Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Surgery
On-line version ISSN 2078-5151
Print version ISSN 0038-2361
S. Afr. j. surg. vol.52 n.4 Cape Town Nov. 2014
http://dx.doi.org/10.7196/sajs.2053
PLASTIC SURGERY
Propeller flaps for lower-limb trauma
A D RogersI; G Dos PassosII
IMB ChB, FC Plast Surg (SA), MMed; Division of Plastic, Reconstructive and Maxillofacial Surgery, Department of Surgery, Groote Schuur Hospital, Cape Town, and Faculty of Health Sciences, University of Cape Town, South Africa
IIMB BCh; Division of Plastic, Reconstructive and Maxillofacial Surgery, Department of Surgery, Groote Schuur Hospital, Cape Town, and Faculty of Health Sciences, University of Cape Town, South Africa
ABSTRACT
The propeller flap has become a versatile and important component in our reconstructive algorithm following complex lower limb trauma. First described by Hyakusoku in 1991, it has since been adapted and modified by Hallock and Teo. This article outlines our experience specifically with perforator pedicled propeller flaps (as per the Tokyo consensus) for traumatic defects of the leg. In this procedure, the reconstructive surgeon skeletonises a single perforator and rotates the skin island on its axis between 90o and 180o to close the defect. The minor blade of the propeller may be designed to close the donor defect completely for the 180o version. The propeller flap has the advantages of local flaps (reliability, contour, texture, 'like-with-like') with additional versatility of design and donor site management, and requires minimal expertise and operative time.
Plastic surgeons are frequently called upon to provide definitive soft-tissue coverage for lower limb defects associated with compound fractures of the tibia or fibula (Gustillo-Anderson IIIB). Usually defects of the upper and middle thirds of the leg are easily managed with either gastrocnemius myocutaneous flaps or hemi-soleus muscle coverage and skin grafting (Fig. 1).
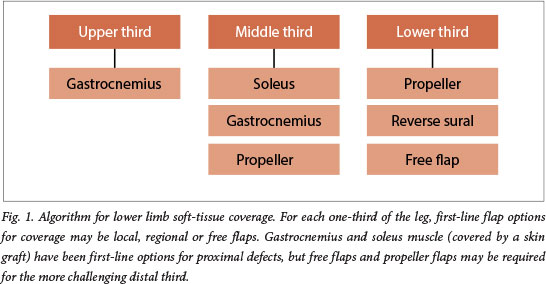
Owing to limited laxity in the lower third, management of defects in this area has been more challenging. Historically even cross-leg flaps, with concomitant staging and the requisite immobility, have sometimes been required. More recently, microsurgical free flaps have featured early on reconstructive algorithms. Free flaps offer versatile and sufficient coverage without the limitations of the pedicle, and elevation and anastomosis can occur well beyond the zone of injury. Unfortunately, as a result of limited operating time and available consultant microsurgeons, only one free flap had been performed for this indication in the Groote Schuur Hospital complex (Cape Town, South Africa) over the 18 months leading up to the time of writing. Registrars have approached these challenges either by making use of negative-pressure dressings or expanding their armamentarium of local flaps. The most useful of these options has undoubtedly been the propeller flap.
The propeller flap has become a versatile and important component in our reconstructive algorithm following complex lower limb trauma. First described by Hyakusoku in 1991, it has since been adapted and modified by Hallock and Teo.[1-3] This article outlines our experience specifically with perforator pedicled propeller flaps (as per the Tokyo consensus[4]) for traumatic defects of the leg.
Methods and technique
This was a prospective study of use of the propeller flap for coverage of lower limb compound fractures over an 18-month period. Demographic details, operative details and outcomes were recorded.
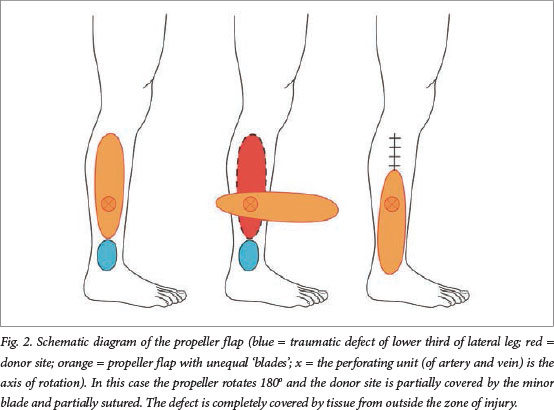
The propeller flap is an islanded fasciocutaneous flap based on a single dissected perforator. The most useful perforators arise from either the posterior tibial artery or the peroneal vessels. The propeller flap has two unequal blades with the perforator forming the pivot point so that when the blades are switched, the long arm fills in the defect. There is minimal tissue laxity in the lower leg, so a proximally based flap may expose the Achilles tendon or the tibia. Flaps therefore often need to be based distally. The propeller flap, pivoted on a single perforator, avoids these problems by importing undamaged tissue from the proximal calf into the primary defect. It transfers the secondary defect to an easily graftable area over the proximal muscle bellies, but frequently enables the surgeon to close the defect primarily (Fig. 2).
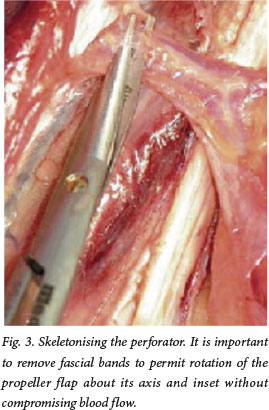
Unlike in other flaps, two pedicles are not better than one in a propeller flap. If there are two pedicles there is likely to be the problem of kinking or twisting, particularly if the propeller needs to be rotated more than 90o. Making the propeller a full island removes the dogears, improves the contour and gives the flap more freedom to pivot and move into the defect. In addition, skeletonising the perforator (Fig. 3) under 4.5 times loupe magnification avoids the compression of fascial strands when the flap is rotated and inset.[1-4]
Use of Doppler ultrasound may be helpful in identifying the most promising perforator artery near the defect. A provisional flap design can then be drawn with the perforator as the pivot point of the flap. The distance between the perforator and the distal edge of the defect is measured; this value is then transposed proximally along the axis of the main source vessel, again measured from the perforator, and 1 cm is added, which would be the proximal limit of the flap. Measuring the width of the defect, adding 1 cm, and then measuring equal distances on each side of the perforator determine the width of the proximal flap needed to cover the defect.[2-3]
The subfascial approach is generally our preferred method to visualise the pedicle, which is selected based on position and calibre, and the flap design is adjusted accordingly. We avoid any perforator in scar tissue, as injury is more likely and dissection more difficult. The perforator should also be fairly near the defect to reduce flap size. The surgeon should follow the perforator and free it back to the source vessel (often 3 cm or more) to allow a gradual twist when the flap is rotated through 180°. Any fascial strands that may compress the perforating artery or vein (once twisted) should be meticulously divided (Fig. 3). Vasodilators such as calcium channel blockers, lignocaine or papaverine may be used to improve the flow through the flap, and a tourniquet may facilitate dissection, although our use of these has been inconsistent.
Once good perfusion is observed, the flap may be moved into the defect. The maximum rotation is 180°, either clockwise or anti-clockwise, whichever direction places the pedicle under less tension. A skin graft may be required to cover the donor area.
Case series
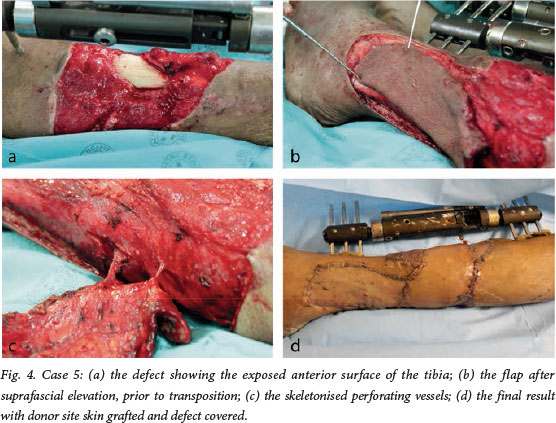
Seven patients underwent surgery for coverage of distal third tibia and/or fibula bones associated with fractures (Gustillo-Anderson IIB defects). Table 1 demonstrates the salient features of these patients and their management. Case 5 is illustrated in Fig. 4. During this procedure, the flap was rotated 130o when inset.
The mean age of the patients was 28.9 years, and all but one was male. The mean defect size was 20.75 cm2. The mean time from injury to surgery was 13 days, and in most cases interval negative-pressure dressings were used prior to definitive cover.
All the wounds were successfully covered using propeller flaps. One patient had partial distal necrosis, which healed with conservative management using negative-pressure dressings. The mean duration of follow-up was 1.7 months.
Conclusion
The perforator pedicled propeller flap is a useful and versatile option for coverage of compound lower limb defects, where the alternative means of coverage would have been a free flap.
The skeletonised perforator enables skin paddle rotation between 90o and 180o. The minor blade may be designed to close the donor site in the 180o version. The propeller flap has the advantage of local flaps (reliability, texture and contour), avoids donor morbidity and risk of free flaps in the subacute phase or in the zone of injury, but also improves versatility by being islanded. Propeller flaps require careful planning, but minimal expertise and operative time.
Acknowledgement. This paper was presented at the annual meeting of the Association of Plastic and Reconstructive Surgeons of Southern Africa (APRSSA) in October 2013.
REFERENCES
1. Hyakusoku H, Yamamoto T, Fumiiri M. The propeller flap method. Br J Plast Surg 1991;44(1):53-54. [http://dx.doi.org/10.1016/0007-1226(91)90179-N] [ Links ]
2. Teo TC. The propeller flap concept. Clin Plast Surg 2010;37(4):615-626. [http://dx.doi.org/10.1016/j.cps.2010.06.003] [ Links ]
3. Teo TC. Perforator local laps in lower limb reconstruction. Cirugía Plástica Ibero-Latinoamericana 2006;32:15-16. [ Links ]
4. Pignatti M, Ogawa R, Hallock GG, et al. The 'Tokyo' Consensus on propeller l aps. Plast Reconstr Surg 2011;127(2):716-722. [http://dx.doi.org/10.1097/PRS.0b013e3181fed6b2] [ Links ]
 Correspondence:
Correspondence:
A D Rogers
(rogersadr@gmail.com)














