Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.99 n.1 Pretoria Jan. 2009
ORIGINAL ARTICLES
STUDENT PAPER
Mechanisms of oedema formation: the minor role of hypoalbuminaemia
C SteylI; R Van Zyl-SmitII
I4th-year medical student, currently 2nd-year intern. University of Pretoria
IIMB BCh, FCP (SA), FRCP, MD. University of Cape Town and New Somerset Hospital, Cape Town
ABSTRACT
OBJECTIVES: Seriously ill patients often suffer from disorders of salt and water balance and present with clinical signs of either dehydration or oedema. The relationship of hypoalbuminaemia to oedema is complex and controversial and formed the central issue of this study.
DESIGN: Prospective study.
SETTING: Medical wards of New Somerset Secondary Hospital, November 2004.
SUBJECTS: 50 patients admitted consecutively to the medicalwards at New Somerset Hospital were evaluated; 26 males and 24 females participated.
OUTCOME MEASURES: An attempt was made to correlate causesof salt and water imbalance with the clinical assessment ofvolume status, oedema formation, nutritional state and serum albumin levels.
RESULTS: Hypoalbuminaemia was not related to oedema inthis study. Of 24 patients with a serum albumin level below 30 g/l, only 6 had oedema. These patients all had other abnormalities that could have resulted in the oedema, notably primary salt retention by failing kidneys, cor pulmonale and malignancy. None of the patients with serum albumin levels below 15 g/l had any signs of oedema. The combined insult of a chronic inflammatory disease and malnutrition had a marked effect on serum albumin levels.
CONCLUSION: Significant hypoalbuminaemia was present in many of our patients, yet oedema was detected infrequently and generally had an easily identifiable cause not related to low albumin levels. Most patients with hypoalbuminaemiapresented with normal or positive water balance. This study supports the notion that hypoalbuminaemia is infrequently associated with oedema and plays a minor role in its formation.
Seriously ill patients often suffer from disorders of salt and water balance and present with the clinical signs of either dehydration or oedema. The relationship of hypoalbuminaemia to oedema is complex and formed the central issue of this study. The concept that hypoalbuminaemia directly causes oedema due to fluid extravasation can be challenged, as there is insufficient intravascular fluid to cause any significant expansion of the interstitial space. Slight intravascular volume contraction as a result of hypoalbuminaemia might cause salt retention secondary to increased aldosterone secretion; however, this mechanism of oedema formation has infrequently been reported in the literature.1
Methods
Fifty patients admitted consecutively to the medical wards of New Somerset Secondary Hospital were studied. An attempt was made to correlate causes of salt and water imbalance with the clinical assessment of volume status, oedema formation, nutritional state and serum albumin levels. Total body fluid balance was assessed by two independent observers who recorded the extent of oedema formation on a scale of 0 to 4, tissue turgor on a scale of 0 to -3, and by paying attention to the jugular venous pressure and pulse rate, blood pressure and signs of pulmonary congestion on the chest radiograph. Dipstix urinalysis was done on all patients. Albumin levels were measured only as part of routine clinical management, so some patients with hypoalbuminaemia would therefore have been missed. All findings were recorded anonymously after patient consent had been obtained. Ethical approval was obtained from the local ethics committee.
Results
Hypoalbuminaemia was noted in 26 patients, largely in association with tuberculosis: all 13 patients with this diagnosis had serum albumin levels below 30 g/l. Despite this, only 2 patients presented with oedema. The first patient, with a serum albumin level of 16 g/l, had pulmonary tuberculosis (TB) with cor pulmonale and a haemoglobin concentration of 8 g/dl. The second patient, with a serum albumin level of 28 g/l, had TB and clear signs of volume overload with an elevated jugular venous pressure and moderate hepatomegaly.
The combined insult of a chronic inflammatory disease and malnutrition appeared to have a marked effect on serum albumin levels. The average serum albumin level for the 19 wasted patients was 22 g/l, compared with an average of 20 g/l for the 11 patients with TB and malnutrition. The average serum albumin level among well-nourished patients was 26 g/l.
Patients with renal impairment and proteinuria (>1 g/l) generally had less severe hypoalbuminaemia, with an average serum albumin level of 27 g/l. Sixteen patients had impaired renal function, yet only 4 of these patients presented with oedema. This finding was surprising as the expectation would be to find salt retention and a higher prevalence of oedema in association with poor renal function.
Hypoalbuminaemia was not related to oedema in the study patients. Of the 24 patients with a serum albumin level below 30 g/l, only 6 presented with oedema (Table I). These patients all had other abnormalities that could have resulted in the oedema, notably primary salt retention by failing kidneys (creatinine levels 676 µmol/l, 1 276 µmol/l and 168 µmol/l respectively), cor pulmonale and malignant disease. None of the patients with serum albumin levels below 15 g/l had any signs of oedema.
This discrepancy between hypoalbuminaemia and oedema is clearly illustrated in the Figs 1 and 2. Fig. 1 shows a patient with marked generalised oedema and nephrotic range proteinuria (3.3 g/d), yet the serum albumin level was relatively high at 28 g/l. The patient had clinical signs of fluid overload and was being treated with furosemide 120 mg/d.

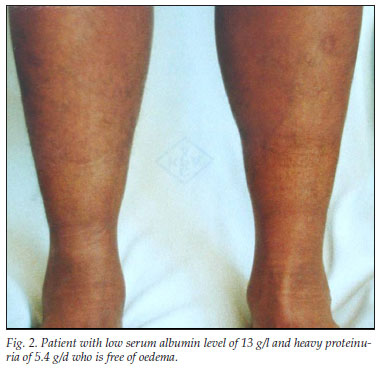
In contrast, Fig. 2 shows a patient with a serum albumin level of 13 g/l and heavy proteinuria of 5.4 g/d, yet this patient is oedema free. This patient was not taking any diuretics.
Most marked oedema was seen in patients with malignancy, cardiac failure and renal failure and was often associated with signs of volume overload.
Discussion
Generalised oedema becomes clinically noticeable when the extracellular fluid space is expanded by about 2 - 3 litres of fluid.1 Excess extracellular fluid accumulates largely in the interstitial space resulting in clinically detectable swelling that is most prominent in dependent parts of the body such as the ankles and the sacral area in bed-bound patients. Fluid accumulation either results from primary 'salt-retaining' types of renal disease, resulting in expansion of both the intra- and extravascular compartments, or is due to homeostatic responses causing salt retention by normal kidneys as is found in patients with cardiac and liver disease.1,2 Factors such as changes in capillary hydrostatic pressure, capillary blockage and inflammation resulting in increased capillary permeability may also be involved, but are more commonly encountered with localised forms of oedema.1
Hypoalbuminaemia cannot directly cause oedema by fluid extravasation as there is insufficient intravascular fluid to cause any significant expansion of the interstitial space.1 The theory that fluid extravasation due to hypoalbuminaemia commonly leads to a contracted intravascular volume that results in stimulating secondary renal salt-retaining mechanisms can be refuted, as the majority of oedematous patients are normovolaemic or have signs of volume expansion.2-4 This was also noted in the current study.
The role of low intravascular oncotic pressure as a cause of outward fluid movement has been overstated, as experimental models and most patients with nephrotic syndrome do not show a decreased transcapillary osmotic gradient.3,5 The plasma oncotic pressure and interstitial oncotic pressure has in fact been shown to have a linear relationship, which protects against a fall in blood volume.3,6,7
Patients with hypoalbuminaemia secondary to heavy proteinuria (nephrotic syndrome) probably all have primary renal salt retention caused by a variety of mechanisms including both the proximal and distal tubules.2 Proteinuria itself may also be an independent factor that induces salt retention in addition to its known capacity to induce direct renal injury.8
Hypoalbuminaemia may also occur in the absence of significant proteinuria or renal disease, as often occurs in association with liver disease, inflammatory states and malnutrition. Such patients do not have a mechanism for primary salt retention and could be evaluated in relation to oedema formation independently from primary renal salt retention.
Conclusion
The supposed direct link between hypoalbuminaemia and oedema is proving to be too simple an explanation for an age-old enigma. Significant hypoalbuminaemia was present in a substantial portion of the patients included in this study, yet oedema was detected infrequently and when present generally had an easily identifiable cause not obviously related to low albumin levels. Most patients with hypoalbuminaemia presented with a normal or positive water balance. This study therefore supports the notion that hypoalbuminaemia is infrequently associated with oedema and plays a minor role in its formation.
References
1. Rose BD, Post TW. Edematous states. In: Clinical Physiology of Acid-Base and Electrolyte Disorders. 5th ed. New York: McGraw-Hill, Health Professions Division, 2001: 478-534. [ Links ]
2. Hamm LL, Batuman C. Edema in the nephrotic syndrome: new aspects of an old enigma. J Am Soc Nephrol 2003; 14(12): 3288-3289. [ Links ]
3. Deschenes G, Feraille E, Doucet A. Mechanisms of edema in nephrotic syndrome: old theories and new ideas. Nephrol Dial Transplant 2003; 18(3): 454-456. [ Links ]
4. Geers AB, Koomans HA, Boer P. Dorhout Mees AJ. Plasma and blood volumes in patients with the nephrotic syndrome. Nephron 1984; 38(3): 170-173. [ Links ]
5. Joles JA, Willekes-Koolschijn N, Braam B, Kortland W, Koomans HA, Dorhout Mees EJ. Colloid osmotic pressure in young analbuminaemic rats. Am J Physiol 1989; 257(1 Pt 2): F23-28. [ Links ]
6. Fauchald P. Transcapillary colloid osmotic pressure gradient and body fluid volumes in renal failure. Kidney Int 1986; 29(4): 895-900. [ Links ]
7. Koomans HA. Pathophysiology in idiopathic nephrotic syndrome. Nephrol Dial Transplant 2003; 18: Suppl 6, vi30-32. [ Links ]
8. Klisic J, Zhang J, Nief V, Reyes L, Moe OW, Ambuhl PM. Albumin regulates the NA+/H+ exhanger in OKP cells. J Am Soc Nephrol 2003; 14(12): 3008-3016. [ Links ]
Accepted 5 November 2008.
Corresponding author: C Steyl (charle_steyl@yahoo.co.uk)
What this paper adds:
What is already known on the subject?
Medical teaching on oedema usually focuses on Starling's equilibrium, ascribing much significance to decreased plasma colloid osmotic pressure in oedema formation. Numerous experimental models have attempted to demonstrate the mechanism of oedema formation. The two prevailing hypotheses are the so-called 'overfill' (oedema formation by primary renal salt and water retention) and 'underfill' (decreased plasma colloid osmotic pressure leading to a fluid shift to the interstitium) hypotheses.
What does this study add?
This study provides clinical evidence in support of the 'overfill' hypothesis and demonstrates that hypoalbuminaemia does not cause oedema directly.














