Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Surgery
On-line version ISSN 2078-5151
Print version ISSN 0038-2361
S. Afr. j. surg. vol.51 n.3 Cape Town Jan. 2013
PAEDIATRIC SURGERY
The use of diverting colostomies in paediatric peri-anal burns: Experience in 45 patients
C E PriceI; S CoxII; H RodeIII
IMB ChB, Department of Plastic and Reconstructive Surgery, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town, South Africa
IIMB ChB, FCS (SA), Department of Paediatric Surgery, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town, South Africa
IIIMB ChB, MMed (Surg), FCS (SA), FRCS (Ed), Department of Paediatric Surgery, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town, South Africa
ABSTRACT
BACKGROUND: Peri-anal burns are common in children. Continual exposure to enteric organisms may lead to a contaminated burn wound, invasive sepsis, graft loss, scar contracture, anal and urinary malfunction, and delayed discharge from hospital. Use of a temporary diverting colostomy has been advocated to reduce complications.
OBJECTIVES: To review the incidence, indications, methods, bacteriology, therapeutic effects and outcomes of colostomy for peri-anal burns.
METHODS: A prospective study of children with peri-anal burns and stomas over a 17-year period. Prophylactic or therapeutic faecal diversion was achieved by temporary, divided sigmoid end-colostomy with Hartmann's closure of the distal end.
RESULTS: Between 1995 and 2012, 45 children with peri-anal burns received a colostomy. The mean total body surface area burned was 35% (range 3 - 80%). There were 28 flame burns, 16 fluid burns and 1 contact burn. Prophylactic colostomies were performed in 29 children, on average on day 6 after admission, and therapeutic colostomies to counteract deep wound infection and septicaemia in 16 patients, on average on day 24. In all but 2 cases there was a marked improvement in clinical appearance, graft take and healing. The bacterial profile changed from gut-derived organisms to Pseudomonas aeruginosa or no pathogens. Complications occurred in 5 patients (11.1%). Three stomas required manual reduction. Two children died of established septic shock, compounded by stomal dehiscence in 1 case. Reversal of the colostomy was performed on average at 4 months.
CONCLUSION: Diverting colostomy has therapeutic advantages in a select group of paediatric burns patients in whom continual faecal soiling poses a threat to both graft and life.
Burns to the gluteal, perineal and upper thigh areas are common in paediatric patients.[1,2] Infants in diapers invariably have soiling of these areas with Gram-negative enteric organisms. Continual exposure may lead to a colonised burn wound or invasive burn-wound sepsis, which may be difficult to treat. Enteral feeding, the preferred method in the infant, may further compound the situation owing to increased stool volumes. Wound care is technically problematic because of anatomical location, the three-dimensionality of this area, the proximity to the anus and difficulty in controlling faecal contamination. Inadequate management of burn wounds in these areas may therefore result in an increased incidence of invasive burn-wound sepsis, graft loss, scar contracture, and anal and urinary malfunction and can delay hospital discharge, increasing both the complexity and the cost of burn care.[3-6] The use of temporary diverting colostomies for a highly select group of children has been advocated by various authors to reduce the incidence of these complications[1,6] The fashioning and subsequent closure of colostomies is not without complications, which include necrosis, bleeding, prolapse or retraction, parastomal hernias, anastamotic breakdown, skin excoriation, wound sepsis and adhesive bowel obstruction and have a documented incidence of 4 - 31% in patients with primary bowel pathology such as anorectal malformations, Hirschsprung's disease, necrotising enterocolitis, trauma and neoplasms.[7-9] Transverse colostomies are more prone to complications than those sited in the left colon.[8] Meticulous technique is essential in keeping complications to a minimum, and experience on the part of the surgeon is essential.
Objective
The aim of this study was to report on our ongoing experience with stoma formation in children who have suffered severe peri-anal, perineal, buttock and upper-thigh burns. The indications for colostomy and the effect of diversion of the faecal stream on the bacterial profile of the burn wounds, wound healing, graft take and long-term complications were reviewed. We postulated that when performed as an elective procedure in the presence of a healthy bowel the complication rate would be lower than when stomas are fashioned for colorectal pathology.
Methods
A prospective study of children who had sustained peri-anal, perineal, gluteal and upper thigh burns was conducted over a 17-year period (1995 - 2012). For clarity, these injuries will all be labelled peri-anal burns. These burns were rarely isolated and in most cases were part of a more extensive thermal injury.
Patients were selected for the study when the decision to create a stoma was made. All details relating to the burn wounds, bacteriology and operative procedures were documented and later analysed. Standard protocols for admission and burn wound management were followed.[10] Burn wounds were superficially debrided when necessary, showered clean and dressed using modern burn wound management techniques.[11] Initial daily application of silver sulfadiazine (Flamazine) or Acticoat (Smith & Nephew, Hull, UK) was done and subsequent topical therapy was dictated by bacteriological profiles of the wounds.
Urinary catheters were passed either as part of the resuscitation phase of management or when there were significant burns of the penis or labia. No urinary catheter was passed if the child could void and a catheter was not required in the course of resuscitation.
Methods to prevent passage of frequent or loose stools included the use of a polymeric diet and constipating agents. Dressings were changed whenever they were soiled. The burn wound was inspected daily for the presence of stool staining (green-brown discoloration of the eschar) and clinical deterioration of the burn. Bacterial surveillance was done by means of semi-quantitative surface swabs performed weekly using the Levine technique.[12]
Partial-thickness burns of the external genitalia, perineum and gluteal and upper thigh areas were allowed to heal spontaneously. Full-thickness gluteal and thigh burns were managed with early escharectomy and autografting. Equivocal burns were biopsied to confirm burn depth prior to excision.
In all but 1 patient the method of faecal diversion was a temporary, divided sigmoid end-colostomy with Hartmann's closure of the distal end. This was performed as part of the debridement and skin grafting procedure, or in a separate setting. The distal rectosigmoid loop was lavaged clean at the time of fashioning the colostomy. Colostomy was performed either prophylactically in children with severe, deep partial- or full-thickness burns of the peri-anal area to prevent soiling of the wound, as well as in those with loose frequent stools, or therapeutically as part of the management where continual faecal soiling had led to progressive wound or systemic sepsis, repeated graft loss or donor site infections despite maximal local and enteral care.
The colostomy was preferably sited in an unburnt area in the left iliac fossa, or if this was not possible, in an area that had previously been grafted. Stomal closure was performed when the burn injury had healed completely and after any perineal reconstructive procedures had been completed. The methods employed to prevent peri-anal complications included early ambulation, splinting of the legs in maximal abduction and active and passive physical therapy.
Outpatient follow-up in this group was done in the general surgery, burn or skin clinics and was continued until no further interventions or scar manipulation were required.
Results
Between 1995 and 2012, 14 110 children were admitted to the Burns Unit at Red Cross War Memorial Children's Hospital, Cape Town, South Africa. Of these patients only 45 underwent colostomies for severe peri-anal, perineal, buttock and upper thigh burns (0.3%). Their mean age was 4.6 years (range newborn to 13 years). The mean total body surface area (TBSA) burned was 36% (range 3 - 86% TBSA). Twenty-eight patients sustained flame burns, 15 hot-water burns or scalds, 1 a contact burn, and 1 rectal and peri-anal burns due to a hot-water enema.
Prophylactic colostomies were performed in 29 children, on average on day 6 after admission (range 1 - 25 days). Therapeutic colostomies were performed in 16 patients, on average at day 24 (range 5 - 74 days). The preferred site for the colostomies was the left lower abdomen. Two sigmoid colostomies were sited in the suprapubic region on unburned skin. One patient had a transverse colostomy because of full-thickness burns to the left lower abdomen. Prior to therapeutic colostomy there was 100% graft loss on peri-anal regions. After therapeutic or prophylactic colostomy was performed, engraftment improved significantly. In all the children there was an improvement in the clinical appearance of the wounds after colostomy, and the granulation tissue was less inflamed, thinner and less friable. It was also no longer stained with faeces. The average number of skin grafts needed to provide cover after the colostomy was formed was 4 (range 1 - 9) to cover the total burn wound. This number reflects the patchy placement of grafts, as healing occurred at different sites, rather than recurrent graft loss. Specifically, peri-anal grafts after colostomy had minimal graft loss, and only a single attempt at cover was required for each area after appropriate wound bed preparation.
Bacteriological cultures confirmed a change from predominantly Gram-negative gut-derived organisms (Escherichia coli, E. cloacae, E. faecalis, Acinetobacter baumannii, Bacteroides sp.) and Candida yeasts before colostomy to predominantly Pseudomonas aeruginosa (25 cultures, 55.6%) or no bacterial growth (14 cultures, 31.1%) after diversion of the faecal stream (Fig. 1).
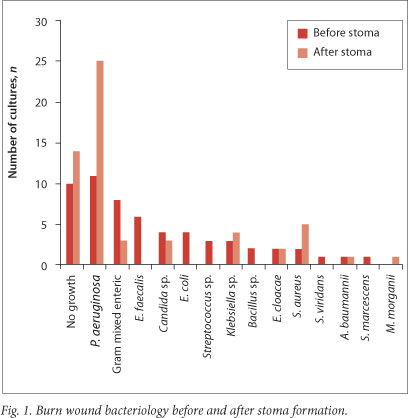
The stomas were reversed an average of 4 months (range 32 days - 14 months) after discharge from the burns unit. Time spent in hospital was on average 1.9 days per % TBSA and ranged from 13 to 168 days (mean 67 days).
Complications directly attributable to colostomy were few and minor (5 patients, 11.1%). A prophylactic colostomy was performed in error in 1 child, where the wound depth was misassessed. Three patients required manual reduction of stoma prolapse. The colostomy bags also leaked on a number of occasions. One patient developed adhesive bowel obstruction 3 months after closure which required a laparotomy. Eight children had to be transferred from the burns ward to the intensive care unit for treatment of systemic sepsis from their unhealed and infected perineal burn wounds.
The genital area generally healed well. Two penises required surgical intervention: 1 was treated with standard circumcision; and 1 had a dorsal slit performed. No testes were lost, although the size of the scrotal sac was significantly reduced in 1 child, resulting in a high-riding testis.
At follow-up, long-term morbidity following gluteal and perineal burns was minimal. None of the children was incontinent of faeces, and 2 required procedures to correct anal stenosis and a transverse perineal contracture. Two children with 40% and 86% TBSA burns respectively died from multiple organ failure. At the time of death both stomas were functional, although 1 patient had stomal breakdown with partial dehiscence of the peristomal area.
Discussion
Burns to the gluteal, perineal and upper thigh areas are common in paediatric patients. As with any burn wound, it is imperative to exclude child abuse or neglect. Although 1 of the patients in this study was burned as a direct result of adult intervention with a hot-water enema, this was not done with the intention of harming the child, but rather as a traditional remedy for abdominal pain (which turned out to be acute appendicitis requiring appendectomy).
The nature of a devitalised burn eschar lends itself to bacterial contamination followed by bacterial proliferation, invasion and ultimately invasive burn wound sepsis when the bacterial count exceeds 105 colony-forming units per gram of tissue. Burns in the gluteal, perineal and upper thigh areas pose unique problems. They are often deep and are prone to faecal contamination, especially in an infant or child who is not yet toilet trained, and it is difficult to apply antiseptics because of the three-dimensional contours. Invasive sepsis can be anticipated if the wound changes in clinical appearance, especially in the presence of clinical staining of the eschar; this is an ominous sign of deep burn wound sepsis and has an estimated mortality of 13%.[5] Penile-scrotal and vulvar burns in isolation were not an indication for diversion. Suprapubic diversion was not used in this series.
The vast majority of these regional burns, both partial and full thickness, will not require a colostomy as they heal exceptionally well and seldom require excision and grafting, despite faecal contamination.[3,4,13-15] Skin grafting is generally required in less than 5% of patients.[13-15] However, 1 - 13% of burns in the peri-anal area prove difficult to treat and are prone to septic complications resulting in graft loss, genital and anal functional impairment and contractures.[13]
Numerous bandaging techniques and various adhesive products have been advocated for use in this area in an attempt to prevent faecal soiling, but to date in certain patients our unit has had difficulty in this respect. Attempts to reduce stool frequency, and hence soiling, have included the use of a polymeric diet and constipating agents, but while they reduce stool frequency and increase consistency they may not solve the problem. The use of non-surgical devices for faecal diversion, which work well in adults, has not met with success in paediatric peri-anal burns.[16] These products are not licensed for use in children and in our experience have caused patulous and atonic anal sphincters in 2 children (these cases are not included in this series).
Diverting colostomy is a useful procedure to facilitate the management of deep perineal, buttock and thigh burns. It is a short, relatively easy procedure with minimal morbidity and mortality.[8] The colostomy can either be performed prophylactically or as therapeutically. Prophylactic colostomies prevent burn-wound infection in severe, deep partial- or full-thickness burns of the peri-anal area, and therapeutic stomas aid with local control of systemic sepsis.
A defunctioning sigmoid end-colostomy with Hartmann's closure of the distal stump is our preferred procedure. This ensures complete interruption of the faecal stream.[1] A loop or diverting colostomy with a distal mucous fistula presents problems, namely prolapse, faecal spill over, a large parietal defect and two stomal orifices requiring space in an already compromised area. In extensive burns the colostomy can be brought out in an area with a superficial skin burn or in a newly grafted area.[1] We prefer to form the stoma in unburned skin in the left iliac fossa. Having a well-sited colostomy eliminates the need for an elemental diet, constipating medications, uncomfortable splinting, multiple dressing changes, trips to the operating room to debride septic wounds, and labour-intensive nursing. In 2 children the colostomy was sited in a recently grafted area, with no resultant graft loss and normal colostomy function. Feeding with a polymeric diet is commenced within 24 hours.
A therapeutic colostomy was performed when there was deterioration in the clinical status of the wound. Appearance included conversion into a deeper wound, wound discoloration, tissue separation, spreading cellulitis, friable bleeding granulation tissue, and failed skin grafts. Diverting the faecal stream not only resulted in a clinical improvement in the wounds; the bacterial profile also changed substantially from overwhelming gut-derived enteric organisms to no growth (26.7% of swabs), or single-organism profiles. P. aeruginosa (55.6% of swabs) is a resident organism in burns units, extremely difficult to eradicate and commonly encountered in chronically unhealed wounds, and probably reflects the prolonged non-epithelisation of these wounds. Purulent exudate was reduced, granulation tissue assumed a healthy appearance, graft loss was minimised, and systemic evidence of infection abated.[1] These results document clear improvements in the patients' condition and therefore promote the use of a colostomy.
Complications were infrequent. Two children died after colostomy from sepsis and multi-organ failure, and because we were unable to complete grafting procedures. There were 3 prolapsed stomas; 2 were easily reduced manually and 1 required operative reduction, and none recurred. The overall complication rate (11.1%) was lower than that for diversions performed for primary bowel pathology.[8,9]
The stomas were reversed an average of 4 months (range 32 days - 14 months) after discharge from the burns unit. The colostomy was only closed once all wounds had healed and reconstructive procedures had been completed. These included the release of a contracture band across the perineum, repair of a single case of anal stenosis, and multiple small skin grafts to the lower limbs to ensure complete skin cover before reversal of the colostomy.
Conclusion
Formation of a defunctioning colostomy in paediatric patients, although infrequent, has a definitive role in burns of the peri-anal, gluteal and upper thigh burns. It can easily be performed with a low complication rate, and the use of a stoma significantly alters the bacterial profile of the involved areas. Specific indications exist for either prophylactic or therapeutic colostomies, but the decision should always be based the individual case and is not protocol driven. When done for the correct indications colostomy formation can be a life-saving procedure that can rapidly result in improved clinical status and improved wound healing. The aim of this paper was to substantiate our original findings in 13 patients.[1] Thirty-two patients were added, and the same conclusions were drawn - the method is valuable in selected patients.
REFERENCES
1. Quarmby CJ, Millar AJ, Rode H. The use of diverting colosomies in paediatric peri-anal burns. Burns 2002;27(5):645-650. [http://dx.doi.org/10.1016/S0305-4179(99)00076-5] [ Links ]
2. Angel C, Tung S, French D, Orihuela E, Lukefahr J, Herndon DN. Genital and perineal burns in children: 10 years of experience at a major burn center. J Pediatr Surg 2002;37(1):99-103. [http://dx.doi.org/10.1053/jpsu.2002.29437] [ Links ]
3. McDougal WS, Petersen HD, Pruit BA, et al. The thermally injured perineum. J Urol 1979;121(3):320-324. [ Links ]
4. Pisarski GP, Greenhagh DG, Warden GD. The management of perineal contractures in children with burns. J Burn Care Rehabil 1994;15(3):256-259. [http://dx.doi.org/10.1097/00004630-199405000-00010] [ Links ]
5. Renz BM, Sherman R. Exposure of buttock burn wounds to stool in scald-abused infants and children. Am Surg 1993;59(6):379-383. [ Links ]
6. Lam A, Wise L, Butcher H. Role of colostomy in perineal burns. Mo Med 1974;71(9):538-541. [ Links ]
7. Gauderer MW, Izant RJ Jr. A technique for temporary control of colostomy prolapse in children. J Pediatr Surg 1985;20(6):653-655. [http://dx.doi.org/10.1016/S0022-3468(85)80017-8] [ Links ]
8. Millar AJ, Lakhoo K, Rode H, Ferreira MW, Brown RA, Cywes S. Bowel stomas in infants and children. A 5-year audit of 203 patients. S Afr J Surg 1993;31(3):110-113. [ Links ]
9. Nour S, Beck J, Stringer MD. Colostomy complications in infants and children. Ann R Coll Surg Engl 1996;78(6):526-530. [ Links ]
10. Karpelowsky JS, Wallis L, Madaree A, Rode H. South African burn stabilisation protocol. S Afr Med J 2007;97(8):574-577. [ Links ]
11. Cox S, Rode H. The modern management of paediatric burns. CME 2010;28(3):113-118. [ Links ]
12. Levine NS, Lindberg RB, Mason AD Jr. The quantitative swab culture and smear: A quick, simple method for determining the number of viable aerobic bacteria on open wounds. J Trauma 1976;16(2):89-94. [ Links ]
13. Constable JD. Management of burn contractures of the perineum. Plast Reconstr Surg 1983;72(6):841-842. [ Links ]
14. Sawney CP. Management of burn contractures of the perineum. Plast Reconstr Surg 1983;72(6):837-840. [ Links ]
15. Peck MD, Boileau MA, Grube BJ, Heimbach DM. The management of burns to the perineum and genitals. J Burn Care Rehabil 1990;11(1):54-56. [http://dx.doi.org/10.1097/00004630-199001000-00012] [ Links ]
16. Bordes J, Goutorbe P, Asencio Y, Meaudre E, Dantzer E. A non-surgical device for faecal diversion in the management of perineal burns. Burns 2008;34(6):840-844. [http://dx.doi.org/10.1016/j.burns.2007.11.009] [ Links ]
 Correspondence:
Correspondence:
C E Price
(cprice2006@gmail.com)














