Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 no.9 Pretoria sep. 2008
ORIGINAL ARTICLES
Closed suction drainage versus closed simple drainage in the management of modified radical mastectomy wounds
E R EzeomeI; C A AdebamowoII
IMB BS, MA, FWACS; Department of Surgery, University of Nigeria Teaching Hospital, Enugu, Nigeria
IIMB BS, ScD, FWACS; Division of Oncology, Department of Surgery, University College Hospital, Ibadan, Nigeria
ABSTRACT
OBJECTIVE: To compare the outcomes of modified radical mastectomy wounds managed by closed wound drainage with suction and without suction.
METHOD: A prospective randomised trial was conducted at the University College Hospital in Ibadan, and the University of Nigeria Teaching Hospital in Enugu. Fifty women who required modified radical mastectomy for breast cancer were randomised to have closed wound drainage with suction (26 patients) and closed wound drainage without suction (24 patients).
RESULTS: There was no significant difference in the intraoperative and postoperative variables. Suction drainage drained less volume of fluid and stayed for a shorter time in the wound, but the differences were not significant. There was no difference in the length of hospital stay, time to stitch removal, and number of dressing changes. More haematomas and wound infections occurred in the simple drain group while more seromas occurred in the suction drain group, but these were not significant. The suction drain was more difficult to manage and the cost was 15 times higher than the simple drainage system.
CONCLUSION: Closed simple drains are not inferior to suction drains in mastectomy wounds and, considering the cost saving and simplicity of postoperative care, they are preferable to suction drains.
Since the first mastectomy was performed for carcinoma of the breast, management of the resulting large wound has always posed problems. Of the measures employed to manage mastectomy wounds, closed suction wound drainage has been accepted as the most effective in reducing morbidity.1,2 However, suction drains are expensive and out of reach of many surgeons in developing countries where open and closed simple drains are in common use. Whereas open drains have been shown to be inferior to both closed suction and closed simple drains,2-4 no study has shown that suction is a necessity in mastectomy drains. A study that compared closed drains with and without suction suggested that there were no significant differences in their performance.5
Our aim was to compare the outcomes of modified radical mastectomy wounds managed by either closed suction drainage or closed simple drainage. The outcomes measured were the frequency and severity of complications directly or indirectly related to fluid collection and drainage in mastectomy wounds; the overall cost of wound management by each drainage system; particular problems of each drainage system; and nursing problems.
Materials and methods
All patients scheduled to undergo modified radical mastectomy at the surgical oncology unit of the University College Hospital, Ibadan, from January 1997 to May 1998, and the University of Nigeria Teaching Hospital, Enugu, from March 1999 to July 2003, were recruited for the study. One of the authors was involved in the management of all the patients. Excluded were patients who requested a particular drain, and patients scheduled for immediate breast reconstruction. Ethical approval was obtained before the commencement of the study.
Preoperative evaluation of patients noted their age, height, weight, body mass index, bra size, breast volume, side of the lesion, and dominant hand. Patients were randomised for either the suction drain or simple drain by drawing lots from a sealed bag containing equal numbers of both options. Preoperative, operative and postoperative management were identical in all patients except for the use of drains. The closed simple drain was fashioned out of a feeding tube with multiple fenestration of the intra-wound segment, and connected to a drainage bag. The suction drain group had Redivac wound closure apparatus No. 1800 from Zimmer Orthopedics Ltd, UK. This drain (the only version of suction drains commonly available in Nigeria) uses a non-compressible plastic bottle and creates high suction pressure in the wound. A motorised suction machine was used to recharge the suction drains each time they lost vacuum.
All the patients had a modified radical mastectomy. Axillary clearance was done up to level III nodal area. At the end of axillary dissection, the selected drain was placed to run from the lateral side of the wound up to the axilla and down to the medial side of the wound. The drain was brought out through a separate stab wound on the lateral side of the wound. All patients had a standard external wound dressing with meshed gauze over the incision site, followed by Gamgee tissue; these were held in place by a circumferential elastic bandage on the chest wall.
Drain volumes were recorded daily. These were initially read off directly from the calibrations on the bottles and bags but were replaced by the use of measuring jars about halfway through the study when we noticed that the calibrations on the simple drainage bags were consistently giving falsely high values. Drains were removed from the wounds once the volume of drainage per day was <20 ml for 2 consecutive days. Dressings were changed every 3 - 4 days, at which time complications were noted and appropriate treatment instituted. Seroma was aspirated as necessary until it resolved. In the case of flap necrosis, the wound was debrided and dressed as necessary. Also recorded were cases of wound infection, wound dehiscence, arm oedema and shoulder movement. Problems related to the functioning and nursing of each drain were noted, including blockage, migration or dislodgement of the drain, need for vacuum recharge, breaks or leaks in the apparatus, as well as difficulties in the management of each drain. Stitches were removed between day 10 and 14. Patients were discharged on assessment of satisfactory recovery from surgery and followed up for at least 1 month in the outpatient clinic.
Statistical analysis
The two groups were compared using the chi-square test and Student's f-test, as appropriate. The level of significance was set at a p-value of 0.05. The analysis was done using SPSS® statistical software version 9.
Results
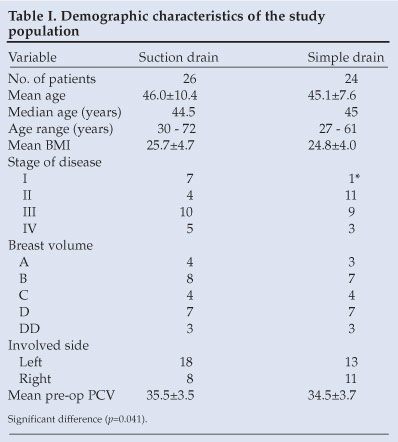
A total of 50 patients was recruited into the study. Twenty-six patients had suction drainage while 24 patients had closed simple drainage. Table I shows the characteristics of patients in the 2 groups. Both groups were similar in age, weight, body mass index and breast size. The simple drain group had more patients with stage II disease while the suction drain group had more patients with stage I disease. All the patients were right-handed but the suction drain group had more patients with left-sided breast tumours while the simple drain group had more right-sided breast tumours. The differences were not significant.
The mean operating time was 123 minutes in the suction group and 128 minutes in the simple drain group. The estimated mean blood loss in the simple drain group (866 ml) was slightly higher than in the suction group (779 ml). A median of 1 unit of blood was transfused in each group (range 0 - 2 units). The average number of nodes palpated within the removed specimen was 10 for both groups, with a range of 2 - 30 nodes for the suction-drained group and 5 - 21 nodes for the simple drain group. The mean postoperative packed cell volume (PCV) was 30% in each group.
Table II shows that the suction system drained less fluid in total (813.8 ml) and less per day (mean 86.1 ml) compared with the simple drain (965.0 ml and 96.1 ml, respectively) but these differences were not significant. The average duration of drainage was shorter in the suction group, but this was not statistically significant (9.4 days v. 9.8 days). The median duration of stay of drains was 8 days in the suction group and 10 days in the simple drain group. The mean length of hospital stay and the mean time to stitch removal was the same in both groups. The mean and median number of dressing changes both with the drains in sifu and after the drains had been removed were similar. Of the complications noted (Table III), the simple drain group had more haematomas (6 cases v. 4 cases) and a higher incidence of wound infection (5 cases v. 4 cases), but these differences were insignificant. Flap necrosis was the same in both groups, but 1 patient in the simple drain group needed debridement. The suction drain group had more seromas (12 cases v. 6 cases); these were repeatedly aspirated until resolution.
There was no difference in the incidence of blocked drains (4 cases v. 4 cases), but more patients in the suction drain group had dislodgement (5 cases v. 3 cases) while more patients in the simple drain group had breakages and leakages (2 cases v. 3 cases). These differences were not statistically significant. The suction drains were recharged on average 1 - 3 times before removal and were more difficult to empty. Overall, the suction drains needed more medical and nursing staff attention to remain effective. The cost of a suction drain was $20.00; the simple drain cost $1.25. Wound management did not differ between the two groups except for the treatment of wound complications.
Discussion
Our study compared the performance of closed suction drains with that of closed simple drains in mastectomy wounds. There was no significant difference in effectiveness, complications and duration of hospital stay between the two systems; however, simple drains were less costly and easier to manage.
Management of the large wounds that follow mastectomy still presents problems to the surgeon. Suction drains have been used for years, but the problems of suction in mastectomy wounds have only recently been appreciated. Fluid accumulation under flaps has not been eradicated, and complications are now noted to be more common; these include flap necrosis, early non-functioning of drains due to obstruction, pocketing of fluids below the flap, pain, and difficulty in removing the drains.1,6 Several alternatives and adjuncts to the use of drains have been explored, including suturing the flaps to the chest wall,7 fibrin sealant,8 external compression dressings9 and intraoperative tetracycline.10 None of these measures led to improved outcomes. More recent attempts have focused on reducing the period of drainage and eliminating other aspects of wound drainage that contribute to morbidity but not to effectiveness. High suction pressure is not superior to low pressure; hence, recent suction devices use low-pressure systems.11,12
Open mastectomy drains have been shown to be inferior to closed drains with or without suction,3,4 but no study has shown closed drains without suction to be inferior to closed drains with suction in mastectomy wounds. A study comparing the two concluded that the performance of siphon drains (drains without suction) is comparable to that of suction drains, without the associated risk of vacuum loss.5 We found no significant difference between the two drains but showed that the simple drains were much cheaper and easier to manage. This is a considerable advantage, especially in resource-poor areas with limited staff.
Our study showed that simple drains had an insignificant but definite tendency to drain more fluid compared with suction drains. Errors introduced by our initial readings, which were made directly from the calibrations on the simple drainage bags and which were falsely high, cannot be ruled out. Most of our patients had right-sided tumours; it was therefore expected that patients in the simple drainage group would have more seromas because their mastectomies were on the same side as the dominant hand. However, the simple drain group had less seromas but more haematomas. This fact suggests that suction drainage empties the initial collection (haematoma) better but creates a false impression of complete drainage, and a consequent tendency to remove drains prematurely. The higher incidence of seromas, on the other hand, suggests that drainage was more complete in the group where simple drains stayed slightly longer in the wounds. In addition, the faster adhesion of the flaps on the chest wall with suction drains might have led to pocketing of fluid under the flaps and hence the increased incidence of seromas.3
We experienced no difference in the incidence of tube blockage, which was contrary to a study where the simple drain had a lower incidence of tube blockage.5 This may be related to the type of materials used for simple drains. The suction drains needed more medical and nursing staff time. On average, the system needed to be recharged 1 - 3 times before each drain was removed. This is a significant problem if suction has to be recreated by a motorised system. More modern versions of suction drains (such as the Haemovac, Jackson-Pratt and Surgivac drains) use compressible bottles to create suction (without the need for motorised suction), which is easier to manage. However, some of them are more expensive (US$15 - 40) compared with the model we used.13
The only significant difference between the two groups was cost and ease of management. Suction drains cost almost 15 times more than simple drains. In terms of complications, one needs to consider the cost of treating haematomas and wound infections with seromas. Since there is no significant difference in the incidence of complications, a difference in the management of complications is not expected. We controlled intraoperative variables by reporting patients managed by only 1 surgeon. This made our study population small and might have 'underpowered' the study in the detection of small differences that occurred in some parameters, such as seroma and haematoma.
We thank the physiotherapy departments at UCH Ibadan and UNTH Enugu for allowing us use of their goniometer in this study.
References
1. Moss JP. Historical and current perspectives on surgical drainage. Surg Gynaecol Obstet 1981; 152: 517-527. [ Links ]
2. Aitken DR, Hunsaker R, James AG. Prevention of seromas following mastectomy and axillary dissection. Surg Gynaecol Obstet 1984; 158: 327-330. [ Links ]
3. Morris AM. Controlled trial of closed wound suction drainage in radical mastectomy. Br J Surg 1973; 60: 357-359. [ Links ]
4. Sheppard MD. Sealed drainage of wounds. Lancet 1952; 1: 1174-1176. [ Links ]
5. Whitfield PC, Rainsbury RM. Suction versus siphon drainage after axillary surgery for breast cancer: a prospective randomized trial. Br J Surg 1994; 81: 546-547. [ Links ]
6. McCullum GT. Some hazards of suction drainage of large skin flaps. Ann Surg 1961; 154: 990-992. [ Links ]
7. O'Dwyer PJ, O'Higgins NJ, James AG. Effect of closing dead space on incidence of seroma after mastectomy. Surg Gynecol Obstet 1991; 172: 51-55. [ Links ]
8. Moore M, Burak WE, Nelson E, Kearney T, Sommons R, Mayers L. Fibrin sealant reduces the duration and amount of fluid drainage after axillary dissection: a randomized prospective clinical trial. J Am Coll Surg 2001; 192: 591-599. [ Links ]
9. O'Hea BJ, Ho MN, Petrek JA. External compression dressing versus standard dressing after axillary lymphadenectomy. Am J Surg 1999; 177: 450-455. [ Links ]
10. Rice DC, Morris SM, Sarr MG, et al. Intraoperative topical tetracycline sclerotherapy following mastectomy: a prospective randomized trial. J Surg Oncol 2000; 73: 224-227. [ Links ]
11. Wedderburn A, Gupta R, Bell N, Royle G. Comparison between low and high pressure suction following axillary clearance. Eur J Clin Oncol 2000; 26: 142-144. [ Links ]
12. Chintamani, Singhal V, Singh JP, Bansal A, Saxena S. Half versus full suction drainage after modified radical mastectomy for breast cancer - a prospective randomized clinical trial. BMC Cancer 2005; 5: 11-16. [ Links ]
13. Buyers guide. Contract HCNS 102405 for the supply of wound drainage systems. Perth: Department of Health, Government of Western Australia. http://www.healthcorporatenetwork.health.wa.gov.au/supply/contract_search.cfm (accessed 7 January 2008). [ Links ]
 Correspondence:
Correspondence:
E R Ezeome
(ezeome95@gmail.com)
Accepted 8 January 2008














