Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.98 no.2 Pretoria Fev. 2008
ORIGINAL ARTICLES
Pharmacologically active: Clinical trials and the pharmaceutical industry
Michael KahnI; Michael GastrowII
IBSc Hons, PhD, MA; Centre for Science, Technology and Innovation Indicators (CeSTII), Human Sciences Research Council, Cape Town
IIBA Hons, MA; Centre for Science, Technology and Innovation Indicators (CeSTII), Human Sciences Research Council, Cape Town
ABSTRACT
Multinational pharmaceutical companies ('pharmas') import and produce pharmaceuticals and also conduct clinical trials which are an important aspect of research and development (R&D). This may raise the question: Is South Africa a guinea pig for the pharmas? The Department of Trade and Industry National Industrial Policy Framework1 designates chemicals, plastic fabrication and pharmaceuticals as a key value chain. So a second question could be: Can South Africa be a manufacturer for the pharmas, or can it leverage strengths in medical research and the conducting of clinical trials so as to develop a discovery-led industry? This paper analyses and quantifies the state of the clinical trials industry in South Africa, and concludes that: (i) a sizeable clinical trials industry exists, and that these trials are predominantly phase 3 and global in scope; (ii) South Africa is not a specific or unique guinea pig - a range of conditions is studied as part of global trials; and (iii) while South Africa has excellent prospects for increased clinical trials activity, R&D investment is too low to make it a major pharmaceutical contender.
The South African pharmaceutical industry
The South African Government's Department of Health (DOH), through the Medicines Control Council (MCC), licenses 75 companies to manufacture pharmaceuticals, 24 to carry out quality control and testing, and 251 to function as importers, wholesalers or retailers of products.2 The MCC also issues licences for the performance of clinical trials. Multinational pharmas dominate the local industry, and 7 of them have local production plants. The South African pharmaceutical industry turnover is estimated at around R14.1 billion, including imports of R7.44 billion and exports3 of approximately R0.77 billion.
Measuring pharmaceutical R&D in South Africa
There is no reliable published value of pharmaceutical industry expenditure on clinical trials. The Pharmaceutical Manufacturers Association, on the basis of a survey of its members, estimated that the 2004 spend was in the order of R650 million (personal communication with Maureen Kirkman, Head: Scientific and Regulatory Affairs, Pharmaceutical Industry Association of South Africa), but it is unclear whether this included phase IV trials as well. Their current estimate is in the order of R900 million.
The Organisation for Economic Co-operation and Development (OECD) survey guidelines for R&D state that: 'Clinical trial phases I, II and III can be treated as R&D. Phase IV clinical trials, which continue testing the drug or treatment after approval and manufacture, should only be treated as R&D if they bring about a further scientific or technological advance'.4
To determine the scale of current pharmaceutical R&D and clinical trials, three sources of data are used: the South African National Clinical Trials Register (SANCTR);2 United States National Institutes of Health website;5 and national R&D surveys that are official statistics under the Statistics Act No. 6 of 1999.
The SANCTR lists 308 active trials by condition without declaring their phase. A contact person with a telephone number is listed, but extensive inquiry yielded almost no further information save a few responses indicating phase III activities. One cannot readily distinguish industry from university or private clinical trials from this information.
The US database, on the other hand, clearly identifies 172 active industry-sponsored trials in South Africa. Another 53 trials are mainly supported by non-industry sources. Unfortunately, neither database provides enrolment per country that could be used to estimate the cost associated with each clinical trial.
The annual National R&D Survey continually updates its register of pharmaceutical R&D performers by direct enquiry, studying the media, and annual reports of listed companies.
Findings
The 2005/2006 R&D Survey (Table I) shows excellent overlap with the US company data and recorded 28 firms (22 'manufacturers' and 6 'contract research organisations' or CROs) with expenditure on R&D (including clinical trials) of some R822 million.
While the 2005/2006 level of pharmaceutical-related R&D represents a 3% real growth from 2004/2005, it represents a slight decline from 11.8% down to 10.2% in relation to total business expenditure on R&D.
Capital expenditure related to R&D is a good indicator of laboratory investment and experimentation. Of the 'manufacturers', only two showed any capital expenditure on R&D, amounting to R119 million, or 14.3%, of the 2005/2006 total. On the other hand, the median R&D-related capital expenditure for the 28 firms is about R1.5 million - an extremely low amount, from which it may be concluded that, by and large, the firms are engaged only in routine pharmaceutical manufacturing, and in the management and conducting of clinical trials.
The firms report R0.3 million of government funding, and R402 million in internal and domestic business sector funding, compared with an inward flow of foreign funds to themselves of R420 million (Table II). These figures highlight that government plays little role in funding business sector pharmaceutical R&D, although the National Industrial Policy Framework may change this.
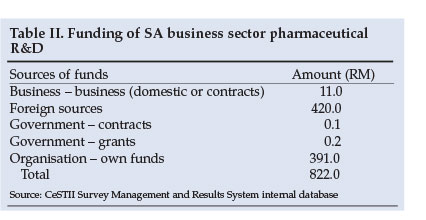
The ratio of the cost of labour to current expenditure for firms in the pharmaceutical sector shows a marked deviation from the 1:1 norm for the business sector. The former ratio, at 2:1, suggests significant unspecified labour costs. This reflects one of the main challenges in measuring clinical trials R&D: underestimation of the headcount of researchers who are operating within extended clinical trials R&D value chains. Companies often do not have a direct way of reporting these headcount data. Accordingly, the 491 full-time equivalent researchers recorded in the pharmaceutical sector is a lower bound and might be less than one half of the researcher workforce involved in clinical trials outside the public sector. This figure should be seen in the context of the 3 700 specialists and 12 000 GPs in the country.
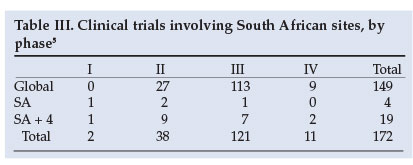
Most trials in which South Africa is one of many participating countries, are global (149), where 'global' is defined as encompassing more than five countries (Table III), as opposed to multinational trials which include South Africa and up to four other countries (19) or those where South Africa is a unique country, which occurs only 4 times. Of the 172 clinical trials, 40 concentrate on cardiovascular conditions, 23 on cancers and 21 on diabetes. The remaining 88 trials cover another 20 conditions. Of the 4 trials unique to South Africa, 3 are on HIV/AIDS; of the 19 multinational trials involving South Africa, 4 more are on HIV/AIDS. There is no particular clustering by condition among the remaining 15 trials in the country, and consequently no evidence to support claims that South Africa is a unique guinea pig in such trialling.
Implications for policy
Pharmaceutical clinical trials in South Africa involve an R&D expenditure of R822 million. This forms part of an overall health-related R&D expenditure of R2.088 billion. The bulk of R&D in the pharmaceutical industry involves foreign-led global phase III clinical trials. Only two phase I clinical trials are being undertaken and these are listed as foreign-supported. The www.clinicaltrials.gov register does not reveal any South African company as the sponsor of a clinical trial.
The R&D survey shows very low R&D-related capital expenditure by business, which is not sufficient for the widespread invention and production of active pharmaceutical ingredients. The R119 million capital expenditure for R&D invested by the industry in 2005/6 originated mostly in one company, and is far too small to drive drug discovery. The focus of pharmaceutical R&D in South Africa is thus in the area of clinical trials, and not discovery.
The health sciences total fulltime researcher availability, including doctoral and post-doctoral students, was estimated at approximately 3 700 (unpublished data extract for the Department of Science and Technology by Centre for Science, Technology and Innovation Indicators). This resource, spread over all medical research fields, should be seen against the resources of a pharma such as AstraZeneca, which employs 12 000 researchers worldwide.6 South Africa neither has the requisite human resources to be internationally competitive in pharmaceutical R&D, nor does it invest nearly enough. This is in line with findings of the National Economic Development and Labour Council,7 which suggest that R&D growth in the South African pharmaceutical industry is more likely to be in clinical trials than in basic R&D.
However, good prospects exist for increased clinical trials activity. There are factors that may attract this, such as well-established credentials in medical research, high-quality personnel, relatively good infrastructure,8 access to a larger African market, a relatively drug-naïve population, and a high burden of disease.
Policies should therefore be directed towards this end, and could include application of new R&D tax incentives, co-financing, access to infrastructure, a proactive regulatory environment, and better application of immigration regulations as a means toward 'brain gain' of highly skilled personnel.
This work is supported by funding provided to the Centre for Science, Technology and Innovation Indicators (CeSTII), by the Department of Science and Technology and the Human Sciences Research Council. The authors acknowledge the assistance of Natalie Vlotman and Maalikah van der Schyff in data gathering and analysis.
References
1. A National Industrial Policy Framework. Pretoria: Department of Trade and Industry, 2007. [ Links ]
2. South African National Clinical Trials Register. Pretoria: Department of Health, 2005. http://www.sanctr.gov.za/SAClinicalbrnbspTrials/tabid/169/Default.aspx (accessed 17 October 2007). [ Links ]
3. The Power of Hope: How Adcock Ingram Adds Value to Life. http://www.adcock.co.za/Resources/June%202007_Roadshow.PDF (accessed 17 October 2007). [ Links ]
4. Proposed Standard Practice for Surveys on Research and Experimental Development Frascati Manual 2002. Paris: Organization for Economic Cooperation and Development, 2002. [ Links ]
5. US Department of Health and Human Services: National Institutes of Health, Clinical Trials._ www.clinicaltrials.gov (accessed 17 October 2007). [ Links ]
6. AstraZeneca's R&D capabilities. http://www.astrazeneca.com/article/500880.aspx (accessed 17 October 2007). [ Links ]
7. FRIDGE Study: Pharmaceutical Manufacturing Sector Study. Johannesburg: National Economic Development and Labour Council, July 2000: 125. [ Links ]
8. Akkermann B. South Africa's untapped clinical potential. Contract Services Europe 1 June 2006. [ Links ]
 Correspondence:
Correspondence:
M Kahn
(mkahn@hsrc.ac.za)
Accepted 12 October 2007.














