Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Surgery
versión On-line ISSN 2078-5151
versión impresa ISSN 0038-2361
S. Afr. j. surg. vol.55 no.3 Cape Town sep. 2017
GENERAL SURGERY
Resection of benign liver tumours: an analysis of 62 consecutive cases treated in an academic referral centre
J E J KrigeI, III, IV; E JonasI, III, IV; S J BeningfieldII, IV; A BoothI; U K KotzeI, III, IV; M BernonI, III, IV; S BurmeisterI, III, IV
IDepartments of Surgery University of Cape Town Health Sciences Faculty
IISurgical and Radiology, University of Cape Town Health Sciences Faculty
III Surgical Gastroenterolgy Unit Groote Schuur Hospital and the Netcare University of Cape Town, Private Academic Hospital, Observatory, Cape Town
IVPrivate Academic Hospital, Observatory, Cape Town
ABSTRACT
BACKGROUND: Benign tumours of the liver are increasingly diagnosed and constitute a substantial proportion of all hepatic tumours evaluated and resected at tertiary referral centres. This study assessed the safety and outcome after resection of benign liver tumours at a major referral centre.
METHODS: All patients with symptomatic benign liver tumours who underwent resection were identified from a prospective departmental database of a total of 474 liver resections (LRs). Demographic data, operative management and morbidity and mortality using the Accordion classification were analysed.
RESULTS: Sixty-two patients (56 women, 6 men, median age 45 years, range 17-82) underwent resection of symptomatic haemangiomata n=23 (37.1%), focal nodular hyperplasia n=19 (30.6%), biliary cystadenoma n=16 (25.8%) and hepatic adenomas n=4 (6.5%). A major resection was required in 25 patients, 14 patients had 4 segments resected, 11 had 3 segments and 37 patients had 2 or fewer segments resected. Median operating time was 169 minutes (range 80-410). Median blood loss was 300 ml (range 50-4500 ml) and an intra-operative blood transfusion was required in 6 patients. Median length of post-operative hospital stay was 7 days (range 4-32). Complications occurred in 11 patients (Accordion grades 1 n=1, 2 n=4, 3 n=1, 4 n=4, 6 n=1). Four patients required re-operation (bleeding n=2, bile leak n=1, small bowel obstruction n=1). An elderly patient died in hospital on day 16 following a postoperative cerebrovascular accident.
CONCLUSIONS: Clinically relevant symptomatic benign liver tumours comprise a substantial proportion of LRs. Our data suggest that resections can be performed safely with minimal blood loss and transfusion requirements. We advocate selective resection according to established indications. Despite the low postoperative mortality rate, the risk of postoperative complications emphasizes the need for careful selection of patients for resection.
Key words: Surgery, benign, liver tumours
Introduction
Benign liver tumours (BLTs) are now identified more frequently with the increased use of radiologic investigations and higher resolution modern imaging methods.1 Most BLTs are discovered incidentally during non-liver directed imaging, whereas a subset of patients present with either abdominal pain or discomfort. While most BLTs can safely be treated conservatively, those causing symptoms or with malignant potential should be resected. Compared to results two decades ago, liver resection (LR) has become a safer operation with low mortality rates. Recent series report improved outcomes following major hepatic resection at high-volume centres with mortality rates below 3%, compared with rates greater than 10% pre-1990.2 The perceived safety of LR has extended the indications to patients with benign liver lesions. However, despite improved overall outcomes, hepatic resection remains a major operation with postoperative morbidity rates as high as 27% when complex resections are undertaken.3 The aim of this study was to assess the spectrum of hepatic resections for BLTs in an academic referral centre, specifically assessing perioperative morbidity and mortality and outcome.
Patients and methods
Patient Selection
The prospective database in the Surgical Gastroenterology and Hepatopancreatobiliary (HPB) Unit at Groote Schuur Hospital was used to identify patients who underwent surgery for BLTs at Groote Schuur Hospital and Netcare University of Cape Town Academic Hospital between 1990 and 2015. Clinical information was collected from the database, hospital files and laboratory and pathology reports. Patient demographics, imaging studies, surgical procedures, postoperative morbidity, histopathological details of the resected specimens, evidence of recurrence and outcome were all reviewed. The Couinaud nomenclature4,5 was used to define the segmental extent of the resection and the expanded Accordion classification of surgical complications was used to score surgical outcomes.6 Indications for surgery were symptomatic tumours or a risk of malignant transformation. This study was approved by the University of Cape Town Human Research Ethics Committee (R024/2014).
Radiologic assessment
The major imaging modalities used in the diagnosis of BLTs in this study included transabdominal ultrasound (US) and triple-or four-phase contrast-enhanced computed tomography (CE-CT) for tumour characterization and segmental orientation, depiction of bilio-vascular anatomy and assessment of tumour encroachment on, or proximity to, vital vascular and biliary structures. Detailed volumetry was performed in patients with a marginal future liver remnant (FLR). When necessary, magnetic resonance imaging (MRI) was performed with liver specific MRI contrast agents, including gadopentetate dimeglumine (Magnevist®; Bayer Pharmaceuticals) and Gadoxetate disodium (Primovist®, Bayer Pharmaceuticals) used at the discretion of the radiologist. The general imaging findings for characterization of haemangioma, focal nodular hyperplasia (FNH), hepatic adenoma (HA) and biliary cystadenoma (BCA) are shown in Table 1.7 The largest diameter of tumours was measured on cross-sectional imaging.
Surgical Technique
Details of the operative technique have been described previously.811 In brief, patients were explored through a subcostal incision positioned in relation to the tumour and the liver to be resected. For large or central tumours a bilateral subcostal incision with a vertical midline extension to the xiphoid cartilage was used. The costal margins were elevated using an Omni-tract or Thompson fixed body wall retractor. Intraoperative US was used to define the relationship of the tumour to vascular structures including portal pedicles, hepatic veins and inferior vena cava (IVC). Mobilization of the liver by division of the peritoneal attachments was performed as appropriate for the planned resection. For right-sided sectoral resections and hemihepatectomies, the relevant hemiliver was fully mobilized, including exposure of the extrahepatic hepatic veins and retrohepatic IVC. The plane of the planned parenchymal transection was marked on the liver surface using diathermy, and parenchymal transection was performed using a Cavitron Ultrasonic Surgical Aspirator (CUSA). Haemostasis was secured using argon beam coagulation and suture or clip ligation for larger vessels. For formal right or left hemihepatectomy, early vascular inflow control was used. Intermittent inflow occlusion applied in cycles (application for 20 minutes and release for 10 minutes) was used selectively for lesser resections depending on blood loss during parenchymal transection. On completion the transected liver surface was inspected for bile leaks and sealed using Tisseel®. The resection area was routinely drained using closed silastic suction drains. Intermittent calf compression stockings and subcutaneous Clexane (40 mg daily) were used as deep vein thrombosis prophylaxis.
Anaesthetic management
For anaesthesia, a defined institutional protocol for LR was used.8 Radial artery and central venous catheters were inserted and arterial and central venous pressures (CVP) were continuously measured. IV fluids were restricted to 1-1.5 mL/kg/h during the extrahepatic dissection with a target CVP < 5 cm H2O during parenchymal transection to minimize hepatic venous congestion and reduce blood loss. At completion of parenchymal transection, the cumulative fluid deficit was replaced to replenish the intravascular volume and preserve renal function.
Statistical analyses
The data were analysed using Stata version 11 (StataCorp. 2009. Stata: Release 11. Statistical Software. College Station, TX: StataCorp LP). For bivariate analysis, the Pearson chi-square or Kruskal-Wallis tests were used for categorical variables, and the non-parametric Wilcoxon rank-sum test for numerical variables. Univariate and multivariate logistic regression models were used to evaluate the odds ratios (OR) and 95% confidence intervals of clinical variables (while excluding collinearity). All statistical tests were two-tailed and a p-value < 0.05 was considered statistically significant.
Results
Between January 1990 and December 2015, 474 elective LRs were performed, of which 62 were for benign indications. During the same period, and not included in this study, were two patients with multiple symptomatic hepatic adenomas who underwent liver transplantation. A further two patients with acute bleeding in hepatic adenomas were treated with selective hepatic artery embolization, after which both adenomas involuted. Patient demographics and operative details are shown in Table 2. The diagnoses were made on imaging in all patients and no patient required preoperative biopsy for characterization. Major LR, defined as resection of three or more segments, was required in 25 patients. Median operating time was 169 minutes (range 80-410). Median blood loss was 300 ml (range 50-4500) and intra-operative blood transfusion was required in 6 patients. Portal vein and hepatic artery inflow occlusion was used in 28 patients with a median total ischaemic time of 46 minutes (range 12-90 minutes). The median length of postoperative hospital stay was 7 days (range 4-32 days). The maximal tumour diameters were (i) BCA: median 60 mm, range 25-100 mm, (ii) FNH: median 58 mm, range 10-100 mm, (iii) cavernous haemangioma: median 50 mm, range 10-130 mm, (iv) HA: median 150 mm, range: 130-180 mm.
Details regarding postoperative care and complications are shown in Table 3. Eleven patients (17.7%) developed 13 postoperative complications (Accordion grades 1 n=1, 2 n=4, 3 n=1, 4 n=4, 6 n=1), with a median hospital stay of 10 days (range 5-32 days) compared to the non-complication group with a median stay of 7 days (range 4-10 days). One patient had minor complications (Accordion Grade 1) which were self-limiting and included a pleural effusion, ascitic leak and prolonged drainage of serous fluid from the drain placed at operation. Four patients developed wound sepsis (Accordion Grade 2). One patient received antibiotics for a respiratory infection (Accordion Grade 2). Three patients had Accordion Grade 4 complications. One patient required a laparotomy for postoperative adhesive small bowel obstruction, another had a laparotomy for an infected bile collection and two a laparotomy to control bleeding from the resection margin. One patient died (Accordion Grade 6) following a right hepatic lobectomy for complex multiple bilobar haemangiomas without clear resection margins which bled and required liver packing. She died on day 16 of a severe cerebrovascular accident.
Discussion
The advent of the modern era of safe LR has been facilitated by advances in imaging, improved anaesthetic and surgical techniques and an appreciation of the importance of the functional reserve and the regenerative capacity of the FLR.12-15 Operative techniques, including accurate delineation of the transection planes by selective inflow control or occlusion and intra-operative US are aimed at reducing blood loss and the need for blood transfusion.8,16,17,18 In the present study, 13% of all patients registered in the elective LR database underwent resection for BLTs which is consistent with reports from other centres. Jarnagian et al. reported a LR rate for BLTs of 9% in an analysis of 1803 hepatectomies at Memorial Sloan-Kettering Hospital19 and Poon et al. a LR rate of 15.4% in 1222 hepatectomies,20 while Finch and colleagues reported a LR rate of 22% in a total of 129 resections at The Royal Infirmary in Edinburgh.21
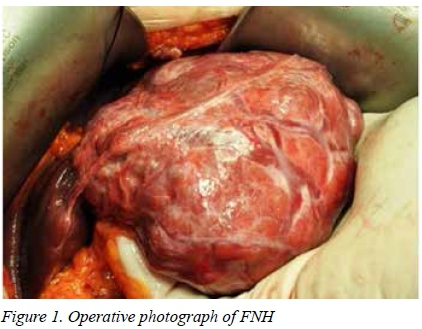
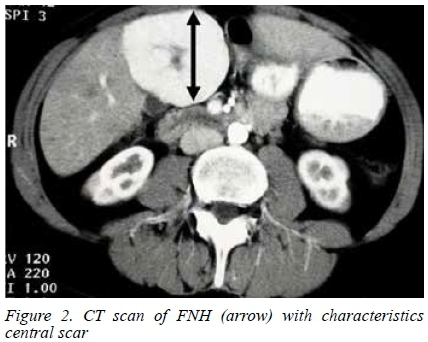
As in other studies our data demonstrate a marked predominance of young women. Both Buell et al.22 and Mizhur and Jarnagan23 report that the most common benign liver lesions resected were haemangiomas, followed by FNH, which is similar to our series. However, in our study BCA exceeded HA which were uncommon. Despite the benign indications a major anatomical LR was required in 40% of patients. Blood loss was limited with less than 10% of our patients requiring a blood transfusion. Complications after LR can be substantial.2 Although patients in our study were relatively young and healthy, 11 (17.7%) developed postoperative complications. The majority of postoperative complications correlated with the extent of LR and not the pathology of the tumour, while minor hepatectomies were associated with a significantly lower risk of postoperative complications.
Haemangiomas are the second most common liver tumours after metastases and account for around 75% of all BLTs.24 Published surgical series are significantly skewed due to selection bias and rates of resection for haemangioma in the literature therefore vary widely, ranging from 3% to 54%.25 There is consensus that haemangioma size does not represent a valid indication for intervention.26 Although non-operative methods of treatment (radiofrequency ablation, radiotherapy, arterial embolization and chemotherapy) have been used in the past, these procedures are now outdated and inappropriate and the only rational treatment, when sound indications exist, is resection.27 The surgical choice is dictated by haemangioma volume and location in relation to major vessels and may require either enucleation or LR using an open or laparoscopic approach, or rarely, liver transplantation. There is general consensus that the only indications for resection are patients with symptoms clearly attributed to the haemangioma.26 Our preferred resection technique is enucleation of the haemangioma in the plane between the lesion capsule and adjacent liver parenchyma to minimise loss of functioning liver, as well as limiting perioperative blood loss and minimising the risk of a bile leak.7 In haemangiomas located centrally or for large tumours near major vascular structures, a formal non-anatomic or anatomic resection may be required.27
FNH was the second most common BLT in this study and occurred predominantly in women. While up to 20% of FNH lack a central scar, bile duct proliferation is consistently present. The previous histological categorization that divided FNH into three subtypes (telangiectatic, mixed, and FNH with cytologic atypia) has since been modified in that telangiectatic FNH is currently regarded as an inflammatory HA subtype.28, 29, 30
The current concept is that FNH develops as a vascular malformation as a result of dysregulation of angiopoietin genes (ANGPT1 and ANGPT2). These cause hyperperfusion and consequently a secondary hyperplastic and regenerative hepatic parenchymal response, mediated by increased expression of vascular endothelial and somatic growth factors that in turn activate hepatic stellate cells.31 Given the low risk of FNH-related complications surgery is recommended only in symptomatic patients. Because FNH is not a premalignant lesion, parenchyma-sparing non-anatomical local resection of the lesion can be performed.
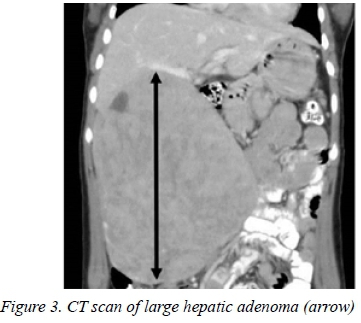
Although HA was the least common tumour in our series, there is evidence that the incidence has increased substantially over the past decades coinciding with the increased use of oral contraception (OC). The new classification based on molecular features divides HA into four subtypes: HNF1A, CTNNB1 (β-catenin), inflammatory, and unclassified.32 This classification is of clinical significance as it identifies the β-catenin subtype which is at higher risk for malignant transformation. MRI has markedly improved diagnosis and can identify the HA subtypes. The indications for surgical resection of HA have been refined during the period covered in this paper, but in general symptomatic patients: HA which increase in size despite discontinuing OC, patients at risk for developing bleeding (tumour size >5 cm) or patients at risk for malignant transformation (men, older women, tumour size >5cm, telangiectatic type inflammatory HA and ß-catenin mutated subtypes). Because of the low risk of vascular invasion and lymph node involvement a wide resection margin and a regional lymphadenectomy are not required. Despite the large size of HA in this series, only one of the four patients required a formal hemihepatectomy (Figure 3).
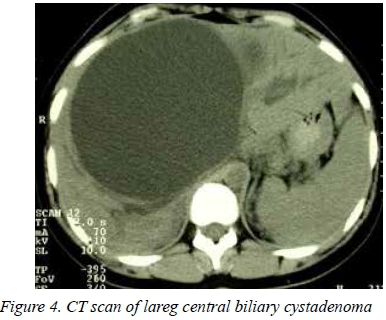
In this series BCA occurred exclusively in women and 38% required a major resection for complete excision. Surgery is indicated even in asymptomatic patients due to the risk of malignant change.33 As BCA and biliary cystadenocarcinoma cannot be reliably differentiated radiologically (Figure 4), formal resection is the treatment of choice. Previous treatments, including marsupialization, internal Roux-en-Y drainage, aspiration or partial resection are oncologically unsound, have high recurrence rates and should not be performed. Pinson et al. have reported cyst enucleation without late recurrence and mortality.34 If, however, attachment of cyst wall to vital structures precludes complete resection, partial cyst resection is an option, provided that complete ablation of the residual cyst surface using argon beam coagulation can be performed.
A suggested algorithm for the management of BLTs in which high-quality and detailed cross-sectional imaging is fundamental, is shown in Figure 5. Modern cross-sectional imaging allowing accurate assessment and diagnosis, combined with improved knowledge of the natural history of specific BLTs can avoid unnecessary resection in many.23. Most authorities concur that surgical intervention in benign disease is indicated in patients with symptoms unresponsive to analgesia, suspicion of or malignancy or those at risk of developing complications or malignancy.35 The risk of complications after any LR should be carefully balanced against the benefits of resection. The relative safety of hepatic resection and the availability of minimally invasive techniques should not dilute the indications for resection of BLTs. Morbidity and mortality rates of LR may be underreported in the literature and should be appreciated, especially considering the benign nature and low risk of complications and malignant transformation of BLTs.
REFERENCES
1. Ibrahim S, Chen CL, Wang SH, Lin CC, Yang CH, Yong CC, Jawan B, Cheng YF. Liver resection for benign liver tumors: indications and outcome. Am J Surg. 2007;193:5-9. doi: 10.1016/j.amjsurg.2006.04.017 [ Links ]
2. Newhook TE, LaPar DJ, Lindberg JM, Bauer TW, Adams RB, Zaydfudim VM. Morbidity and mortality of hepatectomy for benign liver tumors. Am J Surg. 2016;211:102-8. doi: 10.1016/j.amjsurg.2015.06.010 [ Links ]
3. Clarke DL, Currie EJ, Madhavan KK, Parks RW, Garden OJ. Hepatic resection for benign non-cystic liver lesions. HPB (Oxford). 2004;6:115-9. doi: 10.1080/13651820410026326. PMCID: PMC2020664 [ Links ]
4. Couinaud C. Anatomical principles of left and right regulated hepatectomy: technics. J Chir (Paris) 1954;70:933-66. [ Links ]
5. Couinaud C. Liver anatomy: Portal (and suprahepatic) or biliary segmentation. Dig Surg 1999;16(6):459-67. [ Links ]
6. Strasberg SM, Linehan DC, Hawkins WG. The Accordion severity grading system of surgical complications. Ann Surg. 2009;250:177-86. doi: 10.1097/SLA.0b013e3181afde41 [ Links ]
7. Krige JEJ, Beningfield SJ, Bornman PC. Liver and Portal Circulation. In: Burnand K, Young A. Rowlands BJ, Scholfield J, Lucas J (Eds). New AIRD'S Textbook of Surgery. 3rd Edition. Churchill Livingstone London 2005. [ Links ]
8. Terblanche J, Krige JE, Bornman PC. Simplified hepatic resection with the use of prolonged vascular inflow occlusion. Arch Surg. 1991;126:298-301. [ Links ]
9. Bhaijee F, Locketz ML, Krige JE. Fibrolamellar hepatocellular carcinoma at a tertiary centre in South Africa: a case series. S Afr J Surg 2009;47:108-11. [ Links ]
10. Lemmer ER, Krige JE, Hall PM, Bornman PC, Taylor DA, Terblanche J. Surgical resection for hepatocellular carcinoma in Cape Town--a clinical and histopathological study. S Afr Med J. 1998;88:1575-80. [ Links ]
11. Bhaijee F, Krige JE, Locketz ML, Kew MC. Liver resection for non-cirrhotic hepatocellular carcinoma in South African patients. S Afr J Surg. 2011;49:68-74. [ Links ]
12. Krige JE, Thomson SR, Bornman PC. Choosing the optimal tools and techniques for parenchymal liver transection. S Afr J Surg. 2013;51:2-4. doi: 10.7196/sajs.1664 [ Links ]
13. Vollmer CM, Dixon E, Sahajpal A, et al. Water-jet dissection for parenchymal division during hepatectomy. HPB (Oxford) 2006;8:377-85. doi: 10.1080/13651820600839449. PMCID: PMC2020750 [ Links ]
14. Khatri VP, Schneider PD. Liver surgery: Modern concepts and techniques. Surg Clin North Am 2004;84:xv-xvi. [ Links ]
15. Aragon RJ, Solomon NL. Techniques of hepatic resection. J Gastrointest Oncol. 2012;3:28-40. doi: 10.3978/j.issn.2078-6891.2012.006. PMCID: PMC3397635 [ Links ]
16. Poon RT. Current techniques of liver transection. HPB (Oxford) 2007;9:166-73. doi: 10.1080/13651820701216182. PMCID: PMC2063596 [ Links ]
17. Lesurtel M, Selzner M, Petrowsky H, McCormack L, Clavien PA. How should transection of the liver be performed? A prospective randomized study in 100 consecutive patients: Comparing four different transection strategies. Ann Surg 2005;242:814-22. PMCID: PMC1409877 [ Links ]
18. Smyrniotis V, Arkadopoulos N, Kostopanagiotou G, et al. Sharp liver transection versus clamp crushing technique in liver resections: A prospective study. Surgery. 2005;137:306-11. doi: 10.1016/j.surg.2004.09.012 [ Links ]
19. Jarnagin WR, Gonen M, Fong Y, et al. Improvement in perioperative outcome after hepatic resection: Analysis of 1,803 consecutive cases over the past decade. Ann Surg 2002;236:397-406. doi: 10.1097/01.SLA.0000029003.66466.B3. PMCID: MC1422593 [ Links ]
20. Poon RT, Fan ST, Lo CM, Liu CL, Lam CM, Yuen WK, Yeung C, Wong J. Improving perioperative outcome expands the role of hepatectomy in management of benign and malignant hepatobiliary diseases: analysis of 1222 consecutive patients from a prospective database. Ann Surg. 2004;240:698-710.PMCID: PMC1356471 [ Links ]
21. Finch MD, Crosbie JL, Currie E, Garden OJ. An 8-year experience of hepatic resection: indications and outcome. Br J Surg. 1998;85:315-19. doi: 10.1046/j.1365-2168.1998.00585.x [ Links ]
22. Buell JF, Tranchart H, Cannon R, Dagher I. Management of benign hepatic tumors. Surg Clin North Am. 2010;90:719-35. doi: 10.1016/j.suc.2010.04.006 [ Links ]
23. Mezhir JJ, Fourman LT, Do RK, Denton B, Allen PJ, D'Angelica MI, DeMatteo RP, Fong Y, Jarnagin WR. Changes in the management of benign liver tumours: an analysis of 285 patients. HPB (Oxford). 2013;15:156-63. doi: 10.1111/j.1477-2574.2012.00556.x. PMCID: PMC3719923 [ Links ]
24. Toro A, Mahfouz AE, Ardiri A, Malaguarnera M, Malaguarnera G, Loria F, Bertino G, Di Carlo I. What is changing in indications and treatment of hepatic hemangiomas. A review. Ann Hepatol. 2014;13:327-39. [ Links ]
25. Margonis GA, Ejaz A, Spolverato G, Rastegar N, Anders R, Kamel IR, Pawlik TM. Benign solid tumors of the liver: management in the modern era. J Gastrointest Surg. 2015;19:1157-68. doi: 10.1007/s11605-014-2723-x [ Links ]
26. Hoekstra LT, Bieze M, Erdogan D, Roelofs JJ, Beuers UH, van Gulik TM. Management of giant liver hemangiomas: an update. Expert Rev Gastroenterol Hepatol. 2013;7:263-8. doi: 10.1586/egh.13.10 [ Links ]
27. Bornman PC, Terblanche J, Blumgart RL, Jones EP, Pickard H, Kalvaria I. Giant hepatic hemangiomas: diagnostic and therapeutic dilemmas. Surgery. 1987;101:445-9. [ Links ]
28. Marrero JA, Ahn J, Rajender Reddy K; American College of Gastroenterology. ACG clinical guideline: the diagnosis and management of focal liver lesions. Am J Gastroenterol. 2014;109:1328-47. doi: 10.1038/ajg.2014.213 [ Links ]
29. Nguyen BN, Flejou JF, Terris B, Belghiti J, Degott C. Focal nodular hyperplasia of the liver: a comprehensive pathologic study of 305 lesions and recognition of new histologic forms. Am J Surg Pathol. 1999. 23:1441-54. [ Links ]
30. Paradis V, Benzerkri A, Dargere D, Bieche I, Laurendeau I, Vilgrain V, et al. Telengiectatic focal nodular hyperplasia: a variant of hepatocellular adenoma. Gastroenterology. 2004;126(5):1323-9. [ Links ]
31. Sato Y, Harada K, Ikeda H, Fijii T, Sasaki M, Zen Y, Nakanuma Y. Hepatic stellate cells are activated around central scars of focal nodular hyperplasia of the liver-a potential mechanism of central scar formation. Hum Pathol. 2009.40:181-8. doi:10.1016/j.humpath.2008.04.024 [ Links ]
32. Cristiano A, Dietrich A, Spina JC, Ardiles V, de Santibanes E. Focal nodular hyperplasia and hepatic adenoma: current diagnosis and management. Updates Surg. 2014;66:9-21. doi: 10.1007/s13304-013-0222-3 [ Links ]
33. Soares KC, Arnaoutakis DJ, Kamel I, Anders R, Adams RB, Bauer TW, Pawlik TM. Cystic neoplasms of the liver: biliary cystadenoma and cystadenocarcinoma. J Am Coll Surg. 2014;218:119-28. doi: 10.1016/j.jamcollsurg.2013.08.014. MCID: PMC4106371 [ Links ]
34. Pinson CW, Munson JL, Rossi RL, Braasch JW. Enucleation of intrahepatic biliary cystadenomas. Surg Gynecol Obstet1989;168:534-7. [ Links ]
35. Terkivatan T, de Wilt JH, de Man RA, van Rijn RR, Zondervan PE, Tilanus HW, Izermans JN. Indications and long-term outcome of treatment for benign hepatic tumors: a critical apraisal. Arch Surg. 2001;136:1033-8. [ Links ]
 Correspondence:
Correspondence:
Jake Krige
jej.krige@uct.ac.za














