Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Koedoe
versión On-line ISSN 2071-0771
versión impresa ISSN 0075-6458
Koedoe vol.51 no.1 Pretoria ene. 2009
ORIGINAL RESEARCH
Influence of fire frequency on Colophospermum mopane and Combretum apiculatum woodland structure and composition in northern Gonarezhou National Park, Zimbabwe
Edson GandiwaI, II; Shakkie KativuI
ITropical Resource Ecology Programme, University of Zimbabwe, Zimbabwe
IIScientific Services, Zimbabwe Parks and Wildlife Management Authority, Zimbabwe
ABSTRACT
We investigated the long-term effects of fire frequency on Colophospermum mopane and Combretum apiculatum woodland structure and composition in northern Gonarezhou National Park (GNP), Zimbabwe. Fire frequency was categorised as high (every 1–2 years), medium (every 3–4 years) and low (every 5–6 years). The following variables were measured or recorded: plant height, species name, canopy depth and diameter, basal circumference, number of stems per plant, plant status (dead or alive) and number of woody plants in a plot. There was a positive correlation (r = 0.55, P = 0.0007) between annual area burnt (total from January to December) and annual rainfall (average over two rain stations per rain year, July to June) between 1972 and 2005. A total of 64 woody species were recorded from C. mopane and C. apiculatum woodlands. Mean plant height increased from 4.5 to 8.2 meters in C. mopane woodland and from 4.5 to 5.1 meters in C. apiculatum woodland in areas subjected to high and low fire frequencies. In C. mopane woodland, low fire frequency was characterised by a significantly low density of woody plants (P < 0.001), however, with a significantly high mean basal area (P < 0.001). Fire frequency had no significant effect on species diversity (P > 0.05). Our results suggest that C. mopane and C. apiculatum woodlands are in a state of structural transformation. Fire frequency effects, however, appear to be woodland specific. Fire management strategies in GNP should take into consideration annual rainfall and the different vegetation types.
CONSERVATION IMPLICATION: This study provides valuable information on fire frequency effects on woody vegetation in northern GNP, which can be used in fire management programmes for the park. The positive relationship between annual rainfall and annual area burnt emphasises the need for wildlife managers to consider annual rainfall in fire management.
Keywords: fire history; annual area burnt; annual rainfall; savanna; southeast lowveld
INTRODUCTION
For millennia, fire has been a common, natural phenomenon in African savanna systems and can be seen as one of the determinants of savanna ecosystem structure and functioning (Walker 1981). Savannas are characterised by the co-existence of a herb layer, dominated by C4 grasses, and variable densities of trees and shrubs (Knoop & Walker 1985; Scholes & Archer 1997). A fire regime is defined as the combination of frequency, season, intensity and type of fire that prevails in a given area (Van Wilgen & Scholes 1997). Fire frequency is an important characteristic of a fire regime. How frequently a site burns affects many aspects of ecosystem function from nutrient cycling, to grass productivity, to tree recruitment (e.g. Anderson et al. 2007; Beringer et al. 2007; Bird et al. 2000; Bond & Keeley 2005; DeBano et al. 1998; Frost & Robertson 1987; Holdo et al. 2007; Kraaij & Ward 2006; Prior et al. 2009; Scholes 1997). It has been suggested that fire markedly influences the structure of savannas (Higgins et al. 2007) and acts as the most important 'consumer' globally, depressing biomass from its climate potential (Bond et al. 2005). Fire consumes vegetation and acts as a top-down control on ecosystem structure (Bond 2008). Fires thus always reduce plant biomass and, depending on their frequency and severity, can also replace trees with shrublands or grasslands (Bond et al. 2005). Vegetation communities vary in their response to period of exposure to fire. The specific impact of fire varies with the fire regime and the vegetation type affected (Heinl et al. 2007). Some plant species may not survive when fires are too frequent, too early or too late in their growth life (Chandler et al. 1983), and the overall effects of fire on growth and survival of plants vary among and within species (Gambiza et al. 2005).
Earlier studies in savanna ecosystems suggest that fire can have a marked effect on savanna vegetation. For example, in Zambia, complete protection from fire resulted in an increase in woody canopy height and biomass (Trapnell 1959). Chidumayo (1988) recorded lower species diversity in fire-protected plots in the Copperbelt area, Zambia. Scholes and Walker (1993) recorded an increase in woody biomass with reduction in fire frequency and/or intensity in their Nylsvley study, South Africa. Kennan (1972) observed that plots with 14 years of annual burning at the Matopos Research Station, Zimbabwe, showed a 70% reduction in woody phytomass in comparison with plots completely excluded from fire. Kennedy and Potgieter (2003) recorded significant differences in tree height, canopy diameter, mean stem circumference and number of stems with different fire seasons in C. mopane woodland in South Africa. In another study, dry season fires in a C. apiculatum savanna in South Africa led to the formation of short, open, extensively coppiced shrub vegetation while wet season fires produced taller and closed vegetation (Tainton et al. 1993). Enslin et al. (2000) reported that frequent fires in the Kruger National Park (KNP), South Africa, led to reduced height of woody plants through topkill; however, no marked decrease in species composition was observed with increasing fire frequency. Similarly, Govender et al. (2006) recorded no marked effect of fire on woody species composition and in contrast recorded high impacts of fire on the structure of woody communities in the KNP. Shackleton and Scholes (2000) found that high fire frequencies had differing effects on various attributes of vegetation structure, for example reduced woody plant height, increased number of stems per plant and increased plant density. In contrast, Van Wyk (1971) showed that the frequency of burning over a 42-year period appeared not to have had any noticeable effect on woody plant density in the KNP. In the Sengwa Wildlife Research Area, Zimbabwe, late dry season fires altered the height class distribution of shrubs, resulting in an increase in proportions of shrubs (Mapaure 2001). Elsewhere, Stronach (1989) found that late dry season fires in the Serengeti National Park, Tanzania, were more damaging to woody plants than were early dry season fires. In addition, Stronach (1989) observed that woody plant growth was greatest when fire was excluded. In another study, Van de Vijver et al. (1999) found no significant effect of fire on tree density over a 25-year period in Tarangire National Park, northern Tanzania. More recent work in North Australian savannas suggests that an increase in the frequency or severity of fires is likely to change the tree density and stand basal area of savannas (Prior et al. 2009).
Burning is a major management issue in the parks and wildlife estates of Zimbabwe (Robertson 1993), particularly where wildfires are concerned (Mapaure 2001). Very little is known of the fire regime in GNP before the early 1920s. Tsetse fly eradication teams started fires in the dry month of October between 1924 and 1968 (Tafangenyasha 2001). However, data on fire frequency related to tsetse fly eradication fires and the areas burnt are not available. For the period between 1968 and 1985, attempts were made to protect the entire park from burning by maintaining firebreaks and extinguishing fires whenever possible (Sharp 1985). Because most fires recorded in GNP were unprescribed and anthropogenically induced, about 51% of all recorded fires, efforts to protect the park from fires were largely unsuccessful (Department of National Parks and Wild Life Management [DNPWLM] 1998). From 1985 onwards, fire management policies in GNP focused on the reduction of spatial extent of burnt area and frequency of fires through the preparation of border and internal firebreaks, effecting early burning and placement of fire detection systems, and this was accompanied by a specific fire reaction plan (DNPWLM 1998). Elsewhere, changes in fire management policies have been reported to have influenced fire regimes in protected areas. For example, a fire exclusion policy in the KNP between 1948 and 1956 resulted in some inadvertent very large fires (Bond & Archibald 2003). However, Van Wilgen et al. (2004) found no perceivable management effect on annual area burnt. Fire management in the KNP has, however, passed through several phases from 1926 to 2005 (see Van Wilgen et al. 2004, 2008 for details) as compared to GNP since the park was established in 1968 (see DNPWLM 1998 for details).
Changes in woody vegetation structure and composition may have important implications for wildlife habitat, biotic diversity and risk of future disturbances (Peterson & Reich 2001). Fire occurrences have been a common phenomenon in GNP, and concern has been raised about possible increase in annual area burnt and changes in woodland structure and composition. Previous studies in GNP suggest that fire is an important factor that has contributed to woodland changes in the park. Tafangenyasha (1997) reported that frequent fires in GNP led to the decline of canopy woodlands and herbaceous plant cover. In addition, Tafangenyasha (2001) suggested that fire and elephant (Loxodonta africana) damage contributed to the general modification of habitats and, in particular, to the decline of Brachystegia glaucescens woodland in GNP. Because savanna landscapes respond variably to long-term burning (e.g. Higgins et al. 2007) and if fire is to be useful as a management tool, it is imperative that we have a greater predictive understanding of its effects across a wider range of species and settings (Shackleton 2007). GNP provided us with an excellent opportunity for the evaluation of the long-term effects of fire frequency on woody vegetation structure and composition. Fire frequency data were available for a period spanning 34 years (i.e. from 1972 to 2005). Our objectives were to answer the following questions: (1) What were the trends in fire occurrences, the extent of burnt areas and their relationship with rainfall in northern GNP? (2) To what extent did different fire frequencies affect the structure and composition of C. mopane and C. apiculatum woodlands in northern GNP?
METHODS
Study area
GNP is located in the southeast lowveld of Zimbabwe (see Figure 1), between latitudes 21° 00' – 22° 15' S and longitudes 30° 15' – 32° 30' E. GNP has a mean altitude of 400 m above sea level and covers a total area of 5 053 km2 (DNPWLM 1998). Average annual precipitation for the park is 466 mm, and average monthly maximum temperatures are 25.9 °C in July and 36 °C in January. Average monthly minimum temperatures range between 9 °C in June and 24 °C in January. Prevailing winds are southeasterly, with wind speeds of between 11 and 17 km per hour (Robertson 2005). Farrell (1968) provided a description of the vegetation of northern GNP. The major vegetation type is C. mopane woodland, which covers approximately 40% of the park (DNPWLM 1998). Our study sites were located in the northern section of GNP, in the Pombadzi Wilderness Area (see Figure 1). The Pombadzi Wilderness Area has a spatial extent of approximately 755.2 km2 (DNPWLM 1998).
Review of fire and rainfall records
Original fire and rainfall data kept at Chipinda Pools, GNP, were used in the present study. Both planned and unplanned fires in GNP have been recorded since the establishment of the park in 1968. However, for the purposes of this study, only fire data from 1972 to 2005 were used. This was because rainfall data also used in this study were only available from 1972. The details regarding burnt area are, however, not precise but provide a reasonable indication of the position and extent of individual burns. Oral accounts of fires from park rangers in GNP were used to confirm the spatial extent of recorded fires, particularly for the period 2001 to 2005.
We examined data on the number of fires per year, the spatial extent of fires and fire occurrences in C. mopane and C. apiculatum woodlands from annual fire maps and supplementary data for the period 1972–2005. The spatial extents of burnt areas were represented on fire maps through shading, using the geographical location grid-square system on GNP maps. In the present study, the extents of burnt areas were determined by summing the area of shaded grid-squares, because no digitised fire maps were available. In addition, corresponding data on the annual fire maps, for example on the spatial extent of areas burnt, were checked to verify with the shaded extent of burnt area on the fire maps. In order to determine fire frequencies in C. mopane and C. apiculatum woodlands, the spatial extent of each woodland boundary, using a generalised vegetation map of northern GNP, was traced on top of each fire map for the period 1972–2005, using the geographical location grid-square system. The fire frequency spatial scale used in the present study was 1 km2. We then noted the number of times each grid-square (1 km2) falling under the traced extent of C. mopane and C. apiculatum woodlands was burnt over the 34-year period only in the Pombadzi Wilderness Area, northern GNP. We categorised the grid-squares falling under C. mopane and C. apiculatum woodlands based on the number of times they were burnt over the 34 years into three fire frequency classes with the following mean intervals between fires: high fire frequency (burnt every 1–2 years), medium fire frequency (burnt every 3–4 years) and low or rare fire frequency (burnt every 5–6 years). We also utilised the summarised past fire frequency maps, (1) for the entire GNP (see Tafangenyasha 2001) and (2) only for northern GNP (see also Robertson 2005), in standardising our fire frequency classes. In order to identify the burnt areas and study woodlands in the field, we used geographical features to demarcate and identify the burnt areas and woodlands, using 1:50 000 topographical maps. For rainfall, we gathered all the monthly rainfall amounts available for each year from 1972 to 2005 from two rain stations located in northern GNP, that is at Chipinda Pools and Save-Runde Base Camp.
Experimental layout
This study was based on a stratified random design. Areas that differed with respect to fire frequency over the past 34 years but had been last burnt at approximately the same time in the two woodlands were chosen for the study. This was done to avoid confounding the effects of time since fire and frequency of fires on vegetation. The position of the plots followed stratification according to woodland type and fire frequency. We distinguished and identified C. mopane and C. apiculatum woodlands primarily by considering the dominant tree species, height and cover (Lindenmayer et al. 2005) together by referring to a generalised GNP vegetation map. For each of the three fire frequency classes in C. mopane and C. apiculatum woodlands, at least four replicate plots (total of 27 plots), depending on spatial extent of vegetation patches, were identified as a way of maximising representation of the vegetation (see Table 1). Plots were randomly selected for both C. mopane and C. apiculatum woodlands, using random number tables based on the GNP topographical map grid-square intercept system, in relation to the stratified study sites. We avoided placement of sample plots near roads, streams, rock outcrops and the boundaries between woodlands types.

Data collection
The floristic composition and the structure of the woody vegetation component were assessed towards the end of the rainy season between March and May 2006. At this time of the year, species composition is most conspicuous. Plots measuring 20 m × 30 m (0.06 ha) were used throughout the study. This plot size was determined following Walker's (1976) method of having at least 15–20 trees inside a plot. We collected data on woody plant variables only from the tree and shrub layers. A tree was defined as a plant that was > 3.0 m tall, with one or a few dominant trunks, with a basal diameter of > 6.0 cm, whereas a shrub was defined as a plant that was < 3.0 m with one or a few dominant trunks or having a basal diameter of < 6.0 cm. All woody plants rooted within a plot were recorded and measured. Woody plants occurring along plot margins were included if at least half of the rooted system was inside the plot (Walker 1976). For multi-stemmed plants located at the edges of plots, only stems with more than half their base inside the plot were measured. Plots were assessed only once during the study period, and the variables measured are given in Table 2.

Data analyses
Average annual rainfall in the preceding season, hereafter 'annual rainfall', for the period 1972 to 2005 was calculated by averaging over rain year (i.e. July to June) the monthly rainfall data from two rain stations. In order to attain representativeness in annual rainfall data, for months with missing values (average two (2) months per year with missing rainfall values for the period 1972–1978) for one of the rain stations we filled in the missing data with the long-term average monthly rainfall values for those particular months. The total annual burnt area for each year from 1972 to 2005 was calculated by summing all the burnt areas from January to December.
Tree and shrub heights were averaged for each plot on the basis of woody vegetation assessment data. For each plot, the total number of stems was divided by the number of plant stands to give an average number of stems per plant. Stem circumference data were used in calculating stem basal areas for each woody stem using the following formula:
Basal area (m2) = (C²/4π), where C is stem circumference.
Densities for stems, trees and shrubs per hectare for each plot were calculated using the following formula:
Density (y/ha) = [(x×10,000 m2)/(plot area, m2)], where y denotes any of trees, shrubs or stems and x is the recorded number of trees, shrubs and stems.
Canopy depth and canopy diameter data were used in the calculation of canopy volumes for each tree and shrub in a plot. Canopy volumes were calculated using the following formulae (Anderson 1973):
1) Shrub canopy volume (m3) = ¼(π)(Ht)(D1)(D2), where Ht is height, D1 and D2 are two canopy diameters at 90°, and
2) Tree canopy volume (m3) = ¼(CD)(D1)(D2), where CD is canopy depth.
In order to determine the changes in species composition in relation to different fire frequency and similarities in species composition between C. mopane and C. apiculatum woodlands, we calculated the Shannon Index (H' ) and Sørensen Index of community similarity (ISS). The Shannon Index for each plot was calculated using the following formula (Ludwig & Reynolds 1988):
H' = –Σ (pi × ln(pi)), where pi is the fraction of the entire population made up of species i, and ln is the natural logarithm.
The Sørensen Index of community similarity was calculated using the following formula (Mueller-Dombois & Ellenberg 1974):
ISS = [2(C)/(A+B)] × 100%, where C is the number of species common to woodland A and B, A is the total number of species in woodland A, and B is the total number of species in woodland B.
We conducted statistical tests using STATISTICA for Windows, version 6 (StatSoft 2001). Vegetation survey data were tested for normality using the Kolmogorov-Smirnov test. Data on number of dead plants and density of shrubs, trees and stems were log10 (y + 10) transformed, where y is the vegetation variable quantity, in order to satisfy the assumptions of normality and equality of variance. Further, we performed two separate simple linear regression analyses in order to determine the relationship (1) between annual rainfall of the preceding year and annual area burnt and (2) between annual rainfall of the preceding two rainy seasons and annual area burnt. In both analyses, annual rainfall was taken as the dependent variable and annual area burnt as the independent variable.
In order to test the effects of fire frequency on woodland structure and composition for each woodland, we performed one-way ANOVA tests in STATISTICA for Windows. We considered it to be of no significance when the value of the probability of significance (P) was greater than 0.05. Three levels of statistical significance were considered: P < 0.05, P < 0.01 and P < 0.001. In order to determine whether different sites, specifically sample plots, could be distinguished from each other based on fire history, woody species composition and woodland type, we performed two different analyses: first, a principal component analysis (PCA) and, second, a hierarchical cluster analysis (HCA). The PCA was performed in STATISTICA for Windows, using the following variables: woody plant height, number of species per plot, species diversity, basal area, canopy volume, woody plant densities, number of stems per plant and number of dead plants. We performed the HCA using the average linkage method at 95% similarity level with a matrix of 27 plots and only species abundance data of the 64 woody species in Minitab for Windows (Minitab 2000).
RESULTS
Fire history and relationship between annual area burnt and annual rainfall
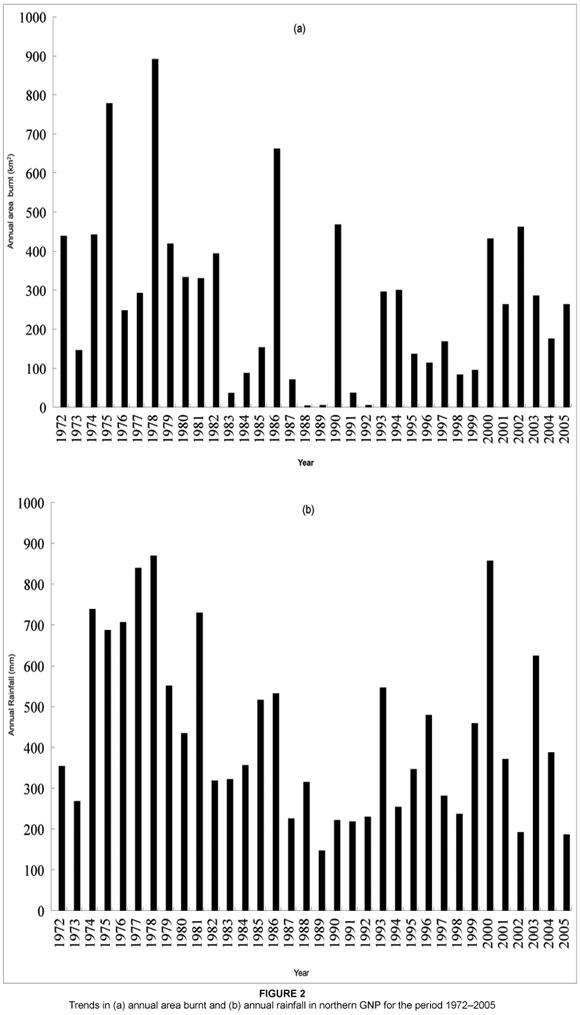
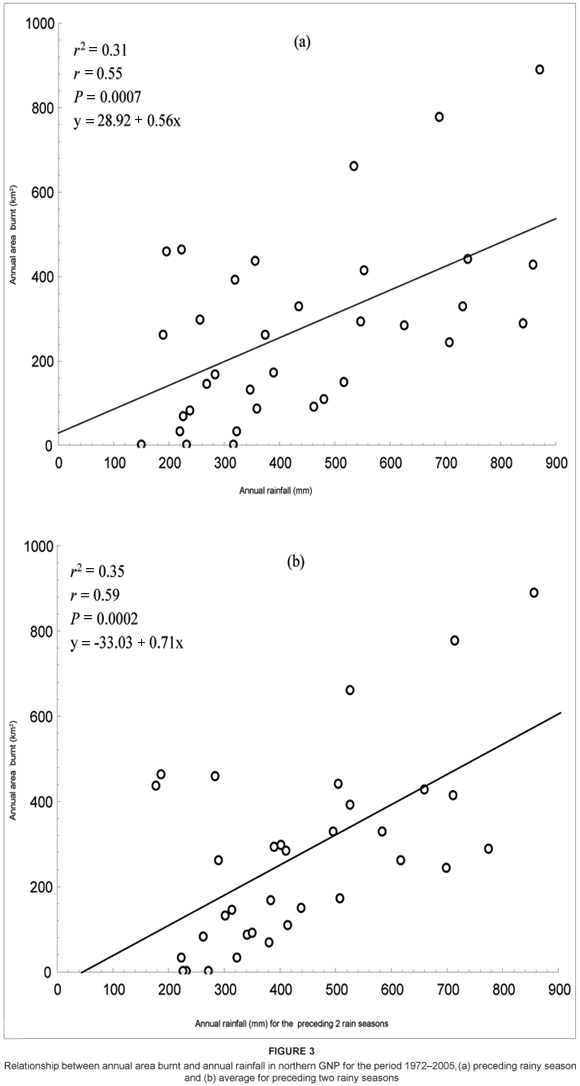
A total of 186 fires (individual ignitions: average 5.5 fires a year) were recorded in GNP during the 34 years between (and including) 1972 to 2005. The average annual area burnt for the period 1972 to 2005 in northern GNP was 274 km2 (median = 264, range = 4.4–891.7 km2). This is equivalent to 25% of the area of northern GNP. The annual area burnt was highest in 1978 with a value of 891.7 km2 and least in 1988 with a value of 4.4 km2 (see Figure 2a). There was high variability in the annual area burnt in the period 1972 to 2005. This variability seemed to correspond to annual rainfall. For example, the period 1974–1979 was characterised by higher annual rainfall (see Figure 2b), and this corresponded to the greater spatial extent of annual area burnt in the same period. In contrast, the period 1982–1992 was generally characterised by lower annual rainfall, and correspondingly lower values of annual area burnt were recorded, however, with the exception of 1986 and 1990 (see Figure 2b). There was a positive correlation (r = 0.55, P = 0.0007) between annual area burnt and annual rainfall in northern GNP between 1972 and 2005 [y (annual area burnt, km2) = 28.92 + 0.56x (annual rainfall, mm)] (see Figure 3a). A relatively stronger positive correlation (r = 0.59, P = 0.0002) between annual area burnt and annual rainfall was produced when using average annual rainfall for the preceding two rainy seasons [y (annual area burnt, km2) = -33.03 + 0.71x (annual rainfall for the preceding two seasons, mm)] (see Figure 3b).
Fire frequency effects on woody vegetation structure and composition
A total of 1 986 individual woody plants with 4 328 stems were assessed in the 27 sampling plots and 64 woody plant species were recorded. Forty-four (44) and 20 woody plant species were recorded in C. apiculatum and C. mopane woodland respectively. Increasing fire frequency altered woodland structure variables in both C. mopane and C. apiculatum woodlands. In C. mopane woodland, (1) mean tree height increased from 4.49 ± 0.48 m in areas subjected to high fire frequency to 8.22 ± 0.60 m in areas subjected to low or rare fire frequency (one-way ANOVA, F2,11 = 62.90, P < 0.001; see Table 3); (2) mean number of dead plants increased from 14 ± 5 to 143 ± 115 plants/ha in areas subjected to low and high fire frequencies (F2,11 = 9.23, P < 0.01); (3) mean basal area decreased from 2.91 ± 0.45 to 1.06 ± 0.30 m2/ha in areas subjected to low and high fire frequencies (F2,11 = 32.99, P < 0.001); (4) mean number of stems/plant increased from 2 ± 1 to 4 ± 1 stems/plant in areas subjected to low and high fire frequencies (F2,11 = 4.89, P < 0.05); (5) mean shrub density increased from 73 ± 27 to 557 ± 167 shrubs/ha in areas subjected to low and high fire frequencies (F2,11 = 56.19, P < 0.001); (6) mean stem density increased from 365 ± 40 to 2 230 ± 836 stems/ha in areas subjected to low and high fire frequencies (F2,11 = 106.02, P < 0.001); and (7) mean tree density increased from 98 ± 4 to 420 ± 100 trees/ha in areas subjected to low and high fire frequencies (F2,11 = 94.23, P < 0.001). In contrast, there were no significant effects of increasing fire frequency on mean shrub height, mean number of woody species per plot, canopy volume and species diversity (H' ) (one-way ANOVA, all P > 0.05; see Table 3) in C. mopane woodland.
In C. apiculatum woodland, (1) mean tree height increased from 4.45 ± 0.44 to 5.08 ± 0.13 m in areas subjected to high and low fire frequencies (one-way ANOVA, F2,10 = 4.80, P < 0.05) and (2) mean canopy volume increased from 17 038.92 ± 6 886.43 to 38 943.6 ± 6 310.4 m3/ha in areas subjected to high and low fire frequencies (F2,10 = 5.50, P < 0.05). However, there were no significant effects of increasing fire frequency on mean shrub height, mean number of woody species per plot, number of dead plants, basal area, number of stems per plant, woody plant densities and species diversity (H' ) (one-way ANOVA, all P > 0.05; see Table 3) in C. apiculatum woodland.
Association of sample plots in relation to fire history
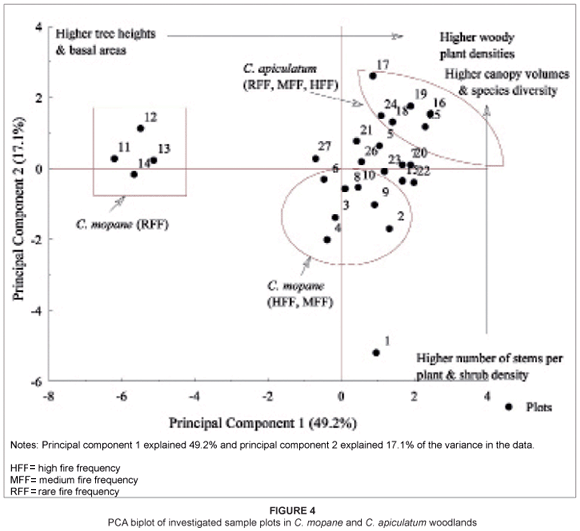
Figure 4 shows a PCA biplot with 27 plots from both C. mopane and C. apiculatum woodlands (also refer to Table 1 for sample plot details). Principal component 1 defines a gradient from sites with taller trees and higher basal areas to sites with high densities of woody plants. Principal component 2 defines a gradient from sites with higher numbers of stems per plant and higher shrub densities to sites with lower numbers of stems per plant but with higher canopy volumes and higher species diversity. There was an association of sample plots from C. mopane woodland in relation to fire history, whereas the same was not evident for sample plots from C. apiculatum woodland. Sample plots occurring in the HFF and MFF sites in C. mopane woodland were comprised of high woody plant densities and high numbers of stems per plant whilst sample plots in the RFF sites of C. mopane woodland had high canopy volumes and basal areas. In addition, a high number of sample plots from C. apiculatum woodland consisted of high woody plant densities, high canopy volumes and species diversity.
Woodland (or community) similarity and sample plot classification based on fire history
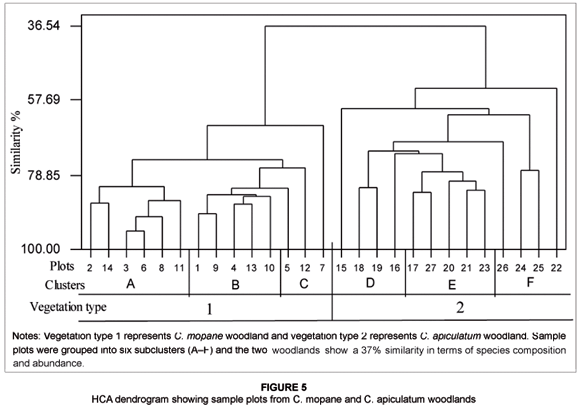
The Sørensen Index of community similarity showed a 31.3% similarity between C. mopane and C. apiculatum woodlands. Only 10 woody species were common in C. mopane and C. apiculatum woodlands and these were Monodora junodii, Euclea natalensis, Burkea africana, C. apiculatum, Combretum imberbe, Combretum molle, Combretum mossambicense, Dichrostachys cinerea, Rhoicissus revoilii and Pterocarpus rotundifolius. Furthermore, the HCA dendrogram distinguished plots from C. mopane and C. apiculatum woodlands and showed a 37% similarity between the two woodlands based on species composition and abundance. The 27 sample plots were grouped into six subclusters (A–F) (see Figure 5; also refer to Table 1 for sample plot details). First, clusters A to C consisted of all sample plots from C. mopane woodland. Cluster A comprised sample plots from MFF and RFF sites with species such as Rhus tenuinervis, Boscia angustifolia, C. mopane and Xeromphis obovata having been recorded. Cluster B was dominated by sample plots from HFF sites, which largely consisted of the following species: C. mopane, Dichrostachys cinerea, Combretum imberbe and Grewia monticola. Cluster C was composed of a mixture of sample plots from HFF, MFF and RFF sites. Second, clusters D–F consisted of all sample plots from C. apiculatum woodland. Cluster D had an association of sample plots from HFF sites and comprised the following dominant species: C. apiculatum, C. molle, Julbernardia globiflora and Terminalia sericea. Cluster E mainly consisted of sample plots from RFF sites which were characterised by woody species such as Dalbergia melanoxylon, Xeroderris stuhlmannii and Pterocarpus brenanii. Last, cluster F consisted of sample plots from RFF and MFF sites and contained the following species: C. apiculatum, Strychnos spinosa, Dalbergia melanoxylon and Rhus gueinzii, among others.
DISCUSSION
Fire history and relationship between annual area burnt and annual rainfall
Fire occurrences have been a common phenomenon in GNP. Annual area burnt trends in northern GNP appear to be strongly affected by annually fluctuating rainfall (see Figure 2). Our results show evidence that annual rainfall is related to the extent of annual area burnt in northern GNP (see Figure 3a). The positive correlation recorded between annual rainfall and annual area burnt in northern GNP suggests that it may be possible to predict future annual burnt area given the annual rainfall of the preceding year. This is so as rainfall determines fuel load, which in the present case is predominantly grass (Norton-Griffiths 1979; Robertson 2005; Skarpe 1992). Grass production occurs in the wet season. This is followed by an extended dry season. The latter leads to a continuous cover of fuel (Higgins et al. 2000). Our results are consistent with earlier observations from other studies in savanna ecosystems. Stronach (1989) observed a corresponding relationship in Serengeti National Park, Tanzania. Balfour and Howison (2002) found a strong correlation between area burnt in one year and average rainfall for two preceding summers in a study in Hluhluwe-Umfolozi Park, South Africa, and Govender et al. (2006) and Van Wilgen et al. (2004) found that at the scale of the entire KNP, the area burnt in any given year was strongly correlated with rainfall (and therefore grass fuels) in the preceding two years. We found that a two-year accumulator worked relatively better in northern GNP (see Figure 3b). Russell-Smith et al. (2007) recorded greater fire frequency and intensity in years with above average rainfall in the preceding one or two years in Australian landscapes. They observed that the area burnt in some Australian landscapes was also greater in places that had greater areas burnt in the preceding one or two years. They further reported that fire in preceding years was positively associated with contemporary fire. This observation, however, is in contrast to the negative association hypothesised on the grounds that prior fire reduces fuel loads and consequently reduces fire size and risk (Russell-Smith et al. 2007).
The rainfall data we used in this study, however, might have influenced the correlation strength between annual rainfall and annual area burnt. This is because rainfall data were taken from only two rain stations. In addition, there were some years in which some monthly rainfall data from one rain station were missing for the period 1972–1978. Data from these two rain stations may, however, have inaccurately represented the spatial variation in rainfall. Factors such as relief and spatial scale may influence the spatial variation in rainfall; hence, interpolation from only two rain stations may not be adequate. Nevertheless, the differences may be negligible given the relatively small size of the study area, in this case only the Pombadzi Wilderness Area, and also the fact that the entire park lies in a low-altitude, semi-arid region.
Effects of fire frequency on woody vegetation structure and composition
We recorded a decreasing trend in mean tree height associated with increasing fire frequency in C. mopane and C. apiculatum woodlands (see Table 3). Other researchers have reported similar findings from studies conducted in savanna ecosystems (e.g. Moreira 2000; Nefabas & Gambiza 2007; Sabiiti & Wein 1988; Shackleton & Scholes 2000; Tafangenyasha 2001; Trapnell 1959). Decreases in woody plant height in frequently burnt sites may be a result of plant tissue topkill (e.g. Enslin et al. 2000; Hoffmann & Solbrig 2003; Peterson & Reich 2001). Topkill, however, decreases with increase in plant size (Frost & Robertson 1987). We recorded more shrubs in the frequently burnt sites. This may probably be due to repeated fires, maintaining a firetrap, which limits growth in height (Bond & Van Wilgen 1996). It has been suggested that savanna trees only recruit into adult population once they escape the zone of grass fires (Higgins et al. 2000) and that the presence of fire-resistant adult trees for many years buffers the system against frequent fires (Hanan et al. 2008).
We recorded a high number of dead plants, mostly shrubs, in HFF sites, particularly in C. mopane woodland. Miller (2000) stated that plant mortality reflects the amount of meristematic tissues killed by heat. It is, however, suggested that high fire frequency is not responsible for the death of large trees as flame heights of a burning grass layer are usually low (Bond & Van Wilgen 1996). In savanna woody species, topkill is much more frequent than complete mortality after fire (Hoffmann & Solbrig 2003). Grass fire, instead, suppresses the recruitment of small individuals to the canopy layer (Bond & Van Wilgen 1996). It is possible that the high number of dead woody plants recorded in C. mopane woodland with increasing fire frequency may be a coincidence, as other factors may have interacted with fire, leading to an increase in woody plant deaths. However, it is also likely that the high number of dead plants, particularly small trees, recorded in C. mopane woodland may have resulted from the cumulative effects of fire over a long time. We attribute the large tree mortalities recorded in the RFF sites to droughts, old age and disease. Tafangenyasha (1998) reported a large drought-related tree dieback in the southeast lowveld of Zimbabwe associated with the 1991/92 drought. Other studies have associated woodland change in semi-arid savanna with fire and droughts (e.g. Fensham et al. 2003; Mosugelo et al. 2002).
The present study suggests a link between fire frequency and basal area among woody plants in C. mopane woodland (see Figure 4). The decreasing basal area associated with increasing fire frequency is attributed to the high number of thin stems recorded in MFF and HFF sites that result from basal coppicing and high shrub densities. In contrast, high mean basal areas were observed in RFF sites. We attribute this to the existence of tall, huge and single-stemmed trees. Our observations are consistent with several other researchers' findings (e.g. Enslin et al. 2000; Gambiza 2001; Peterson & Reich 2001; Shackleton & Scholes 2000; Tafangenyasha 2001). Prior et al. (2009) reported that an increase in mortality leads to a decrease in tree density and tree size through a decrease in average longevity, both of which lead to a decrease in basal area.
The high number of stems per plant recorded in HFF sites, particularly in C. mopane woodland (see Figure 4), is likely to be associated with an increase in basal coppicing (or resprouting) as a response to burning among species such as C. mopane, D. cinerea and G. monticola. This observed trend correlates well with the findings of other researchers (e.g. Kennedy & Potgieter 2003; Walters 2000). Walters (2000) observed an increase in the number of stems per plant with increasing fire frequency in C. mopane woodland in the southeast lowveld of Zimbabwe. In a study in Australia, Marrinan et al. (2005) observed that resprouting was more common in burnt sites than in unburnt sites. Tchie and Gakahn (1989), however, suggested that the number of stems per plant is species specific. The low difference recorded in the present study in the number of stems per plant in C. apiculatum woodland may be attributed to the presence and abundance of commonly multi-stemmed plant species such as Pseudolachnostalysis maprouneifonia, D. cinerea, Grewia spp. and Combretum spp., among others, in most plots occurring in this woodland.
Canopy volume is an important woody plant variable that provides an indication of areas resistant to fire damage (Melville et al. 1999). Low canopy volume values that were recorded, particularly in C. apiculatum woodland (see Table 3) with increasing fire frequency, are attributed to changes in plant height, multi-stemming and topkill. Thus, woody species in this woodland type could have failed to establish themselves as mature trees as a result of repeated exposure to fire (Cauldwelland & Zieger 2000). Significant canopy volume changes, particularly in C. apiculatum woodland with increasing fire frequency, are consistent with earlier observations by Tafangenyasha (2001) who observed that frequent fires and elephant damage led to a significant loss of woody canopy cover of B. glaucescens woodland in northern GNP. In contrast, the insignificant differences in canopy volume in C. mopane woodland are probably due to the fact that more shrubs were recorded with increasing fire frequency and that these made a considerable contribution to canopy volume in frequently burnt sites.
Our results showed that woody plant densities in C. mopane woodland increased with increasing fire frequency (see Table 3). We attribute this recorded trend to the increase in multi-stemming among woody plants as a consequence of basal coppicing. The results of the present study, particularly in the case of woody plant densities in C. mopane woodland, lend support to findings by Nefabas and Gambiza (2007). They recorded a high density of smaller trees with increasing fire frequency at Matopos Research Station, Zimbabwe. We observed that high fire frequency did not induce a significant change in plant densities in C. apiculatum woodland. Similar findings were reported by Higgins et al. (2007) in a long-term fire study in the KNP. Higgins et al. (2007) attributed the non-significant changes in plant density to the ability of woody species to resprout from the base after fire. The contrasting plant densities recorded between C. mopane and C. apiculatum woodlands point to other factors, because both C. mopane and C. apiculatum woodlands occurred on basalt soils. Such factors could be differences in the clustering patterns of tree species that can be attributed to the availability of water and the effects of competition on seedling establishment (e.g. Groen et al. 2008).
Our results revealed that fire frequency had no significant influence on woody species diversity in C. apiculatum and C. mopane woodlands in northern GNP for the period 1972 to 2005. Enslin et al. (2000) also found no relationship between species diversity and increasing fire frequency in the KNP. Similarly, Govender et al. (2006), studying fire effects in the KNP, concluded that fire does not have a marked effect on woody species composition. However, species vary in their responses to fires (Andersen et al. 2005), and fire tolerance among woody species depends on species sensitivity, the duration and intensity of the fire and the physiological and developmental state of individual plants (Frost & Robertson 1987). Our results showed that both the C. mopane and C. apiculatum woodlands had relatively low similarity in terms of woody plant species. The HCA dendrogram showed that C. mopane and C. apiculatum woodlands were 37% similar based on species composition (see Figure 5), whilst the Sørensen Index for community similarity showed a 31.3% similarity. The difference in the two measures of woodland or community similarity in terms of woody species composition can be attributed to the weighting component that was induced in the HCA analyses by the species abundance data.
The influence of large herbivores (e.g. elephants) on woody vegetation has not been ascertained in the present study as low numbers of herbivore-damaged plants (e.g. debarked or broken plants) were observed during the field assessments. Any consideration of herbivory as a confounding factor is only speculative. However, some studies have highlighted the significant interaction between fire and herbivory (e.g. Baxter 2003; Ben-Shahar 1996; Ben-Shahar 1998; Eckhardt et al. 2000; Guy 1989; Mapaure & Campbell 2002; Mourik et al. 2007; Ribeiro et al. 2008; Van Langevelde et al. 2003) and frost in the presence of elephants and/or fire (e.g., Holdo 2005; 2007); as important factors in shaping savanna woodlands and grasslands. Although the possible effects of herbivory following fires have not been considered in the present study, it would appear that herbivore impacts were diluted to a greater extent as large areas in northern GNP were burnt each year in relatively low-herbivore-density areas. In addition, it is also possible that combinations of past tsetse fly elimination and human activities may have influenced woody plant structure and composition in C. mopane and C. apiculatum woodlands. Sankaran et al. (2005) suggested that if disturbances by fire, browsers and humans were absent, large sections of the African continent would switch to a wooded state.
CONCLUSIONS
Our study showed that annual rainfall was positively correlated with annual area burnt in northern GNP between 1972 and 2005. However, the relationship between annual area burnt and annual rainfall has to be interpreted with caution, particularly where large areas are concerned, for example the entire GNP. This is because of possible pronounced spatial heterogeneity in annual rainfall from one year to another, thus influencing fuel load (Brockett et al. 2001). This study revealed that high fire frequency altered the structure of C. mopane and C. apiculatum woodlands. However, the response was different for C. mopane and C. apiculatum woodlands. In contrast, fire frequency appeared not to have altered the species diversity of both C. mopane and C. apiculatum woodlands. C. mopane woodland structure appeared to have been more altered by frequent fires than C. apiculatum woodland. The difference in more pronounced fire effects on C. mopane woodland structure as compared to C. apiculatum woodland may be explained through the physiology of C. mopane plants. C. mopane leaves contain resin, which volatilises and allows shoots to burn even when green (Trollope & Potgieter 1983). It is most likely that fire can convert C. mopane woodland of high stature into scrub C. mopane woodland (Mlambo & Mapaure 2006). Tafangenyasha (2001) identified B. glaucescens woodland as one major vegetation type to have become degraded by fire and elephant damage after the establishment of GNP in 1968. This study has added C. mopane and C. apiculatum woodlands as having been altered partly by repeated fires in northern GNP. Future fire management strategies in GNP should take into consideration annual rainfall and the different vegetation types present in the park. This study examined the effects of fire frequency on only two vegetation types in northern GNP. However, fire frequency effects on woody vegetation need to be studied further, on a larger scale (e.g. the entire park), taking into consideration other vegetation types and the additional effects of herbivory. We end by recommending the following: (1) continued maintenance of a reliable and up-to-date fire database that serves as an important baseline for fire research (Parr & Chown 2003) and (2) establishment of a long-term fire experiment programme in GNP, drawing from the KNP's experience (see Van Wilgen et al. 2007 for details). A long-term fire experiment programme will not only enhance the understanding of ecological processes but will also play an important role in influencing management decisions related to fire management in GNP.
ACKNOWLEDGEMENTS
We wish to express our thanks and gratitude to Dr. M.Z. Mtsambiwa, the Director-General of Zimbabwe Parks and Wildlife Management Authority for permission and support to carry out this study in GNP. We are extremely grateful to Professor C.H.D. Magadza, Professor N.A.G. Moyo, Dr. H. Madzikanda, Dr. F. Murindagomo, Takawira Madawo, Bruce Clegg and Sarah Clegg for their invaluable contributions at various stages of this study. The comments of Dr. L.C. Foxcroft and anonymous reviewers improved the manuscript. We would like to thank Handikoshiwe Muvhuni, Eliah Nhongo and Itai Siabulembu for excellent help in the field. Special thanks to Patience Zisadza for invaluable help in the manuscript preparation. The assistance of Liezel Grunewald with the handling of the manuscript was highly appreciated. Many former and present staff members in GNP have contributed to the collection and updating of fire and rainfall data used in this study, too numerous to mention individually, but we recognise their invaluable contribution. The support of the Frankfurt Zoological Society is gratefully acknowledged.
REFERENCES
Andersen, A.N., Cook, G.D., Corbett, L.K., Douglas, M.M., Eager, R.W., Russell-Smith, J., Setterfield, S.A., Williams, R.J. & Woinarski, J.C.Z. 2005. Fire frequency and biodiversity conservation in Australian tropical savannas: implications from the Kapalga fire experiment. Austral Ecology 30, 155–167. [ Links ]
Anderson, G.D. 1973. Vegetation composition and elephant damage in the major habitat types of the Sengwa Wildlife Research Area of Rhodesia. MSc thesis, Salisbury: University of Rhodesia. [ Links ]
Anderson, T.M., Ritchie, M.E., Mayemba, E., Eby, S., Grace, J.B. & McNaughton, S.J. 2007. Forage nutritive quality in the Serengeti ecosystem: the roles of fire and herbivory. The American Naturalist 170(3), 343–357. [ Links ]
Balfour, D.A. & Howison, O.E. 2002. Spatial and temporal variation in a mesic savanna fire regime: responses to variation in annual rainfall. African Journal of Range and Forage Science 19, 45–53. [ Links ]
Baxter, P.W.J. 2003. Modelling the impact of the African elephant, Loxodonta africana, on woody vegetation in semi-arid savannas. PhD thesis, Berkeley: University of California. [ Links ]
Ben-Shahar, R. 1996. Woodland dynamics under the influence of elephants and fire in northern Botswana. Vegetatio 123, 153–163. [ Links ]
Ben-Shahar, R. 1998. Changes in structure of savanna woodlands in northern Botswana following the impacts of elephants and fire. Plant Ecology 136, 189–194. [ Links ]
Beringer, J., Hutley, L.B, Tapper, N.J. & Cernusak, L.A. 2007. Savanna fires and their impact on net ecosystem productivity in North Australia. Global Change Biology 13, 990–1004. [ Links ]
Bird, M.I., Veenendaal, E.M., Moyo, C., Lloyd, J. & Frost, P. 2000. Effect of fire and soil texture on soil carbon in a sub-humid savanna (Matopos, Zimbabwe). Geoderma 94, 71–90. [ Links ]
Bond, W.J. & Archibald, S. 2003. Confronting complexity: fire policy choices in South African savanna parks. International Journal of Wildland Fire 12, 381–389. [ Links ]
Bond, W.J. & Keeley, J.E. 2005. Fire as a global "herbivore": the ecology and evolution of flammable ecosystems. Trends in Ecology and Evolution 20, 387–394. [ Links ]
Bond, W.J. & Van Wilgen, B.W. 1996. Fire and plants. Population and community biology series 14. London: Chapman and Hall. [ Links ]
Bond, W.J. 2008. What limits trees in C4 grasslands and savannas? Annual Review of Ecology, Evolution and Systematics 39, 641–659. [ Links ]
Bond, W.J., Woodward, F.I. & Midgley, G.F. 2005. The global distribution of ecosystems in a world without fire. New Phytologist 165, 525–538. [ Links ]
Brockett, B.H., Biggs, H.C. & Van Wilgen, B.W. 2001. A patch mosaic burning system for conservation areas in southern African savannas. International Journal of Wildland Fire 10, 169–183. [ Links ]
Cauldwelland, A.E. & Zieger, U. 2000. A reassessment of the fire-tolerance of some miombo woody species in the Central Province, Zambia. African Journal of Ecology 38(2), 138–146. [ Links ]
Chandler, C., Cheney, P., Thomas, P., Traband, L. & Williams, D. 1983. Fire in forestry – volume 1. Forest fire behaviour and effects. New York: John Wiley and Sons. [ Links ]
Chidumayo, N.E. 1988. A re-assessment of effects of fire on Miombo regeneration in the Zambian copperbelt. Journal of Tropical Ecology 4(4), 361–372. [ Links ]
Coates-Palgrave, K. 1997. Trees of southern Africa. 2nd edn. Cape Town: Struik. [ Links ]
DeBano, F.L., Neary, G.D. & Ffolliot, F.P. 1998. Fire's effects on ecosystem. New York: John Wiley and Sons. [ Links ]
Department of National Parks and Wild Life Management. 1998. Gonarezhou National Park management plan 1998–2002. Harare: Government Printers. [ Links ]
Eckhardt, H.C., Van Wilgen, B.W. & Biggs, H.C. 2000. Trends in woody vegetation cover in the Kruger National Park, South Africa, between 1940 and 1998. African Journal of Ecology 38, 108–115. [ Links ]
Enslin, B.W., Potgieter, A.L.F., Biggs, H.C. & Biggs, R. 2000. Long-term effects of fire frequency and season on the woody vegetation dynamics of the Sclerocarya birrea/Acacia nigrescens savanna of the Kruger National Park, South Africa. Koedoe 43(1), 27–37. [ Links ]
Farrell, J.A.K. 1968. Preliminary notes on the vegetation of the lower Sabi-Lundi basin, Rhodesia. Kirkia 6(2), 233–248. [ Links ]
Fensham, R.J., Fairfax, R.J., Butler, D.W. & Bowman, D.M.J.S. 2003. Effects of fire and drought in a tropical eucalypt savanna colonised by rain forest. Journal of Biogeography 30(9), 1405–1414. [ Links ]
Frost, P.G.H. & Robertson, F. 1987. The ecological effects of fire in savannas, in Walker, B.H. (ed.) Determinants of tropical savannas. IUBS monograph series 3. pp. 93–140, Paris: IRL Press. [ Links ]
Gambiza, J. 2001. The regeneration of Zambezi teak forests after logging: influence of fire and herbivory. PhD thesis, Harare: University of Zimbabwe. [ Links ]
Gambiza, J., Campbell, B.M, Moe, S.R. & Frost, P.G.H. 2005. Fire behaviour in a semi-arid Baikiaea plurijuga savanna woodland on Kalahari sands in western Zimbabwe. South African Journal of Science 101, 239–244. [ Links ]
Govender, N., Trollope, W.S.W. & Van Wilgen, B.W. 2006. The effect of fire season, fire frequency, rainfall and management on fire intensity in savanna vegetation in South Africa. Journal of Applied Ecology 43(4), 748–758. [ Links ]
Groen, T.A., Van Langevelde, F., Van de Vijver, C.A.D.M., Govender, N. & Prins, H.H.T. 2008. Soil clay content and fire frequency affect clustering in trees in South African savannas. Journal of Tropical Ecology 24, 269–279. [ Links ]
Guy, P.R. 1989. The influence of elephants and fire on a Brachystegia-Julbernardia woodland in Zimbabwe. Journal of Tropical Ecology 5(2), 215–226. [ Links ]
Hanan, N.P., Sea, W.B., Dangelmayr, G. & Govender, N. 2008. Do fires in savannas consume woody biomass? A comment on approaches to modelling savanna dynamics. The American Naturalist 171(6), 851–856. [ Links ]
Heinl, M., Sliva, J., Murray-Hudson, M. & Tacheba, B. 2007. Post-fire succession on savanna habitats in the Okavango Delta wetland, Botswana. Journal of Tropical Ecology 23, 705–713. [ Links ]
Higgins, S.I., Bond, W.J, February, E.C., Bronn, A., Euston-Brown D.I.W., Enslin, B., Govender, N., Rademan, L., O'regan, S., Potgieter, A.L.F., Scheiter, S., Sowry, R., Trollope, L. & Trollope, W.S.W. 2007. Effects of four decades of fire manipulation on woody vegetation structure in savanna. Ecology 88(5), 1119–1125. [ Links ]
Higgins, S.I., Bond, W.J. & Trollope, W.S.W. 2000. Fire, resprouting and variability: a recipe for grass-tree coexistence in savanna. Journal of Ecology 88, 213–229. [ Links ]
Hoffmann, W.A. & Solbrig, O.T. 2003. The role of topkill in the differential response of savanna woody species to fire. Forest Ecology and Management 180, 273–286. [ Links ]
Holdo, R.M. 2005. Stem mortality following fire in Kalahari sand vegetation: effects of frost, prior damage and tree neighbourhoods. Plant Ecology 180, 77–86. [ Links ]
Holdo, R.M. 2007. Elephants, fire, and frost can determine community structure and composition in Kalahari woodlands. Ecological Applications 17(2), 558–568. [ Links ]
Holdo, R.M., Holt, R.D., Coughenour, M.B. & Ritchie, M.E. 2007. Plant productivity and soil nitrogen as a function of grazing, migration and fire in an African savanna. Journal of Ecology 95, 115–128. [ Links ]
Kennan, T.C.D. 1972. The effects of fire on two vegetation types of Matopos, Rhodesia. Proceedings of the Tall Timbers Fire Ecology Conference 11, 53–98. [ Links ]
Kennedy, A.D. & Potgieter, A.L.F. 2003. Fire season affects size and architecture of Colophospermum mopane in southern African savannas. Plant Ecology 167, 179–192. [ Links ]
Knoop, W. & Walker, B. 1985. Interactions of woody and herbaceous vegetation in a southern African savanna. Journal of Ecology 73, 235–253. [ Links ]
Kraaij, T. & Ward, D. 2006. Effects of rain, nitrogen, fire and grazing on tree recruitment and early survival in bush-encroached savanna, South Africa. Plant Ecology 186, 235–246. [ Links ]
Lindenmayer, D., Crane, M. & Michael, D. 2005. Woodlands: a disappearing landscape. Collingwood, Victoria: CSIRO Publishing [ Links ]
Ludwig, A.J. & Reynolds, J.F. 1988. Statistical ecology: a primer on methods and computing. New York: John Wiley and Sons. [ Links ]
Mapaure, I. 2001. The influence of elephants and fire on the structure and dynamics of miombo woodlands in Sengwe Wildlife Research Area, Zimbabwe. PhD thesis, Harare: University of Zimbabwe. [ Links ]
Mapaure, I.N. & Campbell, B.M. 2002. Changes in miombo woodland cover in and around Sengwa Wildlife Research Area, Zimbabwe, in relation to elephants and fire. African Journal of Ecology 40, 212–219. [ Links ]
Marrinan, M.J., Edwards, W. & Landsberg, J. 2005. Resprouting of saplings following a tropical rainforest fire in north-east Queensland, Australia. Austral Ecology 30(8), 817–826. [ Links ]
Martin, R.B. 1974. Structure, biomass and utilisation of vegetation in the Mopane and Miombo woodlands of the Sengwa Wildlife Research Area. Certificate in Field Ecology Report, Salisbury: University of Rhodesia. [ Links ]
Melville, H.I.A.S., Cauldwell, A.E. & Bothma, J. du P. 1999. Comparisons of two techniques for estimating tree canopy volume. South African Journal of Wildlife Resources 29(4), 113–117. [ Links ]
Miller, M. 2000. autecology, in Brown, J.K. & Smith, J.K. (eds). Wildland fire in ecosystems: effects of fire on flora. General Technical Report RMRS-GTR-42-vol.2. pp. 9–34, Department of Agriculture, Forest Service, Rocky Mountain Research Station, Ogden, UT. [ Links ]
Minitab. 2000. Minitab statistical software, release 12. Coventry: Minitab Limited. [ Links ]
Mlambo, D. & Mapaure I. 2006. Post-fire resprouting of Colophospermum mopane saplings in a southern African savanna. Journal of Tropical Ecology 22, 231–231. [ Links ]
Moreira, A.G. 2000. Effects of fire protection on savanna structure in central Brazil. Journal of Biogeography 27(4), 1021–1029. [ Links ]
Mosugelo, D.K., Moe, S.R., Ringrose, S. & Nellemann, C. 2002. Vegetation changes during a 36-year period in northern Chobe National Park, Botswana. African Journal of Ecology 40(3), 232–240. [ Links ]
Mourik, A.A., Van Langevelde, F., Van Tellingen, E., Heitkönig, I.M.A. & Gaigher, I. 2007. Stability of wooded patches in a South African nutrient-poor grassland: do nutrients, fire or herbivores limit their expansion? Journal of Tropical Ecology 23, 529–537. [ Links ]
Mueller-Dombois, D. & Ellenberg, H. 1974. Aims and methods of vegetation ecology. New York: John Wiley and Sons. [ Links ]
Nefabas, L.L. & Gambiza, J. 2007. Fire-tolerance mechanisms of common woody plant species in a semiarid savanna in south-western Zimbabwe. African journal of Ecology 45, 550–556. [ Links ]
Norton-Griffiths, M. 1979. The influence of grazing and fire on vegetation dynamics of the Serengeti, in Sinclair, A.R.E. & Norton-Griffiths, M. (eds). Serengeti: dynamics of an ecosystem. pp. 310–352, Chicago: University of Chicago Press. [ Links ]
Parr, C.L. & Chown, S.L. 2003. Burning issues for conservation: a critique of faunal fire research in Southern Africa. Austral Ecology 28, 384–395. [ Links ]
Peterson, D.W. & Reich, P.B. 2001. Prescribed fire in oak savanna: fire frequency effects on stand structure and dynamics. Ecological Applications 11(3), 914–927. [ Links ]
Prior, L.D., Murphy, B.P. & Russell-Smith, J. 2009. Environmental and demographic correlates of tree recruitment and mortality in North Australian savannas. Forest Ecology and Management 257(1), 66–74. [ Links ]
Ribeiro, N.S., Shugart, H.H. & Washington-Allen, R. 2008. The effects of fire and elephants on species composition and structure of the Niassa Reserve, northern Mozambique. Forest Ecology and Management 255, 1626–1636. [ Links ]
Robertson, E.F. 1993. Early burning in the Brachystegia woodland of the parks and wildlife estates. The Zimbabwe Science News 27, 68–71. [ Links ]
Robertson, E.F. 2005. Fire in northern Gonarezhou National Park, in Feasibility study for the development of a rhino IPZ in Gonarezhou. pp. 34–44, SADC Regional Programme for Rhino Conservation, semester 12a task 3.1–12.1, Harare. [ Links ]
Russell-Smith, J., Yates, C.P., Whitehead, P.J., Smith, R., Craig, R., Allan, G.E., Thackway, R., Frakes, I., Cridland, S., Meyer, M.C.P. & Gill, A.M. 2007. Bushfires 'down under': patterns and implications of contemporary Australian landscape burning. International Journal of Wildland Fire 16, 361–377. [ Links ]
Sabiiti, E.N. & Wein, R.W. 1988. Fire behaviour and the invasion of Acacia sieberiana into savanna grassland openings. African Journal of Ecology 26, 301–313. [ Links ]
Sankaran, M., Hanan, N.P., Scholes, R.J., Ratnam, J., Augustine, D.J., Cade, B.S., Gignoux, J., Higgins, S.I., Roux, X.L., Ludwig, F., Ardo, J., Banyikwa, F., Bronn, A., Bucini, G., Caylor, K.K., Coughenour, M.B., Diouf, A., Ekaya, W, Feral, C.J., February, E.C., Frost, P.G.H., Hiernaux, P., Hrabar, H., Metzger, K.L., Prins, H.H.T, Ringrose, S., Sea, W., Tews, J., Worden, J. & Zambatis, N. 2005. Determinants of woody cover in African savannas. Nature 438(8), 846–849. [ Links ]
Scholes, R.J. & Archer, S.R. 1997. Tree-grass interactions in savannas. Annual Review of Ecology and Systematics 28, 517–544. [ Links ]
Scholes, R.J. & Walker, B.H. 1993. An African savanna: synthesis of the Nylsvley study. Cambridge: Cambridge University Press. [ Links ]
Scholes, R.J. 1997.Savanna, in Cowling, R.M., Richardson, D.M. & Pierce, S.M. (eds). Vegetation of southern Africa. pp. 258–277, Cambridge: Cambridge University Press. [ Links ]
Shackleton, C.M. & Scholes, R.J. 2000. Impact of fire frequency on woody community structure and soil nutrients in Kruger National Park. Koedoe 43(1), 75–81. [ Links ]
Shackleton, C.M. 2007. The effects of fire on post-fire seed germination of selected savanna woody species. African Journal of Ecology 45, 545–549. [ Links ]
Sharp, G. 1985. Proposed fire management policy – Gonarezhou National Park. Unpublished report ref 1011/1/85. Chiredzi, Department of National Parks and Wildlife Management. [ Links ]
Skarpe, C. 1992. Dynamics of savanna ecosystems. Journal of Vegetation Science 3, 293–300. [ Links ]
StatSoft. 2001. STATISTICA for Windows, version 6. 2300. Tulsa: StatSoft. [ Links ]
Stronach, H.R.N. 1989. Grass fires in Serengeti National Park, Tanzania: characteristics, behaviour and some effects on young trees. PhD thesis, Cambridge: Cambridge University. [ Links ]
Tafangenyasha, C. 1997. Tree loss in the Gonarezhou National Park (Zimbabwe) between 1970 and 1983. Journal of Environmental Management 49, 355–366. [ Links ]
Tafangenyasha, C. 1998. Phenology and mortality of common woody plants during and after severe drought in southeastern Zimbabwe. Transactions of the Zimbabwe Scientific Association 72, 1–6. [ Links ]
Tafangenyasha, C. 2001. Decline of the mountain acacia, Brachystegia glaucescens, in Gonarezhou National Park, southeast Zimbabwe. Journal of Environmental Management 63, 37–50. [ Links ]
Tainton, N.M., Hardy, M.B. & Morris, C.D. 1993. The response of Combretum apiculatum savanna of the Kruger National Park to fire. Bulletin of the Grassland Society of Southern Africa 4, 20–21. [ Links ]
Tchie, N. & Gakahn, G.C. 1989. Responses in important woody species of Kenya's rangeland to prescribed burning. African Journal of Ecology 27, 119–128. [ Links ]
Trapnell, C.G. 1959. Ecological results of woodland burning experiments in northern Rhodesia. Journal of Ecology 47, 129–168. [ Links ]
Trollope, W.S.W. & Potgieter, A.L.F. 1983. Characteristics of fire behaviour in the Kruger National Park. Unpublished report to the University of Fort Hare and the Division of Nature Conservation, South Africa, Kruger National Park. [ Links ]
Van de Vijver, C.A.D.M., Foley, C.A. & Olff, H. 1999. Changes in the woody component of an east African savanna during 25 years. Journal of Tropical Ecology 15, 545–564. [ Links ]
Van Langevelde, F., Van de Vijver, C.A.D.M., Kumar, L., Van de Koppel, J., De Ridder, N., Van Andel, J., Skidmore, A.K., Hearne, J.W., Stroosnijder, L., Bond, W.J., Prins, H.H.T. & Rietkerk, M. 2003. Effects of fire and herbivory on the stability of savanna ecosystems. Ecology 84(2), 337–350. [ Links ]
Van Wilgen, B.W. & Scholes, R.J. 1997. The vegetation and fire regimes of southern hemisphere Africa, in Van Wilgen, B.W., Andreae, M.O., Goldammer, J.G. & Lindesay, J.A. (eds). Fire in southern African savannas: ecological and atmospheric perspectives. pp. 27–46, Johannesburg: Witwatersrand University Press. [ Links ]
Van Wilgen, B.W., Govender, N. & Biggs, H.C. 2007. The contribution of fire research to fire management: a critical review of a long-term experiment in the Kruger National Park, South Africa. International Journal of Wildland Fire 16, 519–530. [ Links ]
Van Wilgen, B.W., Govender, N. & Macfadyen, S. 2008. An assessment of the implementation and outcomes of recent changes to fire management in the Kruger National Park. Koedoe 50(1), 22–31. [ Links ]
Van Wilgen, B.W., Govender, N., Biggs, H.C., Ntsala, D. & Funda, X.N. 2004. Response of savanna fire regimes to changing fire-management policies in a large African national park. Conservation Biology 18(6), 1533–1540. [ Links ]
Van Wyk, P. 1971. Veld burning in Kruger National Park. An interim report of some aspects of research. Proceedings Tall Timbers Fire Ecology Conference 11, 9–31. [ Links ]
Walker, B.H. 1976. An approach to the monitoring of changes in the composition and utilisation of woodland and savanna vegetation. South African Journal of Wildlife Resources 6(1), 1–32. [ Links ]
Walker, B.H. 1981. Is succession a viable concept in African savanna ecosystems?, in West, D.C., Shugart, H.H. & Botkin, D.B. (eds). Forest succession. pp. 431–447, Berlin: Springer Verlag. [ Links ]
Walters, J.M. 2000. Effect of season and type of fire on Colophospermum mopane woodland in the southeastern lowveld of Zimbabwe. MSc thesis, Pietermaritzburg: University of Natal. [ Links ]
 Correspondence to:
Correspondence to:
Edson Gandiwa
Scientific Services, Gonarezhou National Park
Private Bag 7003
Chiredzi, Zimbabwe
e-mail: egandiwa@gmail.com
Received: 10 Jan. 2009
Accepted: 02 Mar. 2009
Published: 04 June 2009
DOI: 10.4102/koedoe. v51i1.685
This article is available at: http://www.koedoe.co.za














