Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 no.11 Pretoria nov. 2009
ORIGINAL ARTICLES
Atopy in HIV-infected children in pretoria
R MasekelaI; T MoodleyII; N MahlabaIII; D F WittenbergIV; P BeckerVII; O KitchinV; R J GreenVI
IMB BCh, MMed (Paeds), Dip Allergology (SA), Cert Pulmonology (SA) Paed, FCCP. Department of Paediatrics and Child Health, University of Pretoria
IIMB ChB, FCPaed, Dip Allergology (SA). Department of Paediatrics and Child Health, University of Pretoria
IIIMedical student. Department of Paediatrics and Child Health, University of Pretoria
IVMD, FCP (SA). Department of Paediatrics and Child Health, University of Pretoria
VMB ChB, FCPaed, Dip Allergology (SA). Department of Paediatrics and Child Health, University of Pretoria
VIMB BCh, FCPaed, MMed (Paed), Dip Allergology (SA), DCH, DTM&H, PhD, FCCP. Department of Paediatrics and Child Health, University of Pretoria
VIIPhD. Biostatistics Unit, Medical Research Council, Pretoria
ABSTRACT
INTRODUCTION: The development or aggravation of a pre-existing atopic state in patients with human immunodeficiency virus (HIV) has not been thoroughly investigated in South Africa. HIV-infected adults have been shown to have a higher prevalence of atopy in some international studies, but this has not been documented in children.
METHODS: A prospective convenience sample of 50 children aged between 3 months and 12 years attending the Tshwane District Hospital Paediatric HIV clinic in Pretoria was recruited. Their personal and family histories of atopy, World Health Organization (WHO) HIV clinical staging and Centers for Disease Control (CDC) immunological staging with CD4 counts were documented. An age- and sex-matched control group of 50 HIV-negative children was included. Skin prick tests (SPTs) to identify common aeroallergens were conducted on all patients.
RESULTS: One hundred children were enrolled, with 50 in each group. Ten per cent of the HIV-infected patients compared with 16% of controls had positive SPTs to aeroallergens. A higher percentage of the HIV-infected patients had chronic rhinitis and eczema (60% and 68%, respectively). There was no relationship between CD4 count and positive SPTs (p=0.61), mean log CD4 count and presence of reported asthma (p=0.71), and CD4 count and presence of reported dermatitis (p=0.84). The CD4 count was not statistically different between children with and without a family history of atopy (p=0.68).
CONCLUSION: It appears that the stage of HIV disease does not influence the development or expression of allergy. There is a high prevalence of dermatitis and chronic rhinitis in HIVinfected children, probably not atopic in origin.
Both atopic and HIV-related diseases are common in South Africa. Atopy is a genetic predisposition to form excessive immunoglobulin E (IgE), leading to a generalised and prolonged hypersensitivity to common environmental allergens - both inhaled and dietary. Atopic individuals manifest one or more of a group of diseases, including asthma, atopic eczema, allergic rhinitis, urticaria and gastrointestinal conditions, which tend to run in families.1 These have been shown to be associated with Th2 cytokines. Regulatory T cells via interleukin-10 (IL-10), a cytokine known to play a pivotal role in the expression of specific immune pathways in a specific individual, are involved.2
The burden of human immunodeficiency virus (HIV) infection in sub-Saharan Africa is high, with 230 000 children born annually to HIV-infected mothers in this region.1 The epidemic proportions of the disease make it essential for appropriate diagnosis and management of affected children. This disease has a significant impact on mortality, with HIVinfected infant death rates as high as 130 -390 per 1 000 live births.3 The availability of highly active antiretroviral treatment (HAART) in the last decade in the developed world has had a significant effect on the survival of patients infected with HIV.4 Patients infected with HIV are therefore now demonstrating morbidity similar to other chronic conditions, including atopic conditions.
HIV is a retrovirus which, after gaining entry into the body, binds to a host of cells involved in innate and adaptive immunity.5 Resulting abnormalities affect both the cellular and humoral immune system. T-cell abnormalities that result in depletion of CD4 cells, as well as polyclonal activation of B cells with hyperglobulinaemia, are well known.6 This hyperglobulinaemia also affects IgE, with a marked elevation of this immunoglobulin. Adult studies have suggested an association between atopy and HIV;6,7 the evidence in children is scanty.
The switch from a Th1 to a Th2 cytokine profile has previously been shown to be a critical step in the progression of HIV infection to acquired immunodeficiency syndrome (AIDS) in adults.8,9 Even before depletion of CD4 cells, there is a qualitative defect in CD4 that results in loss of antigen and mitogen-induced interleukin-2 (IL-2) and interferon gamma (INF-γ) production. IL-2 and INF-γ are important cytokines in the Th1 pathway. A reduction in IL-2 results in a switch to interleukin-4 (IL-4) production, which is a critical step in the switch to a Th2-mediated response.8 IL-4 drives the development and expansion of Th2 cells and mediates downstream effector functions, such as B-cell activation, in particular increased major histocompatibility complex class II expression and isotype switching to IgE production.10 Although the exact mechanism is not well understood, a possible role of HIV antigen gp120 and HIV-1 trans-activating protein (Tat protein) is suspected. Gp120 is thought to act as a superantigen, stimulating the immune system with a bias toward Th2 cytokine production via release of IL-4 and interleukin-13 (IL-13) from human F epsilon R-positive cells (FcεR1+ cells).11 Tat protein may also act as a chemo-attractant for FcεR1+ cells and also upregulate chemokine receptor 3 (CCR3) expression.12
We aimed to study the association between atopy (sensitivity to environmental factors, i.e. allergy and clinical disease states) and HIV infection in children and the role of HIV infection on the development of allergy.
Methods
A prospective convenience sample of children aged 3 months - 12 years attending the Tshwane District Hospital Paediatric HIV Clinic was obtained. Informed consent to take part in the study was one of the inclusion criteria. All HIVinfected patients in the study were receiving antiretroviral therapy. Subjects for whom informed consent could not be obtained were excluded. Information regarding the children's personal and family histories of atopy was recorded from information provided by parents/guardians. An overview of the children's medical history and a general examination of their current state of well-being were conducted. The World Health Organization (WHO) HIV clinical staging, CD4 counts obtained by flow cytometric analysis, and evidence of atopy were also recorded.13 An age- and sex-matched control group of 50 healthy HIV-negative children attending routine follow-up at the cardiology and neurology clinics of Pretoria Academic Hospital were included. Skin prick tests (SPTs) (Alk-Abello) for common aeroallergens were conducted on all patients with negative saline and on positive (histamine-dihydrochloride 10mg/ml) controls. An induration of 3 mm or greater than the negative control was regarded as a positive result. The allergen extracts used were: Bermuda grass, five-grass mix, tree mix, dog hair dander, cat hair dander, standard mite (Dermatophagoides pteronyssinus), and cockroach (Blatana sp).
Diagnoses of asthma, allergic rhinitis and eczema were offered to respondents in the questionnaire but were neither investigated nor proven.
Statistical analysis
A Welch two-sample t-test with unequal variances was employed in the analysis of the CD4 count with regard to family history of atopy, dermatitis and asthma. A p-value of <0.05 was considered statistically significant. Approval for the study was obtained from the Research Ethics Committee of the University of Pretoria. Informed consent was obtained from all parents or guardians of the patients. Assent was also obtained from all subjects >7 years old.
Results
A total of 100 children were enrolled, with half in the study arm and half in the control group. Forty-five (90%) of the 50 HIV-infected children and 42 (84%) of the control group had a negative SPT to common aeroallergens (Tables I and II). There was no statistical difference between groups (p=0.95). The most common allergen identified was D. pteronyssinus in both groups, with 3 of the 5 HIV-infected patients being monosensitive to house dust mite (Fig. 1).

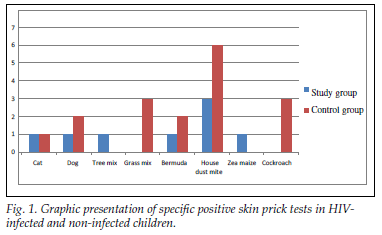
Twelve (24%) of the HIV-infected children tested had a positive family history of atopy, while only 2 of them had a reactive SPT. Eleven (22%) HIV-infected children had been diagnosed with asthma. The majority of these (9 patients) had a negative SPT. Two of the asthmatic patients were WHO HIV stage 1, and only one of them had a positive SPT.
Of the 12 HIV-infected patients who had a family history of atopy, 8 (66.6%) gave a positive history of 'allergic' rhinitis. Thirty HIV-infected children (60%) gave a positive history of allergic rhinitis. All 5 patients who had a positive SPT had allergic rhinitis. Of the children with no family history of atopy, 3 had positive SPTs; this constituted the majority of the total group (60%) with a positive SPT.
Thirty-four (68%) of the HIV-infected group had dermatitis. Of these, 10 had a family history of atopy and 10 had asthma. There was no relationship between CD4 count and SPT positivity, Welch t-test (p=0.61), logarithmic transformed CD4, and reported presence of asthma (p=0.71) (95% confidence interval (Cl): -0.472 -0.674), and log CD4 and reported presence of dermatitis (p=0.84) (95% CI: -0.556 -0.457). There was also no relationship between CD4 count and family history of atopy (p=0.63) (95% CI: -0.329 -0.532) (Table I).
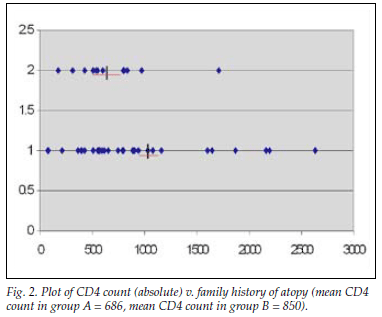
The comparison between family history of atopy and log CD4 count was subject to the Welch two-sample t-test with unequal variances, with no correlation demonstrated (p=0.68) (Fig. 2). Group A (positive family history) and group B (negative family history) did not differ significantly (p=0.63) with regard to log CD4 counts, with mean log transformed CD4 counts 6.385 (95% CI 6.017 -6.0753) and 6.487 (95% CI 6.229 -6.745), respectively. Twenty-eight (56%) of the total HIV-infected group had a CD4 <20%; 17 (35%) of these were WHO clinical stage 3 or 4. Seventeen (34%) were HIV stage 1; of these, only 2 had a family history of atopy. Only 1 of these children had a positive SPT.
Discussion
This pilot study suggests that there is no significant association between atopy and HIV-related disease, with no difference in allergy SPT positivity between HIV-infected children and a control group of healthy children. It also appears that the stage of HIV disease in HIV-infected children does not influence the development of allergy, which may be because the immune mechanisms are truly different. This is consistent with the findings by Bowser et al. in perinatally HIV-infected children.14
Ninety per cent of the HIV-infected children had negative SPTs to environmental allergens, demonstrating the absence of antigen-specific IgE to the measured allergens. Ten per cent of the HIV-infected children, who had evidence of atopy and a reactive SPT, probably have an inherent but independent genetic predisposition to atopy. In a study by Bacot et al., SPTs were positive in 28% of HIV-infected children, although adult studies report an incidence of around 10%.6 Interestingly, the most common aeroallergen in this study was D. pteronyssinus, which has been found to be present in 45% of asthmatics in Johannesburg.15
The pathogenesis of eczema is thought to be related to allergen uptake by the Langerhans cells in the skin via specific IgE bound to the high-affinity IgE receptors on cell surfaces, resulting in an allergen-specific T-cell response in memory CD4 cells. It is well known that HIV-infected patients have a higher incidence of dermatitis;16 this may present as inflammatory or eczematous eruptions. Patients with HIV have dry skins, and this barrier disruption has been postulated by Rudikoff to favour a Th2-mediated response to exogenous allergens.16 Bacot et al. found no correlation between the presence of atopic dermatitis and the level of immunosuppression in CD4 levels.6 Our study confirmed this finding.
Adult studies have, however, demonstrated chronic pruritic or eczema rashes occurring much more frequently with more severe HIV disease with CD4 counts <200/dl.17 HIV-infected patients may display inflammatory or erythematous rashes, including HIV eosinophilic folliculitis, papular urticaria, seborrhoeic dermatitis, psoriasis and pruritus nodularis, which resemble atopic dermatitis. This makes the distinction between atopic and non-atopic dermatitis difficult, particularly pruritus nodularis, which has a pruritic component.16 The presence of dermatitis in our study population was quite striking. Whether or not all these patients had eczema is difficult to delineate. All patients with a reactive SPT also had dermatitis. It therefore seems reasonable to suppose that a fair number of them were truly atopic. Other causes of dermatitis should also be included in a differential diagnosis, especially drug-related eruptions.
Sinusitis and ear infections are more common and are associated with hay fever and chronic nasal symptoms. The incidence of allergic rhinitis has been reported to be 20.7% in South Africa.17 There is a higher prevalence of rhinosinusitis related to a decrease in cellular immunity but unrelated to IgE-mediated hypersensitivity. Most patients in our group had house dust mite allergy. Evidence of causality of rhinitis in patients is complex, as most cases of rhinitis may be the result of an infection.18
The dermatitis and chronic rhinitis prevalent in these individuals is probably due to some other factor, such as infective processes or a dysfunctional immune system. Although there is a suggestion of differences in mean CD4 counts between HIV-infected individuals, with and without a family history of atopy, it is not statistically significant, with overlapping 95% CIs. This measure of atopy is not related to HIV infection.
In the International Study of Asthma and Allergy in Childhood (ISAAC), South Africa reported on asthma prevalence of 13.6% in 13 - 14-year-old children in Cape Town.19 In the current study, 22% of patients were diagnosed with asthma, suggesting a higher prevalence in HIV-infected children. However, the possibility of chronic lung disease with airway reversibility may be contributing to the higher percentage. The absence of objective lung function testing is a weakness of the study. The reasonable correlation of CD4 count with HIV stage is an expected finding, as the CD4 count forms part of this assessment.
Many studies define atopy on the basis of elevation of IgE. In South African children, the burden of parasitic infection is high. One previous study demonstrated a 44% Ascaris lumbricoides positivity in the stool of children.20 Therefore, a total IgE level in this context becomes unreliable. In a study by Koutsonikolis et al., HIV-infected children had a higher total but not specific IgE.21
Our study has several limitations. No attempt was made to assess whether reported rhinitis in patients was truly allergic via Hansel staining. No CAP RAST testing was conducted for inhaled or food allergens. Because of the limitations of using a total IgE in our context where parasitic infection accounts for elevated levels, this was not tested. This study should be regarded as a pilot study of the important association between two extremely common disease states in South Africa. Both conditions are currently experiencing a rising prevalence in the country, and it is logical to assume that they will co-exist in some individuals.
References
1. Haslett C, Chilvers ER, Boon NA, Colledge NR, eds. Davidson's Principles and Practice of Medicine. 19th ed. Edinburgh: Churchill Livingstone, 2002. [ Links ]
2. Wu K, Bi Y, Wang C. IL-10-producing type 1 regulatory T cells and allergy. Cell Mol Immunol 2007; 4(4): 269-275. [ Links ]
3. Jankelevich S. Serious bacterial infections in children with HIV. http://hivinsite.uscf.edu/Insite?=kb-05&doc=kb-05-01-01-01 (accessed 10 April 2008). [ Links ]
4. Guidelines for preventing opportunistic infections among HIV-infected persons. Mor Mortal Wkly Rep 2002; 51(RR-8): 1-51. [ Links ]
5. Hoffman C, Rockstroh JK, Kamps BS, eds. HIV Medicine. Wuppertal, Austria: Steinhauser Verlag, 2005. www.HIVMedicine.com (accessed 15 June 2008). [ Links ]
6. Bacot BK, Paul ME, Navarro M, et al. Objective measures of allergic disease in children with human immunodeficiency virus infection. J Allergy Clin Immunol 1997; 100(5): 707-711. [ Links ]
7. Vigano A, Principi N, Crupi L, et al. Elevation of IgE in HIV-infected children and its correlation with the progression of disease. J Allergy Clin Immunol 1995; 95(2): 627-632. [ Links ]
8. Empson M, Bishop AG, Nightingale B, Garsia R. Atopy, anergic status, and cytokine expression in HIV-infected subjects. J Allergy Clin Immunol 1999; 103(5): 833-842. [ Links ]
9. Clerici M, Shearer GM. The Th1-Th2 switch is a critical step in the etiology of HIV infection. Immunol Today 1993; 14: 107-111. [ Links ]
10. Liu Z, Liu Q, Pesce J, et al. Requirements for the development of IL-4 producing T cells during intestinal nematode infections: what it takes to make a Th2 cell in vivo. Immunol Rev 2004; 201: 57-74. [ Links ]
11. Patella V, Florio G, Petraroli A, Marone G. HIV-1 gp120 cytokines induces IL-4 and IL-13 release from human Fc epsilon R+ cells through interaction with VH3 region of IgE. J Immunol 2000; 164: 589-595. [ Links ]
12. de Paulis A, De Palma R, Di Giola I, et al. Tat protein is an HIV-1 encoded beta-chemokine homolog that promotes migration and up-regulates CCR3 expression on human Fc epsilon R+ cells. J Immunol 2000; 165: 7171-7179. [ Links ]
13. Interim WHO clinical staging of HIV/AIDS and HIV/AIDS case definition for surveillance. www.who.int/hiv/pub/guidelines/clinicalstaging.pdf (accessed 20 May 2008). [ Links ]
14. Bowser CS, Kaye J, Joks RO, et al. IgE and atopy in perinatally HIV-infected children. Pediatr Allergy Immunol 2007; 18: 298-303. [ Links ]
15. Luyt DK, Davis G, Dance M, Simmank K, Patel D. Clinical characteristics of black childhood asthma. S Afr Med J 1995; 85: 999-1001. [ Links ]
16. Rudikoff D. The relationship between HIV infection and atopic dermatitis. Current Allergy and Asthma Reports 2002; 2: 275-281. [ Links ]
17. Strachan DP, Sibbald B, Weiland SK, et al. Worldwide variations in prevalence of symptoms of allergic rhinoconjunctivitis in children: International Study of Asthma and Allergies in Childhood (ISAAC). Pediatr Allergy Immunol 1997; 8: 161-176. [ Links ]
18. Garcia-Rodriguez JF, Corominas M, Fernandez-Vilarich P, et al. Rhinosinusitis and atopy in patients infected with HIV. Laryngoscope 1999; 109(6): 939-944. [ Links ]
19. ISAAC Steering Committee. Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis and atopic eczema. ISAAC. Lancet 1998; 351: 1225-1232. [ Links ]
20. Obihara CC, Beyers N, Gie RP, et al. Inverse association between Mycobacterium tuberculosis infection and atopic rhinitis in children. Allergy 2005; 60: 1121-1125. [ Links ]
21. Koutsonikolis A, Nelson RP, Fernandez-Caldas E. Serum total and specific IgE levels in children with human immunodeficiency virus. J Allergy Clin Immunol 1996; 97: 692-697. [ Links ]
Accepted 6 May 2009.
Corresponding author: R Masekela (refiloe.masekela@up.ac.za)














