Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 n.5 Pretoria May. 2008
SCIENTIFIC LETTERS
Coronary spasm and thrombosis in a bodybuilder using a nutritional supplement containing synephrine, octopamine, tyramine and caffeine
J P SmedemaI; G J MüllerII
IMD, MMed (Int), FCP (SA), FCCP, FESC; Lazaron Heart Clinic, Netcare N1 City Hospital, Goodwood; Netcare Blaauwberg Hospital, Sunningdale; Department of Medicine, University of Cape Town
IIMB ChB, BSc Hons, MMed (Anesth), PhD; Department of Pharmacology, Stellenbosch University, Parow, W Cape
To the Editor: A young, previously healthy bodybuilder suffered an acute myocardial infarction in the absence of known cardiovascular risk factors or demonstrable atherosclerotic plaque. The infarction probably resulted from coronary spasm, platelet activation and in situ thrombosis triggered by the chronic consumption of a 'nutritional supplement' which contains synephrine, octopamine, tyramine and caffeine.
The 39-year-old bodybuilder presented after developing new-onset angina pectoris with vegetative symptoms during a bodybuilding competition. He had no previous medical history or cardiovascular risk factors, and denied ever using androgenic anabolic steroids. He had been involved in competitive bodybuilding for 7 years. He had been taking for several years a 'nutritional supplement' that contains synephrine (oxedrine), octopamine, tyramine (sympathomimetic amines) and caffeine. The preparation also contains several 'nutrients', of which the herb St John's wort has significant pharmacological actions on the nervous system.1 In the 3 months preceding the competition concerned, he had taken a daily dose equivalent to 40 mg synephrine, 400 mg caffeine, and an unspecified amount of tyramine and octopamine. Synephrine is more potent than the other sympathomimetic amines. Its action is similar to that of phenylephrine, an alpha-adrenergic agonist,2 and it has been used for treating hypotension in doses of about 100 mg 3 times a day. Octopamine has about one-hundredth the potency of noradrenaline.
The patient had restricted his fluid intake and increased his carbohydrate intake during the 36 hours before the competition. Physical examination was normal. The electrocardiogram showed a wide right bundle-branch block that resolved after several hours, and 1 mm ST-segment elevation in leads II, III, aVF, and V4-6. There was evidence of renal impairment, with creatinine at 171 µmol/l and urea at 11 mmol/l, and creatine kinase (CK) levels of 8 500 IU/l. The troponin T level was normal on admission, but rose to 0.53 ng/ml after 6 hours, and 1.9 ng/ml after 24 hours. Echocardiography demonstrated a dyskinetic basal interventricular septum, with mild biventricular hypertrophy. Oral aspirin, clopidogrel and bisoprolol and intravenous nitroglycerine, enoxiparine and eptifibatide were initiated, and he was hydrated with intravenous 0.9% saline. Coronary angiography showed a thrombus in the proximal left anterior descending artery, with diffuse spasm in the mid- and distal segments (Fig. 1), which resolved after the administration of intracoronary nitroglycerine. A 4.5 mm bare metal stent covering the lesion was successfully positioned. His low-density lipoprotein, high-density lipoprotein and triglyceride levels were respectively 2.1, 0.31 and 1 mmol/l, the fasting homocysteine level was normal, and anti-phospholipid antibodies were absent. He was discharged fully recovered and remains symptom-free 6 months later.
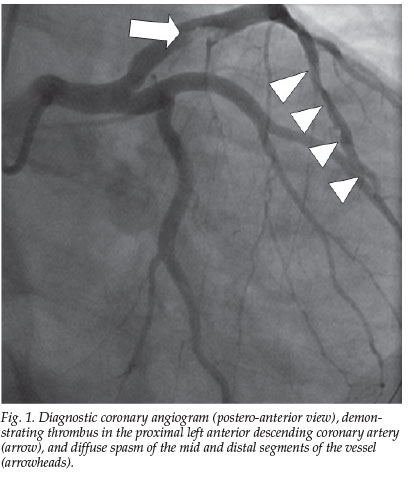
Discussion
The acute myocardial infarction in this young, previously healthy bodybuilder in the absence of known cardiovascular risk factors or demonstrable atherosclerotic plaque was probably caused by coronary spasm, platelet activation and in situ thrombosis.
The USA's Food and Drug Administration (FDA) banned the sale of products containing ephedrine alkaloids in 2004 because of associated cardiovascular toxicity.3 Synephrine and octopamine are structurally similar to norepinephrine, and have been associated with acute myocardial infarction and ischaemic colitis. 4-7 Both are trace endogenous bioamines, agonists of the α1, α2, β1 and β3 adrenoreceptors, are found in human plasma, platelets, sympathic nerves and adrenal tissue, and are present in Citrus aurantium (Seville orange, bitter orange), an ingredient in dietary supplements marketed for weight loss.8-10 In animal studies, synephrine increased cardiac output and caused vasoconstriction and ventricular arrhythmias.11,12 Bitter orange has been identified as a cause of resistant hypertension, syncope, myocardial infarction, tachycardia and ventricular fibrillation, and exacerbated coronary spasm in tobacco smokers.5 Prolonged administration, or the combined consumption, of synephrine with octopamine and caffeine may result in haemodynamic effects.13-16 A single dose of bitter orange extract containing the equivalent of 50 mg synephrine significantly increased the systolic and diastolic blood pressures, as well as the heart rate, of healthy young adults for up to 5 hours.17
The risk of adverse cardiovascular events may be higher in persons with pre-existing underlying cardiovascular disease. Caffeine enhances the cardiovascular and central nervous system effects of adrenergic amines though augmentation of catecholamine release.18-20 The enhanced sympathetic activity increases platelet reactivity. Although an underlying atheromatous plaque has not been excluded with absolute certainty in our patient, it seems very likely that use of a nutritional supplement containing synephrine, octopamine, tyramine and caffeine, combined with intravascular dehydration and impaired renal function, triggered coronary spasm and thrombosis of a major proximal coronary artery. Constituents of St John's wort are inhibitors of serotonin, noradrenaline and dopamine uptake in the synaptic cleft,1 which may potentiate the effects of alpha-adrenergic stimulants and thereby enhance their vasoconstrictor activities.
The safety of over-the-counter supplements containing synephrine has been called into question.4-8 Consumers consider dietary supplements to be safe, but these are currently not subjected to scientific scrutiny, and some contain potentially harmful ingredients. The use of supplements containing the combination of synephrine, octopamine, tyramine and caffeine may constitute a risk of cardiovascular toxicity. There is a need for centralised monitoring of clinical adverse events in consumers using nutritional supplements.
References
1. do Rego JC, Benkiki N, Chosson E, et al. Antidepressant-like effect of hyperfoliatin, a polyisoprenylated phloroglucinol derivative from Hypericum perfoliatum (Clusiaceae) is associated with an inhibition of neuronal monoamines uptake. Eur J Pharmacol 2007; 569: 197-203. [ Links ]
2. Bowman WC, Rand MJ. Textbook of Pharmacology. 2nd ed. Oxford: Blackwell ScientificvPublications, 1980: 11.4 and 11.30. [ Links ]
3. Food and Drug Administration, Health and Human Services. Final rule declaring dietary supplements containing ephedrine alkaloids adulterated because they present an unreasonable risk. Fed Regist 2004; 69: 6787-6854. [ Links ]
4. Nykamp DL, Fackih MN, Compton AL. Possible association of acute lateral-wall myocardial infarction and bitter orange supplement. Ann Pharmacother 2004; 38: 812-816. [ Links ]
5. Jordan S, Murty M, Pilon K. Products containing bitter orange or synephrine: suspected cardiovascular adverse reactions. Can Med Assoc J 2004; 171: 993-994. [ Links ]
6. Gange CA, Madias C, Felix-Getzik EM, et al. Variant angina associated with bitter orange in a dietary supplement. Mayo Clin Proc 2006; 81: 545-548. [ Links ]
7. Sultan S, Spector J, Michell RM. Ischemic colitis associated with use of a bitter orange-containing dietary weight-loss supplement. Mayo Clin Proc 2006; 81: 1630-1631. [ Links ]
8. Fugh-Berman A, Myers A. Mini-review: Citrus aurantium, an ingredient of dietary supplements marketed for weight loss: current status of clinical and basic research. Exp BiolvMed 2004; 229: 698-704. [ Links ]
9. Brown CM, McGrath JC, Midgley JM, et al. Activities of octopamine and synephrine stereoisomer on α-adrenoreceptors. Br J Pharmacol 1988; 93: 417-429. [ Links ]
10. Jordan R, Midgley JM, Thonoor CM, Williams CM. Beta-adrenergic activities of octopamine and synephrine stereoisomer on guinea-pig atria and trachea. Br J Pharmacol 1987; 39: 752-754. [ Links ]
11. Zhao XW, Li JX, Zhu ZR, et al. Anti-shock effects of synthetic effective compositions of Fructus aurantii immaturus: experimental study and clinical observations. Chin Med J 1989; 102: 91-93. [ Links ]
12. Huang YT, Lin HC, Chang YY, et al. Hemodynamic effects of synephrine treatment in portal hypertensive rats. Jpn J Pharmacol 2001; 85: 183-188. [ Links ]
13. Min B, Ciois D, Kluger J, White CM. Absense of QTc-interval-prolonging or hemodynamic effects of a single dose of bitter-orange extract in healthy subjects. Pharmacotherapy 2005; 25: 1719-1724. [ Links ]
14. Haller CA, Benowitz NL, Jacob P III. Hemodynamics of ephedra free weight-loss supplements in humans. Am J Med 2005; 18: 998-1003. [ Links ]
15. Haller CA, Jacob P III, Benowitz NL. Enhanced stimulant and metabolic effects of combined ephedrine and caffeine. Clin Pharmacol Ther 2004; 75: 259-273. [ Links ]
16. Brown NJ, Ryder D, Branch RA. A pharmacodynamic interaction between caffeine and phenylpropanolamine. Clin Pharmacol Ther 1991; 50: 363-371. [ Links ]
17. Bui LT, Nquyen DT, Ambrose PJ. Blood pressure and heart rate effects following a single dose of bitter orange. Ann Pharmacother 2006; 40: 53-57. [ Links ]
18. Benowitz NL. Clinical pharmacology of caffeine. Ann Rev Med 1990; 41: 277-288. [ Links ]
19. Robertson D, Frolich JC, Carr RK, et al. Effects of caffeine on plasma renine activity, catecholamines and blood pressures. N Engl J Med 1978; 298: 181-186. [ Links ]
20. Sachdeva R, Sivasankaran S, Fishman RF, et al. Coronary thrombosis related to use of Xenadrine® RFA. Tex Heart Inst J 2005; 32: 74-77. [ Links ]
 Correspondence:
Correspondence:
J Smedema
(jansmedema@hotmail.com)
Accepted 20 November 2007.














