Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 no.5 Pretoria Mai. 2009
ORIGINAL ARTICLES
Associations between body mass index and serum levels of C-reactive protein
Tung-Wei KaoI; I-Shu LuIII; Kuo-Chen LiaoV; Hsiu-Yun LaiVI; Ching-Hui LohII; Hsu-Ko KuoIV, *
IMD, Department of Family and Community Medicine, Tri-Service General Hospital; School of Medicine, National Defense Medical Center, Taipei, Taiwan
IIMD, PhD, Department of Family and Community Medicine, Tri-Service General Hospital; School of Medicine, National Defense Medical Center, Taipei, Taiwan
IIIMD, Departments of Internal Medicine and Geriatrics and Gerontology, National Taiwan University Hospital, Taipei, Taiwan
IVMD, MPH, Departments of Internal Medicine and Geriatrics and Gerontology, National Taiwan University Hospital, Taipei, Taiwan
VMD, Division of General Medicine and Geriatrics, Department of Internal Medicine, Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Taoyuan, Taiwan
VIMD, Center for Geriatrics and Gerontology, Taipei Veterans General Hospital, Taipei, Taiwan
ABSTRACT
BACKGROUND: Obesity leads to increased risk of cardiovascular disease and glucose intolerance, which are phenomena of chronic inflammation. This study was performed to determine whether a higher body mass index (BMI) and central obesity are associated with low-grade inflammation.
METHODS: An analysis of 8 453 adults aged >20 years was performed. Every subject completed a household interview and a questionnaire regarding personal health, and their BMI and serum C-reactive protein (CRP) level were measured. The BMI data were divided into quintiles, using multiple linear regression to estimate the relationship between CRP level and BMI quintiles. An extended-model approach was used for covariate adjustment. The association between central obesity and CRP level was examined by this method as well.
RESULTS: After controlling for demographics, chronic diseases, health behaviours and levels of folate and vitamin B12, the β coefficient (which represents the change of natural-log-transformed levels of CRP for each kg/m2 increase in BMI) was 0.078 (p<0.001). The CRP levels also increased across increasing quintiles of BMI (p for trend <0.001). The β coefficient, representing the change of natural-log-transformed levels of CRP comparing subjects with central obesity to those without, was 0.876 (p<0.001).
CONCLUSION: Higher BMIs as well as central obesity are independently associated with higher levels of CRP.
C-reactive protein (CRP) (an acute phase protein) has been thought to be synthesised in the liver, with a plasma half-life of 18 hours.1 However, the extrahepatic expression of CRP has been detected in macrophages and smooth-muscle cells from atherosclerotic plaques.2 It plays an important role in the inflammatory process and is recognised as a useful biochemical marker of inflammation. Increasing epidemiological evidence supports the notion that low-grade inflammation, as reflected by elevated levels of CRP, is associated with glucose intolerance3 and various vascular diseases4-7 including atherosclerosis, stroke, ischaemic heart disease, and peripheral vascular disease. Evidence-based systemic reviews have suggested that elevated CRP is related to an increased risk of stroke and cognitive impairment.8
There is evidence for the presence of CRP in human adipose tissue9,10 and growing evidence that adipose tissue can induce chronic low-grade inflammation by producing pro-inflammatory cytokines such as interleukin-6.11 Overweight or obese individuals have an increased risk of developing cardiovascular diseases12 and insulin resistance.13 A certain degree of inflammatory process in subjects with obesity could be suspected, but data examining the direct association between body mass index (BMI) and CRP is sparse. This study aimed to evaluate whether higher BMI and central obesity are associated with low-grade systemic inflammation as measured by serum CRP levels, using data from the National Health and Nutrition Examination Survey (NHANES), 1999 - 2002.
Materials and methods
Participants
The National Health and Nutrition Examination Survey (NHANES) is a population-based survey to collect information on the health and nutrition of the USA's domestic population. NHANES used a stratified, multistage and cluster sampling design to obtain a representative sample of the non-institutionalised civilian USA population by conducting detailed home interviews and health examinations in a mobile examination centre. Since 1999, NHANES became a continuous annual survey rather than its past form of a periodic survey. Our study population comprised adults aged >20 years who participated in the NHANES 1999 - 2002. Detailed survey operations manuals, consent documents and brochures of NHANES 1999 - 2002 are available on its website.14,15
A total of 10 291 participants aged >20 years were included. Body measurements in some participants could not be obtained because of safety concerns, refusals or physical limitations (such as the participant being confined to a wheelchair); of these, 1 402 were excluded from the analysis because of missing BMI values (N=1 227) and waist measurements (N=175), and 436 were excluded because of unavailable data on CRP. Therefore, the final sample comprised 8 453 adults who completed a household interview and laboratory and clinical examinations.
Body measurements
Generally, height and weight were not obtained for subjects using a wheelchair in the mobile examination centre. Height was measured with a stadiometer to the nearest 0.1 cm. Weight was measured with a Toledo digital scale and recorded to the nearest 0.01 kg. BMI was calculated as mass in kilograms divided by the square of height in metres. Waist circumference was measured on the horizontal plane at the level of the uppermost border of the bilateral iliac crests.
C-reactive protein measurements
Blood specimens were collected at the mobile examination centres and analysed at the Department of Laboratory Medicine, University of Washington. CRP was analysed by a highly sensitive assay technique. Standard phlebotomy techniques were used to obtain specimens, which were held at –20°C until used for laboratory analysis. CRP was quantified by utilising latex-enhanced nephelometry with a Behring nephelometer (Deerfield, USA). Results were reported to the nearest hundredth (0.01) unit. The lowest reportable CRP result is approximately 0.02 mg/dl; this varies slightly with different calibrator lots. The assay does not have a maximum reportable limit since the instrument automatically prepares a higher dilution and retests specimens with results above the linearity of the assay to obtain reactions within the linear range for the assay. Detailed specimen collection and processing instructions are discussed in the NHANES Laboratory Procedures Manual.
Covariates
Age, sex, race/ethnicity, and smoking status were self-reported. Diabetes was defined by the self-reporting of a physician's diagnosis, or random plasma glucose >200 mg/dl, or the use of diabetic medications (including insulin injection or oral hypoglycaemic agents). Three, and sometimes four, blood pressure (BP) tests were done, using a mercury sphygmomanometer, by a NHANES physician. BP was measured via the right arm unless specific conditions prohibited its use. Average systolic and diastolic BPs were obtained. The presence of hypertension was defined by a self-reported doctor's diagnosis, the use of antihypertensive medications, or average blood pressure >140/90 mmHg. Medical histories of myocardial infarction (>6 weeks), congestive heart failure, angina, chronic bronchitis, emphysema and arthritis were ascertained by self-report. Alcohol intake was determined by the question: 'In any one year, have you had >12 drinks of any type of alcohol beverage?' Serum folate levels were measured using a commercially available radioprotein binding assay kit, the Quantaphase II Folate/vitamin B12 radio-assay kit (Bio Rad Laboratories, Hercules, CA, USA). Detailed specimen collection and processing instructions are in the NHANES Laboratory Procedures Manual and on their website.15
Statistical analysis
The distributions of CRP serum levels in the population were right-skewed. Therefore, the values of CRP were natural-log-transformed, providing a best-fitting model for analysis in which the serum CRP levels were treated as a continuous variable. We used multiple linear regression to determine the change of natural-log-transformed levels of CRP for each kg/m2 increase in BMI, and quintile-based analysis by dividing BMI into quintiles with the subjects in the lowest BMI quintile as the reference group. The cut-off levels for BMI quintiles were: Q1 – BMI <23.32 kg/m2; Q2 – BMI >23.32 - 26.07 kg/m2; Q3 – BMI >26.08 - 28.73 kg/m2; Q4 – BMI >28.74 - 32.53 kg/m2; Q5 – BMI >32.53 kg/m2. The β coefficients were interpreted as the differences in mean natural-log-transformed CRP levels comparing subjects in the upper four BMI quintiles with those in the lowest quintile. Central obesity was defined as waist circumference >102 cm in males, and >88 cm in females. The β coefficients were interpreted as the differences in mean natural-log-transformed CRP levels comparing subjects with central obesity with those without. An extended-model approach was used for covariate adjustment: model 1= age, gender, race; model 2 = model 1 plus levels of vitamin B12 and folate; model 3 = model 2 plus chronic diseases (hypertension, diabetes mellitus, heart disease, stroke) and health behaviours (smoking status and alcohol consumption). All analyses were conducted using STATA version 8.0 (Stata Corporation, College Station, TX, USA).
Results
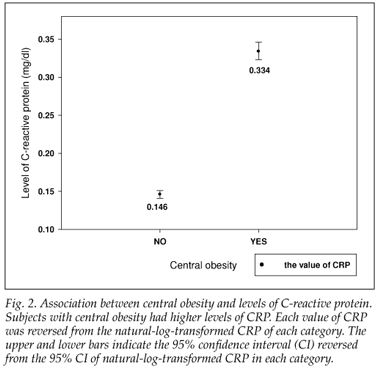
The characteristics of the study subjects are summarised in Table I. Concerning chronic diseases, 38.9% of the subjects were hypertensive and 11.4% had diabetes mellitus; 49.1% were non-Hispanic Caucasian; and participants with higher BMIs tended to have a high level of CRP.
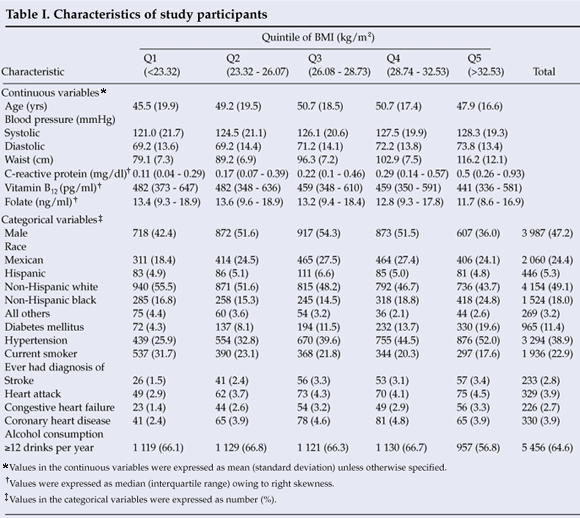
In the linear model, CRP levels positively correlated with BMI. After adjusting for age, gender and race (model 1), the β coefficient, representing the change of natural-log-transformed levels of CRP for each kg/m2 increase in BMI, was 0.080 (p<0.001) (Table II). The correlation remained unchanged after additionally adjusting for other covariates in models 2 and 3 (Table II). The results of BMI quintile-based multiple logistic regression analysis are shown in Table III. From model 1 to model 3, we observed positive correlations between BMI and CRP levels. Subjects in the higher quintiles of BMI tended to have higher CRP levels (Fig. 1). The trends of CRP levels across BMI quintiles were all statistically significant.


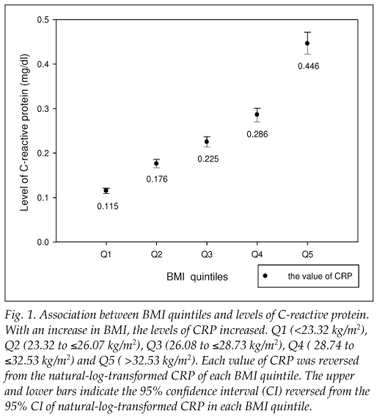
CRP levels were higher in subjects with central obesity (Fig. 2). After controlling for age, gender and race, the β coefficient, reflecting the change of natural-log-transformed levels of CRP comparing subjects with central obesity with those without, was 0.876 (p<0.001). Additional adjustment of covariates did not change the correlation (Table IV).

Discussion
This study demonstrates a positive correlation between BMI and serum CRP, supporting and extending previous studies showing that elevated plasma levels of CRP are associated with obesity.16, 17 Unlike this study, previous studies had weaknesses in terms of weight categorisation, with strikingly uneven subject numbers among different weight groups.16,17 Stratifying the BMIs into quintiles, we tried to quantify the correlation between BMI and inflammation, and to investigate whether fat distribution, especially abdominal girth, is independently associated with CRP levels. Our finding that subjects with central obesity seemed to have higher CRP levels is compatible with other reports suggesting that a high waist-to-hip ratio and increased abdominal visceral fat have links with insulin resistance, hypertriglyceridaemia, hypertension, dyslipidaemia18 and chronic low-grade inflammation.16,18 Combining our observations with those in previous reports, it can be concluded that the high prevalence of chronic diseases among overweight and obese subjects – including insulin resistance, diabetes and cardiovascular disease – may be explained by the finding of higher CRP levels, indicating that a certain degree of inflammation is present in these populations.
Subjects with obesity tend to have more abdominal visceral adipocytes, which are supposed to produce almost 25% of systemic interleukin-6 in vivo;19 and CRP could be stimulated by cytokines such as interleukin-1-beta, interleukin-6 and tumor necrosis factor-α. Consequently, adipose tissue could play a role in the regulation of circulating CRP levels via interleukin-6 production; this may partially explain our study's findings of why subjects with central obesity have higher serum levels of CRP.
Our study has the following implications: firstly, subjects with a higher BMI tend to have higher CRP levels, indicating that overweight or obese adults are vulnerable to adverse outcomes of chronic inflammation such as myocardial infarction and stroke. Weight reduction has been demonstrated to reduce CRP20 or interleukin-6 levels.21 Aggressive weight control could potentially minimise the threat of cardiovascular diseases. Further prospective studies to investigate the influence of weight reduction, lifestyle modifications and weight control agents on inflammatory markers are necessary. The second implication is that a strong correlation exists between central obesity and CRP levels, after adjusting for possible confounding factors. Abdominal girth is viewed as one of the criteria for metabolic syndrome, and reduction of waist circumference is considered to be crucial in reducing the risk of inflammation.
There are some limitations to our study. Because of the cross-sectional design, causality between obesity and serum CRP level could not be established. Additional data regarding interleukin-6 or tumour necrosis factor-α is absent from NHANES. Since these factors play major roles in the inflammatory process, a concrete association between interleukin-6, tumour necrosis factor-α and weight status could possibly be obtained from further examination.
Conclusion
Higher BMIs are associated with higher CRP levels in this population-based cross-sectional study. Subjects with central obesity tend to have higher levels of CRP independent of major confounding variables. Aggressive weight reduction could be a worthwhile intervention to reduce inflammation-associated adverse outcomes in overweight and obese subjects.
References
1. Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest 2003; 111: 1805-1812. [ Links ]
2. Yasojima K, Schwab C, McGeer EG, McGeer PL. Generation of C-reactive protein and complement components in atherosclerotic plaques. Am J Pathol 2001; 158: 1039-1051. [ Links ]
3. McMillan DE. Increased levels of acute-phase serum proteins in diabetes. Metabolism 1989; 38: 1042-1046. [ Links ]
4. Ridker PM, Buring JE, Shih J, Matias M, Hennekens CH. Prospective study of C-reactive protein and the risk of future cardiovascular events among apparently healthy women. Circulation 1998; 98: 731-733. [ Links ]
5. Koenig W, Sund M, Frohlich M, et al. C-Reactive protein, a sensitive marker of inflammation, predicts future risk of coronary heart disease in initially healthy middle-aged men: results from the MONICA (Monitoring Trends and Determinants in Cardiovascular Disease) Augsburg Cohort Study, 1984 to 1992. Circulation 1999; 99: 237-242. [ Links ]
6. Ridker PM, Cushman M, Stampfer MJ, Tracy RP, Hennekens CH. Plasma concentration of C-reactive protein and risk of developing peripheral vascular disease. Circulation 1998; 97: 425-428. [ Links ]
7. Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med 2000; 342: 836-843. [ Links ]
8. Kuo HK, Yen CJ, Chang CH, Kuo CK, Chen JH, Sorond F. Relation of C-reactive protein to stroke, cognitive disorders, and depression in the general population: systematic review and meta-analysis. Lancet Neurol 2005; 4: 371-380. [ Links ]
9. Ouchi N, Kihara S, Funahashi T, et al. Reciprocal association of C-reactive protein with adiponectin in blood stream and adipose tissue. Circulation 2003; 107: 671-674. [ Links ]
10. Calabro P, Chang DW, Willerson JT, Yeh ET. Release of C-reactive protein in response to inflammatory cytokines by human adipocytes: linking obesity to vascular inflammation. J Am Coll Cardiol 2005; 46: 1112-1113. [ Links ]
11. Bastard JP, Jardel C, Delattre J, Hainque B, Bruckert E, Oberlin F. Evidence for a link between adipose tissue interleukin-6 content and serum C-reactive protein concentrations in obese subjects. Circulation 1999; 99: 2221-2222. [ Links ]
12. Kim KS, Owen WL, Williams D, Adams-Campbell LL. A comparison between BMI and Conicity index on predicting coronary heart disease: the Framingham Heart Study. Ann Epidemiol 2000; 10: 424-431. [ Links ]
13. Boden G. Free fatty acids-the link between obesity and insulin resistance. Endocr Pract 2001; 7: 44-51. [ Links ]
14. National Center for Health Statistics. National Health and Nutrition Examination Survey (NHANES), 1999-2000. http://www.cdc.gov/nchs/about/major/nhanes/nhanes99_00.htm (accessed 20 July 2007). [ Links ]
15. National Center for Health Statistics. National Health and Nutrition Examination Survey (NHANES), 2001-2002. http://www.cdc.gov/nchs/about/major/nhanes/nhanes01_02.htm (accessed 20 July 2007). [ Links ]
16. Visser M, Bouter LM, McQuillan GM, Wener MH, Harris TB. Elevated C-reactive protein levels in overweight and obese adults. JAMA 1999; 282: 2131-2135. [ Links ]
17. Ford ES. Body mass index, diabetes, and C-reactive protein among U.S. adults. Diabetes Care 1999; 22: 1971-1977. [ Links ]
18. Lafontan M, Berlan M. Do regional differences in adipocyte biology provide new pathophysiological insights? Trends Pharmacol Sci 2003; 24: 276-283. [ Links ]
19. Mohamed-Ali V, Goodrick S, Rawesh A, et al. Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J Clin Endocrinol Metab 1997; 82: 4196-4200. [ Links ]
20. Heilbronn LK, Noakes M, Clifton PM. Energy restriction and weight loss on very-low-fat diets reduce C-reactive protein concentrations in obese, healthy women. Arterioscler Thromb Vasc Biol 2001; 21: 968-970. [ Links ]
21. Gallistl S, Sudi KM, Aigner R, Borkenstein M. Changes in serum interleukin-6 concentrations in obese children and adolescents during a weight reduction program. Int J Obes Relat Metab Disord 2001; 25: 1640-1643. [ Links ]
Accepted 19 January 2009.
* Corresponding author: Hsu-Ko Kuo (hsukokuo@yahoo.com)














