Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 no.11 Pretoria nov. 2008
ORIGINAL ARTICLES
Neuropathic pain in AIDS patients prior to antiretroviral therapy
S A HitchcockI; H P MeyerII; E GwytherIII
IMB ChB, M Prax Med, M Phil Pall Med. Department of Family Medicine, Kalafong Hospital, University of Pretoria
IIMB ChB, M Prax Med, MFGP (SA). Department of Family Medicine, Kalafong Hospital, University of Pretoria
IIIMB ChB, FCFP, MSc Pall Med. Department of Family Medicine, University of Cape Town
ABSTRACT
OBJECTIVE: To measure the prevalence, severity and morbidity of neuropathic pain in AIDS patients, prior to the initiation of antiretroviral (ARV) therapy.
DESIGN: A prospective, cross-sectional and descriptive-analytical study.
SETTING: The Kalafong Hospital HIV Clinic in Pretoria.
SUBJECTS: All patients with confirmed AIDS who were referred to the Kalafong HIV clinic to be initiated on ARV therapy during the period August 2006 to March 2007.
OUTCOME MEASURES: Data were collected regarding the presence and severity of neuropathic pain in each subject. Pain of predominantly neuropathic origin (POPNO) was identified using the Neuropathic Pain Diagnostic Questionnaire (DN4). Numerical rating scales (NRS), adapted from the Brief Pain Inventory, were used to measure pain severity and pain-related interference with six aspects of daily living.
RESULTS: Of the 354 patients studied, 20.9% (95% confidence interval (CI) 16.8 - 25.2%) had POPNO. This pain was significantly more frequent in patients who were male, had lower CD4+ counts or higher viral load levels, and those on TB treatment. Eighty per cent of patients with POPNO experienced significant pain (worst pain severity >5 out of 10 on a NRS). Pain-related interference was highest for enjoyment of life, mood and ability to work. There was a significant positive correlation between severity of pain and pain-related interference for all domains of daily living evaluated.
CONCLUSIONS: POPNO results in significant suffering and impaired functioning in patients with AIDS. It is therefore imperative that clinicians assess patients with AIDS for the presence and severity of neuropathic pain and manage it, using the most recent evidence-based guidelines.
The human immunodeficiency virus (HIV) has a predilection for nerve tissue; therefore, the peripheral nerves, spinal cord and brain are frequently affected in patients with HIV/AIDS, resulting in a high prevalence of neuropathic pain. Neuropathic pain is defined by the International Association for the Study of Pain (IASP) as '... pain initiated or caused by a primary lesion or dysfunction of the nervous system'.1 The lesion may occur anywhere within the central or peripheral nervous system and may lead to structural and functional changes in the nervous system, resulting in altered central neural processing of sensory input (central sensitisation). Consequently, pain may persist long after healing of the original injury. Neuropathic pain may therefore be regarded as abnormal activation of pain pathways.
Neuropathic pain syndromes typically present with both negative and positive sensory symptoms and signs. The usual qualities of neuropathic pain are burning, pricking, shooting or aching. This may be accompanied by hyperalgesia, allodynia, dysaesthesia or paraesthesiae. Neuropathic pain is not necessarily an all-or-nothing phenomenon but may be part of a spectrum where the pain is 'more or less neuropathic' in origin, hence the concept of pain of predominantly neuropathic origin (POPNO).2,3
Several instruments, specifically designed for the identification of neuropathic pain, were reviewed. The Neuropathic Pain Diagnostic Questionnaire (DN4), developed by the French Neuropathic Pain Group, was selected for use in this study, because of its brevity and simple application. It has been validated in a single study and found to have a sensitivity of 82.9% and specificity of 89.9% for identifying neuropathic pain.4
The most frequent cause of neuropathic pain in patients with HIV infection is a distal sensory polyneuropathy (DSP), which may either be the result of the HIV infection itself or of neurotoxicity from drugs used in treating patients with AIDS. The combined effect of the potentially neurotoxic antiretroviral drugs, d4T and ddI, with isoniazid used in the treatment of tuberculosis, may cause significant problems. Malignancies, central neuropathic pain secondary to cerebrovascular incidents, herpes zoster and post-herpetic neuralgia are additional causes of neuropathic pain in HIV-infected patients.
There is limited evidence in the literature regarding the prevalence of neuropathic pain in AIDS patients. Several studies have investigated the prevalence of symptomatic DSP in AIDS patients, which ranges from 35% to 45%.5,6
The prevalence of neuropathic pain in AIDS patients in South Africa is unknown, and little is known about its severity or impact on their lives. Although neuropathic pain in AIDS patients is common, it is frequently under-diagnosed and under-treated, and a lack of awareness of the extent of the problem could contribute to the inadequate management.7 Failure to recognise when neuropathic pain mechanisms predominate and the lack of knowledge of appropriate treatment, results in failure to treat patients appropriately.
Our aim was to identify the prevalence, severity and effects on quality of life by pain that is predominantly neuropathic in origin in patients with AIDS, prior to the initiation of ARV therapy.
Methods
Study setting and population
A prospective survey of new patients referred to the Kalafong Hospital HIV Clinic in Pretoria was conducted from August 2006 to March 2007. Only patients with confirmed AIDS and who require ARV therapy are referred to this clinic. Each new patient was invited to participate in the study. All subjects met the 1993 Centers for Disease Control (CDC) case definition criteria for AIDS, namely a history of an AIDS-defining clinical condition or a CD4 lymphocyte count <200 cells/µl.8 Patients already using ARV medication, or who had cognitive impairment such that they would be unable to answer the questionnaire, were excluded. The study was approved by the Research and Ethics Committees of the Universities of Cape Town and Pretoria. Patients were included only after they had given informed, written consent.
Data collection
Data were collected using an interviewer-administered questionnaire and focused neurological examination (Table I).

Analysis of data
Data were analysed using SPSS for Windows statistical software. Descriptive statistics were produced to examine the study sample, prevalence and severity of POPNO and pain-related interference. Means ± standard deviations were used for continuous variables, and frequency distributions for categorical variables. Factors associated with the presence of POPNO were identified using chi-squared tests. Kruskal-Wallis statistics were used to evaluate significant linear relationships between the severity of POPNO and interference items. Statistical significance was evaluated at the 95% confidence level.
Results
This study included 354 patients diagnosed with AIDS, with a mean age of 36.3 years (±8.6) and ranging from 18 to 67 years. Female patients (mean age 35.0 years) were significantly younger than male patients (mean age 40.0 years) (p<0.001). Patients had a mean CD4+ count of 111 (±70.8). Of the sample, 21% had recently received or were currently receiving tuberculosis treatment.
The distribution of pain within the whole sample (N=354) is displayed in Fig. 1. The prevalence of significant pain of any type within the whole sample was 62.1% (220 patients). Male patients experienced significantly more pain, with 72.9% having pain, compared with 58.1% of female patients (p=0.011).

Of the total sample, 20.9% (74 patients) (95% CI 16.8 - 25.2%) had POPNO, based on achieving a score of four or more (range 0 - 10), using the DN4. Chi-squared analysis demonstrated a significantly higher prevalence of POPNO in male patients (31.3%), compared with 17.1% in female patients (p=0.003). Patients with lower CD4+ counts (p=0.009) and those with higher viral load values (p=0.006) showed a significantly higher prevalence of POPNO, as did patients with a history of recent or current treatment for tuberculosis (p<0.001). The POPNO was located in the lower limbs in 84% (62 out of 74) patients. It is therefore likely that most of the neuropathic pain in this study was as a result of a peripheral neuropathy.
Regarding the severity of POPNO, a numerical rating scale (NRS) score for worst pain of 1 - 4 was regarded as mild pain (20.3% of patients), NRS 5 - 7 as moderate pain (39.2%), and NRS 8 - 10 as severe pain (40.5%). The mean score for pain at its worst was 6.31 (±2.57).
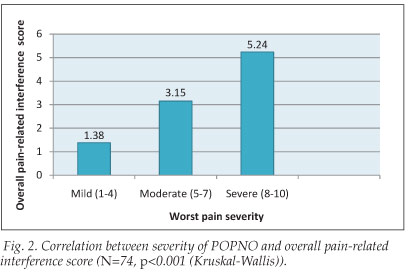
Pain-related interference was measured for 6 aspects of daily functioning. The domains of 'enjoyment of life', 'mood' and 'ability to work' were most affected by the neuropathic pain. At least 25% of the POPNO patients experienced severe pain-related interference in these three aspects of functioning. In addition, sleep and walking were found to be severely affected by the pain in 23% and 17.6% of patients, respectively. A significant positive correlation was demonstrated between pain severity and pain-related interference (Fig. 2).
Of the 74 patients with POPNO, 91.9% displayed at least 1 or more clinical abnormalities on neurological examination, the most frequent being abnormal pinprick sense (73% of patients), abnormal tactile sense (68.9%) and decreased or absent vibration sense (62.1%). Hypoaesthesia for pinprick and tactile sense occurred more commonly than hyperaesthesia.
Discussion
This study's POPNO prevalence of 20.9% is lower than expected in comparison with previous studies in Africa and the USA, where a prevalence of symptomatic DSP in AIDS patients of 35 - 45% was found.5,6 The higher prevalence of POPNO found in male patients, those with a lower CD4 cell count and a higher viral load, and those on tuberculosis treatment, confirms previous findings, where lower CD4+ counts and male gender were significantly associated with a higher prevalence of polyneuropathy.9 This correlation implies that the timely use of ARV therapy could lower the incidence of neuropathic pain in this population.
An objective of this study was to increase awareness among clinicians of the severity and impact of neuropathic pain in AIDS patients. This and previous pain studies on HIV patients describe 'worst pain' (rated as 5 or above) as significant pain, and report it as contributing to disproportionately more functional impairment.7,10 In this study, 79.7% of POPNO patients rated their worst pain as 5 or more, which is higher than a USA study of patients with metastatic cancer.10 It has been shown3 that the general health status measure (SF-36) of patients with POPNO was comparable with that of patients with serious somatic diseases, including coronary artery disease, recent myocardial infarction and poorly controlled diabetes.11
The burden carried by patients as a result of POPNO may be more accurately understood by measurement of impairment of function and reduced quality of life. The pain-related interference ratings in this study are similar to those in a USA study of post-herpetic neuralgia in older persons.12 Controlling POPNO has the potential to greatly improve functioning and quality of life; however, analgesic and adjuvant drug side-effects must be taken into account.
It is significant that 91.9% of patients with POPNO demonstrated an objective clinical abnormality, of which decreased tactile, pinprick and vibration sense were the most common. A focused neurological examination which includes these modalities may therefore be of particular value in AIDS patients with suspected neuropathic pain.
Limitations of study
Representation was limited to patients with AIDS who were willing and able to attend this public health facility for ARV therapy. It excluded those who were unable or unwilling to use ARV therapy or unable to attend due to physical disabilities and/or socio-economic factors. These factors could have resulted in a disproportionate number of patients who were most affected by AIDS having been excluded. Furthermore, the sample was restricted to patients reliant on public health care and patients not yet using ARV therapy. Further research should be done, involving more varied populations and patients on ARV therapy. It would be of value to determine the influence of ARV therapy - in particular, the widely used nucleoside reverse transcriptase inhibitors (NRTIs) - on the prevalence of neuropathic pain in AIDS patients.
Conclusions and recommendations
This study demonstrates a significant prevalence and impact of neuropathic pain in patients with AIDS. These findings underscore the need for improved pain control in AIDS patients to improve their quality of life and assist them in remaining economically active. While the timely use of ARV therapy can contribute to the management of AIDS-related neuropathic pain, additional measures are frequently required. These include appropriate assessment of pain with regard to the type, severity and related functional impairment and appropriate pain management, alongside ARV therapy.
In contrast to pain management in general, neuropathic pain requires a more rigorous approach, involving a multidimensional bio-psycho-social approach that involves the use of appropriate pharmacological agents, exercise, behavioural therapy, attention to sleep quality, patient education and return to work if possible.13 Pharmacological management places particular emphasis on adjuvant drugs, added to non-opioid and opioid analgesics. Clinicians should avail themselves of current evidence regarding the most effective adjuvant drugs for neuropathic pain, with the fewest side-effects. Several classes of adjuvant drugs have proven efficacy in neuropathic pain, although further studies are required to prove their efficacy specifically in HIV-related neuropathic pain. These include the tricyclic antidepressants (TCAs), anticonvulsants (in particular the a2-S ligands, such as gabapentin and pregabalin) and the newer serotonin and noradrenaline re-uptake inhibitors (SNRIs).14
In South Africa, primary health care providers are in the most favourable position to assess and manage patients presenting with POPNO, utilising a multidisciplinary approach, and have a responsibility to develop the required knowledge and skills. Patients with complicated presentations and those who do not respond to a first-line approach should be referred to appropriate specialists.13 Given the extent of the problem of AIDS in South Africa, a patient-centred approach is imperative to alleviate suffering and improve the lives of patients with AIDS and neuropathic pain.
References
1. IASP task force on taxonomy. Part III: Pain terms, a current list with definitions and notes on usage. In: Merskey H, Bogduk N, eds. Classification of Chronic Pain. 2nd ed. Seattle: IASP Press, 1994: 209-214. [ Links ]
2. Attal N, Bouhassira D. Can pain be more or less neuropathic? Pain 2004; 110: 510-511. [ Links ]
3. Smith BH, Torrance N, Bennett MI, Lee AJ. Health and quality of life associated with chronic pain of predominantly neuropathic origin in the community. Clin J Pain 2007; 23(2): 143-149. [ Links ]
4. Jensen MP. Using pain quality assessment measures for selecting analgesic agents. Clin J Pain 2005; 22: S9-S13. [ Links ]
5. Schiffito G, McDermont MP, McArthur JC, et al. Incidence of and risk factors for HIV-associated distal sensory polyneuropathy. Neurology 2002; 58: 1764-1768. [ Links ]
6. Mbuya SO, Kwasa TO, Amayo EO, Kioy PG, Bhatt SM. Peripheral neuropathy in AIDS patients at Kenyatta National Hospital. E Afr Med J 1996; 73(8): 538-540. [ Links ]
7. Larue F, Fontaine A, Colleau SM. Underestimation and undertreatment of pain in HIV disease: multicentre study. BMJ 1997; 314: 23-28. [ Links ]
8. Centers for Disease Control. 1993 Revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Morb Mortal Wkly Rep 1992; 41(RR17): 1-19. [ Links ]
9. Tagliati M, Grinell J, Godbold J, Simpson DM. Peripheral nerve function in HIV infection: clinical, electrophysiologic and laboratory findings. Arch Neurol 1999; 56: 84-89. [ Links ]
10. Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 1994; 330: 592-596. [ Links ]
11. McHorney CA, Ware JE, Raczek AE. The MOS 36-item short form health survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 1993; 31: 247-263. [ Links ]
12. Oster G, Dukes E, Harding G, Edelsberg J, Cleary PD. Pain, medication use, and health-related quality of life in older persons with post-herpetic neuralgia: results from a population-based survey. J Pain 2005; 6(6): 356-363. [ Links ]
13. Meyer HP. Pain management in primary care - current perspectives. S Afr Fam Pract 2007; 49(7): 19-25. [ Links ]
14. Dworkin RH, O'Connor AB, Backonja B, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain 2007; 132: 237-251. [ Links ]
 Correspondence:
Correspondence:
S Hitchcock
(soma.Mtchcocld@up.ac.za)
Accepted 10 July 2008.














