Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Surgery
On-line version ISSN 2078-5151
Print version ISSN 0038-2361
S. Afr. j. surg. vol.53 n.3-4 Cape Town Dec. 2015
GENERAL SURGERY
A closer look at burn injuries and epilepsy in a developing world burn service
M P Faurie; N L Allorto; C Aldous; D L Clarke
Pietermaritzburg Metropolitan Complex, Edendale Hospital, Department of General Surgery, University of Kwa-Zulu Natal, Nelson R Mandela School of Medicine, South Africa
ABSTRACT
BACKGROUND: Burn injuries in South Africa result in significant morbidity and mortality, and specific vulnerable groups of patients are at increased risk of sustaining a burn injury. Epileptic patients are one such vulnerable group. The spectrum of burn injuries sustained by epileptic patients in a South African township and the pattern of injury, mechanism and outcome were reviewed in this study.
METHOD: A retrospective review of all epileptic patients admitted to the burn service of Edendale Hospital was undertaken for the period July 2011 to June 2013.
RESULTS: One hundred and ninety-seven adult patients were admitted with burns over this period. There were 39 epileptic patients in this cohort, of whom 26 were female. The average age of the patients was 36 years (a range of 21-40 years). The majority of patients sustained a small total body surface area burn. The most common mechanism of burn was from a fire or flames, followed by hot water scalding. Coal or wood fires were the predominant energy source used for heating and cooking at home.
CONCLUSION: Epileptics comprise a significant proportion of patients who sustain a burn injury. Typically, they sustain burns during a seizure. These are mostly caused by open flames in the South African environment, and are deep. They tend to be confined to the upper torso, upper limbs and hands. Injury prevention programmes should target epileptics as a vulnerable group.
People with epilepsy are at increased risk of sustaining an accidental injury compared to the general population, and burn injuries are a particular challenge in this population group. This is even more challenging in the developing world.1-6 Edendale Hospital is a regional hospital outside Pietermaritzburg, and serves a large indigent rural and peri-urban population. A high incidence of epileptic patients who sustained a burn injury was reported in a previous review of the spectrum and outcome of burns at our institution.7 A more detailed review of the problem was undertaken in light of this to elucidate the pattern of burn injuries and the mechanism of injury associated with burn injuries in epileptic patients.
Method
Data were taken from the burn admission forms for the 24-month period, starting in July 2011 and ending in June 2013. The data were collated and reviewed. Collected data included the patient's demographic details, and whether or not the patients had had a seizure at the time of the burn. The mechanism of the burn was noted, as were events surrounding the incident, such as the activity in which the patient was engaged at the time of the burn, and the energy source used. The extent, distribution and depth of the burn were also recorded, as well as the treatment required, the number and type of surgeries, and length of hospital stay.
Results
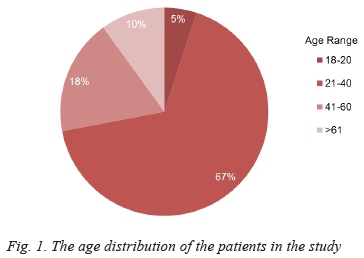
One hundred and ninety-seven adult patients were admitted with a burn injury during the 24-month period under review. Of this cohort, 39 (20%) were epileptics. Two thirds of these epileptic patients were female (26/39), and the majority (67%) were between the ages of 21 and 40 years, as demonstrated in Figure 1.
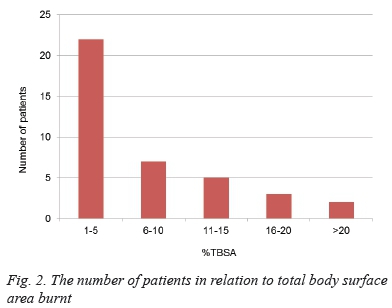
Most patients sustained a small total body surface area (TBSA) burn. Only two sustained burns of more than 20% TBSA. The extent of the burns is illustrated in Figure 2.
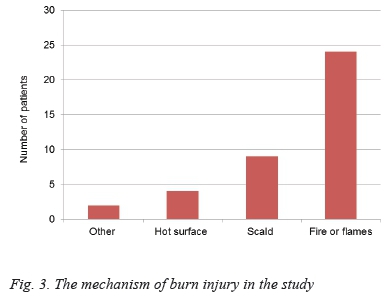
Burns that were larger than 15% TBSA were predominantly due to open flames (83%). Twenty-four patients sustained flame burns, nine hot water scalding, four were burnt by a hot surface, one patient by a chemical burn, and one by hot oil. The mechanism of injury is documented in Figure 3.
Just under half of the patients (19/39) sustained the burn during a seizure. None of burns were sustained at work, but over two thirds (28/39) were sustained at home. Of these patients, five of them were burnt while cooking, eight patients were busy with "other" activities, and three patients were burnt as part of an assault. The energy source used in the home was not well documented in 18 cases. In cases where the energy source was documented, almost two thirds (12/21) used fire as the source of energy for heating and cooking, followed by electricity in six cases. "Other" energy sources were used in three cases. There was a wide variation in the distribution of the burns, but the majority were to the arms and hands. Multiple different sites were burnt in most of the patients, although the TBSA burnt still remained low in the majority of cases. A breakdown of the burnt areas is provided in Table 1. There were no superficial burns. Forty-two per cent were partial-thickness, 29% deep burns and a further 29% a combination of deep- and partial-thickness burns. There was a wide distribution of burnt areas, but the upper limb and hands were by far the most common areas to be burnt (Table 1). Three quarters of these patients required surgical intervention. Most of the patients underwent more than one procedure, i.e. either debridement, split skin grafting or both. The total length of stay ranged from 1-140 days. The average length of stay was 32 days.

Discussion
Chronic disease in sub-Saharan Africa is often poorly controlled for a number of logistical and socio-political reasons. Generally, there is a paucity of research on these conditions, and this is certainly the case with epilepsy.8,9 Almost half of the epileptics in the subcontinent are children who develop epilepsy secondary to adverse perinatal events, and two thirds of seizures begin in childhood.8 Adult-onset epilepsy is mostly secondary to parasitic disease and head trauma.8,9 It is estimated that less than one third of epileptic patients in the region receive appropriate antiepileptic treatment, and the subsequent poor control of seizures exacerbates the risk of accidental injury in these patients.8
In contrast to a previous audit on the spectrum of burns at our institution, in which the incidence of epilepsy-related burn injury was shown to be half of all burn victims admitted to our institution, the current study demonstrated an incidence of 20%.7 While this incidence is lower than that reported in our previous study, it still comprises a fifth of burn admissions. The reason for this discrepancy is unclear, and could be attributed to under-reporting of epilepsy as a co-morbidity in the current database. Previously, all patients who experienced a seizure at the time of the burn were considered to be epileptics, whereas epilepsy was only documented in the current database if the patient had a previous diagnosis of epilepsy.
More female epileptics sustain burns than their male counterparts.3,4,10-12 It is most likely that this relates to the activity at the time of the burn, as most burns were sustained at home.3,4,11-16 Almost a quarter of the female patients in this series were burnt while cooking. Rural women in South Africa are predominantly the homemakers, and thus tend to be at higher risk of being burnt while cooking. The international literature also reports a higher incidence of burns in female epileptics, and the only study to differ reports a male predominance of 56%, and emanated from the American state of Arizona.3,4,11,12 The reasons for this difference are unclear. Just under half of the patients sustained their burns during a seizure. This may reflect both poor compliance and the epileptogenic potential of open flames.3,4,11,12 The majority of the burns in our population group were caused by flames, which contrasts with other studies where hot water scalds predominate.3,6,10-12,15 This can be attributed to the large dependence on open fires in the South African rural population.
Most burns sustained by this group of patients were confined to a small TBSA, which is in keeping with the international literature.10-12,15 These small burns comprise a combination of hot water scalding and flames, possibly due to patients spilling boiling water on themselves, or exposing a body part to flames during a seizure. Burns > 15% TBSA were predominantly due to open flames (83%), and this is probably as a result of the clothing catching fire during the incident. The burns tend to be deep, and this is because of the inability of epileptic patients to withdraw from the heat source during a seizure. Surgical therapy is necessitated by the deep nature of the burns, and this results in a prolonged hospital stay. The average length of hospital stay for epileptic patients who sustain a burn is 32 days, in comparison to the average of 24 days for non-epileptics with similar burns. This is owing to a number of factors, such as the increased need for rehabilitation in patients who are already disabled prior to sustaining a burn.
Conclusion
Epileptic patients were identified as a vulnerable group for burn injury in the South African environment in our study. Female epileptics are at greater risk than males of sustaining such an injury as they are most frequently perform domestic chores, like cooking. The reliance on open flames as an energy source in rural South Africa increases the risk. The majority of burns are sustained during a seizure, and the upper torso, upper limbs and hands are the most frequently affected parts of the body. These injuries are morbid, and may impact on long-term functionality. Further research into the relationship between epilepsy and burn injury is needed to inform burn injury prevention programmes.
Conflicts of interest: None
Declaration: None.
REFERENCES
1. Beghi E. Accidents and injuries in patients with epilepsy. Expert Rev Neurother. 2009;9(2):291-299. [ Links ]
2. Sapna CS, Sheeba SL, John J, Thomas SV. Accidents and injuries in people with epilepsy attending a tertiary care center in India. Epileptic Disord. 2008;10(4):427-481. [ Links ]
3. Ansari Z, Brown K, Carson N. Association of epilepsy and burns - a case control study. Aust Fam Physician. 2008;37(7):584-589. [ Links ]
4. Minn YK. Who burned and how to prevent? Identification of risk for and prevention of burns among epileptic patients. Burns. 2007;33(1):127-128. [ Links ]
5. Wirrell EC. Epilepsy-related injuries. Epilepsia. 2006;47 Suppl 1:70-86. [ Links ]
6. Kinton L, Duncan JS. Frequency, causes and consequences of burns in patients with epilepsy. J Neurol Neurosurg Psychiatry. 1998;65(3):404-405. [ Links ]
7. Allorto NL, Oosthuizen GV, Clarke DL, Muckart DJ. The spectrum and outcome of burns at a regional hospital in South Africa. Burns. 2009:35(7):1004-1008. [ Links ]
8. Kariuki SM, Matuja W, Akpalu A, et al. Clinical features, proximate causes, and consequences of active convulsive epilepsy in Africa. Epilepsia. 2014;55(1):76-85. [ Links ]
9. Ngugi AK, Bottomley C, Kleinschmidt I, et al. Prevalence of active convulsive epilepsy in sub-Saharan Africa and associated risk factors: cross-sectional and case-control studies. Lancet Neurol. 2013;12(3):253-263. [ Links ]
10. Rimmer RB, Bay RC, Foster KN, et al. Thermal injury in patients with seizure disorders: an opportunity for prevention. J Burn Care Res. 2007;28(2):318-323. [ Links ]
11. Jang YC, Lee JW, Han KW, Han TH. Burns in epilepsy: seven years of experience from the Hallym Burn Centre in Korea. J Burn Care Res. 2006; 27(6):877-881. [ Links ]
12. Al-Qattan MM. Burns in epileptics in Saudi Arabia. Burns. 2000;26(6):561-563. [ Links ]
13. Tiamkao S, Shorvon SD. Seizure-related injury in an adult tertiary epilepsy clinic. Hong Kong Med J. 2006;12(4):260-263. [ Links ]
14. Tiamkao S, Amornsin O, Pongchaiyakul C, et al. Siezure-related injuries in northeast Thailand. J Med Assoc Thai. 2006:89(5):608-613. [ Links ]
15. Josty IC, Narayanan V, Dickson WA. Burns in patients with epilepsy: changes in epidemiology and implications for burn treatment and prevention. Epilepsia. 2000;41(4):453-456. [ Links ]
16. Spitz MC, Towbin JA, Shantz D, Adler LE. Risk factors for burns as a consequence of siezures in persons with epilepsy. Epilepsia. 1994;35(4):764-767. [ Links ]
 Correspondence:
Correspondence:
M Faurie
michael.faurie@gmail.com














