Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Surgery
On-line version ISSN 2078-5151
Print version ISSN 0038-2361
S. Afr. j. surg. vol.52 n.3 Cape Town Aug. 2014
http://dx.doi.org/10.7196/sajs.1954
GENERAL SURGERY
Intraoperative examination of sentinel lymph nodes using scrape cytology
A MannellI; B WiumII; C ThatcherIII
IFRACS, FRCS (Lond), MS; Department of Surgery, University of the Witwatersrand, Johannesburg, South Africa
IIBSc (Med) (Hons), MB BCh, FCPath (SA); Drs Gritzman & Thatcher Inc. Laboratory, 4 Main Street, Bordeaux, Johannesburg, South Africa
IIIMB BCh, FCPath (SA); Drs Gritzman & Thatcher Inc. Laboratory, 4 Main Street, Bordeaux, Johannesburg, South Africa
ABSTRACT
BACKGROUND: In breast cancer, sentinel lymph node biopsy (SLNB) is widely used to assess the axilla when the nodes appear normal on palpation and ultrasonography. When the sentinel lymph nodes (SLNs) are negative, no further dissection is required. Surgical dissection or radiotherapy of the axilla is indicated for macrometastases, as well as adjuvant therapy for macrometastases and micrometastasic spread.
AIM: To determine the accuracy of scrape cytology (SC) for intraoperative examination of the SLNs.
METHODS: SLNB was performed in 100 patients with early breast cancer in whom the axillary nodes appeared normal on clinical examination and ultrasonography. None of the patients with negative SLNs or SLNs with micrometastases only (N1mic, 0.2 - 2 mm) had further axillary dissection. Patients with SLNs containing macrometastases (N1, >2 mm) underwent axillary lymph node dissection. The results of intraoperative cytology were compared with the histopathological findings on examination of serial paraffin-embedded sections of the SLNs.
RESULTS: Intraoperative SC identified SLN metastases in 20 patients: 17 had macrometastases and 3 micrometastases. Histopathological examination of the SLN s found macrometastases in 18 patients and micrometastases in 6. SC had a sensitivity of 94% and specificity of 100% for the detection of macrometastases. For micrometastatic spread, the sensitivity and specificity were 50% and 100%, respectively.
CONCLUSION: SC is a rapid, sensitive technique for detection of macrometastases in the SLNs of women with breast cancer. The overall accuracy in identification of any metastatic spread (N1mic , N1) to the SLNs was 85%, rising to 94% in SLNs with macrometastases.
In patients with breast cancer, the presence of lymphatic metastases is one of the most significant factors affecting prognosis and indicates the need for adjuvant therapy to reduce risk of locoregional recurrence and distant disease.[1] Sentinel lymph node biopsy (SLNB) is a widely used technique for the intraoperative examination of axillary lymph nodes when these appear normal on clinical examination and on ultrasonography but may still contain metastases.[2,3] The finding of lymph node metastases can be followed by axillary lymph node dissection (ALND) for local control at the same time as definitive breast surgery.[3-5] Conversely, when the sentinel lymph nodes (SLNs) are free of cancer, ALND is not indicated and the risk of complications such as seroma, infection, pain and lymphoedema is significantly reduced.
Various methods have been used for intraoperative analysis of SLNs, including frozen section (FS), touch imprint cytology (TIC), scrape/scrimp cytology (SC) and immunohistochemistry (IHC). FS is a well-known method but requires an expensive fixed cryostat, a microtome and cylinders of carbon dioxide for freezing. This equipment is not easily portable and may not be available in small centres. Both TIC and SC need only glass slides, bottles of haematoxylin and eosin (H&E) stains, alcohol fixative and a microscope. Where FS equipment is available in the operating theatre complex, FS analysis can be performed in 15 minutes. TIC and SC are even more rapid methods, completed in 5 - 6 minutes, and since the cellular yield by scraping is higher than that with touch imprints, SC[6-8] has been the preferred method for intraoperative examination of the SLNs. IHC has the major disadvantage of requiring at least an hour for incubation of tissue and the antibody complex.
Aim
To assess the sensitivity, specificity[9] and accuracy of SC in intraoperative examination of the SLNs. The intraoperative results were compared with the definitive histopathological evaluation of paraffin-embedded serial sections of the SLNs.
Methods
This prospective study comprised 101 breast cancer patients with clinically normal and ultrasonographically negative axillae treated from 2001 to 2006. All had early breast cancer (stages 0, I or II) of ductal origin (Table 1). No patient had received neoadjuvant hormonal treatment or chemotherapy. The SLNs were identified using radioscintigraphy and Patent Blue V dye. On the morning of the operation, one millicurie of 99m Tc-labelled nanocolloid was routinely given as an areolar intradermal and peritumoural injection in the nuclear medicine facilities at Linksfield Park or Rosebank private clinics in Johannesburg, where the operative procedures were also performed. At operation 2 ml of reconstituted Patent Blue V dye was injected into the areolar dermis after induction of anaesthesia. The results of the radioscintigraphic study were scrutinised and the ipsilateral axilla then explored through an appropriately placed small incision before the definitive breast surgery. A lymph node was recognised as an SLN when it was stained blue, had a blue afferent lymphatic and/or had a radioactive count measured by the gamma probe to be >10% of the highest count at the radiocolloid injection site. The SLNs were excised and bivalved along their long axes. The cut surfaces were gently scraped with a scalpel blade onto glass slides which were fixed with 95% alcohol and stained with H&E. Smear preparations were made, which were then fixed in 95% alcohol and stained with H&E.

None of the patients with negative SLNs or SLNs with micrometastases only (N , 0.2 - 2 mm diameter)[10] underwent a completion ALND. Patients with SLNs containing macrometastases (N1, >2 mm diameter)[10] underwent ALND. The one patient with macrometastases in the SLN and in whom SC was falsely negative underwent delayed ALND.
Results
SLNs were located in 100 of the 101 patients (identification rate 99.0%). The single patient in whom both radioscintigraphy and blue dye failed to identify the SLN underwent ALND. The mean age of the study group was 53 years (Table 1). Of the patients, 92 had invasive ductal carcinoma of the breast and 8 an area of ductal carcinoma in situ (DCIS).
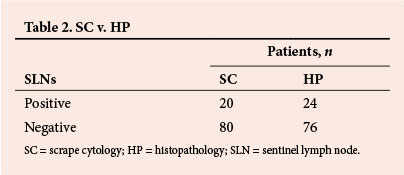
Intraoperative SC identified SLN metastases in 20 patients, 17 of whom had macrometastases and 3 micrometastases. Histopathological examination of the SLN paraffin-embedded sections revealed that 18 patients had macrometastatic spread to the SLNs and 6 had micrometastases in the SLNs (Table 2). The intraoperative diagnoses of lymph node metastases were correct in 96 patients, giving SC a sensitivity and specificity of 83% and 100%, respectively. The positive predictive value (PPV) and negative predictive value (NPV) were 100% and 95%, respectively. SC for macrometastatic spread had a sensitivity of 94% and a specificity of 100% (PPV 100%, NPV 99%). For micrometastases, sensitivity, specificity, PPV and NPV were 50%, 100%, 100% and 96%, respectively (Table 3).

Discussion
Intraoperative examination of SLNs is a widely accepted technique used to assess the clinically node-negative axilla in breast cancer patients, and various methods are in use. These include FS, TIC, SC and IHC.[5-8,11-13]
In this study SC proved to be a reliable method to identify SLN macrometastases, with a sensitivity of 94% and a specificity of 100%. FS has a similar sensitivity and specificity and is widely used, but requires expensive fixed equipment, results in some loss of tissue, and can lead to tissue artefacts during freezing.[12-14] The technique of TIC involves touching the cut surfaces of the SLN onto glass slides, fixing and staining. This method is quicker than FS, requires only simple materials and does not destroy tissue, which is then available for further analysis.[15] However, the sensitivity of TIC varies considerably, ranging from 47% to 75%.[11] A recent series found that TIC had a sensitivity of 34% for macrometastases.[14] By scraping the cut surfaces of the SLN onto glass slides, SC combines the ease and rapidity of TIC with an improved cellular yield.[6] This has resulted in an improved sensitivity for macrometastases.[17,8] Both methods require pathologists with cytology training.
When time under anaesthesia and expense are considerations, IHC is a technique of limited use for intraoperative analysis of the SLNs[14] and is most appropriately performed in the laboratory on paraffin sections. It is, however, of particular value when metastatic lobular carcinoma is suspected, and to detect isolated tumour cells.[16]
When the SLNs are normal, there is no indication for further dissection in the axilla and the complications of ALND, including pain, seroma formation, infection, disabling lymphoedema and shoulder stiffness, are minimised. However, when macrometastases are found in the SLNs, additional treatment for the axilla must be considered. The standard method of management of the positive axilla is ALND to remove all macroscopic disease.[3] This helps to achieve local control and when done at the same time as definitive breast surgery spares the patient the expense and anxiety of a second operation.[3,15]
To avoid the complications of ALND, other methods of management of the positive axilla have evolved. These include radiation to the axilla as part of breast-conserving surgery (BCS) or following mastectomy.
BCS entails wide local excison of the cancer followed by whole-breast irradiation, for which tangential fields are commonly used. This results in radiation to axillary levels I and part of II.[3] It is likely that radiation to the axilla contributed to the results of the American College of Surgeons Oncology Group Z0011 randomised controlled trial.[17] In this study women with small breast cancers and clinically negative axillae underwent BCS. When only one or two SLNs were found to be positive, the patients were then randomised to receive ALND or no further surgery to the axilla. Almost all women in both groups received adjuvant chemotherapy and/or hormonal manipulation. Not surprisingly, there was no significant difference in locoregional recurrence rates or overall survival between the groups after a 5-year follow-up.[17, 18] The results of long-term follow-up of these patients are awaited, as are the results of similar studies which include patients with a low volume of axillary disease undergoing mastectomy without radiation. The After the Mapping of the Axilla: Radiotherapy or Surgery (AMAROS) trial[19] has compared ALND and axillary radiation (AR) when the SLNs were positive. The most recent analysis of the AMAROS trial has shown that both modalities were effective for local control of the axilla with a small volume of metastatic disease.[19] AR resulted in fewer cases of lymphoedema than in patients undergoing ALND, although patients in the ALND group suffered less restriction of shoulder movement.
There is little debate over whether women with clinically suspect or matted axillary nodes and those with macrometastases present in three or more SLNs require definitive treatment of the axilla. This necessitates ALND and/or complete AR to achieve local control, as well as adjuvant systemic therapy to reduce the risk of distant relapse. However, management of the axilla when only micrometastases are identified in the SLNs remains controversial. Several studies have evaluated the outcome in patients who did not undergo completion ALND for micrometastatic spread, and found that none of them suffered an axillary recurrence.[20,21] SC in the current per-patient analysis of SLN was found to have a reduced sensitivity for micrometastases (N1mic) (Table 3) of 50%, in keeping with that of other methods of intraoperative SLN examination. In this series, ALND was not performed either at the initial operation or as a delayed procedure when SLNs were found to be N1mic only. Although it is unlikely that the axilla requires local treatment for N , several series[21,22] have reported worse overall survival in patients with micrometastatic disease[12] if no adjuvant systemic treatment is given. In a population of over 10 000 breast cancer patients with long-term follow-up, Kuijt et al.l23] identified 4.1% of cases with micrometastases. The 10-year survival of this group was 56.1% compared with 69.7% in patients with node-negative breast cancer. These findings support those of Tan et al.,l24] who demonstrated that micrometastases to the axillary nodes had a significant effect on 15-year disease-free survival.
In the current study there were 20 patients with SLN micrometastases, all of whom received adjuvant systemic therapy. Follow-up data are incomplete, but longer-term studiesl22,23] suggest that, irrespective of the size of the lymph node metastasis,l22-24] when lymphatic spread has occurred the patient is at increased risk of distant disease and systemic adjuvant therapy is mandatory.
Conclusion
SC is a rapid, simple and sensitive technique for intraoperative examination of the SLNs. Requiring no expensive fixed equipment, it is particularly appropriate for a resource-constrained environment.
In this study the overall accuracy in identification of any metastatic spread (N1mic , N1) to the SLNs was 85%, rising to 94% in SLNs with macrometastases. This allows the surgeon to proceed to ALND at the same time as definitive breast surgery, if this is the method selected to manage the positive axilla. In addition, the prompt referral[25,26] of patients for adjuvant systemic therapy is facilitated.
Acknowledgements. The data in this article formed part of a presentation by the authors to the Breast Cancer Forum, Durban, South Africa, in October 2011. Approval from the Human Research Ethics Committee (University of the Witwatersrand) was obtained for this study (clearance certificate No. M140238).
REFERENCES
1. Carter C, Allen C, Heeson D. Relation of tumour size, lymph node status and survival in 24,740 breast cancer cases. Cancer 1989;63(1):181-187. [http://dx.doi.org/10.1002/1097-0142(19890101)63:1<181::AID-CNCR2820630129>3.0.CO;2-H] [ Links ]
2. Carlson GW, Wood WC. Management of axillary lymph node metastasis in breast cancer. JAMA 2011;305(6):606-607. [http://dx.doi.org/10.1001/jama.2011.131] [ Links ]
3. Morris T, Weitzig N, Sinclair S, et al. Evaluation of implementation of sentinel node biopsy in Australia. Aust N Z J Surg 2012;82(7/8):541-547. [http://dx.doi.org/10.1111/5.1445-2197,2012.06111.X] [ Links ]
4. Yeoh EK, Denham JW, Davies SA, Spittle MF. Primary breast cancer complications of axillary management. Acta Radiol Oncol 1986;25(2):105-108. [ Links ]
5. Leong Chong CY, Mirbagheri N, Harris M, Fox J. Sentinel node micrometastases in breast cancer: A survey of Australia and New Zealand breast surgeons. Aust N Z J Surg 2012;82(1):766-767. [http://dx.doi.org/10.1111/J.1445-2197.2012.06763.X] [ Links ]
6. Owings M. Rapid cytologic examination of surgical specimens: A valuable technique in the surgical pathology laboratory. Hum Pathol 1984;15(7):605-664. [http://dx.doi.org/10.1016/S0046-8177(84)80283-X] [ Links ]
7. Smidt ML, Besseling R, Wanters CAP, Strobbe LJA. Intraoperative scrape cytology of the sentinel lymph node in patients with breast cancer. Br J Surg 2002;89(10):1290-1293. [http://dx.doi.org/10.1046/J.1365-2168.2002.07189.X] [ Links ]
8. Teal CB, Tabbara J, Kelly TA. Evaluation of intraoperative scrape cytology for sentinel lymph node biopsy in patients with breast cancer. Breast J 2007;13(2):155-157. [http://dx.doi.org/10.1111/j.1524-4741.2007.00392.x] [ Links ]
9. Sunny JK. http://www.josephsunny.com/medsoft/sensitivity_specificity.html (accessed 13 July 2011). [ Links ]
10. Edge SB, Bynd DR, Compton CC, et al., eds. AJCC Cancer Staging Handbook. 7th ed. New York: Springer, 2010. [ Links ]
11. Tew K, Irwig L, Mathews A, et al. Meta-analysis of sentinel node imprint cytology in breast cancer. Br J Surg 2005;92(9):1068-1080. [http://dx.doi.org/10.1002/BJSK.5139] [ Links ]
12. Mori M, Twada K, Ikenaga M, et al. Frozen section is superior to imprint cytology for the intraoperative assessment of sentinel lymph node metastasis in stage 1 breast cancer patients. World J Surg Oncol 2006;4:26-32. [http://dx.doi.org/10.1186/1477-7819-4-26] [ Links ]
13. Safai A, Razeghi A, Monabathi A, Azarpira N, Talei A. Comparing touch imprint cytology, frozen section analysis and cytokeratin immunostaining for intraoperative evaluation of axillary sentinel nodes in breast cancer. Indian J Pathology 2012;55(2):183-186. [http://dx.doi.org/10.4103/0377-4929.97859] [ Links ]
14. Richards ADM, Lakhoni SR, James DT, Ung OA. Intraoperative imprint cytology for breast cancer sentinel nodes: Is it worth it? Aust N Z J Surg 2013;83(7/8):539-544. [http://dx.doi.org/10.1111/J.1445-2197.06293.X] [ Links ]
15. Krishnamurthy S, Meric-Bernstan F, Lucci A, et al. A prospective study comparing touch imprint cytology, frozen section analysis and rapid cytokeratin immunostain for intraoperative evaluation of axillary sentinel lymph nodes in breast cancer. Cancer 2009;115(7):1553-1562. [http://dx.doi.org/10.1002/cncr.24182. [ Links ]
16. Weinberg ES, Dickson D, Whit L, et al. Cytokeratin staining for intraoperative evaluation of sentinel lymph nodes in patients with invasive lobular carcinoma. Am J Surg 2004;188(4):419-422. [http://dx.doi.org/10.1016/J.AMJSURG.2004.06.019] [ Links ]
17. Giuliano AE, Hunt KK, Ballman KV, et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis. JAMA 2011;305(6):569-575 [http://dx.doi.org/10.1001/JAMA.2011.90] [ Links ]
18. Straver ME, Meijnen P, van Tienhoven G, et al. Role of axillary clearance after a tumour-positive sentinel in the administration of adjuvant therapy in early breast cancer. J Clin Oncol 2010;28(5):731-737. [http://dx.doi.org/10.1200/JCO.2008.21.7554] EPUB.2009DRC28] [ Links ]
19. Rutgens EJT, Meijnen P, Bonnefoi H. Clinical trials update of the European Organization for Research and Treatment of Cancer Breast Cancer Group. Breast Cancer Res 2004;6(4):165-169. [http://dx.doi.org/10.1186/bcr 906] [ Links ]
20. Damie S, Teal CB. Can axillary lymph node dissection be safely omitted for early-stage breast cancer patients with sentinel lymph node micrometastases? Ann Surg Oncol 2009;16(12):3215-3216. [http://dx.doi.org/10.1245/s10434-009-0702-3] [ Links ]
21. Pernas S, Benilez GM, Bazen MT, et al. Avoiding axillary treatment in sentinel lymph node micrometastases of breast cancer: A prospective analysis of axillary or distant recurrence. Ann Surg Oncol 2010;17(3):772-777. [http://dx.doi.org/10.1245/S10434-009/0804/Y] [ Links ]
22. Pepels MJ, de Boer M, Bult P, et al. Regional recurrence in breast cancer patients with sentinel node micrometastases and isolated tumour cells. Ann Surg 2013;255(1):116-121. [http://dx.doi.org/10.1097/SLA.06013e3182dc616] [ Links ]
23. Kuijt GP, Voogd AC, van de Poll-France L, et al. The prognostic significance of axillary lymph node micrometastases in breast cancer patients. Eur J Surg Oncol 2005;31(5):500-505. [http://dx.doi.org/10.1016/J.EJSO.2005.01.001] [ Links ]
24. Tan LK, Giri D, Hummer AJ, et al. Occult axillary node metastases in breast cancer are prognostically significant: Results in 368 node-negative patients with 20-year follow-up. J Clin Oncol 2008;10;26(11):1803-1809. [http://dx.doi.org/10.1200/JCO.2007.12.6425.EPUB2008.MAR10] [ Links ]
25. Fox PW, Chatfeld MD, Beith JM, et al. Factors delaying chemotherapy for breast cancer in four urban and rural oncology units. Aust N Z J Surg 2013;83(7/8):533-538. [http://dx.doi.org/10.1111/J.1445-2197.2012.06254X] [ Links ]
26. Biagi JJ, Rapheal M, King WD, et al. The effect of delay in time to adjuvant chemotherapy on survival in breast cancer: A systematic review and meta-analysis (abstract). J Clin Oncol 2011;29(15, Suppl):1128. [ Links ]
 Correspondence:
Correspondence:
A Mannell
(mannell@mweb.co.za)














