Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Koedoe
versión On-line ISSN 2071-0771
versión impresa ISSN 0075-6458
Koedoe vol.57 no.1 Pretoria 2015
http://dx.doi.org/10.4102/KOEDOE.V57I1.1237
CHECKLIST
A checklist of the plants of the forests and grasslands in the Weza district, southern KwaZulu-Natal and a review of their status in the Red Data List
Graham R.H. Grieve; Colleen T. Downs
School of Life Sciences, University of KwaZulu-Natal, South Africa
ABSTRACT
Eastern mistbelt forests are naturally fragmented forests with grassland which occur from the Eastern Cape to KwaZulu-Natal, South Africa. These were heavily logged by colonial settlers and continue to be harvested despite being protected. Consequently we documented a checklist of the plants of the forests and grasslands in the Weza district (3029DA WEZA), southern KwaZulu-Natal, including Ngeli Forest and nearby indigenous forest patches to highlight their biodiversity status and need for conservation. We also reviewed their status in the Red Data List. Of the 1554 records included in this summary of plant species for the Weza district, there were 6 lichens (0.4%), 46 bryophytes (3.0%), 58 pteridophytes (3.7%), 6 gymnosperms (0.4%) and the remaining 1424 species angiosperms (92.5%). Of the angiosperms, 27.3% were monocotyledons and 72.7% were dicotyledons. The most species-rich family was Asteraceae (239 species) followed by Fabaceae (115 species), Liliaceae (used for purposes of comparison against older studies - 89 species), Orchidaceae (89 species), Iridaceae (59 species), Poaceae (58 species), Asclepidaceae (again used for purposes of comparison against older studies - 57 species), Scrophulariaceae (42 species), Euphorbiaceae (32 species), Lamiaceae (32 species) and Rubiaceae (27 species). These 10 families each comprised more than 2% of the species in the list. Together they contributed 55% of the angiosperm species and 34.1% of the angiosperm genera. The biodiversity and conservation value of the study area are conserved pockets of eastern mistbelt forest, Drakensberg foothill moist grassland and mistbelt grassland. More than 4% of the species are under some degree of threat, as was evidenced by the number of species regarded as endangered (5), vulnerable (18), near threatened (10), critically rare (1), rare (20) or declining (11) amongst the 1554 species covered in the list.
Conservation implications: In terms of taxa under some degree of threat, number of endemic and near endemic species the biodiversity and conservation value of the Ngele (3029DA WEZA) area is reinforced. This necessitates that the area is appropriately protected from invasive alien species, and uncontrolled and illegal use by the neighbouring communities.
Introduction
Globally Africa currently has one of the highest levels of deforestation, which affects both habitat quality and species diversity (Bodart et al. 2013; FAO 2010; Lawes, Macfarlane & Eeley 2004; Wade et al. 2003). In South Africa the forest biome is the smallest of the eight biomes (Mucina & Geldenhuys 2006). Eastern mistbelt forests, which form part of the southern mistbelt forest group, are naturally fragmented forests with grassland which occur from the Eastern Cape to KwaZulu-Natal (KZN) (Mucina & Geldenhuys 2006). These forests are dominated by Afrocarpus/Podocarpus species (yellowwoods) (Adie, Rushworth & Lawes 2013; Moll 1972). Drakensberg foothill moist grassland and Midlands mistbelt grassland comprise the largest part of the untransformed grassland habitat in the study area and are home to a wide variety of plants (Mucina & Geldenhuys 2006).
The first forest legislation in KZN was passed in 1853, a forest service was started in 1891, and a functional forestry department in 1902 (Fourcade 1889; King 1941). However, most of the forests had already been exploited by European settlers, including colonial woodcutters (Cawe & McKenzie 1989; Cooper 1985; King1941). Fourcade (circa 1889) was seconded to the Natal Government to prepare a report on the condition of indigenous forests on Crown and Trust lands (Fourcade 1889). His report subsequently led to the decision in 1891 to protect the Crown Forests (Fourcade 1889; Storrar 1990).
Remnants of sawpits can still be seen in Ngeli Forest and others, and although the forests appear to have recovered substantially since protection was afforded (pers. obs.), regeneration is slow and the canopies are generally irregular and areas reduced (Geldenhuys 2007; Lawes et al. 2004; Moll 1972). Following the colonial logging exploitation in the mid-1800s and intermittently to the 1940s, these forests in KZN have been allowed to recover by natural succession processes (Adie et al. 2013). However, as most of these forests are in close proximity to communal lands, subsistence wood harvesting and use of the forest and grasslands to graze cattle has continued (Adie et al. 2013; Lawes, Griffiths & Boudreau 2007; Robertson & Lawes 2005; Wirminghaus et al.1999; pers. obs.). Despite being protected, generally conservation of these forests and their associated grasslands are difficult (Adie et al. 2013; Cawe & McKenzie 1989; Downs 2005; Wirminghaus et al. 1999).
It is clearly difficult to determine whether the present plant species diversity in Ngeli Forest and nearby forest patches and associated grasslands is similar to what existed before the exploitation that began in the mid to late 19th century, although some of the specimens collected and included in this species list date from that period. Therefore we documented a checklist of the plants of the forests and grasslands in the Weza district (3029DA WEZA), southern KZN, including Ngeli Forest and nearby indigenous forest patches to highlight the biodiversity importance of this area and need for conservation. The list could serve as an important conservation management tool in future. We also reviewed the plants' status in the Red Data List.
Methods
Study area
The study area was the 3029DA quarter-degree square described as 3029DA WEZA (Figure 1; Table 1). This includes the village of Weza in the southern interior of KZN, South Africa, close to the border with the Eastern Cape, situated between Harding and Kokstad. It is a centre for timber growing and has a sawmill to process the pine Pinus sp. logs extracted from the plantations in the surrounding hills. The Weza State Forests, which include pine plantations as well as indigenous forest patches and grassland (firebreaks and the tops of the mountain range), lie to the south-east of the Ngeli mountain range. The range runs from a peak at 30°34.3's, 29°35.5'E on the Eastern Cape border, past the dominant peak of Ngeli mountain at 2267.7 m and slowly reduces in height towards the north-east. There is a high point to the north of the N2 highway on which fire lookout towers are situated at 30°31.2's, 29°41.9'E. The study area lies generally to the south-east of the axis joining these extremes.
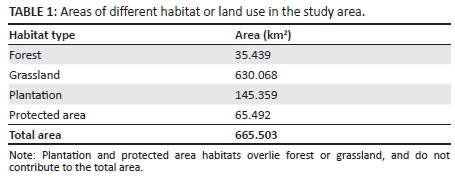
The Ngeli Forest and nearby forest patches and associated grasslands lie between an altitude of 950 m adjacent to Weza and the altitude of the higher peaks at about 2200 m. The Ngeli mountain range is regarded as an outlier of the Drakensberg Alpine Centre (Carbutt & Edwards 2006). One of the larger forest patches straddles the N2 highway roughly midway between Harding and Kokstad, but the bulk of the study area is to the south-west of the N2 highway and south-east of the Ngeli range. The Umtamvuna River rises in the Ngeli range and flows in a south-easterly direction to reach the sea just south of Port Edward. Apart from a short section in the Ngeli range, the Umtamvuna forms the border between the Eastern Cape and KZN. The forests in this area fall under the FOz 3 southern mistbelt forest category, whilst the grasslands are categorised as Gs 9 Midlands mistbelt grassland and Gs 10 Drakensberg foothill moist grassland (Mucina & Rutherford 2006).
The average rainfall in this area is 991 mm/a, although there appears to have been periods of below-average (664 mm/a) and above-average (1476 mm/a) rainfall during the period covered by the data (Whyle, unpublished data). If the rainfall is considered as the precipitation during the rainy season from the beginning of July until the end of June in the following year, there are similar peaks and troughs but there are also much greater swings in total rainfall, from a low of 580 mm to a high of 1624 mm (Whyle, unpublished data).
The main parts of the exposed rocks of the Ngeli range are composed of dolerite rocks and associated soil types (Wirminghaus et al. 2001). Norman (2012) notes that the dolerite occurs in sills and dykes, and these are mixed with un-deformed sedimentary strata from the Lower Karoo age (Permian). The sedimentary horizons and sills are almost horizontal.
Data sources
The plant species list for the study area is based on data mainly from voucher specimens collected by a wide range of botanists, although there are several species, mostly naturalised invasive alien species, for which only 'observation' data are currently available. In addition, voucher specimens were collected from 2009 to 2014 whilst conducting monthly research in the area (Hart, Grieve & Downs 2013) and specimens were identified with the aid of Pooley (1998, 2003) and Boon (2010).
The voucher specimens included in this species list are housed at a number of herbaria around the country, namely KBG, NBG, NH, NU, PCE and PRE. An attempt was made to obtain an extract of data from the Schweikert Herbarium at Pretoria University (as many records of A. Abbott and A.E. van Wyk are known to be housed in the Schweikert Herbarium), but they advised they were in the process of migrating their database records from PRECIS to BRAHMS and were unable to assist at the time the request was made. Although the collecting of species on which this study is based took place over an extended period (1933-2014), there may still be species which have been overlooked.
The initial stimulus for this work was provided by T. Abbott, who made a plant data list of Ngeli species available (Abbott 2010). The South African National Biodiversity Institute (SANBI) was asked to make similar lists available for the collections at PRE and NH, and use was made of the SIBIS (SANBI's Integrated Biodiversity Information System) website (http://sibis.sanbi.org/) for further data. Information was received in discrete subsets which had to be consolidated and duplication removed. Where necessary, additional information on collector ID, voucher number and location of voucher were also derived from the SIBIS site.
Species list arrangement
The arrangement of the species list and species nomenclature follows the Red List of South African plants(Raimondo et al. 2009). This was used as it was felt important to determine the Red List status of as many of the species in the 3029DA quarter-degree square as possible to facilitate and reinforce conservation measures for the area. However, this publication only covers vascular plants. In the case of the bryophytes, use was made of 'A checklist of South African plants' (Germishuizen et al. 2006), and for the few lichens amongst the data, Google searches had to suffice; in neither of these cases was there any information on conservation status of these species.
Use was made of the lists of endemic and near endemic species for the Drakensberg Alpine Centre (Carbutt & Edwards 2006) to show which species in the Ngeli area are endemic and near endemic. This information was included, where applicable, for the angiosperms in the species list (Online Appendix 1). Naturalised exotic species (weeds) were included and appropriately indicated.
The web-based Red Data List (RDL) (Raimondo et al. 2009) was used to determine the status of each of the vascular plant species recorded for the study area. Percentages of each RDL category falling within the order breakdown were determined.
Results and discussion
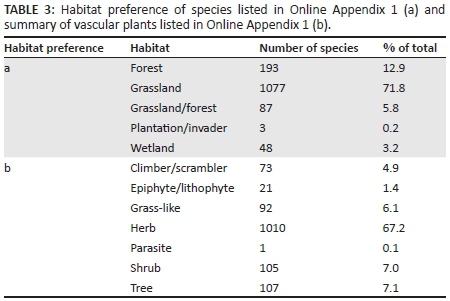
Of the 1554 records included in this summary of plant species for the Weza district, there were 6 lichens (0.4%), 46 bryophytes (3.0%), 58 pteridophytes (3.7%), 6 gymnosperms (0.4%), and 1438 angiosperms (92.5%) (Table 2). The number of plant species was highest in the grasslands, and most of these were herbaceous species (Table 3). Of the angiosperms, 27.3% were monocotyledons and 72.7% were dicotyledons. The ratio of monocotyledons to dicotyledons for Ngeli was 1:2.7. This was higher than reported for Mahwaqa Mountain (1:2.1) (Meter et al. 2002).
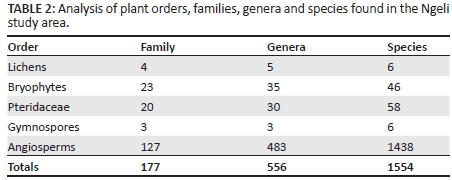
Recently, the flora of the Great Winterberg-Amatholes area (7382 km2 in extent) was described (Clark et al.2014). Whilst this region has similar numbers of families for the pteridophytes, gymnosperms, monocotyledons and dicotyledons, the numbers of genera and species are higher, possibly because of the larger area studied compared with the current study area. The number of endemics recorded in this study (43) exceeds the number of endemics in the Clark et al. (2014) study (35) in a much larger area.
Only plant families with more than 1% of the total number of species for the Ngeli quarter-degree square were included (Table 4). To make this comparable with a similar list (Meter et al. 2002), the family structure of Asclepidaceae before consolidation into Apocynaceae was used (Table 4). The 24 families contributed 71.0% of the species and 57.2% of the genera of this list.
As is the case with Mahwaqa Mountain, KZN (Meter et al. 2002), the most species-rich family for 3029DA WEZA was Asteraceae, with 239 species (Table 4, Online Appendix 1). There were 14 endemics (5.9%) and 67 near endemics (28.0%) amongst the listed Asteraceae. Amongst the Asteraceae, the following species fell into 'endangered' categories in the RDL: Berkheya pannosa (rare, endemic); Felicia wrightii (critically rare, endemic);Gnaphalium griquense (rare, near endemic); Helichrysum tenax var. palidum (rare, near endemic);Phymaspermum villosum (rare); and Senecio poseidonis (threatened, near endemic) (Table 5). Asteraceae also included 11 (4.6%) naturalised species.
The second most species-rich family for 3029DA WEZA was Fabaceae, with 115 species (Table 4, Online Appendix 1), in contrast to Poaceae in Meter et al. (2002) (Table 6). However, this was consistent with the ranking of Fabaceae as the second most important species contributor to the KZN flora (Hilliard & Burtt 1987). There was one endemic (0.9%) and 11 near endemic species (9.6%) amongst the listed Fabaceae. Amongst the Fabaceae, the following species fell into 'endangered' categories in the RDL (Table 5): Crotalaria dura subsp.dura (near threatened) and Psoralea abbottii (vulnerable).
Using the old family structure Liliaceae for comparative purposes, this family is the third most species-rich family, with 109 species (Table 4). There were 5 endemic (4.6%) and 14 near endemic species (12.8%) amongst the listed Liliaceae. Amongst the Liliaceae, the following species fell into 'endangered' categories in the RDL (Table 5): Aloe kniphofioides (vulnerable), Aloe linearifolia (near threatened), Sandersonia aurantiaca(declining), Bowiea volubilis (vulnerable), Eucomis bicolor (near threatened), Eucomis comosa (declining) andMerwilla plumbea (near threatened).
The Orchidaceae was the fourth most species-rich family in 3029DA WEZA, with 89 species (Table 4, Online Appendix 1). There were three endemics and 25 near endemics amongst the listed Orchidaceae. Four orchid species collected were listed as rare. These were: Disa oreophila subsp. erecta (rare, endemic); Disa tysonii(rare, near endemic); Satyrium microrhynchum (rare, near endemic) and Schizochilus flexuosus (rare, near endemic).
The Iridaceae was the fifth most species-rich family in 3029DA WEZA, with 59 species (Table 4, Online Appendix 1) Three of these were endemic (5.1%) and 21 were near endemic (35.6%) (Table 5). One of these species was listed as rare, one as vulnerable, one as declining and one as endangered; they were: Dierama ambiguum(endangered, near endemic) (Table 5); Dierama tysonii (vulnerable, near endemic); Gladiolus oppositiflorussubsp. salmoneus (declining, near endemic) and Hesperantha ingeliensis (rare, near endemic).
The Poaceae ranked sixth, with 58 species in 3029DA WEZA (Table 4, Online Appendix 1). A comparison of the relative richness of Poaceae at Ngeli against that reported for Mahwaqa Mountain, the southern Drakensberg (Meter et al. 2002) and KZN as a whole suggest that Poaceae might have been under-collected at Ngeli (Table 6). Indeed, once a base species list was available, it proved to be relatively easy to collect previously uncollected grass species in the study area; there are undoubtedly further grass species awaiting collection there. This historical under-collecting of Poaceae could explain the difference in monocotyledon to dicotyledon ratio between the present study and that of Meter et al. (2002) (Table 6). One species was endemic (1.7%) and three were near endemic (5.2%) (Table 3). Poaceae included 5 naturalised species (Table 4, Online Appendix 1).There were no 'endangered' species in this list (Table 5, Online Appendix 1).
The Asclepiadaceae (57 species) (Table 4, Online Appendix 1), again used for purposes of comparison against older studies, was the seventh most numerous family in 3029DA WEZA. Three of these species were endemic (5.3%) and 17 near endemic (29.8%). Five species were listed as rare and three as vulnerable. These were:Asclepias xysmaloboides (rare, endemic); Aspidonepsis cognate (rare, near endemic); Brachystelma molaventi(vulnerable); Brachystelma tenellum (vulnerable); Emplectanthus gerrardii (rare); Schizoglossum elingue subsp. purpureum (rare, endemic); Schizoglossum montanum (rare, endemic); Schizoglossum singulare (vulnerable).1
The Scrophulariaceae had 42 species in 3029DA WEZA (Table 4, Online Appendix 1). Of these, 4 were endemic (9.5%) and 11 were near endemic (26.1%) (Table 3). None of these Scrophulariaceae were listed as endangered in the RDL (Table 5).
The Euphorbiaceae in 3029DA WEZA had 32 species (Table 4, Online Appendix 1). Amongst these were two near endemic species (Table 5), one near threatened species (Alchornia hirtella forma glabrata) and one vulnerable species (Euphorbia flanaganii).
The Lamiaceae had 32 species in 3029DA WEZA (Table 4, Online Appendix 1). Amongst these were four near endemic species (Table 5). None of these Lamiaceae were in 'endangered' categories in the RDL, but there were two naturalised species (Table 5).
The 10 families above all comprised more than 2% of the species in the list (Online Appendix 1). Together they contributed 53.5% of the species and 41.9% of the genera.
Comparative data for this study, for Mahwaqa Mountain, for the southern Drakensberg (Meter et al. 2002) and for KZN as a whole, are shown in Table 6. As mentioned before, the Poaceae in 3029DA WEZA appeared to be comparatively under-represented, despite additional collecting. Fabaceae, in contrast, appeared to be over-represented when compared with Mahwaqa and the southern Drakensberg (Meter et al. 2002), although as a proportion of all recorded species, their data were more in line with the data for KZN as a whole. These apparent anomalies might justify a review of the Poaceae (some work already done) and Fabaceae to determine whether these relative proportions are real or as a result of collecting bias. The latter is unlikely given the significant number of collectors involved here.
Conservation status and management
The biodiversity and conservation value of the study area lie in the conserved pockets of eastern mistbelt forest and Drakensberg foothills moist grassland and eastern mistbelt grassland. This was evidenced by the number of species regarded as endangered (5), vulnerable (18), near threatened (10), critically rare (1), rare (20) or declining (11) amongst the 1554 species covered in the list, that is, more than 4% of the species are under some degree of threat. This, together with the fact that, amongst the angiosperms, there are 44 (3.1%) endemic species (11 monocotyledons and 33 dicotyledons) and 256 (17.8%) near endemic species (79 monocotyledons and 177 dicotyledons), reinforces the biodiversity and conservation value of the 3029DA WEZA area. Furthermore, this area supports important fauna (Adie et al. 2013; C. Potgieter [University of KwaZulu-Natal], pers. comm.). This necessitates that the responsible authorities ensure that the area is appropriately protected from invasive alien species, and uncontrolled and illegal use by the neighbouring communities (Adie et al. 2013). An interesting question to be investigated is whether all the species collected in the period leading up to 1891, when protection was afforded this and other KZN forests (McCracken 2004), are still present.
Acknowledgements
W. Rozani, O. Zikhali and B. Dzidza from the Department of Agriculture, Forestry and Fisheries (DAFF) are thanked for permission to undertake the research work in the Ngeli forests. The DAFF forestry security guards S. Cwele, D. Masuku and R. Madwe are thanked for their considerable assistance in the field during data collection. T. Abbott kindly supplied a significant portion of the data on which this paper is based, and assisted with tree identifications at the outset of the project. He also kindly facilitated the first author's involvement with the Nicholson/Abbott Herbarium at the Umtumvuna Nature Reserve which led to the preparation of the most recent specimens and accession of these into that collection. B. Bytebier provided valuable guidance on the structuring of the species list. Y. Ehlers-Smith kindly generated the maps used.
Competing interests
The authors declare that they have no financial or personal relationships which may have inappropriately influenced them in writing this article.
Authors' contributions
Both G.R.H.G. (University of KwaZulu-Natal) and C.T.D. (University of KwaZulu-Natal) conceived the project and wrote the manuscript. In addition, C.T.D. sourced funding and G.R.H.G. collected and analysed the data.
References
Abbott, A.T.D., 2010, Checklist of Ngele Mountain and environs Weza State Forest 3029DA collectors. Unpublished report, Nicholson/Abbott Herbarium, Port Edward. [ Links ]
Adie, H., Rushworth, I. & Lawes, M.J., 2013, 'Pervasive, long-lasting impact of historical logging on composition, diversity and above ground carbon stocks in Afrotemperate forest', Forest Ecology and Management 310, 887-895. http://dx.doi.org/10.1016/j.foreco.2013.09.037 [ Links ]
Bodart, C., Brink, A.B., Donnay, F., Lupi, A., Mayaux, P. & Achard, F., 2013, 'Continental estimates of forest cover and forest cover changes in the dry ecosystems of Africa between 1990 and 2000', Journal of Biogeography 40, 1036-1047. http://dx.doi.org/10.1111/jbi.12084 [ Links ]
Boon, R., 2010, Pooley's trees of eastern South Africa, Flora & Fauna Publications Trust, Durban. [ Links ]
Carbutt, C. & Edwards, T.E., 2006, 'The endemic and near endemic angiosperms of the Drakensberg Alpine Centre', South African Journal of Botany 72, 105-132. http://dx.doi.org/10.1016/j.sajb.2005.06.001 [ Links ]
Cawe, S.G. & McKenzie, B., 1989, 'The afromontane forests of Transkei, southern Africa II. A floristic classification', South African Journal of Botany 55, 31-39. [ Links ]
Clark, V.R., Dold, A.P., McMaster, C., McGregor, G., Bredencamp, C. & Barker, N.P., 2014, 'Rich sister, poor cousin: Plant diversity and endemism in the great Winterberg-Amatholes (Great Escarpment, Eastern Cape, South Africa)', South African Journal of Botany 92, 159-174. http://dx.doi.org/10.1016/j.sajb.2014.01.008 [ Links ]
Cooper, K.H., 1985, The conservation status of indigenous forests in Transvaal, Natal and O.F.S., South Africa, Wildlife Society of Southern Africa, Durban. [ Links ]
Downs, C.T., 2005, 'Abundance of the endangered Cape Parrot, Poicephalus robustus, in South Africa: Implications for its survival', African Zoology 40, 15-24. [ Links ]
Food and Agriculture Organization of the United Nations (FAO), 2010, Global Forest Resources Assessment, Forestry Paper 163, FAO, Rome. [ Links ]
Fourcade, H.G., 1889, Report on the Natal Forests, Watson, Pietermaritzburg. http://dx.doi.org/10.5962/bhl.title.23002 [ Links ]
Geldenhuys, C.J., 2007, 'Growth, ingrowth and mortality patterns over stands and species in the forest growth sites at Weza and Ngome indigenous forests, KwaZulu-Natal: Report on first remeasurement, 2003/2004', project no. 2008-450, Department of Water Affairs and Forestry, Pretoria. [ Links ]
Germishuizen, G., Meyer, N.L., Steenkamp, Y. & Keith, M., 2006, 'A checklist of South African plants', South African National Biodiversity Network report no. 41, SABONET, Pretoria. [ Links ]
Hart, L., Grieve, G. & Downs, C.T. 2013, 'Fruiting phenology and implications of fruit availability in the fragmented Ngeli Forest Complex, KwaZulu-Natal, South Africa', South African Journal of Botany 88, 296-305. http://dx.doi.org/10.1016/j.sajb.2013.08.003 [ Links ]
Hilliard, O.M. & Burtt, B.L., 1987, The botany of the southern Natal Drakensberg, Annals Series, Vol 15, National Botanical Gardens, Cape Town. [ Links ]
King, N.L., 1941, 'The exploitation of the indigenous forests of South Africa', Journal of the South African Forestry Association 6, 26-48. http://dx.doi.org/10.1080/03759873.1941.9631098 [ Links ]
Lawes, M.J., 1990, 'The distribution of the samango monkey (Cercopithecus mitis erythrarchus Peters, 1852 and Cercopithecus mitis labiatus I. Geoffroy, 1843) and forest history in southern Africa', Journal of Biogeography17, 669-680. http://dx.doi.org/10.2307/2845148 [ Links ]
Lawes, M.J., Griffiths, M.E. & Boudreau, S., 2007, 'Colonial logging and recent subsistence harvesting affect the composition and physiognomy of a podocarp dominated afrotemperate forest', Forest Ecology and Management248, 48-60. http://dx.doi.org/10.1016/j.foreco.2007.04.012 [ Links ]
Lawes, M.J., Macfarlane, D.M. & Eeley, H.A.C., 2004, 'Forest landscape pattern in the KwaZulu-Natal midlands, South Africa: 50 years of change or stasis?', Austral Ecology 29, 613-623. http://dx.doi.org/10.1111/j.1442-9993.2004.01396.x [ Links ]
McCracken, D.P., 2004, 'Dependence, destruction and development: A history of indigenous timber use in South Africa', in M.J. Lawes, H.A.C. Eeley, C.M. Shackleton & B.G.S. Geach (eds.), Indigenous forests and woodlands in South Africa, pp. 77-308, University of KwaZulu-Natal Press, Pietermaritzburg. [ Links ]
Meter, E.B., Edwards, T.J., Rennie, M.A. & Granger, J.E., 2002, 'A checklist of the plants of Mahwaqa Mountain, KwaZulu-Natal', Bothalia 32, 101-115. [ Links ]
Moll, E.J., 1972, 'The current status of mistbelt mixed Podocarpus forest in Natal', Bothalia 10, 595-598. [ Links ]
Mucina, L. & Geldenhuys, C.J., 2006, 'Afrotemperate, subtropical and azonal forests', in L. Mucina & M.C. Rutherford (eds.), The vegetation of South Africa, Strelitzia 19, pp. 586-655, South African National Biodiversity Institute, Pretoria. [ Links ]
Mucina, L. & Rutherford, M.C., 2006, The vegetation of South Africa, Lesotho and Swaziland, Strelitzia 19, South African National Biodiversity Institute, Pretoria. [ Links ]
Norman, P.G., 2012, 'The geology of the Ngele area: A brief geological description', unpublished report, Southport Geology and Natural History Society, Uvongo. [ Links ]
Pooley, E.A., 1998, Field guide to wild flowers KwaZulu-Natal and the Eastern Region, Natal Flora Publications Trust, Durban. [ Links ]
Pooley, E.A., 2003, Mountain flowers: A field guide to the flora of the Drakensberg and Lesotho, Flora Publications Trust, Durban. [ Links ]
Raimondo, D., Von Staden, L., Foden, W., Victor, J.E., Helme, N.A., Turner, R.C. et al., 2009, Red list of South African plants, Strelitzia 25, South African National Biodiversity Institute, Pretoria. [ Links ]
Robertson, J. & Lawes, M.J., 2005, 'User perceptions of conservation and participatory management of iGxalingenwa forest, South Africa', Environmental Conservation 32, 64-75. http://dx.doi.org/10.1017/S0376892905001979 [ Links ]
Storrar, C., 1990, The four faces of Fourcade: The biography of a remarkable scientist, Maskew Miller Longman, Cape Town. [ Links ]
Wade, T.G., Riitters, K.H., Wickham, J.D. & Jones, K.B., 2003, 'Distribution and causes of global forest fragmentation', Conservation Ecology 7(2), 7. [ Links ]
Wirminghaus, J.O., Downs, C.T., Symes, C.T. & Perrin, M.R., 1999, 'Conservation of the Cape Parrot in southern Africa', South African Journal of Wildlife Research 29, 118-129. [ Links ]
Wirminghaus, J.O., Downs, C.T., Symes, C.T. & Perrin, M.R., 2001, 'Fruiting in two afromontane forests in KwaZulu-Natal, South Africa: The habitat type of the endangered Cape Parrot Poicephalus robustus', South African Journal of Botany 67, 329-336. [ Links ]
 Correspondence:
Correspondence:
Colleen Downs
Private Bag X01
Pietermaritzburg 3209
South Africa
Email: downs@ukzn.ac.za
Received: 30 June 2014
Accepted: 07 Jan. 2015
Published: 16 Sept. 2015
Note: Additional supporting information may be found in the online version of this article as an Online Appendix:http://dx.doi.org/10.4102/koedoe.v57i1.1237-1.
1 In a subsequent collecting trip with two other postgraduate researchers, collections were made of several Asclepiadaceae believed to be undescribed species. Additional work done by these other researchers may lead to the publication of new species descriptions.














