Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 n.9 Pretoria Sep. 2008
SCIENTIFIC LETTERS
Intestinal parasitic infections in adult patients in KwaZulu-Natal
Z L KwitshanaI; J M TsokaII; M L H MabasoIII
INat Dip MT, Sp Dip Chem Path, NHD MT, MSc (Med); Helminthiasis Research Group, Nutritional Intervention Research Unit, Medical Research Council, Durban
IIBSc Hons, MSc, MPH, PhD; Research Management Division, Medical Research Council, Cape Town
IIIBSc, BSc Hons, MSc, PhD; Malaria Research Lead Programme, Medical Research Council, Durban
To the Editor: Intestinal parasitic infections are among the most common chronic human infections in developing countries, particularly in the tropical and subtropical regions. The major groups of parasites include geohelminths, schistosomes and protozoans1 that are associated with malnutrition, iron deficiency anaemia, and impaired growth and cognitive development caused by decreased appetite, nutrient loss, malabsorption and decreased nutrient utilisation. However, intestinal parasitic infections receive little attention as most are asymptomatic and generally considered to be of less clinical significance than bacterial and viral infections.1
The geographical distribution of intestinal parasites has been shown to coincide with that of HIV/AIDS under conditions of poverty in most countries in sub-Saharan Africa. Interest has therefore increased in the pathological interaction between parasitic infections and HIV/AIDS, particularly in adults.2,3 Regrettably, there are few data on the prevalence of intestinal parasites in the adult population since most surveys focus on school-age children who carry the heaviest morbidity and mortality burden.4 Similarly, in KwaZulu-Natal (KZN), the third poorest province in South Africa, with a high prevalence of HIV/AIDS in teenagers and middle-aged adult populations, there are scant data on the prevalence of helminth and protozoan infections.
We therefore studied the occurrence of helminth and protozoan infections in adult patients throughout KZN from stool samples obtained from regional laboratory services. Ideally, a community-based household survey would provide such information but it would have been difficult and costly to collect stool samples from households in the entire province. Hence, readily available stool samples were sourced.
Methods
Stool samples from adults >18 years of age were obtained from 32 randomly selected public hospital laboratories in all 8 former health regions of the province. The stool samples were processed in the laboratory for intestinal parasites using the formol ether concentration technique,5 and screened for intestinal parasites using microscopy. The samples were also cultured in Robinson's medium to enhance detection of Entamoeba species.6
Results
The overall prevalence of parasites in 5 733 screened stool samples was 20.4%. Ascaris lumbricoides (10.7%) and Trichuris trichiuria (6.7%) were the most common helminth infections, followed by hookworm and Schistosoma mansoni. Entamoeba coli and Endolimax nana were the most commonly detected protozoan parasites, while Isospora, Cryptosporidia and other Coccidia species were less common (Table I). The prevalence of intestinal parasites varied geographically, with the highest infection rates in coastal regions (Fig. 1); this ranged from 30.3% in Jozini (coastal) to 11.2% in Newcastle (inland). The highest levels of A. lumbricoides were recorded in Port Shepstone, Empangeni and Jozini (18%, 15% and 14% respectively). T. trichiura was most prevalent in Jozini, Port Shepstone and Durban regions. Hookworm and schistosoma species were most common in the Jozini region (Table I).
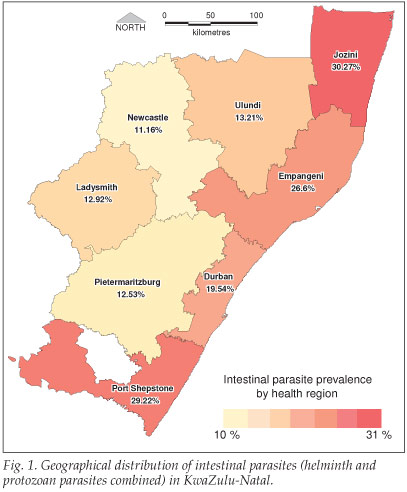
Discussion
Our findings suggest that the occurrence and distribution of intestinal parasites among the adult population varies widely across KZN. The most common parasite in all regions was A. lumbricoides, except for Jozini in the northern region where T. trichiuria was the most prevalent. This region also had the highest prevalence of hookworm infections. S. mansoni was highest in the coastal regions of Port Shepstone, Durban, Empangeni and Jozini, compared with inland regions such as Pietermaritzburg and Newcastle, where none was detected. The distribution of intestinal parasites in South Africa has been attributed to the occurrence of suitable climatic and/ or environmental conditions.7,8 Socio-economic differences between rural, urban and peri-urban areas are also important determinants of the risk of infection.9
The geographical distribution of the most prevalent protozoan parasites also varied widely across the province, with the highest rate of E. coli infections found in the Newcastle and Jozini regions, while E. nana was highest in Ladysmith, followed by the Jozini and Durban regions. Poor environmental sanitation including polluted water and food and direct faecal contamination are the main determinants of their occurrence and distribution.9 The detection of the less common protozoan parasites such as Cryptosporidia, Isospora and other Coccidia is interesting since they are often associated with an increased prevalence of HIV/AIDS.10
In conclusion, while health facility-based data may be less representative of the actual adult population in the respective health regions in the province, this study nevertheless gives an indication of regions with relatively high prevalences of intestinal parasites in the adult population. This is important for the purpose of developing and implementing effective interventions in the light of the escalating HIV/AIDS pandemic, which has been suggested to be related to helminth parasitic infections in Africa,3 and also provides useful data for diagnostic laboratories' parasite screening policies. At the time of data collection, only 8 of the 32 laboratories confirmed that they routinely screened all the submitted stools for parasites. The other participating laboratories indicated that parasite screening is only done upon request by the clinician. Intervention programmes could benefit from routine screening of all stools including the adult population, particularly in areas which are endemic to intestinal parasites and HIV/AIDS.
This study was funded by the South African Medical Research Council. We are also grateful to: Mr J M P Fraser, then Deputy Director, Regional Laboratory Services; the Heads of the Regional Laboratory Services; the Control Technologists-In-Charge of all the participating KZN laboratories; Professor T F H G Jackson and the Amoebiasis Research Unit staff, MRC; Mrs Natashia Morris, Health GIS Centre, Malaria Research Programme, MRC; and Dr David Katerere, PROMEC unit, MRC.
References
1. World Health Organization. Deworming for Health and Development. Report of the Third Global Meeting of the Partnners for Parasite Control. Geneva: World Health Organization, 2005. [ Links ]
2. UNAIDS and World Health Organization. United Nations }oint Report on HIV/AIDS. Report on the Global HIV/AIDS Epidemic. Geneva: UNAIDS and World Health Organization, 1997. [ Links ]
3. Bentwich Z, Maartens G, Torten D, Lal AA, Lal RB. Concurrent infections and HIV pathogenesis. AIDS 2000; 14: 2071-2081. [ Links ]
4. Bundy DAP, Shaeffer S, Jukes M, et al. School-Based Health and Nutrition Programs. In: Jamison DT, Breman JG, Maesham AR, et al., eds. Disease Control Priorities in Developing Countries. 2nd ed. Washington: World Bank and Oxford University Press, 2006: 1091-1108. [ Links ]
5. Ritchie LS. Ether sedimentation technique for routine stool examination. Bull US Army Med Depart 1948; 8: 326. [ Links ]
6. Robinson GL. Laboratory cultivation of some human parasitic amoebae. J Gen Microbiol 1968; 53: 67-79. [ Links ]
7. Appleton CC, Maurihungirire M, Gouws E. The distribution of helminth infections along the coastal plain of Kwazulu-Natal province, South Africa. Ann Trop Med Parasitol 1999; 93: 859-868. [ Links ]
8. Moodley I, Kleinschmidt I, Sharp B, Craig M, Appleton CC. Temperature-suitability maps for schistosomiasis in South Africa. Am Trop Med Parasitol 2003; 97: 617-627. [ Links ]
9. Crompton DWT, Savioli L. Intestinal parasitic infections and urbanization. Bull World Health Organ 2000; 78(1): 1-7. [ Links ]
10. Lockwood DNJ, Weber JN. Parasitic infections in AIDS. Parasitol Today 1989; 5: 310-316. [ Links ]
Accepted 17 April 2008














