Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 no.10 Pretoria Out. 2009
ORIGINAL ARTICLES
Childhood tuberculosis infection and disease: a spatial and temporal transmission analysis in a South African township
Keren MiddelkoopI; Linda-Gail BekkerII; Carl MorrowIII; Eugene ZwaneIV; Robin WoodV
IMB ChB. Desmond Tutu HIV Centre, Institute of Infectious Diseases and Molecular Medicine, and Department of Medicine, University of Cape Town
IIMB ChB, FCP, PhD. Desmond Tutu HIV Centre, Institute of Infectious Diseases and Molecular Medicine, and Department of Medicine, University of Cape Town
IIIBM, BCh, FCP, MMed. Desmond Tutu HIV Centre, Institute of Infectious Diseases and Molecular Medicine, and Department of Medicine, University of Cape Town
IVPhD. Desmond Tutu HIV Centre, Institute of Infectious Diseases and Molecular Medicine, University of Cape Town
VBA, MSc, PhD. Infectious Diseases Epidemiology Unit, School of Public Health and Family Medicine, University of Cape Town
ABSTRACT
BACKGROUND: Tuberculosis (TB) remains a leading cause of mortality and morbidity in South Africa. While adult TB results from both recent and past infection, childhood TB results from recent infection and reflects ongoing transmission despite current TB control strategies.
SETTING: A South African community with high rates of TB and HIV disease.
OUTCOMES: A Geographic Information System was used to spatially and temporally define the relationships between TB exposure, infection and disease in children <15 years of age with exposure to adult HIV-positive and HIV-negative TB disease on residential plots between 1997 and 2007.
RESULTS: During the study period the annual adult TB notification rate increased from 629 to 2 106/100 000 and the rate in children aged <15 years ranged between 664/100 000 and 1 044/100 000. The mean number of exposures to adult TB for TB-uninfected children, latently TB-infected children and TB cases were 5.1%, 5.4% and 33% per annum and the mean number of adult smear-positive cases per exposed child was 1.0, 1.6 and 1.9, respectively. Acquisition of TB infection was not associated with HIV status of the adult TB case to which the child was exposed, and 36% of child TB cases were diagnosed before the temporally closest adult case on their plot.
CONCLUSIONS: Childhood infection and disease were quantitatively linked to infectious adult TB prevalence in an immediate social network. Childhood infection should be monitored in high-burden settings as a marker of ongoing TB transmission. Improved knowledge of township childhood and adult social networks could also facilitate targeted active case finding, which may provide an adjunct to currently failing TB control strategies.
Tuberculosis (TB) is a leading cause of mortality and morbidity globally. South Africa has the largest per capita TB burden in the world, and its TB notification rates are highest in crowded urban areas.1,2 Although DOTS programmes (directly observed therapy, short course) have reduced TB prevalence and death rates in many regions of the world, implementation has not significantly impacted on TB transmission and incidence in Africa.1 Adult TB disease comprises a combination of recent infections and reactivation of latent TB, and therefore represents both recent and past TB exposure. HIV co-infection increases the progression to disease of both recent and latent TB infection.3,4 In contrast, childhood infection is acquired predominantly from adults, as child-to-child transmission is uncommon,5 and young children with TB infection have a greater risk for progression to active disease than adults.6 Infection and disease in young children are therefore a measure of recent TB transmission.
Country-specific TB rates in children are usually reported as smear-positive TB rates per 100 000 of the <15-year-old population.1 This may substantially underestimate the true child TB burden due in part to the low proportion of smear-positive cases in children.7 Furthermore, childhood TB disease occurs most frequently in children <2 years, resulting in the numerator of disease for this small age group being diluted by the larger population denominator (0 - 14-year-olds). This may explain why, despite significant increases in overall TB rates in the Africa region, the reported rates of childhood TB disease for South Africa have remained relatively unchanged since 2002.1,8
We have previously reported a growing TB epidemic in a peri-urban township heavily affected by HIV in Cape Town, South Africa. Recently urbanised, overcrowded, consisting largely of informal dwellings and with a population of low socio-economic status, the township is a well-demarcated, geographically isolated community. The HIV prevalence is 23% among adults9 and 10% among adolescents aged 11 -19 years.10 The National TB Control Programme, based on WHO DOTS recommendations, appears to be functioning well at the community's single clinic, with treatment completion and cure rates of sputum-positive cases of approximately 80%2 and case-finding rates of 67% in HIV-negative smear-positive patients.9 However, local TB notification data show a rapid escalation in TB notification rates in adults. In particular the incidence rate of smear-positive TB increased from 326/100 000 in 1996 to 1 307/100 000 in 2005.11,9 During the same period childhood TB notification rates have increased, but less markedly. We also reported a remarkably high TB prevalence rate of 26% in children aged 5 - 8 years, and a mean annual risk of TB infection in children of 4.1%.12
There are few data on TB exposure, infection and disease in children in a high TB and HIV disease burden setting. Adult TB notification data from this community and Geographic Information System (GIS) software were used to correlate the relationships between TB exposure, infection and disease in children with exposure to adult HIV-positive and HIV-negative TB disease on residential plots over time. The temporal association between adult cases and childhood disease was also explored.
Methods
The study community consists of a formal sector with demarcated, individually numbered serviced plots and an informal sector of shacks sharing communal services. Childhood TB disease cases from 1997 to 2007 were extracted from the community TB register. Childhood TB infection data were obtained from school-based tuberculin skin test (TST) surveys performed in the community in 2006 and 2007.12
The analysis was restricted to residents in the formal sector (approximately 78% of the community) who use the assigned plot number as their address; 1 - 20 houses are built on a single plot (average of 4 houses/plot). All adult TB cases notified by the community TB clinic between 1997 and 2007 were cross-referenced with both childhood TB cases and TST results from the school surveys. Children diagnosed with childhood TB disease during the study period were excluded from the TST database for this analysis. Adult TB cases were defined as patients >15 years of age and children as <15 years. Childhood TB infection was defined as an induration >10 mm in response to 2 units of purified protein derivative (PPD) administered intradermally.12 Demographic and clinical data including age, gender, TB clinical diagnosis and HIV status were collected from the TB register and clinic records. These studies were approved by the University of Cape Town's Research Ethics Committee.
Data were analysed using STATA 9.0 (StataCorp, College Station, Texas). Bivariate analyses employed Student's t-test, Wilcoxon's sum rank test and the chi-square test, as appropriate. Rates of notified child disease were calculated using population denominators from community census data obtained in 1996, and biennially from 2002 to 2008 using linear interpolation. The time difference between adult and child cases was calculated based on the year of case diagnosis. Analysis of childhood TB cases initially included all adult TB exposures on the same residential plot, followed by an analysis of the adult TB exposure with the closest temporal link to each childhood TB case. Multiple logistic regression models were developed to determine characteristics of adult TB cases that were associated with positive TST results. As the age of the child was the only known factor associated with a positive TST result in the TST sample,12 logistic regression models assessing factors associated with TST positivity included this variable and models were adjusted for clustering effect on plots. TB case characteristics were included as binary exposures (any exposure to a TB case with a particular characteristic) and as counts of exposures to a TB case with a particular characteristic, per child. All statistical tests were 2-sided at α=0.05. Average annual exposure rates for disease, infection and non-infection were calculated by dividing the percentage exposed to adult disease by the median age of the group of interest.
The ArcMap 9.2 (EsriTM) Geographic Information System was used to assess the spatial distribution of childhood TB disease, infection and exposure with linked adult cases in the community.
Results
The population childhood TB notification rate varied between 315/100 000 and 1 105/100 000 over the study period, and adult TB rates increased from 629/100 000 to 2 106/100 000. Of the 1 708 notified TB cases 1 386 (81%) were resident in the formal sector. Of these 1 386 notifications 1 212 were adult and 171 were childhood cases, with 3 cases excluded from analysis because no age was recorded. TB cases in the formal sector did not differ from those in the informal sector in terms of age (p=0.21), gender (p=0.83), site of TB disease (i.e. pulmonary v. extrapulmonary disease (EPTB); p=0.60), HIV status among those tested (p=0.58) and outcomes of TB treatment (p=0.16). However, TB cases from the formal sector were more likely to be transferred out of the community than those from the informal areas (11% v. 6% respectively, p=0.016).
Childhood TB disease and exposure to adult TB at residence
The average annual rate of childhood TB disease in the formal sector of the community was 721/100 000 for all TB notifications and 65/100 000 for smear-positive TB. The median age of the 171 childhood TB cases was 2 years (interquartile range (IQR) 1 - 6 years), and 58% were female. In total 120 (70%) of cases were classified as primary TB, with 28 cases (16%) of pulmonary TB (PTB) and 23 cases (13%) of EPTB. The HIV testing was performed in 52 children (33 positive, 19 negative). Childhood TB cases occurred on 144 plots in the community, with a range of 1 - 3 cases per plot (mean 1.19 cases per plot).
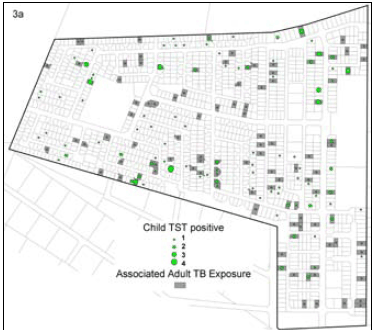
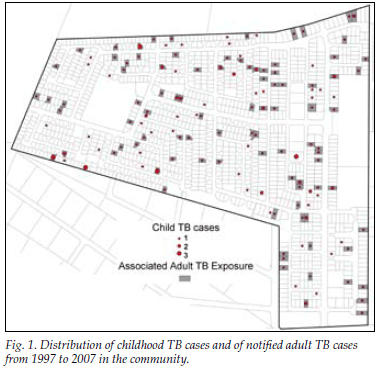
Overall 113 (66%) of the childhood TB patients had one or more exposures to a notified adult TB case on their residential plot during their lifetime, with an annual exposure rate of 33%. In 28% of cases a family name was shared with one of the adults notified on the plot. In total 105 children (61%) were exposed to adult PTB cases, 91 being exposed to a mean of 1.9 smear-positive PTB cases and 40 to smear-negative PTB cases (53% of all child TB cases v. 23%, p<0.001). Fig. 1 shows the distribution of child TB cases and adult TB cases in the community. Children who were exposed to TB cases on their residential plot did not differ from those children not exposed in terms of age (p=0.23), gender (p=0.51) or HIV status (p=0.76). However, childhood TB cases not exposed on their residential plot were more likely to be retreatment TB cases than those exposed on the plot (12% v. 4%, p=0.03).
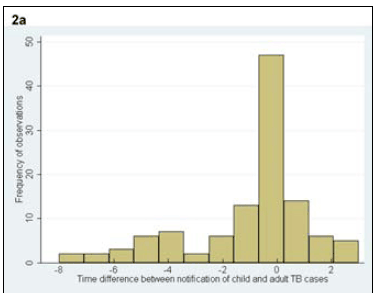
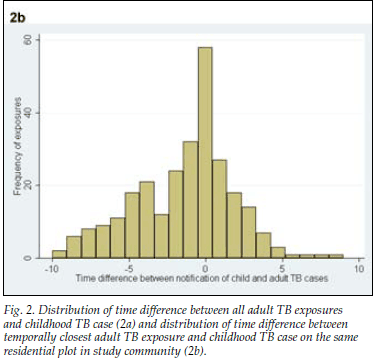
A total of 47 (42%) adult cases occurred in the same year as the child cases, 25 (22%) occurred before the child case and 41 (36%) occurred after the child case. When restricting the analysis to the adult TB case most closely linked temporally to each child case, the time interval between cases varied from 3 years before the child TB case to 8 years after the child TB case (Fig. 2, a). The disease characteristics of the closest adult TB cases by time are shown in Table I. The adult cases that occurred in the same year, before the child TB case and after the child case, did not differ from each other with respect to age (p=0.91), gender (p=0.55), HIV status (p=0.87) or treatment outcome (p=0.40). The time interval between diagnosis of all adult exposure cases and the child case ranged from 9 years before the diagnosis of the child TB case to 10 years after (median 1 year after child case; IQR 1 year before to 4 years after) (Fig. 2, b).
Childhood TB infection and exposure to adult TB at residence
Of the 831 children analysed in the TST survey, 651 (78%) lived in the formal sector and 640 (77%) of these had not had childhood TB. The subset of 640 children used in this study did not differ from the total TST study sample in terms of age (p=0.95), gender (p=0.25), BCG status (p=1.00) or positive TST reaction (p=0.69).
Of the 640 formal sector TST survey participants, 359 (56%) had been exposed to an adult TB case on their plot, and the proportion exposed was significantly higher among TST-positive compared with TST-negative children (65 v. 51%; p=0.001). The average annual exposure rate for children with infection was 5.4% per annum and for children without infection 5.1% per annum. Table II shows the distribution of type of adult TB case by TST result.
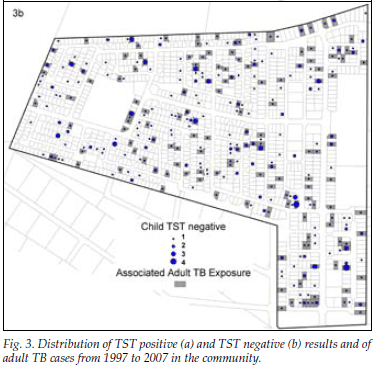
Fig. 3 (a and b) show the distribution of adult TB cases with TST-positive and TST-negative results, respectively. The odds of a positive TST result was 71% higher in those children exposed to any TB case on their plot (odds ratio (OR) 1.71; 95% confidence interval (CI) 1.20 - 2.44), 88% higher in those exposed to PTB (OR 1.88, 95% CI 1.32 - 2.68), and 110% higher in those exposed to smear-positive PTB disease (OR 2.10, 95% CI 1.46 - 3.01) compared with no plot-based exposure to adult TB. Exposure to smear-negative TB was not associated with a positive TST result (OR 0.95, 95% CI 0.59 - 1.54), and this finding persisted after adjusting for HIV status of notified adult TB cases (OR 0.68, 95% CI 0.21 - 2.18).
Multiple exposures increased risk further, and the odds of a positive TST result increased by 60% for every additional exposure to a smear-positive PTB adult (OR 1.60, 95% CI 1.21 - 2.11). The mean number of adult smear-positive exposures for TST-positive children was significantly higher than for TST-negative children (1.6 v. 1.0, p<0.001).
Discussion
We have shown that children in this community have extremely high exposure to adult TB on their residential plot. The strongest association of childhood TB infection and disease was with adult cases of smear-positive PTB rather than smear-negative PTB or EPTB. There was no independent risk of infection associated with exposure to adults with HIV-associated TB, in keeping with the previous finding in this community that child TB notification rates have not increased parallel with adult HIV/TB notification rates.12 It is therefore probable that the HIV epidemic may indirectly impact on childhood TB disease rates by increasing the burden of adult smear-positive TB cases in children's immediate social network.11
While TST surveys can assess rates of TB infection in children, our residential plot analysis was unique in allowing an assessment of annual rate of exposure to adult TB cases. The average annual exposure to adult TB case on residential plots was high for all children living on serviced plots in this community (5.1%), but was extremely high for children with TB disease (33% per annum). These exposure rates probably drive the high rates of child TB infection and disease in this community. By age of 6 years, at school entry, approximately 26% of children are latently infected with TB12 and most adolescents are infected before sexual debut.12,10 The average annual TB notification rate for children under 15 years in this community was 721/100 000 and their smear-positive rates were nearly 3-fold higher than the South African national rates (83 v. 30/100 000).1 The very high rate of HIV-related TB in young adulthood may also result from the high proportion of young adolescents latently infected with TB.
The residential plots contain multiple households, which share communal water and sanitation services and constitute a geographically defined unit for social interaction between adults and children outside the immediate household. Family names differed between children and all their adult exposures in 72% of cases.
This study was restricted to the formally serviced sector in which 78% of the community is resident. We could not define the geographical area of interaction between adults and children living in the informal sectors, and shack identifiers in this sector were not constant over the study period. For this analysis we also assumed that the children had all lived in the community since 1997 or from birth. Some children may have moved into the community subsequent to these dates; however, this would have decreased the strength of association between adult and child TB exposures and resulted in an under-estimation, rather than over-estimation, of the contact rate between adult and children with TB infection and disease.
The majority of childhood TB disease (64%) was diagnosed in the same year or soon after an index adult TB case was notified to the TB control programme. However, over a third of child TB cases in this study were notified before the temporally closest notified adult case on their plot. Adult cases were diagnosed as much as 8 years after the childhood case of TB. These more temporally distant cases probably represent secondary or even tertiary cases due to ongoing adult-to-adult transmission. Potential explanations for this delayed adult presentation include a primary source that may have remained subclinical, who subsequently moved off the plot, or died before diagnosis. However, these data illustrate a significant prevalence of undetected and untreated adult TB on these plots.
Molecular epidemiology from this community shows that adult-to-adult transmission occurs predominantly outside the residential plot.13 The use of molecular epidemiology among children is complicated by the low frequency of smear- and culture-negative disease. However, we have demonstrated that child TB infection and disease rates are strongly associated with exposure to adult TB within their restricted social environment as defined by their residential plot. The high rates of child TB infection and disease therefore indicate failure to control prevalent infectious adult cases. To reduce TB exposure, enhanced and active case finding (ACF) must be combined with the DOTS-based programme. The yield of ACF will vary in different settings. A random, community-based survey in this community showed that 0.1% of the adult population had undiagnosed smear-positive TB and an equal proportion had undiagnosed culture-positive TB, similar to the range of 0.1 - 0.2% in case finding surveys in other developing countries.14,15 We postulate that ACF targeted at residential plots, rather than only households, may have a higher yield than these cross-sectional surveys and may decrease the prevalence of infectious cases, resulting in a lower rate of transmission to children. In addition to the TB case detection yield, efficacy of such a targeted intervention over time could be assessed through repeated TST surveys in children, to ascertain the impact on childhood infection rates.
In conclusion, TB transmission within households and close social networks is an important component of both childhood infection and disease in this community. Childhood TB is a sentinel of infectious adult prevalence and therefore childhood infection and disease rates need to be monitored in these high prevalence settings in order to ascertain the true burden of infection and disease. Targeted ACF, aimed at close social networks of child TB cases may be an appropriate intervention in this setting.
Financial support. National Institutes of Health (Comprehensive Integrated Programme of Research on AIDS) grant 1U19AI053217 to KM, LGB and RW), and NIH CIPRA grant 1U19AI05321 and NIH RO1 grant AI058736-02 to RW.
Potential conflicts of interest. All authors no conflicts.
References
1. World Health Organization. Global tuberculosis control: surveillance, planning, financing. 2008. http://www.who.int/tb/publications/global_report/2008/download_centre/en/index.html (accessed 10 February 2009). [ Links ]
2. Cape Town TB Control. Progress Report 1997-2003. Cape Town: Health Systems Trust, 2004. www.hst.org.za./publications/618 (accessed 19 January 2009). [ Links ]
3. Selwyn P A, Hartel D, Lewis VA, et al. A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection. N Engl J Med 1989; 320(9): 545-550. [ Links ]
4. Badri M, Ehrlich R, Wood R, Pulerwitz T, Maartens G. Association between tuberculosis and HIV disease progression in a high tuberculosis prevalence area. Int J Tuberc Lung Dis 2001; 5(3): 225-232. [ Links ]
5. Loeffler AM. Pediatric tuberculosis. Semin Respir Infect 2003; 18(4): 272-291. [ Links ]
6. Starke JR, Jacobs RF, Jereb J. Resurgence of tuberculosis in children. J Pediatr 1992; 120(6): 839-855. [ Links ]
7. Feja K, Saiman L. Tuberculosis in children. Clin Chest Med 2005; 26(2): 295-312, vii. [ Links ]
8. World Health Organization. Global tuberculosis control: surveillance, planning, financing. 2004. http://www.who.int/tb/publications/global_report/2004/en/index.html (accessed 20 February 2009). [ Links ]
9. Wood R, Middelkoop K, Myer L, et al. Undiagnosed tuberculosis in a community with high HIV prevalence: implications for tuberculosis control. Am J Respir Crit Care Med 2007; 175(1): 87-93. [ Links ]
10. Jaspan HB, Berwick JR, Myer L, et al. Adolescent HIV prevalence, sexual risk, and willingness to participate in HIV vaccine trials. J Adolesc Health 2006; 39(5): 642-648. [ Links ]
11. Lawn SD, Bekker LG, Middelkoop K, Myer L, Wood R. Impact of HIV infection on the epidemiology of tuberculosis in a peri-urban community in South Africa: the need for age-specific interventions. Clin Infect Dis 2006; 42(7): 1040-1047. [ Links ]
12. Middelkoop K, Bekker LG, Myer L, Dawson R, Wood R. Rates of tuberculosis transmission to children and adolescents in a community with a high prevalence of HIV infection among adults. Clin Infect Dis 2008; 47(3): 349-355. [ Links ]
13. Middelkoop K, Bekker LG, Mathema B, et al. Molecular epidemiology of Mycobacterium tuberculosis in a South African community with high HIV prevalence. J Infect Dis (in press). [ Links ]
14. Narang P, Tyagi NK, Mendiratta DK, Jajoo UN, Bharambhe MS, Nayar S. Prevalence of sputum-positive pulmonary tuberculosis in tribal and non-tribal populations of the Ashti and Karanja tahsils in Wardha district, Maharashtra State, India. Int J Tuberc Lung Dis 1999; 3(6): 478-482. [ Links ]
15. Demissie M, Zenebere B, Berhane Y, Lindtjorn B. A rapid survey to determine the prevalence of smear-positive tuberculosis in Addis Ababa. Int J Tuberc Lung Dis 2002; 6(7): 580-584. [ Links ]
Accepted 9 August 2009.
Corresponding author: K Middelkoop (keren.middelkoop@hiv-research.org.za)