Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 n.7 Pretoria Jul. 2008
SCIENTIFIC LETTERS
The mechanism of bleomycin in inducing haemangioma regression
P MabetaI; P F DavisII
IMSc; Department of Physiology, Faculty of Health Sciences, University of Pretoria
IIPhD; Department of Medicine, Wellington School of Medicine, New Zealand
To the Editor: Haemangiomas are neoplasms of the vasculature frequently encountered in paediatrics which, although benign, may present with serious complications.1-3 The potential beneficial effects of intralesional bleomycin injection (IBI) in the treatment of haemangiomas were initially reported by Kullendorf4 and Sarihan et al.5 More recently, the effectiveness of IBI was evaluated in 37 patients with haemangiomas in a study conducted by the Pretoria Vascular Malformation Study Group. Complete resolution or significant improvement was seen in 87% of the patients.6
In another study undertaken at the Cape Town Red Cross Children's Hospital, following the treatment of 30 haemangioma patients with IBI, a response rate of 75 - 100% was attained in 73% of the patients; a response rate of 50 - 75% was reported for the rest of the patients.7
Despite the promising results observed following IBI treatment of haemangiomas, bleomycin's mechanism of action in haemangiomas remains unknown. As an initial study into how bleomycin may cause haemangioma regression, we investigated the drug's effect on human haemangioma biopsies.
Materials and methods
Fresh operative human haemangioma tissue biopsies were cultured in fibrin gel and MCDB 131 medium and exposed to bleomycin (0 - 100 µg/ml). Biopsies were monitored daily using an inverted light microscope fitted with a Pixera digital image camera. For quantitative analysis, the Image 1.62b7 software program was used. Ethical clearance for the use of patient samples was granted by the Wellington Review Board, New Zealand.
Results
Cultured haemangioma tissue biopsies gave rise to an array of microvessels (Fig. 1, A and B). However, fewer neovessels were observed in tissue fragments treated with varying doses of bleomycin (Fig. 1, B).2 Quantitative analysis showed that bleomycin inhibited neovessel growth in a dose-dependent and time-dependent manner.
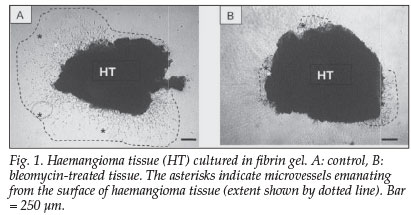
Discussion
The treatment of infantile haemangiomas has remained unsatisfactory. More recently, in South Africa and parts of Asia, intralesional bleomycin has been employed to treat these tumours, with very good results. However, concerns remain about the use of chemotherapy to treat benign tumours and the possible development of bleomycin-induced pulmonary fibrosis in such patients. Therefore, elucidation of bleomycin's mechanism of action in haemangiomas is of clinical relevance.
In this study, neovascularisation was observed in all tumour biopsies. Previously, using this model, endothelial cell markers Von Willebrand factor (vWF) and CD31 were localised to the neovessels, confirming that the outgrowths were indeed blood vessels.2
Furthermore, comparison of the number, localisation and phenotype of endothelial and mast cells and the distribution of basement membrane constituents (type IV collagen and laminins) in the biopsy tissue before and after culture showed that many of the characteristics of the original haemangioma tissue were retained in culture.2
Excessive angiogenesis is considered a central event underlying haemangioma development.3 In addition, pro-angiogenic growth factors VEGF and bFGF were previously detected in proliferating lesions.2 The development of neovessels observed in cultured haemangiomas in the present study may therefore be reminiscent of angiogenesis occurring in paediatric tumours.
Interestingly, fewer neovessels were observed in tissue fragments treated with bleomycin than in untreated fragments. Studies conducted in our laboratory have also shown that bleomycin possesses anti-angiogenic activity (manuscript in preparation).
Our findings therefore indicate that bleomycin inhibits haemangioma growth by inhibiting angiogenesis. The evidence that bleomycin inhibits haemangioma growth by inhibiting neovascularisation offers an opportunity for the use of therapeutic strategies based on the inhibition of angiogenesis.
We thank Dr S T Tan, Hutt Hospital, Wellington, New Zealand, for providing patient biopsies. This study was funded by the National Research Foundation and Bristol-Myers Squibb.
References
1. Tan ST, Wallis RA, He Y, Davis PF. Mast cells and hemangioma. Plast Reconstr Surg 2004; 113: 999-1011. [ Links ]
2. Tan ST, Hasan Q, Velickovic M, et al. A novel in vitro human model of haemangioma. Mod Pathol 2000; 13: 92-99. [ Links ]
3. Sterescu AE, Rousseau-Harsany E, Farrell C, et al. The potential efficacy of ω-3 fatty acids as anti-angiogenic agents in benign vascular tumors of infancy. Med Hypoth 2005; 66: 1121-1124. [ Links ]
4. Kullendorf CM. Efficacy of bleomycin treatment for symptomatic hemangiomas in children Pediatr Surg Int 1997; 12: 526-528. [ Links ]
5. Sarihan H, Mocan H, Yildiz K, Abes M, Akyazici R. A new treatment with bleomycin for complicated cutaneous hemangiomas in children. Eur J Pediatr Surg 1997; 7: 158-162. [ Links ]
6. Muir T, Kirsten M, Fourie P, et al. Intralesional bleomycin injection (IBI) treatment for haemangiomas and congenital vascular malformations. Pediatr Surg Int 2004; 19: 766-773. [ Links ]
7. Pienaar C, Graham R, Geldenhuys S, Hudson DA. Intralesional bleomycin for the treatment of hemangiomas. Plast Reconstr Surg 2006; 117: 221-226. [ Links ]
 Correspondence:
Correspondence:
P Mabeta
(peace.mabeta@up.ac.za)
Accepted 12 February 2008














