therapy if already on medium doses of ICS.20 If still uncontrolled, refer to a specialist.
The need for additional treatment should be re-evaluated at each visit and maintained for as short a period as possible.
If asthma is well controlled for at least 3 months, reduce treatment. There are few studies to guide step-down therapy. A suggested regimen for step-down treatment is: first discontinue oral steroids; next reduce the dosage of ICS (if on high dose); and thereafter discontinue additional controller treatment. The child should be maintained on the lowest effective dose of ICS. Assess control on a regular basis.
There are many factors in poor asthma control (Table X), including lack of adherence, incorrect inhaler technique, inadequate therapy for the degree of severity of asthma, environmental triggers, and presence of co-morbid conditions. Alternative causes of recurrent wheezing must also be sought. Before stepping up or changing treatment, these factors should be excluded.

12. Non-adherence
Studies of adults and children have shown that approximately 50% of those on long-term therapy fail to take medications as directed at least part of the time.86 Non-adherence may be defined in a non-judgemental way as failing to take treatment as agreed upon by the patient and the health care professional. Non-adherence may be identified by prescription monitoring, pill counting or drug assay, but at a clinical level it is best detected by asking about therapy in a way that acknowledges the likelihood of incomplete adherence (e.g. 'So that we may plan therapy, do you mind telling me how often you actually take the medicine?' or 'How many times in a normal week do you forget to take the medicine?'). Specific drug and non-drug factors involved in non-adherence are listed in Table XI.
Strategies to promote adherence include:
• prescribing a simple dosage regimen (once or twice daily) and as few drugs as possible
• selecting treatment that achieves outcomes and addresses preferences that are important to the patient, and reminding patients that adherence will help to achieve goals of treatment
• assessing the patient's and family's level of social support, and encourage family involvement
• tailoring the self-management approach to the needs and literacy levels of the patient, and maintaining sensitivity to cultural beliefs and ethno-cultural practices. Individualised self-management of asthma has been shown to improve medication adherence and asthma control.87
13. Co-morbid conditions
It is necessary to identify and treat co-morbid conditions, such as those below, that may affect asthma control. If these conditions are treated appropriately, asthma control may improve.
• Rhinitis or sinusitis symptoms or diagnosis should be evaluated in patients who have asthma, because of the interrelationship of the upper and lower airways. There is sufficient evidence that co-existing allergic rhinitis or sinusitis may impair asthma control in adults.88 In addition, therapy for allergic rhinitis or sinusitis has been shown to improve asthma control. Treatment of allergic rhinitis includes intranasal corticosteroids, antihistamine therapy, and, in selected cases, immunotherapy.89 Treatment of sinusitis includes intranasal corticosteroids and antibiotics. Evidence is inconclusive regarding the effect of sinus surgery on asthma in patients with chronic rhinosinusitis.
• Gastro-oesophageal reflux disease (GORD) treatment may benefit patients who have asthma and complain of frequent heartburn, particularly those who have frequent nocturnal asthma symptoms. Even in the absence of suggestive GORD symptoms, consider evaluation for GORD in patients who have poorly controlled asthma.
• Allergic bronchopulmonary aspergillosis may be considered in patients who have asthma and a history of pulmonary infiltrates, have IgE sensitisation to Aspergillus, and/or are corticosteroid-dependent. Diagnostic criteria include a positive immediate skin test and elevated serum IgE and/or IgG to Aspergillus and central bronchiectasis.
• Obese patients who have asthma may be advised that weight loss, in addition to improving overall health, could also improve asthma control.
• Stress and depression that not well controlled should be considered in patients who have asthma. Additional education to improve self-management and coping skills may be helpful. Referral to a child psychiatrist/psychologist may be necessary.
14. Referral to a specialist
The majority of asthmatics can be managed optimally in a primary health care facility, provided that the elements of the asthma guideline are followed. Some patients may require referral to a specialist; this is recommended if the goals of management are not achieved, or for the following reasons:
• diagnosis is in doubt
• unstable asthma
• parents or general practitioners need further support
• child on a high dose of ICS (>400 µg beclomethasone equivalent per day)
• oral steroids are required regularly
• after a life-threatening episode
• frequent hospitalisations or visits to an emergency room
• if immunotherapy or omalizumab is considered.
Disclaimer
This national clinical guideline is for reference and educational purposes only and is not intended to be a substitute for the advice of appropriate health care professionals or for independent research and judgement. Some of the medications mentioned in this guideline are used off-label but with the best evidence available.
Acknowledgements
The contribution of the following working group members is gratefully acknowledged:
Professor Cassim Motala (WC) (chairperson), Professors Robin Green (Gauteng), Paul Potter (WC) and Heather Zar (WC); Drs Andrew Halkas (Gauteng), Refiloe Masekela (Gauteng), Essop Moosa (Gauteng), Steve Ponde (Gauteng), Allan Puterman (WC), Mike Levin (WC), Di Hawarden (WC), Gloria Davis (Gauteng), Karin Simmank (Gauteng), Humphrey Lewis (Gauteng), Madeleine Mercer (WC), Sam Logedi (Gauteng), Wilhelm Karshagen (FS), Ahmed Manjra (KZN), Martin Davis (Gauteng), Fred Mokgoadi (Limpopo), Mervin Ossip (Gauteng), Farouk Jooma (KZN) and Andre van Niekerk (Gauteng).
The editors express their appreciation to Dr Sharon Kling and Professors Robert Gie, Elvis Irusen and Eugene Weinberg for reviewing the guideline document. Their constructive comments and suggestions were taken into consideration in compiling the final document. The authors also express their gratitude to Ms Debbie Paulse, Mrs Ruwayda Adams and Mrs Lindy Foot for technical support.
Guideline sponsor
The meetings of the working group were sponsored by the Allergy Society of South Africa (ALLSA).
Disclosure of potential conflict of interest
C Motala is an executive member of ALLSA and an advisory board member of The National Asthma Education Programme (NAEP), GEO Aeroallergen and Health Advisory Group, AstraZeneca, GlaxoSmithKline, MSD and Pharmaplan, and has served on the speakers bureau for GlaxoSmithKline, MSD and Schering-Plough.
R J Green is an executive member of ALLSA, the South African Thoracic Society (SATS) and the NAEP, an advisory board member of AstraZeneca, GlaxoSmithKline, MSD and Wyeth, and has served on the speakers bureau for AstraZeneca, GlaxoSmithKline, MSD and Pfizer.
A I Manjra is an executive member of ALLSA and its current chairman, an advisory board member of AstraZeneca, GlaxoSmithKline and MSD, and has served on the speakers bureau for GlaxoSmithKline, MSD and Schering-Plough.
P C Potter is a member of the board of directors of the World Allergy Organisation and serves on its Sublingual Immunotherapy committee, is an advisory board member of GlaxoSmithKline and MSD, and has served on the speakers bureau for GlaxoSmithKline and MSD.
H J Zar is an executive member and president-elect of SATS, a member of the Global Initiative for Asthma (GINA) Scientific Committee, vice-president of the Pan African Thoracic Society, and was an executive member of ALLSA until July 2009; she is an advisory board member of NAEP, GlaxoSmithKline, Novartis and Wyeth, and has served on the speakers bureau for GlaxoSmithKline, MSD and Schering-Plough.
Endorsement
This guideline has been endorsed by SATS, NAEP, SAPA and the South African Academy of Family Practice.
References
1. South African Childhood Asthma Working Group. Management of childhood and adolescent asthma - 1991 consensus. S Afr Med J 1992; 81: 38-40. [ Links ]
2. South African Childhood Asthma Working Group. Management of chronic childhood and adolescent asthma - 1994 consensus. S Afr Med J 1994; 84: 862-866. [ Links ]
3. South African Childhood Asthma Working Group. Guideline for the management of chronic asthma in children - 2000 update. S Afr Med J 2000; 90(5): 524-539. [ Links ]
4. Guyatt GH, Oxman AD, Kuaz R, et al. Going from evidence to recommendations. BMJ 2008; 336: 1049-1051. [ Links ]
5. Zar HJ, Ehrlich RI, Workman L, Weinberg EG. The changing prevalence of asthma, allergic rhinitis and atopic eczema in African adolescents from 1995 to 2002. Pediatr Allergy Immunol 2007; 18(7): 560-565. [ Links ]
6. Sly PD, Boner AL, Björksten B, et al. Early identification of atopy in the prediction of persisten asthma in children. Lancet 2008; 372(9643): 1100-1106. [ Links ]
7. Konig P. Hidden asthma in children. Am J Dis Child 1981; 135: 1053-1055. [ Links ]
8. Ninan TK, Macdonald L, Russell G. Persistent nocturnal cough in childhood: a population based study. Arch Dis Child 1995; 73: 403-407. [ Links ]
9. Martinez FD, Wright AL, Taussig LM, Holberg CJ, Halonen M, Morgan WJ. Asthma and wheezing in the first six years of life. Group Health Medical Associates. N Engl J Med 1995; 332: 133-138. [ Links ]
10. Castro-Rodriguez JA, Holberg CJ, Wright AL, Martinez FD. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med 2000; 162(4 Pt 1): 1403-1406. [ Links ]
11. Brand PLP, Baraldi E, Bisgaard H, et al. Definition, assessment and treatment of wheezing disorders in preschool children: an evidence-based approach. Eur Respir J 2008; 32: 1096-1110. [ Links ]
12. Klein M. Respiratory system disorders. In: Harrison VC, ed. Handbook of Paediatrics. Cape Town: Oxford University Press, 2004. [ Links ]
13. Beydon N, Davis JD, Lombardi E, et al. An official American Thoracic Society/European Respiratory Society statement: Pulmonary function testing in pre-school children. Am J Respir Crit Care Med 2007; 175: 1304-1345. [ Links ]
14. Platts-Mills TAE. The role of allergens in allergic airway disease. J Allergy Clin Immunol 1998; 101(2): S36. [ Links ]
15. Jooma OF, Weinberg EG, Berman D, Potter PC. Accumulation of mite Der p 1 levels on mattress covers. S Afr Med J 1995; 85: 1002-1005. [ Links ]
16. Arbes SL, Sever M, Archer L, Adler P, Sandal M, Selden DC. Abatement of cockroach antigen (Bla g 1) in low income urban housing: a randomised trial. J Allergy Clin Immunol 2003; 112: 339-345. [ Links ]
17. Arliam L, Neal J, Morgan M, Rapp CM, Clobes AL. Distribution and removal of cat, dog and mite allergens on smooth surfaces in homes with and without pets. Ann Allergy Asthma Immunol 2001; 87: 296-302. [ Links ]
18. Barnes CS, Kennedy K, Johnson L, et al. Use of dilute sodium hypochlorite spray and home cleaning to reduce indoor allergen levels and improve asthma health parameters. Ann Allergy Asthma Immunol 2008; 101(5): 551-552. [ Links ]
19. Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention. Workshop Report. 2006. http://www.ginasthma.com (accessed 18 September 2009). [ Links ]
20. Liu AH, Zeiger R, Sorkness C, et al. Development and cross-sectional validation of the Childhood Asthma Control Test. J Allergy Clin Immunol 2006; 12: 662-817. [ Links ]
21. Jones KP, Mullee MA, Middleton, Chapman E, Holgate ST. Peak flow based asthma self-management: a randomized controlled study in general practice. British Thoracic Society Research Committee. Thorax 1995; 50: 851-857. [ Links ]
22. Pijnenburg MW, Hofhuis W, Hop WC, De Jongste JC. Exhaled nitric oxide predicts asthma relapse in children with clinical asthma remission. Thorax 2005; 60: 215-218. [ Links ]
23. Pedersen S. Inhalers and nebulizers: which to choose and why. Respir Med 1996; 90(2): 69-77. [ Links ]
24. Zar HJ, Weinberg EG. Spacer devices for the developing world. ACI International 2002; 14(1): 13-16. [ Links ]
25. Cates CJ, Crilly JA, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev 2006(2): CD000052. [ Links ]
26. Castro-Rodriguez JA, Rodrigo GJ. Beta-agonists through metered-dose inhaler with valved holding chamber versus nebulizer for acute exacerbation of wheezing or asthma in children under 5 years of age: a systematic review with meta-analysis. J Pediatr 2004; 145: 172-177. [ Links ]
27. Zar HJ, Asmus MJ, Weinberg EG. A 500ml plastic bottle - an effective spacer for children with asthma. Pediatr Allergy Immunol 2002; 13(3): 217-222. [ Links ]
28. Adams NP, Bestall JC, Jones PW, Lasserson TJ, Griffiths B, Cates C. Inhaled fluticasone at different doses for chronic asthma in adults and children. Cochrane Database Syst Rev 2005(3): CD003534. [ Links ]
29. Powell H, Gibson PG. High dose versus low dose inhaled corticosteroid as initial starting dose for asthma in adults and children. Cochrane Database Syst Rev 2004(2): CD004109. [ Links ]
30. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med 2006; 354(19): 1985-1997. [ Links ]
31. Castro-Rodriguez JA, Rodrigo GJ. Efficacy of inhaled corticosteroids in infants and preschoolers with recurrent wheezing and asthma: A systematic review with meta-analysis. Pediatrics 2009; 123: e519-e525. [ Links ]
32. Murray CS, Woodcock A, Langley SJ, Morris J, Custovic A. Secondary prevention of asthma by the use of Inhaled Fluticasone propionate in Wheezy Infants (IFWIN): double-blind, randomised, controlled study. Lancet 2006; 368: 754-762. [ Links ]
33. Agertoft L, Pedersen S. Effect of long-term treatment with inhaled budesonide on adult height in children with asthma. N Engl J Med 2000; 343(15): 1064-1069. [ Links ]
34. Global Initiative for Asthma. Global strategy for diagnosis and management of asthma in children 5 years and younger. http://www.ginasthma.org (accessed 18 September 2009). [ Links ]
35. Baker JW, Mellon M, Wald J, Welch M, Cruz-Rivera M, Walton-Bowen K. A multiple-dosing, placebo-controlled study of budesonide inhalation suspension given once or twice daily for treatment of persistent asthma in young children and infants. Pediatrics 1999; 103: 414-421. [ Links ]
36. Teper AM, Kofman CD, Szulman GA, Vidarrueta SM, Maffey AF. Fluticasone improves pulmonary function in children under 2 years old with risk factors for asthma. Am J Respir Crit Care Med 2005; 171(6): 587-590. [ Links ]
37. McKean M, Ducharme F. Inhaled steroids for episodic viral wheeze of childhood. Cochrane Database Syst Rev 2000(2): CD001107. [ Links ]
38. Kaditis AG, Winnie G, Syrogiannopoulos GA. Anti-inflammatory pharmacotherapy for wheezing in preschool children. Pediatr Pulmonol 2007; 42: 407-420. [ Links ]
39. Bisgaard H, Hermansen MN, Loland L, Halkjaer LB, Buchvald F. Intermittent inhaled corticosteroids in infants with episodic wheezing. N Engl J Med 2006; 354(19): 1998-2005. [ Links ]
40. Ni Chroinin M, Lasserson TJ, Greenstone I, Ducharme FM. Addition of long-acting beta-agonists to inhaled corticosteroids for chronic asthma in children. Cochrane Database Syst Rev 2009(3): CD007949. [ Links ]
41. Bisgaard H. Effect of long-acting β2-agonists on exacerbation rates of asthma in children. Pediatr Pulmonol 2003; 36: 391-398. [ Links ]
42. Beasley R, Martinez FD, Hackshaw A, et al. Safety of long-acting β-agonists: urgent need to clear the air remains. Eur Resp J 2009; 33: 3-5. [ Links ]
43. Russell G, Williams DA, Weller P, Price JF. Salmeterol xinafoate in children on high dose inhaled steroids. Ann Allergy Asthma Immunol 1995; 75(5): 423-428. [ Links ]
44. Malone R, LaForce C, Nimmagadda S, et al. The safety of twice-daily treatment with fluticasone propionate and salmeterol in pediatric patients with persistent asthma. Ann Allergy Asthma Immunol 2005; 95(1): 66-71. [ Links ]
45. Zimmerman B, D'Urzo A, Berube D. Efficacy and safety of formoterol Turbuhaler when added to inhaled corticosteroid treatment in children with asthma. Pediatr Pulmonol 2004; 37(2): 122-127. [ Links ]
46. Verberne AAPH, Frost C, Duiverman EJ, Grol MH, Kerribijn KF. Addition of salmeterol versus doubling the dose of beclomethasone in children with asthma. Am J Respir Crit Care Med 1998; 158: 213-219. [ Links ]
47. Ortega-Cisnero M, Maldonado-Alaniz ML, Rosas Vargas MA, Sierra-Monge JJL. Salmeterol and inhaled beclomethasone versus high dose inhaled beclomethasone in the control of pediatric patients with moderate asthma. Ann Allergy Asthma Immunol 1998; 80: 131. [ Links ]
48. Israel E, Rubin P, Kemp JP, et al. The effect of inhibition of 5-lipoxygenase by zileuton in mild-to-moderate asthma. Ann Intern Med 1993; 119: 1059-1066. [ Links ]
49. Dahlen B, Margolskee DJ, Zetterstrom O, Dahlen SE. Effect of the leukotriene antagonist MK-0679 on baseline pulmonary function in aspirin sensitive asthmatic subjects. Thorax 1993; 48: 1205-1210. [ Links ]
50. Van Adelsberg J, Moy J, Wei LX, Tozzi CA, Knorr B, Reiss TF. Safety, tolerability and exploratory efficacy of montelukast in 6-24 month-old patients with asthma. Curr Med Res Opin 2005; 21: 971-979. [ Links ]
51. Storms W, Michele TM, Knorr B, et al. Clinical safety and tolerability of montelukast, A LTRA, in controlled clinical trials in patients aged 76 years. Clin Exp Allergy 2001; 31: 77-87. [ Links ]
52. Szefler SJ, Phillips BR, Martinez FD, et al. Characterization of within-subject responses to fluticasone and montelukast in childhood asthma. J Allergy Clin Immunol 2005; 115(2): 233-242. [ Links ]
53. Ostrom NK, Decotiis BA, Lincourt WR, et al. Comparative efficacy and safety of low-dose fluticasone propionate and montelukast in children with persistent asthma. J Pediatr 2005; 147(2): 213-220. [ Links ]
54. Garcia ML, Wahn U, Gilles L, Swern A, Tozzi CA, Polos P. Montelukast compared with fluticasone, for control of asthma amoung 6- to 14-year-old patients with mild asthma: the MOSAIC study. Pediatrics 2005; 116(2): 360-369. [ Links ]
55. Ng D, Salvio F, Hicks G. Anti-leukotriene agents compared to inhaled cortisteroids in the management of recurrent and/or chronic asthma in adults and children. Cochrane Database Syst Rev 2004(2): CD002314. [ Links ]
56. Kemp JP, Dochkorn RJ, Shapiro GG, et al. Montelukast once daily inhibits exercise-induced bronchoconstriction in 6- to14-year-old children with asthma. J Pediatr 1998; 133(3): 424-428. [ Links ]
57. Vidal C, Fernandez-Ovide E, Pinerio J, Nunez R, Gonzalez-Quintela A. Comparison of montelukast versus budesonide in the treatment of exercise-induced bronchoconstriction. Ann Allergy Asthma Immunol 2001; 86(6): 655-658. [ Links ]
58. Simons FE, Villa JR, Lee BW, et al. Montelukast added to budesonide in children with persistent asthma: a randomized, double-blind, crossover study. J Pediatr 2001; 138(5): 694-698. [ Links ]
59. Meyer KA, Arduino JM, Santanello NC, Knorr BA, Bisgaard H. Response to montelukast among subgroups of children aged 2 to 14 years with asthma. J Allergy Clin Immunol 2003; 111(4): 757-762. [ Links ]
60. Knorr B, Franchi LM, Bisgaard H, et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 2001; 108: E48. [ Links ]
61. Bisgaard H, Nielsen KG. Bronchoprotection with a leukotriene receptor antagonist in asthmatic preschool children. Am J Respir Crit Care Med 2000; 162: 187-190. [ Links ]
62. Hakim F, Vilozni D, Adler A, Livnat G, Tal A, Bentur L. The effect of montelukast on bronchial hyperreactivity in preschool children. Chest 2007; 131: 180-186. [ Links ]
63. Straub DA, Moeller A, Hamacher J, Wilhaber JH . The effect of montelukast on exhaled nitric oxide and lung function in asthmatic children 2-5 yrs old. Chest 2005; 127: 509-514. [ Links ]
64. Johnston NW, Mandhane PJ, Dai J, et al. Attenuation of the September epidemic of asthma exacerbations in children: a randomized, controlled trial of montelukast added to usual therapy. Pediatrics 2007; 120: 702-712. [ Links ]
65. Straub DA, Moeller A, Minocchieri S, et al. The effect of montelukast on lung function and exhaled nitric oxide in infants with early childhood asthma. Eur Respir J 2005; 25: 289-294. [ Links ]
66. Van Adelsberg J, Moy J, Wei LX, Tozzi CA, Knorr B, Reiss TF. Safety, tolerability and exploratory efficacy of montelukast in 6- to 24-month-old patients with asthma. Curr Med Res Opin 2005; 21(6): 971-979. [ Links ]
67. Wahn U, Dass SB. Review of recent results of montelukast use as a monotherapy in children with mild asthma. Clin Ther 2008; 30: 1026-1035. [ Links ]
68. Philip G, Malmstrom K, Hampel FC Jr, et al. Montelukast for treating seasonal allergic rhinitis: a randomized, double blind, placebo-controlled trial performed in spring. Clin Exp Allergy 2002; 32: 1020-1028. [ Links ]
69. Van Adelsberg J, Philip G, Pedinoff AJ, et al. Montelukast improves symptoms of seasonal allergic rhinitis over a 4-week treatment period. Allergy 2003; 58: 1268-1276. [ Links ]
70. Van Adelsberg J, Philip G, La Force CF, et al. Randomized controlled trial evaluating the clinical benefit of montelukast for treating spring seasonal allergic rhinitis. Ann Allergy Asthma Immunol 2003; 90: 214-222. [ Links ]
71. Katz RM, Rachelefsky GS, Siegel S. The effectiveness of the short- and long-term use of crystallized theophylline in asthmatic children. J Pediatr 1978; 92(4): 663-667. [ Links ]
72. Bierman CW, Pierson WE, Shapiro GG, Furukawa CT. Is a uniform round-the-clock theophylline blood level necessary for optimal asthma therapy in the adolescent patient? Am J Med 1988; 85(1B): 17-20. [ Links ]
73. Pedersen S. Treatment of nocturnal asthma in children with a single dose of sustained-release theophylline taken after supper. Clin Allergy 1985; 15(1): 79-85. [ Links ]
74. South African Childhood Asthma Working Group. Use of theophylline in childhood and adolescent asthma addendum to 1991 consensus statement. S Afr Med J 1993; 83: 913-914. [ Links ]
75. Seddon P, Bara A, Ducharme FM, Lasserson TJ. Oral xanthines as maintenance treatment for asthma in children. Cochrane Database Syst Rev 2006: CD002885. [ Links ]
76. Expert Panel Report 3. Guidelines for the Diagnosis and Management of Asthma. Full Report 2007. National Heart, Lung and Blood Institute. National Asthma Education and Prevention Programme. August 2007. NIH, NHLBI. http://www.nhlbi.nih.gov/guidelines/asthma/index.htm (accessed 12 July 2009). [ Links ]
77. Abramson MJ, Puy RM, Weiner JM. Allergen immunotherapy for asthma. Cochrane Database Syst Rev 2003; 4: CDC001186. [ Links ]
78. Di Rienzo V, Marcucci F, Puccinelli P, et al. Long lasting effect of sublingual immunotherapy in children with asthma due to house dust mites: A 10-year prospective study. Clin Exp Allergy 2003; 33(2): 206-210. [ Links ]
79. Penago M, Compelati E, Tarantini F, et al. Efficacy of sublingual immunotherapy in the treatment of allergic rhinitis in paediatric patients 3-18 years of age: A meta analysis of randomised controlled double blind trials. Ann Allergy Asthma Immunol 2006; 97: 141-148. [ Links ]
80. Silvestri M, Spaldarossa D, Buttisini E. Changes in inflammatory and clinical parameters and in bronchial hyperreactivity in asthmatic children sensitised to house dust mites following SLIT. J Invest Allergol Clin Immunol 2002; 12: 52-59. [ Links ]
81. Rienzo Di V, Pagani A, Parmiani S, Passalacqua G, Canonica G. Post marketing surveillance study of the safety of sublingual immunotherapy in paediatric patients. Allergy 1999; 54: 1220-1223. [ Links ]
82. Richeldi L, Ferrara G, Fabbri LM, Lasseron TJ, Gibson PG. Macrolides for chronic asthma. Cochrane Database Syst Rev 2005; 3: CD002997. [ Links ]
83. Boyd M, Lasserson TJ, McKean MC, Gibson PG, Ducharme FM, Haby M. Interventions for educating children who are at risk of asthma-related emergency department attendance. Cochrane Database Syst Rev 2009; 2: CD001290. [ Links ]
84. Brown G, Levin ME. Asthma education. Curr Allergy Clin Immunol 2005; 18(1): 14-15. [ Links ]
85. Bailey EJ, Cates CJ, Kruske SG, Morris PS, Brown N, Chang AB. Culture-specific programs for children and adults from minority groups who have asthma. Cochrane Database Syst Rev 2009; 2: CD006580. [ Links ]
86. Coutts JA, Gibson NA, Paton JY. Measuring compliance with inhaled medication in asthma. Arch Dis Child 1992; 67(3): 332-333. [ Links ]
87. Janson SL, McGrath KW, Covington JK, Cheng SC, Boushey HA. Individualized asthma self-management improves medication adherence and markers of asthma control. J Allergy Clin Immunol 2009; 123(4): 840-846. [ Links ]
88. Bousquet J, Khaltaev N, Cruz AA, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 Update (in collaboration with the World Health Organization, GA2LEN and Allergen) Allergy 2008; 63(Suppl 86): 8-160. [ Links ]
89. Potter PC, Carte G, Davis G, et al. Clinical management of allergic rhinitis - the Allergy Society of South Africa Consensus Update. S Afr Med J 2006; 96(12 Pt2): 1269-1272. [ Links ]
 Correspondence to:
Correspondence to:
Professor C Motala
Division of Allergy, Department of Paediatrics and Child Health, University of Cape Town and Red Cross War Memorial Children's Hospital
Cape Town. PO Box 2060, Cape Town 8000
Tel. 021 658 5305, cell 082 783 3788
e-mail cassim.motala@uct.ac.za