Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Surgery
versión On-line ISSN 2078-5151
versión impresa ISSN 0038-2361
S. Afr. j. surg. vol.60 no.4 Cape Town dic. 2022
http://dx.doi.org/10.17159/2078-5151/SAJS3803
COLORECTAL SURGERY
Colorectal cancer in South Africa study on the effect of delayed diagnosis to treatment intervals on survival
C BouterI; B PuttergillII; GY HymanIII; S MaphosaI; P GaylardIV; H EtheredgeI; J FabianI; P RuffI; B BebingtonI, VII
IWits Donald Gordon Medical Centre, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, South Africa
IIDepartment of Surgery, Chris Hani Baragwanath Academic Hospital, South Africa
IIIDepartment of Surgery, School of Clinical Medicine, University of the Witwatersrand, South Africa
IVData Management and Statistical Analysis, South Africa
VDepartment of Internal Medicine, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, South Africa
VIDivision of Medical Oncology, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, South Africa
VIIDepartment of Surgery, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, South Africa
ABSTRACT
BACKGROUND: Data on colorectal cancer (CRC) diagnosis to treatment interval (DTI), an index of quality assurance in high-income countries (HICs) is lacking in South Africa. This study aimed to determine DTIs and their impact on CRC survival in a South African cohort
METHODS: Participants (n = 289) from the Colorectal Cancer in South Africa (CRCSA) cohort were identified for inclusion. The DTI was defined as the duration between the diagnosis and initial definitive treatment and categorised into approximate quartiles (Q1-4). The DTI quartiles were 0-14 days, 15-28 days, 29-70 days, and > 71 days. Overall survival (OS) was illustrated using the Kaplan-Meier method and compared between DTI groups using Cox proportional hazards (PH) regression.
RESULTS: There was no significant impact of the DTI (as quartiles) on overall CRC survival. The median length of time between DTI in this cohort was 29 days. Significant associations were identified between the DTI and self-reported ethnicity (p-value = 0.025), the site of the malignancy (colon vs rectum) (p-value < 0.0001), multidisciplinary team (MDT) review (p-value = 0.015) and the initial treatm.ent modality (p-value < 0.0001).
CONCLUSION: Prolonged DTIs did not significantly impact survival for those with CRC in the CRCSA cohort. Symptom to diagnosis time should be investigated as a determinant of survival.
Keywords: pathways to care, colorectal cancer, diagnosis to treatment delay, survival
Introduction
The South African National Cancer Registry (NCR), a division of the National Health Laboratory Service (NHLS), is a pathology-based cancer registry based on inputs from public and private health facilities, the South African Oncology Consortium, Hospice Association, Childhood Cancer Foundation South Africa and the NHLS. Data from the NCR shows that incidence and mortality from colorectal cancer (CRC) are rising. Between 2002 and 2014, the NCR reported a 2.5% rise in age-standardised incidence rates (ASIR) and a 1.3% rise in age-standardised mortality rates (ASMR).1 CRC has the third-highest incidence of all cancers in the NCR. It is the second most common cause of cancer deaths worldwide, with excess mortality in resource-limited settings, namely low-middle-income countries (LMICs) and low socioeconomic status (SES) communities in high-income countries (HICs).2-5 Delays in diagnosis and access to appropriate care reinforce the need for monitoring indices of quality care for those with CRC. These indices include timely diagnosis and commencement of therapeutic interventions; the latter described as "diagnosis to treatment interval (DTI)."
Healthcare policymakers in several HICs use the DTI as a marker of quality care, advocating for a limit on the number of days from diagnosis to treatment of CRC.6-8 The question of whether delays to treatment have any impact on CRC outcomes has engendered controversy in the European context, where healthcare planning imposes maximum limits to the DTI. For example, the Netherlands set a maximum waiting time target of 35 days from diagnosis to treatment for 80% of patients. The remaining patients should be treated within 49 days. This is despite evidence that extended DTIs do not impact survival in those with CRC.9-11 In some studies counter-intuitively, there appears to be an inverse relationship between the DTI and survival - that is, the longer the DTI, the better the survival.91012 It may be in the patients' interest to delay the implementation of treatment which allows them to navigate through the complexities of the treatment plan and allows the attending team to optimise patient health before curative surgery.9
Despite the controversy surrounding the impact (or not) of the DTI on CRC survival, few studies have explored the effects of the DTI on CRC in South Africa.1314 In a prior study investigating the impact of comorbidity on outcomes in CRC, it was demonstrated that there were poorer patient outcomes with DTIs greater than 40 days.15 Although not the study's primary objective, the data from the study differed from the recommended DTI benchmarks in clinical practice guidelines from HICs. The purpose of this study was to conduct an analysis of DTIs in CRC patients treated with curative intent in the Colorectal Cancer in South Africa (CRCSA) cohort. The specific objectives were to determine which factors impacted the DTI and to ascertain whether the DTI affected CRC survival.
Methods
The CRCSA cohort is a longitudinal cohort of adults with CRC, treated in Johannesburg, as described in Bouter et al.15 The variables collected in the study pertain to the clinical presentation, management and outcomes of CRC. The study sites were Charlotte Maxeke Johannesburg Academic Hospital (CMJAH), Chris Hani Baragwanath Academic Hospital (CHBAH), Edenvale Hospital and the Klerkdorp/ Tshepong Complex, and the Wits Donald Gordon Medical Centre (WDGMC). At the baseline visit, consented participants completed a comprehensive questionnaire administered by trained research assistants. Research assistants followed up participants telephonically at 6, 12, 24, and 36 months after the first visit for details related to completed therapy and vital status. Study participant information was corroborated at multidisciplinary team (MDT) meetings and from the clinical records. Study data were collected and managed using REDCap electronic data capture tools hosted at the University of the Witwatersrand.1617
Study sample
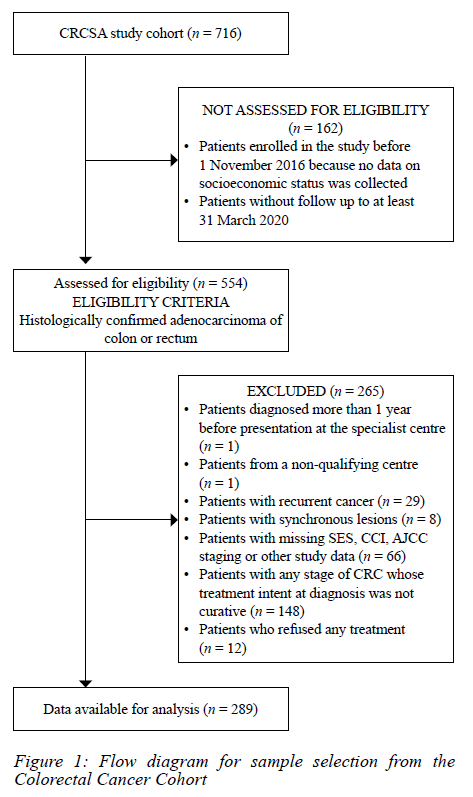
The selection of the study sample is summarised in the flow diagram in Figure 1. Participants enrolled between 1 November 2016 and 31 December 2019, with follow-up until 31 March 2020, and histologically confirmed CRC were eligible for inclusion. Participants with any of the following were excluded: recurrent CRC, confirmed synchronous lesions, missing data regarding socioeconomic status, comorbidity, and American Joint Committee on Cancer classification (AJCC) staging,18 those whose treatment intent at diagnosis was not curative, those who declined treatment, those diagnosed more than one year before presentation at a specialist centre and those referred from a non-qualifying centre. Curative intent was defined as surgical resection aimed at complete removal of the cancer. In patients with AJCC stage 4 disease, management with curative intent was considered possible where the metastases outside of locoregional disease could be surgically resected. The following variables were included in the analysis: age at diagnosis; sex; self-reported ethnicity; SES indicators: number of household members, employment status, the highest level of education (HLOE), and a list of assets in each household;19 site of recruitment (hospital); health care sector (public/private); comorbidities determined by the Charlson comorbidity index (CCI);20 date of CRC diagnosis; management that included review by a MDT; AJCC tumour stage; first potentially curative treatment modality (radiotherapy, chemotherapy, surgical); date of first potentially curative treatment; date of death (all-cause mortality) or date last seen.
Sample size
Sample size estimation was based on the key research objective - to examine the effect of the DTI on CRC survival. Recently published survival data for a CRCSA study group of 60% at three years was used. Approximately four equally sized DTI groups were assumed, with accrual and final follow-up periods of 2.5 years and 0.5 years, respectively.21,22 To detect a hazard ratio (HR) for the risk of death of at least 1.6 with 80% power at the 5% significance level, the required minimum sample size was 263 participants.
Data analysis
The DTI was calculated as the duration, in days, between the date of diagnosis and the date of initial definitive treatment and categorised into approximate quartiles (Q1-4). The DTI quartiles were defined as 0-14 days, 15-28 days, 29-70 days, and > 71 days. Overall survival (OS) was illustrated using the Kaplan-Meier method and compared between DTI groups using Cox proportional hazards (PH) regression.
This model was adjusted for age at diagnosis, sex, SES, CCI, AJCC stage, MDT involvement, location of malignancy and type of initial definitive treatment received (chemotherapy/ radiation/surgery). Data analysis was carried out using SAS version 9.4 for Windows. A 5% significance level was used.
Results
Of 554 CRCSA participants, 289 fulfilled the inclusion criteria for this study, 13 were excluded because they received no treatment, leaving 276 patients for analysis. Overall, the mean age was 57 years (SD 13 years) and sociodemographic and clinical characteristics are summarised in Table I. One hundred and forty-eight participants were excluded because the treatment intent was not curative. Of the 153 patients whose initial treatment was surgical: eight were emergency, 11 were urgent and 122 were elective (12 were unknown) at presentation. Overall, 19/276 (6.9%) were emergency/ urgent cases. The median DTI in this cohort was 29 days. For participants with rectal cancer, 70% waited more than 28 days between their diagnosis and treatment, compared to 28% of participants with colon cancer. Significant associations were identified between the DTI and self-reported ethnicity (p-value = 0.025), the site of the malignancy (colon vs rectum) (p-value < 0.0001), MDT review (p-value = 0.015), and the initial treatment modality (p-value < 0.0001) (Table I). The median follow-up time for participants was 1.3 years, and overall one-year survival was 89%. There was no significant impact of the DTI (as quartiles) on overall CRC survival (Figure 2). The sub-analysis of participant survival by stage and location of malignancy revealed the same results. When adjusting for selected covariates, males compared to females (HR 1.88; 95% CI 1.03-3.44), those with poor SES compared to wealthier participants (HR 4.75; 95% CI 1.78-12.62), and those receiving chemotherapy as curative treatment compared to those receiving surgery or radiation (HR 2.69; 95% CI 1.14-6.34) were at higher risk of death.
Discussion
Patients with CRC have complex care pathways, coordinating multiple specialists and requiring several diagnostic and therapeutic interventions.23 To optimise outcomes, clinicians need time to negotiate diagnostic and treatment decisions. Additionally, patients may require preoperative rehabilitation prior to commencing therapy, so that outcomes of such treatment are more favourable.9 However, some studies have shown that delaying treatment might impact patient survival, albeit few.14,24 It has been postulated that delayed DTIs might cause psychological distress for the patient, placing undue pressure on clinicians to expedite management decisions.9 The complexity of multimodal care in CRC treatment requires navigation between these two extremes.25,26 In this study, lengthy DTIs do not significantly impact on survival of CRC patients for whom the treatment intent was curative. The results of this study are most likely different from the prior study because participants treated for palliation were excluded from this analysis.15 Therefore, the data suggest that more time could be taken to optimise patient health and discuss treatment options in multidisciplinary teams, resulting in the best treatment decision for the patient.
On the contrary, it cannot be concluded that patients will not benefit from an expedited DTI, as there are plausible explanations as to why longer DTIs might not significantly impact survival in this cohort. One reason is that the average duration between diagnosis and treatment is not long enough to derive an effect since CRC may take up to 10 years to develop. Therefore an extra ± 30 days is unlikely to change the status of the disease.9 Lee et al.14 established that the risk of death increased 1.5 times when treatment was started between 31-150 days, compared to 30 days or less. Another potential reason that might explain the results relates to the stage of CRC at clinical presentation in this cohort. A greater proportion of participants presented late: 49% with stage 3 and 22% with stage 4 CRC at their first visit. Amri et al.,10 Pruitt et al.,12 Lee et al.14 and Strous et al.9 agree that the DTIs have less impact on survival in those presenting with advanced CRC because the stage at diagnosis is the single most predictive factor for survival. Additionally, all of these studies reported proportions of advanced-stage cancer between 30-39%, similar to this study (22%).9·10·12·14·27
The National Patient Safety Agency (NPSA) in the UK suggests that up to 60% of delays in accessing appropriate treatment for CRC happen early in the patient care pathway. These delays arise because patients do not recognise symptoms and fail to present to a healthcare facility for further investigation.28 The only way to address these issues is to look at public health education around the early symptoms of CRC and implement rigorous screening programmes to detect CRC. It is logical to assume, and confirmed in the literature, that centres with screening programmes would have higher proportions of patients presenting early in their disease. Consequently, these patients would have the option of elective (as opposed to emergency) surgery and better survival rates.29 In the South African setting, longer delays in care for CRC patients might be expected. However, the results from this study seem to be comparable with HICs with abundant access to timely healthcare, such as the United States and the Netherlands, where the median DTIs were 13 and 32 days, respectively.9,12 Another study from Massachusetts (USA) reported a mean DTI of 31.5 days. Generally, in most of these studies (and this study) rectal cancer patients waited longer to commence treatment. Although delayed DTI in rectal cancer patients did not impact their survival, delays might arise from the relative complexity of rectal cancer and the need for more specific diagnostic modalities compared to colon cancer.
In keeping with the results of this study, Roder et al.27 conclude that surgery had the shortest DTIs (compared to radiation and chemotherapy), and those who received MDTs, understandably, have a longer time to treatment given the time needed for an MDT to come together. Neither of these variables affected survival, but those that did affect survival were male sex, poor SES and chemotherapy as a first-line curative treatment. All of these are understood and accepted worldwide as predictors of mortality in CRC. Patients receiving chemotherapy as first-line treatment may have more advanced CRC. People with poor SES face more barriers to accessing health care, such as less education on the appraisal of early symptoms and a lack of access to screening for CRC, thus predisposing them to late presentation.4,5,30,31
There are limitations to this study, including its restriction to a single, urban site in Johannesburg, South Africa, limiting generalisation. Additionally, there was not sufficient data to permit an analysis of delays in the health-seeking behaviour of the CRCSA participants from onset of symptoms to seeking care, which is an important aspect of the patient journey, and this should be the focus of future work.
Conclusion
In this CRC cohort from Johannesburg, delays to initiation of treatment did not significantly impact survival for those with CRC. The significantly increased risk for poor survival was associated with male sex, poor SES and chemotherapy as first-line curative treatment. The data from this study support the need for public education regarding the interpretation of CRC symptoms and consideration of the introduction of national CRC screening programmes for earlier diagnosis of CRC. While many present for care later than would be preferred, there is time to allow for MDT review, optimise the patient for curative intervention and plan complex therapeutic interventions without adversely affecting survival.
Conflict of interest
The authors declare no conflict of interest.
Funding source
The CRCSA study is funded by the Medical Research Council of South Africa, through the Wits/SAMRC Common Epithelial Cancer Research Centre (CECRC) Grant (Paul Ruff, principal investigator). WDGMC funded the data analysis.
Ethical approval
Ethical approval for the study was obtained from the Human Research Ethics Committee (Medical) of the University of the Witwatersrand (ref. no. M150446).
ORCID
C Bouter https://orcid.org/0000-0001-8814-820X
Β Puttergill https://orcid.org/0000-0002-1759-1033
GY Hyman https://orcid.org/0000-0002-2541-0933
S Maphosa https://orcid.org/0000-0003-3933-8378
Ρ Gaylard https://orcid.org/0000-0002-0055-9168
Η Etheredge https://orcid.org/0000-0002-2581-5754
J Fabian https://orcid.org/0000-0001-7130-9142
Ρ Ruff https://orcid.org/0000-0003-1055-1730
References
1. Motsuku L, Chen WC, Muchengeti MM, et al. Colorectal cancer incidence and mortality trends by sex and population group in South Africa: 2002-2014. BMC Cancer. 2021;21(1):1-11. https://doi.org/10.1186/s12885-021-07853-1. [ Links ]
2. Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424. https://doi.org/10.3322/caac.21492. [ Links ]
3. Favoriti P, Carbone G, Greco M, et al. Worldwide burden of colorectal cancer: a review. Updates Surg. 2016;68(1):7-11. https://doi.org/10.1007/s13304-016-0359-y. [ Links ]
4. Le H, Ziogas A, Lipkin SM, Zell JA. Effects of socioeconomic status and treatment disparities in colorectal cancer survival. Cancer Epidemiol Biomarkers Prev. 2008;17(8):1950-62. https://doi.org/10.1158/1055-9965.EPI-07-2774. [ Links ]
5. Wrigley H, Roderick P, George S, et al. Inequalities in survival from colorectal cancer - a comparison of the impact of deprivation, treatment, and host factors on observed and cause specific survival. J Epidemiol Community Health. 2003;57(4):301-9. https://doi.org/10.1136/jech.57A301. [ Links ]
6. Wilkens J, Thulesius H, Schmidt I, Carlsson C. The 2015 National Cancer Programme in Sweden: introducing standardised care pathways in a decentralised system. Health Policy. 2016;120(12):1378-82. https://doi.org/10.1016/j.healthpol.2016.09.008. [ Links ]
7. Probst HB, Hussain ZB, Andersen O. Cancer patient pathways in Denmark as a joint effort between bureaucrats, health professionals and politicians - a national Danish project. Health Policy. 2012;105:65-70. https://doi.org/10.1016/j.healthpol.2011.11.001. [ Links ]
8. Van Steenbergen LN, Lemmens VEPP, Rutten HJT, Martijn H, Coebergh JWW. Was there shortening of the interval between diagnosis and treatment of colorectal cancer in southern Netherlands between 2005 and 2008? World J Surg. 2010;34:1071-9. https://doi.org/10.1007/s00268-010-0480-x. [ Links ]
9. Strous MTA, Janssen-Heijnen MLG, Vogelaar FJ. Impact of therapeutic delay in colorectal cancer on overall survival and cancer recurrence - is there a safe timeframe for prehabilitation? Eur J Surg Oncol. 2019;45(12):2295-301. https://doi.org/10.1016/j.ejso.2019.07.009. [ Links ]
10. Amri R, Bordeianou LG, Sylla P, Berger DL. Treatment delay in surgically-treated colon cancer: does it affect outcomes? Ann Surg Oncol. 2014;21(12):3909-16. https://doi.org/10.1245/s10434-014-3800-9. [ Links ]
11. Pita-Fernández S, González-Sáez L, López-Calviño B, et al. Effect of diagnostic delay on survival in patients with colorectal cancer: a retrospective cohort study. BMC Cancer. 2016;16(1):664. https://doi.org/10.1186/s12885-016-2717-z. [ Links ]
12. Pruitt SL, Harzke AJ, Davidson NO, Schootman M. Do diagnostic and treatment delays for colorectal cancer increase risk of death? Cancer Causes Control. 2013;24(5):961-77. https://doi.org/10.1007/s10552-013-0172-6. [ Links ]
13. Simunovic M, Rempel E, Thériault ME, et al. Influence of delays to nonemergent colon cancer surgery on operative mortality, disease-specific survival and overall survival. Can J Surg. 2009;52(4):E79-E86. [ Links ]
14. Lee YH, Kung PT, Wang YH, et al. Effect of length of time from diagnosis to treatment on colorectal cancer survival: a population-based study. PLoS ONE. 2019;14(1):1-16. https://doi.org/10.1371/journal.pone.0210465. [ Links ]
15. Bouter C, Bebington B, Maphosa S, et al. It's contrary -comorbidity does not affect survival of South Africans with colorectal cancer: an analysis from the Colorectal Cancer in South Africa cohort. S Afr Med J. 2020;110(5):382-8. https://doi.org/10.7196/SAMJ.2020.v110i5.14252. [ Links ]
16. Harris PA, Taylor R, Thielke R, et al. Research Electronic Data Capture (REDCap) - a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377-81. https://doi.org/10.1016/jjbi.2008.08.010. [ Links ]
17. Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform X. 2019;95:103208. https://doi.org/10.1016/j.jbi.2019.103208. [ Links ]
18. American Joint Committee on Cancer. Colon and Rectum Cancer Staging [Internet]. American Joint Committee on Cancer; 2009. Available from: https://cancerstaging.org/references-tools/quickreferences/documents/colonmedium.pdf. Accessed 24 Apr 2019. [ Links ]
19. Kabudula CW, Houle B, Collinson MA, et al. Assessing changes in household socioeconomic status in rural South Africa, 2001-2013: a distributional analysis using household asset indicators. Soc Indic Res. 2017;133(3):1047-73. https://doi.org/10.1007/s11205-016-1397-z. [ Links ]
20. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-83. https://doi.org/10.1016/0021-9681(87)90171-8. [ Links ]
21. Brand M, Gaylard P, Ramos J. Colorectal cancer in South Africa: an assessment of disease presentation, treatment pathways and 5-year survival. S Afr Med J. 2018;108(2):11822. https://doi.org/10.7196/SAMJ.2017.v108i2.12338. [ Links ]
22. Collett D. Modelling survival data in medical research [Internet]. Chapman and Hall/CRC; 2015. Available from: https://www.taylorfrancis.com/books/9780429196294. https://doi.org/10.1201/b18041. Accessed 9 May 2019. [ Links ]
23. Hayes D. Pathways to care for cancer patients. Health Serv Rep. 1974;89(2):119-27. https://doi.org/10.2307/4594999. [ Links ]
24. Whittaker TM, Abdelrazek MEG, Fitzpatrick AJ, et al. Delay to elective colorectal cancer surgery and implications for survival: a systematic review and meta-analysis. Colorectal Dis. 2021;23(7):1699-711. https://doi.org/10.1111/codi.15625. [ Links ]
25. Miles A, McClements PL, Steele RJC, et al. Perceived diagnostic delay and cancer-related distress: a cross-sectional study of patients with colorectal cancer. Psychooncology. 2017;26(1):29-36. https://doi.org/10.1002/pon.4093. [ Links ]
26. Pita-Fernández S, Pértega-Díaz S, López-Calviño B, et al. Diagnostic and treatment delay, quality of life and satisfaction with care in colorectal cancer patient: a study protocol. Health Qual Life Outcomes. 2013;11:117. https://doi.org/10.1186/1477-7525-11-117. [ Links ]
27. Roder D, Karapetis CS, Olver I, et al. Time from diagnosis to treatment of colorectal cancer in a South Australian clinical registry cohort: how it varies and relates to survival. BMJ Open. 2019;9(9):e031421. https://doi.org/10.1136/bmjopen-2019-031421. [ Links ]
28. The National Patient Safety Agency (NPSA). Delayed diagnosis of cancer - thematic review [Internet]. The Health Foundation; 2014. Available from: https://patientsafety.health.org.uk/resources/delayed-diagnosis-of-cancer-thematic-review. Accessed 2 Apr 2019. [ Links ]
29. National Institute for Clinical Excellence (Great Britain). Improving outcomes in colorectal cancer: manual update [Internet]. National Institute for Clinical Excellence; 2004. Available from: https://www.nice.org.uk/guidance/CSG5. Accessed 8 Apr 2019. [ Links ]
30. White A, Ironmonger L, Steele RJC, et al. A review of sex-related differences in colorectal cancer incidence, screening uptake, routes to diagnosis, cancer stage and survival in the UK. BMC Cancer. 2018;18(1):906. https://doi.org/10.1186/s12885-018-4786-7. [ Links ]
31. Douaiher J, Ravipati A, Grams B, et al. Colorectal cancer-global burden, trends, and geographical variations. J Surg Oncol. 2017;115(5):619-30. https://doi.org/10.1002/jso.24578. [ Links ]
 Correspondence:
Correspondence:
carolynbouter@outlook.com














