Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 n.3 Pretoria Mar. 2009
ORIGINAL ARTICLES
Antimicrobial susceptibility of methicillin-resistant Staphylococcus aureus isolates from South Africa
E MaraisI; N AithmaII; O PerovicIII; W F OosthuysenIV; E MusengeV; A G DuséVI
IBSc, BSc (Hons), MSc, PhD. Department of Clinical Microbiology and Infectious Diseases, University of the Witwatersrand and National Health Laboratory Services, Johannesburg
IIMD, DTM&H, FCPath (SA) Micro, MMed (Micro). Department of Clinical Microbiology and Infectious Diseases, University of the Witwatersrand and National Health Laboratory Services, Johannesburg
IIIBSc, BSc (Hons), MSc (Med). Department of Clinical Microbiology and Infectious Diseases, University of the Witwatersrand and National Health Laboratory Services, Johannesburg
IVMB BCh, DTM&H, MMed (Micro), FCPath (SA) Micro. Department of Clinical Microbiology and Infectious Diseases, University of the Witwatersrand and National Health Laboratory Services, Johannesburg
VMT. National Health Laboratory Services, Johannesburg
VIBSc, PGD Stats, MA, MSc (Med). Epidemiology Data Centre, University of the Witwatersrand, Johannesburg
ABSTRACT
OBJECTIVE: Trends in the antibiotic susceptibility of methicillinresistant Staphylococcus aureus (MRSA) are regularly investigated in many countries, but minimal countrywide data are available for South Africa. The aim of this study was to describe the antibiotic susceptibility patterns of MRSA isolates collected in South Africa.
DESIGN: Susceptibility testing of 248 MRSA isolates collected from 15 National Health Laboratory Services (NHLS) and 8 private laboratories against 17 antibiotics was performed using the disc diffusion method. Demographic data were collected and correlated with antibiotic resistance patterns.
RESULTS: Antibiotic resistance of MRSA to erythromycin, tetracycline, trimethoprim/sulfamethoxazole, gentamicin and ciprofloxacin ranged between 55% and 78%, while all isolates were susceptible to teicoplanin, linezolid, vancomycin and quinopristin/dalfopristin. A significant difference in the resistance pattern of the isolates towards certain antimicrobial agents was identified among adults and children, as well as between isolates collected from the private and NHLS laboratories.
CONCLUSION: This is the first extensive report on the antimicrobial susceptibilities of South African MRSA isolates. These data can assist treatment decisions and form a baseline for further surveillance.
Staphylococcus aureus is a Gram-positive bacterium frequently implicated in a variety of infectious processes ranging from relatively benign skin infections to life-threatening systemic illnesses. The organism possesses several properties that contribute to its ability to cause serious disease, including the production of toxins. Since S. aureus is a significant cause of nosocomial and community-acquired infections, the rising rates of antibiotic resistance that have been noted globally are of concern.
Methicillin resistance in S. aureus is primarily due to the expression of penicillin-binding protein 2a (PBP2a/ PBP2â²) which is encoded by the mecA gene of the mobile staphylococcal chromosomal cassette (SCCmec). Several SCCmec types exist that may encode resistance genes to other antibiotics as well. In addition to hospital-acquired methicillinresistant S. aureus (MRSA) (HA-MRSA), community-acquired MRSA (CA-MRSA) has increasingly been found to cause severe disease.
Information on the antibiotic susceptibility patterns of MRSA isolates is important for tracking the trends in antimicrobial resistance, for treatment approaches, and for establishing effective infection control measures. Baseline data from the Pan-European Antimicrobial Resistance Using Local Surveillance (PEARLS) study of 2001 - 2002 showed that South Africa had a MRSA prevalence of 33.3%.1 A more recent survey of patients with S. aureus bacteraemia at two academic hospitals in Johannesburg identified a MRSA rate of 23%.2 Data on the antibiotic susceptibility of MRSA isolates in South Africa are limited to a recent study of 61 isolates from KwaZulu-Natal (KZN)3 and from international studies performed several years ago.1,4
The goal of our study was to describe the antibiotic susceptibility profiles of MRSA isolates collected from South African laboratories and to correlate the results with patient demographic data.
Materials and methods
Participating laboratories were requested to collect non-duplicated, consecutive MRSA isolates; these were submitted from 15 National Health Laboratory Services (NHLS) and 8 private diagnostic laboratories from all 9 provinces of South Africa between August 2005 and November 2006. The NHLS is a parastatal organisation providing diagnostic laboratory services to public sector hospitals throughout South Africa. Patient demographic data comprised age and gender. Date of sampling and source of isolate were also collected. Ethics clearance was obtained from the University of the Witwatersrand Committee on Human Research.
S. aureus isolates were identified by standard microbiological methods. Genomic DNA for polymerase chain reaction (PCR) was isolated using a previously published 'rapid lysis' procedure.5 The mecA gene was detected using published primers.6 Antibiotic susceptibility was performed by the Kirby-Bauer disc diffusion method according to CLSI guidelines:7 15 µg erythromycin, 2 µg clindamycin, 25 µg trimethoprim/sulfamethoxazole, 30 µg linezolid, 30 µg vancomycin, 30 µg teicoplanin, 30 µg chloramphenicol, 5 µg ciprofloxacin, 5 µg rifampicin, 10 µg gentamicin, 15 µg quinopristin/dalfopristin, 50 µg nitrofurantoin, 30 µg amikacin, 30 µg minocycline and 30 µg tetracycline (Oxoid, Hampshire, England). The inducible MLSβ phenotype was detected using the double erythromycin-clindamycin disc test (D-test). Mupirocin (5 µg and 200 µg) and 10 µg fusidic acid testing was performed as described elsewhere.8,9 S. aureus strain ATCC 25923 was used as a control. Isolates were classified as multidrug resistant (MDR) if, in addition to the β-lactams, they were resistant to >3 classes based on susceptibility to erythromycin, clindamycin, chloramphenicol, ciprofloxacin, tetracycline, trimethoprimsulfamethoxazole and rifampicin.10
Isolates showing intermediate resistance to teicoplanin were tested further using a PC20 panel on a Microscan Walkaway 40SI (Dade Behring, CA, USA). Etest strips (AB Biodisk, Solna, Sweden) were used to confirm high-level mupirocin resistance (>512 µg/ml) v. low-level resistance (8 - 256 µg/ml).
The data were analysed using Pearson's chi-square test. Where the sample size was small, we used appropriate exact tests - either the binomial test (test of 2 proportions) or Fisher's exact test (for contingency tables).
Results
A total of 248 non-duplicated, laboratory-confirmed mecA-positive MRSA isolates were available for antibiotic susceptibility testing - 142 isolates from NHLS laboratories and 106 isolates from private laboratories. Of the submitted samples, 236 had complete data (age, gender, date of sample collection and specimen source).
Demographic data reflected 101 females (42.2%) and 137 males (57.6%); gender was not recorded for 10 patients. The average age was 38.7 (SD 24.0) years, 22 patients were <1 year old, 46 patients <18 years old, 190 patients >18 years old, and in 12 isolates the patient age was missing.
Eighty-eight (35.5%) isolates were retrieved from blood, 82 (33.0%) from pus, 33 (13.3%) from skin wounds, 14 (5.6%) from the respiratory tract, and 3 (1.2%) from cerebrospinal fluid. In 22 (8.9%) of cases, the source was not specified.
Antibiotic resistance of MRSA to erythromycin, trimethoprim/sulfamethoxazole, tetracycline, gentamicin and ciprofloxacin ranged between 55% and 78% (Table I). All isolates were sensitive to vancomycin, teicoplanin, linezolid, quinopristin/dalfopristin and fusidic acid. Although 35% of isolates were resistant to clindamycin by single-disc testing, the D-test identified erythromycin-induced clindamycin resistance in 68 isolates, bringing the effective total figure for clinical clindamycin resistance to 62.5%. Of the 9 isolates found to be resistant to 200 µg mupirocin by disc diffusion testing, 6 were confirmed to have high-level resistance (>1 024 µg/ml) using Etest strips.
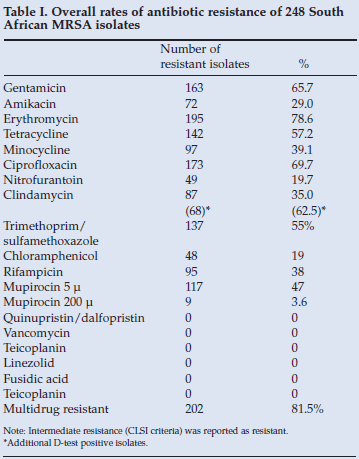
To assess whether antibiotic resistance phenotypes clustered together, antibiograms were assigned using the designation DECLTsRQGA (D for tetracycline, E for erythromycin, C for chloramphenicol, L for clindamycin, Ts for trimethoprim/sulfamethoxazole, R for rifampicin, Q for ciprofloxacin, G for gentamicin, and A for amikacin). Isolates could be grouped into 61 antibiogram patterns. Six predominant groups were found: ELQ (19%), E (11%), DETRG (9%), DECTQGA (7%), DTRG (6%) and DECLTsQGA (5%) (Table II). Of the 248 isolates, 202 (81.5%) were found to be MDR.

The 236 isolates with patient age information were divided into adult (>18 years of age) and children (<18 years of age) to compare resistance profiles. Resistance profiles to most of the antimicrobial agents were similar for adults and children, but significantly lower rates of resistance in children were noted for erythromycin (58% v. 80%, p=0.0015) and clindamycin (21% v. 40%, p=0.018). However, resistance rates to nitrofurantion (38% v.18%, p=0.027) and gentamicin (85% v. 60%, p=0.001) were higher among children than adults.
The rates of antibiotic resistance in NHLS laboratories were compared with those from private laboratories. It was found that significantly more isolates were resistant to rifampicin, gentamicin, trimethoprim/sulfamethoxazole, chloramphenicol, tetracycline, minocycline and nitrofurantoin in the NHLS laboratories than the private laboratories, where resistance was higher for ciprofloxacin, clindamycin, 5 µg mupirocin and erythromycin (Table III).
Discussion
This study presents the first comprehensive data on antibiotic susceptibility patterns for MRSA in South Africa. Associations between resistance and private versus public systems and patient demographic characteristics were investigated.
The MRSA isolates were largely resistant to ciprofloxacin, erythromycin, gentamicin, tetracycline and trimethoprim/sulfamethoxazole. Rates of resistance in this study were in some cases lower than those seen in the KZN study,3 especially for gentamicin (65.7% v. 97%), tetracycline (57.2% v. 90%), minocycline (39.1% v. 90%) and rifampicin (38 v. 74%). Only resistance to ciprofloxacin was notably higher in our study (69.7% v. 18%). These discrepancies may result from the low number of methicillin-resistant isolates collected for the KZN study. Our results correspond more closely with reports from northern Taiwan, in which MRSA resistance to ciprofloxacin and gentamicin was 65% and 73.3% respectively.11
No isolates resistant to vancomycin were detected in our study. Two isolates that were intermediately resistant to teicoplanin by disc diffusion were confirmed to be sensitive on the Microscan. Since the disc diffusion method is not sensitive for detecting intermediate resistance, additional susceptibility testing methods are necessary to determine the true rates of glycopeptide resistance in South Africa.
Eight (3%) isolates showed resistance to 200 µg mupirocin, a topical agent often used to eradicate nasal carriage of S. aureus. Of these, 6 were found to have high-level resistance to mupirocin by means of Etest (MIC >256 µg/ml). Resistance to high doses of mupirocin has been shown to be due to a plasmid-mediated mechanism, and may be of clinical importance. Forty-seven per cent of isolates showed resistance to low levels of mupirocin (using a 5 µg disc), but this may have limited significance as the mechanism involved may not be a precursor to high-level resistance.
Although 61 different antibiogram patterns were found, 5 types made up 57% of the isolates. Previous studies have shown differences in antibiotic resistance profiles among children versus adults that are postulated to be due to differences in host defence during colonisation, the environment and antimicrobial drug selection.12 The lower rate of clindamycin resistance seen in our study in children, as opposed to adults, has been noted before.12,13 However, unlike these studies, the rate of gentamicin resistance in children in
our study was higher than that in adults. Higher gentamicin resistance in children may be explained by the use of first-generation β-lactam antibiotics such as ampicillin, which is recommended as an empirical treatment option for children with infections. It is postulated that certain types of enzymatic resistance to aminoglycosides are linked with the regulatory elements of a TEM-1 type β-lactamase gene carried by a transposable element.14
The differences in antimicrobial resistance patterns of isolates from private versus NHLS channels could be due to the differing pathologies of patients attending these sites and the treatment regimens used. Patients with pulmonary tuberculosis present more frequently at public hospitals and are usually given rifampicin as part of the standard treatment. This could result in the selection of resistant organisms, as reflected in the higher rate of resistance to this antibiotic in this sector. Similarly, the high rates of resistance to ciprofloxacin and erythromycin observed in the private sector could be related to the frequent use of macrolides and quinolones in this setting.
Conclusion
MRSA isolates in South Africa show high rates of resistance to a number of antibiotics, and most isolates are multi-drug resistant. Our data confirm the trend of resistance to the macrolides, tetracycline and the aminoglycosides, while showing that vancomycin, teicoplanin, linezolid and quinopristin/dalfopristin are appropriate agents for clinical use. Differences in resistance profiles among adults and children and isolates from private versus NHLS public systems were found. These data may provide an empirical treatment guideline in clinical settings as well as a baseline for further antibiotic susceptibility studies.
This study was funded by the Medical Research Council of South Africa and the National Health Laboratory Service of South Africa. We also thank for their contributions the members of the South African MRSA Surveillance Group, namely WF Oosthuysen, E Marais, A Dusé (CMID); A Brink (Ampath); G Coetzee, J Mogale, S Mahlati (NHLS); L Badenhorst, K Fick (Ampath Bloemfontein); E Botha (NHLS Welkom); C Jordaan, MN Janse van Rensburg (NHLS Universitas); M Botha, L Ho (Ampath Johannesburg); F Botha, D Hari-Makkan (Ampath Pretoria); O Perovic, D Antelme (NHLS New Johannesburg Hospital); K Lindeque (NHLS, Tshwane Academic Complex, Pretoria); R van der Linde, P Nickleson (Ampath Nelspruit); G Hoyland (NHLS Nelspruit); J Kruger (NHLS Ermelo); Ms Pumeza, C Hammans (NHLS Witbank); D Bothes, S de Wit (Ampath Limpopo); Z Kola (NHLS Polokwane); J Weenink (NHLS Kimberley); M Enslin, H Kuhn (Lancet Johannesburg for NWP); D Cilliers (NHLS Rustenburg); E du Plessis, I Khantsi (NHLS Potchefstroom); W Swart, C Kuhn (Pathcare Port Elizabeth); V Pearce, O Bosch (NHLS Port Elizabeth); M Senekal, S Lalloo (Pathcare Cape Town); S Oliver, I Shankland (NHLS Groote Schuur Hospital); B Jarvis (NHLS George); N Miller, J Bredenkamp (Ampath Durban); N Mkhize, N Khedzie (KwaZulu-Natal Health); Mr Thembe, Ms Mackenza (RK Khan Hospital, KwaZulu-Natal).
References
1. Bouchillon SK, Johnson BM, Hoban DJ, et al. Determining incidence of extended spectrum beta-lactamase producing Enterobacteriaceae, vancomycin-resistant Enterococcus faecium and methicillin-resistant Staphylococcus aureus in 38 centres from 17 countries: the PEARLS study 2001-2002. Int J Antimicrob Agents 2004; 24: 119-124. [ Links ]
2. Perovic O, Koornhof H, Black V, et al. Staphylococcus aureus bacteraemia at two academic hospitals in Johannesburg. S Afr Med J 2006; 96: 714-717. [ Links ]
3. Shittu AO, Lin J. Antimicrobial susceptibility patterns and characterization of clinical isolates of Staphylococcus aureus in KwaZulu-Natal province, South Africa. BMC Infect Dis 2006; 6: 125. [ Links ]
4. Zinn CS, Westh H, Rosdahl VT. An international multicenter study of antimicrobial resistance and typing of hospital Staphylococcus aureus isolates from 21 laboratories in 19 countries or states. Microb Drug Resist 2004; 10: 160-168. [ Links ]
5. Ünal S, Hoskins J, Flokowitsch JE, et al. Detection of methicillin-resistant Staphylococci by using the polymerase chain reaction. J Clin Microbiol 1992; 30: 1685-1691. [ Links ]
6. Oliveira DC, de Lencastre H. Multiplex PCR strategy for rapid identification of structural types and variants of the mec element in methicillin-resistant isolates of Staphylococcus aureus. Antimicrob Agents Chemother 2002; 49: 366-379. [ Links ]
7. Performance Standards for Antimicrobial Susceptibility Testing; Seventeenth Informational Supplement. M100-S17. Vol 27(1). Wayne, PA, USA: Clinical and Laboratory Standards Institute, 2007. [ Links ]
8. Udo EE, Al-Sweih N, Mokaddas E, et al. Antibacterial resistance and their genetic location in MRSA isolated in Kuwait hospitals, 1994-2004. BMC Infect Dis 2006; 6: 168. [ Links ]
9. Skov R, Frimodt-Moller N, Espersen F. Correlation of MIC methods and tentative interpretive criteria for disk diffusion susceptibility testing using NCCLS methodology for fusidic acid. Diagn Microbiol Infect Dis 2001; 40: 111-116. [ Links ]
10. Pillar CM, Draghi DC, Sheehan DJ, et al. Prevalence of multidrug-resistant, methicillinresistant Staphylococcus aureus in the United States: findings of the stratified analysis of the 2004 to 2005 LEADER Surveillance Programs. Diagn Microbiol Infect Dis 2007; 60(2): 221-224. [ Links ]
11. Tai PW, Huang CH, Lin QD, et al. Molecular pattern and antimicrobial susceptibility of methicillin-resistant Staphylococcus aureus isolates at a teaching hospital in Northern Taiwan. J Microbiol Immunol Infect 2006; 39: 225-230. [ Links ]
12. Park JY, Jin JS, Kang HY, et al. A comparison of adult and pediatric methicillin-resistant Staphylococcus aureus isolates collected from patients at a university hospital in Korea. J Microbiol 2007; 45: 447-452. [ Links ]
13. Kilic A, Li H, Stratton CW, et al. Antimicrobial susceptibility patterns and staphylococcal cassette chromosome mec types of, as well as Panton-Valentine leukocidin occurrence among, methicillin-resistant Staphylococcus aureus isolates from children and adults in middle Tennessee. J Clin Microbiol 2006; 44: 4436-4440. [ Links ]
14. Acar JF, Goldstein FW, Menard R, Bleriot JP. Strategies in aminoglycoside use and impact upon resistance. Am J Med 1986; 80: 82-87 [ Links ]
Accepted 22 October 2008.
Corresponding author: E Marais (else.marais@wits.ac.za)














