Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 n.11 Pretoria Nov. 2009
SCIENTIFIC LETTERS
Primary health workers' proficiency in using malaria rapid diagnostic tests in Limpopo province
Devanand MoonasarI; Ameena Ebrahim GogaIII; John FreanV; Philip KrugerVI; Rajendra MaharajIV; Daniel ChandramohanII
IMSc (Clinical Parasitology), DrPh. London School of Hygiene and Tropical Medicine, London, UK
IIMB BS, MSc, PhD. London School of Hygiene and Tropical Medicine, London, UK
IIIMB ChB, DTM&H, DCH, FCPaeds, MSc (MCH), MSc (Epidemiology). Medical Research Council of South Africa, Cape Town
IVMSc (Entomology), MSc (Infectious Diseases), PhD. Medical Research Council of South Africa, Cape Town
VMB BCh, MMed, MSc, DTM&H, FACTM, FFTM RCPS (Glasgow). National Institute for Communicable Diseases, National Health Laboratory Service, Johannesburg
VIDip Public Health. Limpopo Department of Health and Social Development, Polokwane, Limpopo
To the Editor: Malaria rapid diagnostic tests (RDTs) have been used for the diagnosis of malaria in Limpopo Province since 2001. A key determinant of the effectiveness of malaria RDTs is end-user proficiency. Since this had not previously been assessed in the area, we undertook a cross-sectional survey among randomly selected end-users of RDTs at primary health care settings in January 2007. Only 9% of participants checked the expiry date of RDTs, 32% did not add the correct volume of buffer, 24% did not wait for at least 15 minutes before reading the test result, and 24% were unable to read the test correctly. The percentage of participants who correctly interpreted RDT positive results was 85% (177/200), and the percentage who correctly interpreted RDT negative results was 96% (24/25). To achieve the full potential of RDTs as a tool for diagnosis of malaria, end-user proficiency must be improved.
Background
The misdiagnosis of malaria can lead to severe illness and death, or to unnecessary exposure to antimalarial drugs, which may lead to drug toxicity and resistance and, in turn, lack of confidence in health care services.1 Malaria RDTs provide results quickly, do not require sophisticated technology, are easy to use and therefore a useful diagnostic tool in remote areas where malaria diagnostic laboratories are sparse or nonexistent.1 However, the quality and usage of RDTs depend on several factors, including manufacturing standards, storage, transport and end-user performance.2,3
In South Africa, malaria treatment is initiated after a definitive diagnosis by microscopy or RDT. Limpopo Province, one of 3 malaria-affected provinces in South Africa, has been using RDTs for malaria diagnosis since 2001. We undertook this study as RDT end-user performance has not been rigorously investigated in the area.
Method
The study was conducted in the Vhembe District of Limpopo Province among end-users of RDTs in clinics and health centres. Based on the average annual number of malaria cases reported from 2004 to 2006, all clinics and health centres (N=50) were classified as low transmission (10 -19 malaria cases per year), medium transmission (20 -50 cases per year), and high transmission (>50 malaria cases per year) clinics. Fifty per cent of clinics and health centres (N=25) were randomly selected from each group (11/22 from low; 5/10 from medium, and 9/18 from high). From each clinic/health centre, one RDT end-user (participant) was randomly selected and their demographic and professional experience data were collected. Their ability to perform RDTs was assessed by using a nonpatient volunteer and a checklist of observations adapted from a previous study,4 and their ability to interpret test results was assessed using photographs of 10 (4 strong positive, 4 weak positive, 1 negative and 1 invalid result) pre-prepared RDT results. Ethical permission was obtained from the University of Limpopo Research Ethics Committee, the Limpopo Department of Health and Social Development, and the London School of Hygiene and Tropical Medicine.
Results
Characteristics of participants
The median age of the participants was 40; 18 were qualified nurses and 7 were nursing assistants. All participants had performed RDTs previously (median years of experience = 5) but only 18 had any experience in treating malaria. Only 24% (6 of 25) of participants had received RDT training from the malaria control programme; the balance of 76% had received in-house training from colleagues.
Adherence to RDT procedures and interpretation of RDTs from a non-patient volunteer
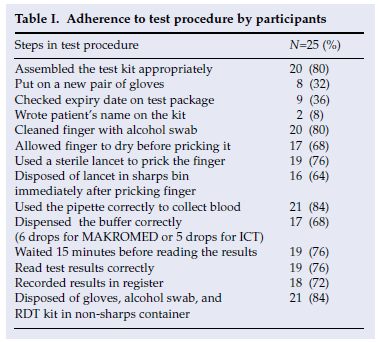
Adherence to the recommended RDT procedures was not optimal (Table I). Although 20 (80%) of participants assembled the kit properly, only 9 (36%) checked its expiry date, and 2 (8%) recorded the patient's identity on the kit. Only 19 (76%) used a sterile lancet to obtain blood; of these, 16 (84%) disposed of the lancet correctly; 8 (32%) did not add the correct volume of buffer; 24% did not wait for at least 15 minutes before reading the test results; and 24% were unable to interpret the test correctly.
Interpretation of the pre-prepared battery of tests
The ability of participants to interpret correctly the true strong or weak positive results (sensitivity) was 85% (177/200), and 96% (24/25) for correctly interpreting the true negative results (specificity). Consequently, the false negative rate was 15%, and the false positive rate was 4%. The positive and negative predictive values of the participants' interpretation of test results were 99% (177/179) and 57% (24/42) respectively.
Discussion
We showed that end-users were not totally proficient in performing and interpreting RDTs. Participants did not use the recommended sterile procedures, and many used a needle to collect blood rather than the lancet included in the kits, which is of grave concern, given the 11.1% HIV prevalence in the Vhembe district.5 Inadequate amounts of buffer were used, and results were read before the stipulated 15 minutes had elapsed. Participants who used a needle (instead of the lancet) to collect blood stated that pricking with a lancet usually yielded insufficient blood. However, we observed that many participants used too much blood for the test, which leads to false negative results, particularly among weak positives in the early stages of the infection.1 The use of inadequate amounts of buffer, and/or reading the RDT prematurely, can also produce false negative results.
The 15% false negative interpretation of the pre-prepared battery of tests was mainly misinterpretation of weak positive RDTs; participants believed that the strength of the test and the control lines on the strip should be equal to declare a test positive. Our rate of observed false negatives was similar to that found in other studies of end-users' RDT proficiency.6-8 In low malaria transmission areas such as South Africa, most early infections are symptomatic and rapidly progress to severe disease. Such early infections often have low parasite density and may yield weak positive RDT results, which end-users may interpret as negative - with harmful consequences.
The strength of our study lies in our inclusion and observation of routine health care staff in a high-risk malaria district. However, we acknowledge the following limitations: direct observation might have biased performance in either direction; photographs may not be the best gold standard to determine end-user proficiency in RDT interpretation - in particular, photographs of weak positive results might have appeared lighter than they truly were, which could have distorted end-users' interpretation of the results; and our sample size was small. Our study results must therefore be interpreted with caution. However, our sample was carefully chosen, using multi-stage stratified random sampling, and is therefore representative of RDT end-users in the study area. Moreover, our study raises similar concerns about weak positive results as other studies.6-8 Our findings may therefore be relevant to similar settings in South Africa and indeed in Africa, where RDTs are used.
RDT end-user proficiency must be improved because enduser training is critical in improving their RDT proficiency.1 The impact of job aids, such as wall charts illustrating all possible test results, and on-the-job training in performing and interpreting RDTs, needs to be evaluated1,6,7 in all settings where RDTs are used. RDT end-user proficiency should be regularly assessed, and interventions to improve proficiency should be implemented and evaluated.
References
1. World Health Organization. Malaria Rapid Diagnosis, Making it Work, RS/2003/GE/05(PHL). http://www.who.int/malaria/cmc_upload/0/000/016/750/rdt2.pdf (accessed 30 June 2006). [ Links ]
2. Bell D, Peeling RW. Evaluation of rapid diagnostic tests: malaria. Nat Rev Microbiol 2006; 4(9): Suppl S34-38. [ Links ]
3. Moonasar D, Goga AE, Frean J, Kruger P, Chandramohan D. An exploratory study of factors that affect the performance and usage of rapid diagnostic tests for malaria in the Limpopo Province, South Africa. Malar J 2007; 6: 74. [ Links ]
4. Tavrow PEA, Knebel E, Cogswell L. Using quality design to improve malaria rapid diagnostic tests in Malawi. Operations Research Results 2000; 1(4). [ Links ]
5. Statistics South Africa. Provincial Profile 2004 Limpopo. Pretoria: Statistics South Africa, 2006. [ Links ]
6. Rennie W, Phetsouvanh R, Lupisan S, et al. Minimising human error in malaria rapid diagnosis: clarity of written instructions and health worker performance. Trans R Soc Trop Med Hyg 2007; 101(1): 9-18. [ Links ]
7. Trachsler M, Schlagenhauf P, Steffen R. Feasibility of a rapid dipstick antigen-capture assay for self-testing of travellers' malaria. Trop Med Int Health 1999; 4(6): 442-447. [ Links ]
8. Funk M, Schlagenhauf P, Tschopp A, Steffen R. MalaQuick versus ParaSight F as a diagnostic aid in travellers' malaria. Trans R Soc Trop Med Hyg 1999; 93(3): 268-272. [ Links ]
Accepted 29 December 2008.
Corresponding author: D Moonasar (moonad@health.gov.za)














