Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 n.2 Pretoria Feb. 2009
SCIENTIFIC LETTER
Prevalence of gastric mucosal interleukin-1 polymorphisms in Kenyan patients with advanced gastric cancer
Kimang'a NyerereI; Shahin SayedII; Gunturu RevathiIII; Peter OjwangIV; Viviene MatiruV; Smita DevaniVI; Mahesh ShahVII; Samuel KariukiVIII
IMSc. Department of Pathology, Aga Khan University Hospital, and Department of Medical Microbiology, Jomo Kenyatta University, Nairobi, Kenya
IIMB ChB, MMed. Department of Pathology, Aga Khan University Hospital
IIIMB BS, MD. Department of Pathology, Aga Khan University Hospital
IVMB ChB, FRCPath, FCPath (SA). Department of Pathology, Aga Khan University Hospital
VBEd, MSc, PhD. Department of Medical Microbiology, Jomo Kenyatta University, Nairobi, Kenya
VIMB ChB, MRCP. Department of Medicine, Aga Khan University Hospital
VIIMB ChB, MMed. Department of Medicine, Aga Khan University Hospital
VIIIBVM, MSc, PhD. Kenya Medical Research Institute, Centre for Microbiology Research (CMR) Nairobi, Kenya
To the Editor: Helicobacter pylori is the main cause of peptic ulceration, distal gastric adenocarcinoma, and gastric lymphoma.1 Worldwide, gastric cancer is the second most common malignancy in men and women.1 According to data from the Nairobi Cancer Registry, gastric cancer is the fourth most common malignancy in adult males and the fifth most common in adult females. However, this may not represent the true situation because of under-reporting of cases. In the development of gastric cancer, environmental factors such as smoking, diet and, in particular, infection with H. pylori are significant.1 Based on epidemiological studies, the International Agency for Research on Cancer (IARC) identified H. pylori as a 'group 1 agent (definite carcinogen)'.2 H. pylori infection can result in decreased acid secretion with subsequent mucosal atrophy and intestinal metaplasia.1 Another precondition for mucosal atrophy is autoimmunity against parietal cells, which can mimic classic autoimmune gastritis with the presence of various autoantibodies in up to 40% of H. pylori-infected individuals.1
The occurrence of intestinal metaplasia, for which a relationship with gastric cancer is strongly suggested, has been demonstrated in approximately 60% of patients with H. pylori infection.1 The metaplasia may then progress to gastric cancer, especially to tumours of the intestinal type.1 Findings by Uemura et al. support the importance of these histological findings as a precancerous condition in H. pyloriassociated gastritis.3 However, only a minority of H. pylori-infected patients develop gastric cancer, which underscores the notion that the host genetic background could be of critical importance.
Data strongly suggest that the susceptibility to infection from H. pylor i is mainly conferred by genes involved in inflammatory processes following colonisation with H. pylori.1 Chronic gastritis is characterised by the release of pro-inflammatory cytokines such as interleukin-1β (IL-1β) or tumour-necrosis factor alpha (TNFα), which are potent inhibitors of gastric acid secretion.1 Advanced-stage gastric cancer has been repeatedly associated with polymorphisms of the IL-1 gene cluster on chromosome 2q, which contains 3 related genes within a 430 kb region (IL-1A, IL-1B, and IL-1RN), encoding for IL-1α , IL-1β, and the endogenous receptor antagonist IL-1ra, respectively. It was hypothesised that genetic differences within these genes could influence the immune response against pathogens such as H. pylori and the development of premalignant histological alterations in the gastric mucosa.1 In patients with advanced-stage gastric cancer, an increased frequency of the IL-1B-31C and IL-1B-511T alleles and the uncommon IL-1B-31C/IL-1B-511T haplotype was demonstrated. In addition, the IL-1RN*2 allele and the homozygous genotype IL-1RN*2/2 were found in increased prevalence in gastric cancers.1 Subsequent studies confirmed these genetic associations.1 El-Omar et al. genotyped patients with gastric cancer according to tumour localisation (cardia v. non-cardia) and oesophageal cancers (adenocarcinomas v. squamous cell carcinomas) for various polymorphisms of genes encoding for pro- and anti-inflammatory cytokines.4 They described an increased risk for non-cardia gastric cancer in carriers of the IL-1B-511T allele, IL-1RN*2 homozygotes, carriers of the TNF-A-308A allele and the haplotype IL-101082A/-819T/-592A. The cumulative risk depends on the number of high-risk alleles or genotypes per patient.4 A previous study confirmed the risk increase for development of gastric carcinoma in carriers of multiple proinflammatory genotypes.1 The alleles IL-1RN*2 and IL-1B-511T are associated with increased synthesis of the proinflammatory cytokine IL-1β, and the allele TNFA-308A results in an increased production of the proinflammatory cytokine TNF.1
Patients and methods
The study used 41 archival paraffin-embedded gastric cancer tissue blocks obtained in 2007 from endoscopic biopsies and gastrectomies. Haematoxylin and eosin-stained slides were reviewed and the tumours classified according to the WHO Center for the International Histological Classification of Tumors. DNA was extracted using the QIAamp DNA kit. The study was approved by the Aga Khan University (East Africa) Research and Ethics Committee. The H. pylori status was determined by polymerase chain reaction (PCR) using the ureA gene according to the method of Miyabayashi et al.5 The ureA region was amplified using the following primers: 5'-AAGCTTTTAGGGGTGTTAGGGGTTT-3'. The IL-1B fragment was amplified using the following primers: 5'- TGGCATTGATCTGGTTCATC-3' and 5'- GTTTAGGAATCTTCCCACTT-3', while the IL-1RN intron 2 containing a penta-allelic 86-bp tandem repeats (VNTR) DNA oligonucleotides 5'-CTCAGCAACACTCCTAT-3' and 5'-TCCTGGTCTGCAGGTAA-3' flanking this region were used as primers.
Data analysis
Statistical analysis to determine the relationship between variables was performed using the chi-square test with p-values less than 0.005 considered as significant. Linear regression determined how independent factors predicted the outcome of dependent factors.
Results
We analysed 41 gastric biopsy samples that were preserved in formalin and embedded in paraffin wax, obtained over a year. All the biopsies were of adult Kenyan patients with advanced gastric cancer; 31 (76%) were of the intestinal type and 24% were of the diffuse 'signet ring' type; 26 (63%) were from male and 15 (37%) from female patients; their ages ranged from 36 years to 76 years, with 56% <50 years of age. We screened for the frequencies of genotypes and alleles of IL-1B and IL-1RN in these cancer tissues. Overall, 19 (46%) patients had IL-1RN while 18 (43%) had IL-1B. When analysed, the IL-1RN found in the Kenyan gastric cancer population only showed allele IL 1RN 1*4 (Fig. 1).
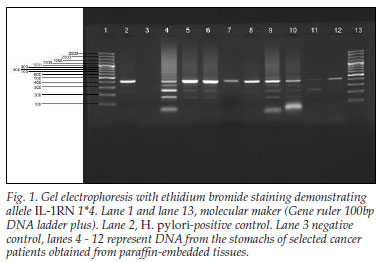
In contrast, when IL-1B was analysed by PCR-RFLP (restriction fragment length polymorphism) method, it gave products that either remained intact (allele 2; IL-1B-511*2) or were cut into two fragments of 190 and 114 bp (allele 1; IL-1B511*1) (Fig. 2).
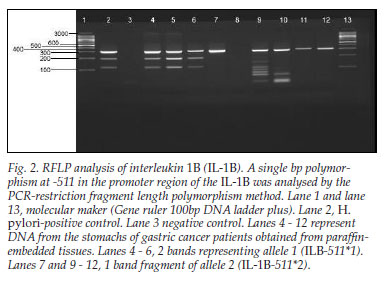
The H. pylori gene was present in only 6 of the 41 samples. This was expected because, when H. pylori has been present for many years and the stomach becomes atrophic and hypochlorhydric, the intragastric environment is no longer optimal for H. pylori. Competing faecal-type organisms are then able to colonise the stomach. Furthermore, H. pylori is able to colonise only normal gastric cells, and not those that have differentiated into intestinal metaplasia. Therefore, H. pylori disappears from the stomach when atrophy and intestinal metaplasia supervene. In patients who are most likely to develop gastric cancer (those with extensive intestinal metaplasia and atrophy) and those already with advanced cancer, as in our cases, the serology is often negative and the genes will almost always not be detectable by PCR.
No significant relationship was found between age and either IL-1RN or IL-1B in gastric carcinoma from Kenyan patients; the chi-square test for both was not significant: p=0.9 for IL-1RN and p=1.0 for IL-1B, against a significance level of 0.3 and 0.2 respectively. This indicates that these variables are probably independent of each other. Similarly, there was no relationship between gender and the occurrence of either IL 1RN or IL-1B. Linear regression also showed that neither age nor gender influenced the expression of IL-1RN or IL-1B.
The situation differs, however, concerning the relationship between IL-1RN and IL-1B. The large chi-square statistic (8.021) and a small significance level (0.005) indicate that it is unlikely that these variables are independent of each other. Therefore, we concluded that there is a relationship between the occurrence of IL-1RN and IL-1B. Linear regression showed an R-value of 0.45, which indicates that a great deal of variance is shared by IL-1RN and IL-1B. Analysis of variance (ANOVA) obtained from the regression F=9.5 appears to indicate that the predictor variable IL-1RN is not equal to the dependent factor IL-1B, and that the presence of IL-IRN could be used to predict the presence of IL-1B.
Discussion
Instrumental in the development of H. pylori-related gastric carcinogenesis are some pro-inflammatory cytokines such as interleukin 1 beta (IL-1β) and interleukin 1 receptor antagonist (IL-1ra). The interleukin-1 (IL-1) gene cluster on chromosome 2q contains 3 related genes within a 340-kilobase (kb) region, IL-1A, IL-1B and IL-1RN, which encodes the pro-inflammatory cytokines IL-1α and IL-1β as well as their endogenous receptor antagonist IL-1ra respectively.6 IL-1β is up-regulated in the presence of H. pylori and is important in initiating and amplifying the inflammatory response to this infection. IL-1β is also a potent inhibitor of gastric acid secretion. The IL-1RN gene has penta-allelic 86-bp tandem repeats (VNTR) in intron 2, of which the less common allele 2 (IL-1RN*2) is associated with a wide range of chronic inflammatory and autoimmune conditions. IL-1RN*2 is associated with enhanced IL-1β production.6 Carriers of IL-1RN 2/*2 and IL-1B-511 T have been reported to be at increased risk of hypochlorhydria and gastric cancer.6
We tested the hypothesis that IL-1 genetic polymorphisms are circulating in the Kenyan population and that they influenced the presence of IL-1B in H. pylori-associated gastric cancer. Intron 2 of the IL-1RN gene was detected and amplified by PCR. Homozygous genotype IL-1RN1/*4 was found to be present. We also observed the IL-1B gene with heterozygous genotype IL-1B-511*1 and IL-1B-511*2 in our cases of advanced gastric cancer. This is in contrast with reports from elsewhere in the world, where IL-1RN 1/*2 has been detected with increased frequency.7
Il-Ran Hwang et al. found that carriers of IL-1B-511 T/T genotypes, denoting a single band of 155 bp or IL-1RN*2, had higher mucosal IL-1β cytokine levels compared with non-carriers.6 They also observed that carriers of IL-1RN 1/*2 had higher mucosal IL-1β levels than those of IL-1RN 1/*1 consistent with an effect of this polymorphism on mucosal IL-1β. Also, IL-1RN 1/*2 carriers have atrophy scores approximately 1 point higher than IL-1RN 1/*1 carriers, which implies that in IL-1RN 1/*2 carriers atrophy is accelerated such that the mucosa are functionally 20 years older than the gastric mucosa of IL-1RN 1/*1 carriers.
IL-1RN*2 allele occurs in the Kenyan population at a much higher frequency (46.3%), in contrast with reports on Western and Asian populations.7 For example, the frequency of the IL-1RN*2 allele in East Asian countries was lowest (3 - 6%), while that of the Thai population was 12%. The frequency in Western populations was reported as 22 - 24%.
Our frequency of the IL-1B-511 allele was lower (18%) than that reported in the Thai population (34%) but falls within the threshold of other populations (15 - 25%).7 These results are consistent with the notion that in any population, the dominant factor or combination of factors in relation to gastric cancer risk among H. pylori-infected individuals will vary and must be examined directly. Only 6 of our cases tested positive for H. pylori by gene analysis despite the fact that Ogutu et al.8 reported a prevalence of 80.5% in Kenyan adults presenting with dyspeptic symptoms and normal endoscopic findings. This anomaly can be explained by the fact that all our cases involved advanced carcinomas, and H. pylori is known to disappear as atrophy and intestinal metaplasia supervene over a prolonged period of infection.9
In conclusion, this pioneer study of genotype polymorphisms for interleukin 1 in the Kenyan population provides evidence that individuals with IL-1B and IL-1RN polymorphisms associated with H. pylori infection have an elevated risk of developing gastric cancer. Since it was a pilot study, the observed significant association could be a chance observation because of lack of complete clinical information on the patients whose tissues were used. Our findings require confirmation by prospective analysis of a larger sample, looking at other related co-factors.
This study was supported by Dr Varocha Mahachai and Dr Somying Tumwasorn of the Department of Medicine and Microbiology, Chulalongkorn University Hospital, Bangkok, Thailand; and Dr Ratha-Korn of the Gastroenterology Unit, Department of Medicine, Thammasat University Hospital, Pathumthani, Thailand; and it was authorised by the Aga Khan University Hospital, Nairobi, Kenya and the Jomo Kenyatta University of Science and Technology, Nairobi, Kenya.
References
1. John C. Atherton. The pathogenesis of Helicobacter pylori-induced gastro-duodenal diseases. Annu Rev Pathol Mech Dis 2006; 1: 63-96. [ Links ]
2. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, 7 - 14 June, Lyon. IARC Monogr Eval Carcinog Risks Hum 1994; 61: 1-241. [ Links ]
3. Uemura N, Okamoto S, Yamamoto S, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med 2001; 345: 784-789. [ Links ]
4. El-Omar EM, Rabkin CS, Gammon MD, et al. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology 2003; 124: 1193-1201. [ Links ]
5. Miyabayashi H, Furihata K, Shimizu T, Ueno I, Akamatsu T. Influence of oral Helicobacter pylori on the success of erradication therapy against gastric Helicobacter pylori. Helicobacter 2000; 5: 30-37. [ Links ]
6. Hwang IR, Kodama T, Kikuchi S, et al. Effect of interleukin-1 polymorphisms on gastric mucosal interleukin 1beta production in Helicobacter pylori infection. Gastroenterology 2002; 123: 1793-1803. [ Links ]
7. Vilaichone RK, Mahachai V, Tumwasorn S, Wu J-Y, Graham DY, Yamaoka Y. Molecular epidemiology and outcome of Helicobacter pylori infection in Thailand: a Cultural Cross Roads. Scand J Gastroenterol 2005; 40: 520-539. [ Links ]
8. Ogutu E, Kang'ethe S, Nyabola L. Endoscopic findings and prevalence of H. pylori in Kenyan patients with dyspepsia. East Afr Med J 1998; 75: 85-89. [ Links ]
9. Kikuchi S, Wada O, Nakajima T, et al. Serum anti-Helicobacter pylori antibody and gastric carcinoma among young adults. Cancer 1995; 75: 2789-2793. [ Links ]
Accepted 14 October 2008.
Corresponding author: S Shahin (shaheen.sayed@aku.edu)














