Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 no.8 Pretoria ago. 2008
ORIGINAL ARTICLES
Pseudomonas aeruginosa bacteraemia in an academic hospital in South Africa
O PerovicI; H H Crewe-BrownII; A G DuseIII; H J KoornhofIV; W van NieropV; J S GalpinVI
IMD (Belgrade), DTM&H, FCPath (SA) (Microbiol), MMed (Microbiol); Department of Clinical Microbiology and Infectious Diseases, National Health Laboratory Services and University of the Witwatersrand, Johannesburg
IIMB BCh, DCH (RCPSE), DTM&H, FCPath (SA) (Microbiol); Department of Clinical Microbiology and Infectious Diseases, National Health Laboratory Services and University of the Witwatersrand, Johannesburg
IIIMT, MB BCh, MMed (Microbiol), DTM&H, FCPath (SA) (Microbiol); Department of Clinical Microbiology and Infectious Diseases, National Health Laboratory Services and University of the Witwatersrand, Johannesburg
IVMB ChB, DCP (Lond), Dip Bact (Lond), FRCPath (Lond); National Institute for Communicable Diseases, Johannesburg
VMSc; Diagnostics Media Products and National Institute for Communicable Diseases, Johannesburg
VIBSc, BSc Hons, MSc, DSc; School of Statistics and Actuarial Science, University of the Witwatersrand
ABSTRACT
OBJECTIVES: The aim of the study was to determine the clinical manifestations, outcome of and prognostic factors associated with Pseudomonas aeruginosa bacteraemia at Chris Hani Baragwanath Hospital, Johannesburg, during the period 1998 -1999, to describe and quantify resistance to antipseudomonal drugs, to characterise bacteraemic isolates, and to investigate the clustering and genotype distribution of drug-susceptible and multiply-resistant strains in the hospital.
METHODS: Clinical and laboratory investigations, culture and antimicrobial susceptibility testing were performed. Bacteraemic isolates were typed by endonuclease macrorestriction. Those with >97% band pattern similarity were assigned genotype status.
RESULTS: Of 91 P. aeruginosa blood isolates, 52 (57.1%) were nosocomially acquired. Underlying conditions associated with episodes were burns in 24 (28%) and HIV infection in 21 (24%). Multidrug resistance was present in 14 (15.4%) of isolates from 91 episodes. In 79 episodes where the outcome was known, 37 (46.8%) of the patients died. Case fatality rates were 75% in adults and 25% in children. Being a child, receiving appropriate antimicrobial treatment and admission to a specialised care unit were significantly associated with improved prognosis.
CONCLUSION: P. aeruginosa bacteraemia was associated with outbreaks caused by two multiply-resistant genotypes. Eighteen antimicrobial-susceptible isolates from bacteraemic episodes in paediatric wards, 9 in HIV-seropositive children, could be linked to small outbreaks in both hospitalised and community-based children.
The incidence of Pseudomonas aeruginosa bacteraemia (PAB) has remained stable over the last few decades.1-3 Although it is still primarily a nososcomial infection, the number of cases of community-acquired bacteraemia caused by this organism has increased, notably affecting patients with AIDS4,5 and neutropenic patients treated for neoplastic disease who received outpatient management.6 Predisposing conditions for PAB include compromised immunity, neutropenia, intensive care, surgical procedures, central venous and urinary catheters and previous cephalosporin therapy.1,3-5,6 Common factors predictive of a fatal outcome reported in the literature are septic shock, neutropenia, immunocompromised state, severe underlying disease, and in the elderly pneumonia, septic metastases, previous therapy and inappropriate choice of antimicrobial drugs for definitive treatment.1,6,7
P. aeruginosa has also emerged as an important bacteraemic pathogen in immunocompromised children,6,8,9 including very-low-birth-weight infants,9 and as a cause of community-acquired pseudomonal sepsis in previously healthy infants and children often initially presenting with diarrhoea.10
During the pre-1990 period when PAB in hospitals peaked, improved outcome of this condition was reported widely, e.g. PAB mortality declined significantly from 53% to 29% from the period 1976 - 1982 to 1992 - 1996.1 Mortality resulting from PAB, however, remains high in certain hospital settings, e.g. 50% in very-low-birth-weight infants,9 33% in AIDS patients in a tertiary care hospital, and 39% at a university hospital in the early 2000s.7
Since PAB, including episodes caused by a multidrug-resistant (MDR) strain, is an important problem at Chris Hani Baragwanath Hospital (CHBH), Johannesburg, a clinical and epidemiological investigation of PAB was conducted in 1998 - 1999. Typing of P. aeruginosa isolates by pulsed-field gel electrophoresis (PFGE) following chromosomal macrorestriction has been shown to be epidemiologically useful for strain differentiation,11-13 and this technique was employed to investigate P. aeruginosa transmission patterns in the hospital setting.
The study aimed to assess the clinical manifestations, outcome and prognostic factors associated with PAB, to describe and quantify resistance to antipseudomonal drugs, and to perform genotypic characterisation of P. aeruginosa isolates causing bacteraemia at CHBH.
Materials and methods
Setting
CHBH is a 3 000-bed tertiary care hospital serving an estimated 1.5 million people, mainly from Soweto, a densely populated part of greater Johannesburg, South Africa, mainly inhabited by black persons of low socioeconomic status. The hospital has surgical, medical and paediatric wards, a large general intensive care unit (ICU) and a sophisticated burns centre with separate units for paediatric (PBU) and adult (ABU) patients. During the study period no antiretroviral treatment was available at CHBH and other public hospitals and the Annual Antenatal Survey prevalence of HIV among pregnant women aged 15 - 24 years was 23% (Department of Health - unpublished data).
Study outline
This was a retrospective study in which P. aeruginosa isolates from blood cultures were collected and stored for future analysis. All patients admitted to CHBH in 1999, as well as all adults admitted in 1998 with a positive blood culture for PAB, were entered into the study. Records from enrolled patients were reviewed. Episodes of PAB were included for investigation when the possibility of contamination or pseudobacteraemia could be excluded and patients' clinical features were compatible with sepsis. Subsequent episodes were recorded in the same patient when the clinical and bacteriological response suggested that the previous episode had been cured or the organism/s eradicated. All positive blood cultures from the same patient obtained within 2 weeks of the index culture of an episode were regarded as part of the same episode and were kept in reserve but not analysed. Polymicrobial episodes were included in the study. All P. aeruginosa cultures were tested for antimicrobial susceptibility and kept on semi-solid nutrient agar for genetic typing.
Genotyping of P. aeruginosa isolates was linked to episodes in relation to time and patient location in the hospital. Treatment details including duration of antimicrobial administration were also available from patients' records.
Categories and definitions
By definition an episode of sepsis required at least two of the following systemic features in addition to a positive blood culture: temperature >38°C or <36°C; pulse rate >90 beats/min; respiratory rate >20 breaths/min (adults) and white cell count >12x109/l or <4x109/l . PAB acquired 2 days or more after admission was defined as nosocomial.
Microbiology
Blood samples were processed using the BacT/Alert system (Organon Teknika, USA). Sets of aerobic and anaerobic blood cultures were processed and incubated routinely for 7 days.
P. aeruginosa was identified according to standard microbiological procedures. Susceptibility to 10 single antipseudomonal antibiotics (piperacillin, ceftazidime, cefepime, imipenem, meropenem, ciprofloxacin, ofloxacin, amikacin, gentamicin and tobramycin) and the piperacillin/ tazobactam (PIP/TAZ) combination, all supplied by Mast Diagnostics (Mast House, Derby Road, Bootle, UK) was determined by the disc diffusion method, minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) determinations. The micro-dilution method was performed according to Clinical Laboratory Standards Institute (CLSI)14 criteria. Multidrug resistance (MDR) was defined as resistance to two or more classes of antipseudomonal agents represented by cephalosporins (ceftazidime and fourth-generation cefepime), ureido-penicillins (PIP/TAZ), carbapenems, aminoglycosides and fluoroquinolones.
Polymicrobial bacteraemia episodes were recorded when additional microbial pathogens together with P. aeruginosa were recovered from blood cultures during an episode of sepsis. Underlying diseases or conditions associated with PAB, including polymicrobial episodes, were noted and included burns, HIV infection, haematological malignancies, neonatal status, diabetes mellitus and malnutrition. If HIV status was not available, clinical criteria (World Health Organization) for AIDS were implemented. Underlying factors specific to PAB included for analysis were invasive procedures, systemic corticosteroid therapy, cytotoxic therapy, surgery, intravascular catheters, granulocytopenia and ICU admission. For each bacteraemic episode, evidence of localised endogenous sources of infection was recorded from patients' clinical records. More than one source per episode was recorded when suggested on clinical grounds.
Antibiotic therapy was considered empiric if it was started before the results of cultures and sensitivity testing were known and definitive when it was instituted after the P. aeruginosa causation and drug susceptibility were established. Antimicrobial therapy was defined as appropriate if the strain isolated was susceptible to the administered antipseudomonal antibiotic/s and the doses and routes of administration were adequate.
Appropriate use of antimicrobial therapy for P. aeruginosa bloodstream infections in relation to treatment outcome was assessed in patients receiving monotherapy, defined as the use of carbapenems, an antipseudomonal β-lactam, a fluoroquinolone or an aminoglycoside agent, and in patients given combination therapy, comprising an antipseudomonal β-lactam agent plus an aminoglycoside or a fluoroquinolone. Patients who died within 24 hours of admission were excluded from analysis of appropriate use of antimicrobial agents.
Death was attributed to PAB if clinical deterioration and death occurred in the presence of active signs of P. aeruginosa infection.
Epidemiological investigations
For genotyping, restriction fragment length polymorphism (RFLP) of isolate DNA using pulsed-field gel electrophoresis (PFGE) was performed. The typing approach used by Struelens et al.11 was followed with minor modifications. Whole chromosomal DNA in agarose was digested with the Xba I enzyme and the restriction fragments separated in a CHEF DR II apparatus (Bio-Rad, Richmond, CA). The gel was run at 14°C, 6 V/cm and a 120°C switch angle for 18 hours. The preparation was then stained with ethidium bromide and photographed under ultraviolet illumination to detect similarities and differences in banding patterns.
Statistical analysis
Logistic regression was used to obtain odds ratios, using SPSS statistical software. Other analyses were done using contingency tables.
Similarity between band patterns of isolates was determined using a computerised scanner (software, Jelcompar version 4.1; Applied Mats-Belgium). Findings were recorded in the form of dendrograms exhibiting the degree of similarity between isolates expressed in percentages. Strains with similarity of 97% (3 - 6 band differences) are equivalent to Tenover et al.'s12 category of 'possibly related' strains and isolates within a >97% similarity cluster were assigned genotype status.11
Results
Clinical features
During the 2-year study period P. aeruginosa isolates from 91 bacteraemic episodes in 86 patients were collected (1 patient had three episodes and 3 patients had two separate episodes each, all at least 2 weeks apart). Demographic and clinical data are presented in Table I, as are underlying conditions that potentially contributed to PAB.
Fifty-seven per cent of bacteraemic episodes were nosocomial and 35.2% community-acquired (unknown 7.7%). The most common underlying conditions were burns (28%), HIV infection (24%), trauma (14%) and haematological malignancies (9%).
The probable source of infection was based on clinical information from hospital records in 79 episodes (86.8%) and unknown in 12 (13.2%). More than a third of episodes (35%) were catheter-related. Of the 32 community-acquired infections, 15 affected the respiratory tract, 14 with clinical features suggesting pneumonia and 1 with chronic obstructive pulmonary disease. In contrast, a clinical diagnosis of pneumonia accounted for 18 of the 52 nosocomial infections, and 11 of these patients required assisted ventilation. Nine of the 32 community-acquired infections appeared to have an intestinal origin, as evidenced by the development of diarrhoea, mainly in children.
A total of 34 bacterial pathogens other than P. aeruginosa were identified in 25 polymicrobial episodes (27.5%), all nosocomially acquired. There were 7 isolates of Acinetobacter baumannii, 7 Staphylococcus spp. (3 methicillin-resistant S. aureus (MRSA), 2 methicillin-resistant S. epidermidis (MRSE), 2 coagulase-negative staphylococci), 12 members of Enterobacteriaceae, 5 Enterococcus spp., 1 group B streptococcus, 1 P. rubrum, and 1 viridans streptococcus.
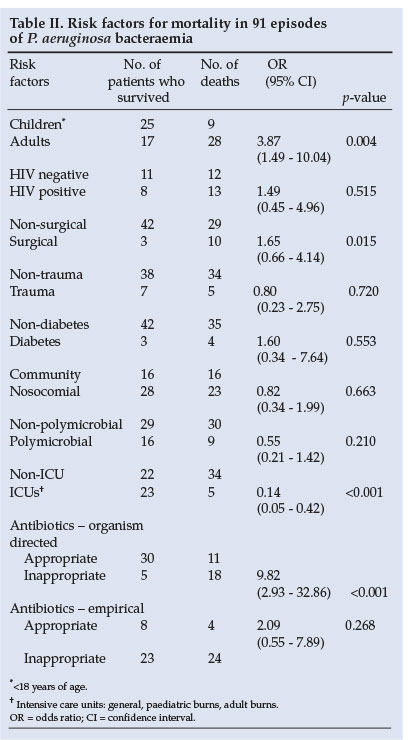
Of the risk factors that contributed to mortality, inappropriate organism-directed treatment was most prominent, with a p-value of <0.001. ICU admission was associated with a decrease in mortality compared with non-ICU patients (p<0.001) (Table II).
Of the 91 isolates, 21 (23.1%) were resistant to the antipseudomonal antibiotics tested. CLSI MIC breakpoint concentrations showed resistance to individual antibiotics to be 16.5% for each of piperacillin and PIP/TAZ, 14.3% for cefepime, 13.2% for each of ceftazidime and carbapenems, 4.4% for amikacin and gentamicin, and 3.3% for tobramycin, while only 1 isolate (1.1%), an untypable MDR strain, was resistant to the fluoroquinolones tested.
Information relating to organism-directed treatment was available for 64 patients. Of those receiving appropriate organism-directed therapy, 15 patients on monotherapy (75%) survived and 5 (25%) died, while of patients receiving combination therapy 17 (81%) survived and 4 (19%) died, showing no statistically significant difference. All patients with concomitant MRSA/E isolates received vancomycin in addition to Pseudomonas cover. Patients who received inappropriate monotherapy did poorly and 14 (73.7%) died. Empirical use of appropriate antibiotics did not significantly decrease mortality (Table II). Organism-directed therapy however, significantly decreased mortality (p<0.001) when appropriate antibiotics were used (Table II).
Epidemiological findings
Genetic analysis was performed on 82 P. aeruginosa isolates. Pulsed-field gel electrophoresis (PFGE) band patterns of isolates from ICU (isolates numbered 1 - 9), ABU (isolates 10 -16) and PBU (isolates 17 - 24) are shown in Fig. 1. A composite dendrogram indicating the degree of genetic relatedness of isolates from specialised ICUs (labelled 1 - 24) and paediatric (labelled P1 - P26), medical (M1 - M23) and surgical wards (S1- S11) is shown in Fig. 2. Seven isolates, including one MDR isolate from a 62-year-old patient in renal failure, showed poor band definition and are not included in the dendrogram.
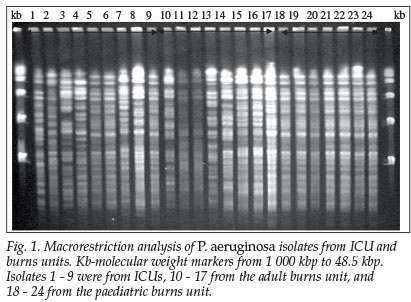

Thirteen of the 14 MDR isolates were from ICU and PBU patients. Eleven of these were linked to two genotypes (clusters with >97% similarity), accommodating isolates 5, 6, 9, 18, 19 and 20 in genotype 1 (3 from ICU and 3 from PBU with 99% and 100% similarity respectively) and isolates 17, 21, 22, 23 and 24 in genotype 2 (Table III). The temporal relationship of MDR genotype clusters expressed in 3-month periods (quarters) is shown in Table III. All 14 MDR strains were isolated during 1999 and with one exception (an adult surgical patient) were in the ICU and special burn units. Distinct temporal associations can be seen, providing strong evidence of transmission involving endemic clones or patient-to-patient transmission. Interestingly, a 9-month-old infant whose isolate (number 6) belonged to genotype 1 (5, 6, 9, 18, 19, 20) was admitted to the PBU on 1 March 1999 and transferred to the ICU on 10 March 1999, where a blood culture taken 1 day later was positive for P. aeruginosa. This isolate was probably acquired in the PBU, as were isolates 18, 19 and 20. However, isolate 5 from an 18-year-old patient in ICU was recovered on the same day as isolate 6, suggesting that this genotype was already present in the ICU by 11 March 1999.
Eight clusters (genotypes) involving 18 isolates (9 from episodes in HIV-seropositive children clinically compatible with AIDS) and 2 - 4 isolates per cluster were identified in paediatric wards. Most clusters showed temporal associations and 3 clusters involved neonates. One community-acquired cluster involved 4 infants suffering from AIDS. Four small clusters comprising 2 isolates each, all linked by time, were demonstrated in medical wards while no clustering was found in 7 surgical patients with PAB.
In paediatric wards 26 PAB isolates were typed. Of these isolates, 18 grouped into 8 clusters (genotypes), varying in size from 2 to 4 isolates. Two of the 8 clusters included isolates from other areas of the hospital (ICU and an adult medical ward). Importantly, 16 of the 18 isolates were drug-susceptible (non-MDR), while 9 of the 18 bacteraemic episodes occurred in HIV-seropositive children with clinical presentations compatible with AIDS. Most clusters developed over relatively short periods and 3 clusters occurred in neonates. One hospital-linked cluster involved 4 infants with AIDS, and their PABs were judged to be community-acquired.
Four small clusters comprising 2 isolates each, all temporally linked, were demonstrated in medical wards, while no clustering was found in 7 surgical patients with PAB.
Discussion
Pseudomonas bacteraemia is a major problem at CHBH. In the present series of 91 bacteraemic episodes the mortality rate was 46.8% for the 79 patients whose outcome was known. In a comprehensive review by Maschmeyer and Braveny15 and a more recent paper by Kang et al? mortality rates of 28% and 48% respectively were reported.
In the present study the age of the patients had an effect on outcome and mortality was higher in adult patients (p< 0.001). The lower mortality rate of 11.4% in children may be attributed to the nature of their underlying conditions, including burns and newborn status, where early diagnosis and specialised care were more accessible and appropriate antimicrobial treatment more likely. In two university hospitals in Taiwan, 23% of previously healthy infants and children, who in most cases (>70%) initially presented with diarrhoea, died as a consequence of P. aeruginosa bacteraemia.10
In the present study, inappropriate treatment was significantly associated with an increased risk of death (p<0.001).
Underlying conditions that contributed substantially to deaths from pseudomonas bacteraemia in this study were burns (28%) and HIV infection 24% (in 86 patients).
P. aeruginosa infections are often endemic in burn patients, and in a large study a mortality rate of 80% was reported.16 In the present study mortality was 36% in burn patients, but only 14.3% among those admitted to a specialised burns unit.
The role of P. aeruginosa as an important pathogen in children, especially in premature infants, has been known for 50 years.17 Of the paediatric patients in the present study, 6 were neonates. In contrast to the report of Tsai et al., none of our patients presented with necrotising enterocolitis. Diarrhoea, however, reported by others, was noted in community-acquired PAB in our series.10,18,19
MDR P. aeruginosa strains were identified in 15.4% of 91 isolates in our study with evidence of clonal spread in ICU and burns units.
Empirical treatment was found not to influence patient outcome significantly (p=0.268). There was no statistical difference in outcome between patients on appropriate monotherapy versus those on appropriate combination therapy (p=0.6). Empirical monotherapy with antipseudomonal antibiotics was found to have a significant impact on patient outcome compared with inappropriate use of a single agent (p=0.002).
For organism-directed therapy, appropriate treatment was significantly associated with reduced mortality (p<0.001) (Table II). As mentioned above, appropriate monotherapy with antipseudomonal drugs was also effective and in the relatively small number of cases available for analysis the outcome was statistically similar to that of combination therapy with a β-lactam and aminogycoside. Published studies indicate that combination therapy is not necessarily more advantageous than monotherapy, but infectious disease experts still favour the use of combination therapy.20,21
With regard to epidemiological findings, molecular typing using PFGE of macrorestriction fragments following digestion by Xba I endonuclease producing 25 - 35 fragments in the 10 - 30 kb range has been used by Belgian and French workers to good effect.13Spe I endonuclease has been preferred by other authors because of slightly superior band separation (20 - 25 fragments).12 PFGE of Xba I cut fragments used by us proved to be very useful, especially in establishing genetic linkage between MDR strains in specialised ICU settings and in paediatric wards where genotype analysis produced strong evidence of clonal spread either through transmission between patients or by means of single-source outbreaks involving environmentally derived clones. For example, MDR strains 5 and 6 at 99% PFGE similarity were isolated from the ICU in March 1999 at the same time as strain 18 from the PBU, which was indistinguishable from strains 19 and 20, both isolated in May and June 1999 (2nd quarter, see Table III). This cluster, which at 98% PFGE similarity also incorporated the MDR strain 9 isolated in May 1999, represents an outbreak in infants and children in ICU and PBU, caused by an MDR clone over a 6-month period. A somewhat different and more complex situation applies to the cluster 15, 16, 21, 23 at 98% similarity, 15 and 16 being susceptible and 21 and 23 multiply resistant. Based on epidemiological evidence, this large 7-member cluster at 97% similarity, (genotype 2, see Table III) provides evidence of another MDR-associated outbreak involving predominantly infants and children and may well present an expanded clone initially present in ABU as susceptible strains 15 and 16 and subsequently as MDR strains in PBU (strains 21, 22 and 23 showed identical resistance profiles and strain 24 was multiply resistant but susceptible to ceftazidime) (Table III). These findings suggest the acquisition and transmission of mobile resistance gene cassettes in the form of integrons,22 involving the expanded clone (genotype 2).
The presence of MDR strains and evidence of transmission in specialised ICU facilities at the hospital suggest that extensive use of antimicrobial agents may have played a selective role, while prolonged stay in these facilities, especially in burns units where wound colonisation with P. aeruginosa was common, created ample opportunities for selection of MDR strains and transmission.
Other workers have also studied MDR transmission patterns by PFGE typing. Pitten et al.23 described a large outbreak in a German academic hospital where all the isolates showed an identical band pattern, Richard et al.,24 using PFGE of exotoxin A gene, showed that previous treatment with ceftazidime at a low dose of 3 g daily in a burns unit was a risk factor for MDR emergence, and Hsueh et al.25 described a small outbreak caused by a MDR P. aeruginosa clone in an intensive care burn unit where the clone persisted in different body sites and colonisation was followed by severe PAB in 2 out of 4 patients.
Limitations of our study relate mainly to the retrospective retrieval of clinical information and include insufficient data on the clinical status of patients and outcome of bacteraemic episodes in a few instances and a lack of detailed information on previously administered antimicrobial treatment.
Conclusions
In summary, based on DNA fingerprinting and temporal linkage our study provides convincing evidence of nosocomial transmission of MDR strains of P. aeruginosa in ICU and PBU. Furthermore, we have been able to show endemicity of these strains.
The mortality rate of our patients was 46.8 %. As expected, failure to employ appropriate organism-directed antibiotic treatment produced the poorest outcome (p=0.001). The most common underlying conditions in our patients were burns/ trauma and HIV infection. A high number of community-acquired Pseudomonas infections were noted in this study.
At CHBH clones of MDR P. aeruginosa emerged in children hospitalised for prolonged periods, causing outbreaks in burns and other ICUs where antimicrobial agents were used extensively, while drug-susceptible genotypes caused outbreaks in children with AIDS in the hospital and in the community.
We thank the technical support group, M Khoosal and M Kassel, for being so helpful, T Gomes for doing PFGE and G Coulson for band pattern analysis by software use. This protocol was cleared by the Committee for Research on Human Subjects of the University of the Witwatersrand in 1999.
References
1. Kuikka A, Valtonen VV. Factors associated with improved outcome of Pseudomonas aeruginosa bacteremia in a Finnish university hospital. Eur J Clin Microbiol Infect Dis 1998; 17: 701-708. [ Links ]
2. Pittet D, Wenzel RP. Nosocomial bloodstream infections. Circular trends in rates, mortality and contribution to total hospital deaths. Arch Intern Med 1995; 155: 1177-1184. [ Links ]
3. Weinstein MP, Towns ML, Quartey SM, et al. The clinical significance of positive blood cultures in the 1990s: a prospective comprehensive evaluation of the microbiology, epidemiology and outcome of bacteremia and fungemia in adults. Clin Infect Dis 1997; 24: 584-602. [ Links ]
4. Dropulic LK, Leslie JM, Lois J, Eldred LJ, Zenilman J, Sears CL. Clinical manifestations and risk factors of Pseudomonas aeruginosa infection in patients with AIDS. J Infect Dis 1995; 171: 930-937. [ Links ]
5. Vidal F, Mensa J, Martinez JA, et al. Pseudomonas aeruginosa bacteremia in patients infected with human immunodeficiency virus type 1. Eur J Clin Microbiol Infect Dis 1999; 18: 473-477. [ Links ]
6. Fergie JE, Shema SJ, Lott L, Crawford R, Patrick CC. Pseudomonas aeruginosa bacteraemia in immunocompromised children: analysis of factors associated with a poor outcome. Clin Infect Dis 1994; 18: 390-394. [ Links ]
7. Kang C-I, Kim S-H, Kim H-B, et al. Pseudomonas aeruginosa bacteraemia: Risk factors for mortality and influence of delayed receipt of effective antimicrobial therapy or clinical outcome. Clin Infect Dis 2003; 37: 745-751. [ Links ]
8. Flores G. Stavola JJ, Noel GJ. Bacteraemia due to Pseudomonas aeruginosa in childbirth with AIDS. Clin Infect Dis 1993; 16: 706-708. [ Links ]
9. Leigh L, Stoll BJ, Rahman M, McGowan J. Pseudomonas aeruginosa infection in very low birth weight infants: a case-control study. Pediatr Infect Dis J 1995; 14 (5): 367-371. [ Links ]
10. Huang Y-C, Lin T-Y, Wang C-H. Community-acquired Pseudomonas aeruginosa sepsis in previously healthy infants and children: analysis of forty-three episodes. Pediatr Infect Dis J 2002; 21: 1049-1052. [ Links ]
11. Struelens MJ. Schwam V, Deplano A, Baran D. Genome macrorestriction analysis of diversity and variability of Pseudomonas aeruginosa strains infecting cystic fibrosis patients. J Clin Microbiol 1993; 31(9): 2320-2326. [ Links ]
12. Tenover FC, Arbeit RD, Goering RV, et al. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol 1995; 33 (9): 2233-2239. [ Links ]
13. Talon D, Cailleaux V, Thouverez M, Michel-Brand Y. Discriminatory power and usefulness of pulsed-field gel electrophoresis in epidemiological studies of Pseudomonas aeruginosa. J Hospital Infect 1996; 32: 135-145. [ Links ]
14. National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; Approved standard M7-A4, 1997. [ Links ]
15. Maschmeyer G, Braveny I. Review of the incidence and prognosis of Pseudomonas aeruginosa infectious in cancer patients in the 1990s. Eur J Clin Microbiol Infect Dis 2000; 19(12): 915-925. [ Links ]
16. McManus AT, Mason AD, McManus WF, Privitt JBA. Twenty-five year review of Pseudomonas aeruginosa bacteraemia in a burn center. Eur J Clin Microbiol 1985; 4: 219-223. [ Links ]
17. Foca M, Jakob K, Whitter S, et al. Endemic Pseudomonas aeruginosa infection in a neonatal intensive care unit. N Engl J Med 2000; 343: 695-700. [ Links ]
18. Tsai MJ, Teng CJ, Teng RJ, Lee PI, Chang MH. Necrotizing bowel lesions complicated by Pseudomonas septicaemia in previously healthy infants. Eur J Pediatr 1996; 40: 233-236. [ Links ]
19. Yeung CK, Lee KH. Community-acquired fulminant Pseudomonas infection of the gastrointestinal tract in previously healthy infants. J Pediatr Child Health 1998; 34: 584-587. [ Links ]
20. Lode H. Hamacher L. Eller J. Schaberg T. Changing role of carbapenems in the treatment of lower respiratory tract infections. Scand J Infect Dis 1995; 96: suppl, 17-23. [ Links ]
21. Par GB, Ramphal R. Pseudomonas aeruginosa. In: Mandell GL, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases. 6th ed. Philadelphia: Elsevier Churchill Livingstone, 1996: 2587-2615. [ Links ]
22. Norrby SN. Integrons: Adding another threat to the use of antibiotic therapy. Clin Infect Dis 2005; 41: 10-11. [ Links ]
23. Pitten F-A, Panzig B, Schroder G, Tietze K, Kramer A. Transmission of a multiresistant Pseudomonas aeruginosa strain at a German university hospital. J Hosp Infect 2001; 47: 125-130. [ Links ]
24. Richard P, Le Floch R, Chamoux C, Parrier M, Espaze E, Richet H. Pseudomonas aeruginosa outbreak in a burn unit: role of antimicrobials in the emergence of multiply resistant strains. J Infect Dis 1994; 170: 377-384. [ Links ]
25. Hsueh P-R, Teng L-J, Yang P-C, Chen Y-C, Ho S-W, Luh K-T. Persistence of a multidrug- resistant Pseudomonas aeruginosa clone in an intensive care burn unit. J Clin Microbiol 1998; 36: 1347-1351. [ Links ]
 Correspondence:
Correspondence:
O Perovic
(olga.perovic@nhls.ac.za)
Accepted 14 August 2007.














