Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Surgery
versão On-line ISSN 2078-5151
versão impressa ISSN 0038-2361
S. Afr. j. surg. vol.51 no.3 Cape Town Jan. 2013
GENERAL SURGERY
Base deficit as an early marker of coagulopathy in trauma
S CheddieI; D J J MuckartII; T C HardcastleIII
IMMed (Surg), FCS (SA), Department of General Surgery, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
IIFRCS, MMSc, Department of General Surgery, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
IIIMMed, FCS (SA), Department of General Surgery, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
ABSTRACT
BACKGROUND: The acute coagulopathy of trauma is associated with hypoperfusion, metabolic acidosis and an increased mortality rate. Biochemical markers of hypoperfusion, namely base deficit (BD) and lactate, are commonly used to assess the degree of hypoperfusion. Early identification of hypoperfusion and acidosis using BD and lactate may help predict the development of coagulopathy in trauma patients and direct therapy.
OBJECTIVES: To identify whether a correlation exists between BD, lactate, injury severity, early-onset coagulopathy and mortality.
METHODS: A retrospective chart analysis was undertaken of patients transferred directly from scene to the level I trauma unit at Inkosi Albert Luthuli Central Hospital, Durban, South Africa, from 2007 to 2008. Patients with evidence of hypoperfusion were selected. Hypoperfusion was defined as a base deficit >-2 and coagulopathy as an International Normalized Ratio (INR) of >1.2. BD, lactate, chloride, temperature, Injury Severity Score (ISS), INR and mortality were recorded in this cohort. Student's t-test and Fisher's exact test were used for continuous and categorical variables, respectively. Correlation curves were used to determine the degree of association between the variables BD, lactate and ISS with respect to the INR. A p-value of <0.05 was considered statistically significant.
RESULTS: Of the 28 patients, males (n=18) accounted for 64.3% of admissions. The mean age was 31 years (range 1 - 75 years, median 30 years). The mechanism of injury was penetrating trauma in 5 cases (17.9%) and blunt trauma in 23 (82.1%). The median ISS was 24 (range 4 - 59). In 16 patients (57.1%) the INR was within normal limits, but in 12 (42.9%) it was over 1.2. There was a significant correlation between BD, ISS and INR (r=0.393; p=0.019 and r=0.565, respectively; p<0.001). Lactate showed a weak and non-significant association with the INR (r=0.232; p=0.18). There were a total of 12 deaths (42.8%) in this cohort of patients with biochemical evidence of hypoperfusion. There was a significant increase in mortality in patients with evidence of hypoperfusion and an elevated INR (75.0% v. 18.7%; p=0.006).
CONCLUSION: BD but not lactate correlates with the development of the coagulopathy of trauma. The ISS showed a significant correlation with coagulation disturbances, and the combination of hypoperfusion and coagulopathy was associated with a significant increase in mortality.
The acute coagulopathy of trauma is a complex pathophysiological state that may be initiated by a number of factors. As an integral part of the 'deadly triad, it may be perpetuated by concomitant acidosis and hypothermia;[1] excessive pre-hospital fluid resuscitation may lead to haemodilution of clotting factors and worsening of the coagulopathy; and exposure of tissue factor when endothelium or brain matter is denuded results in release of thromboplastins.[2] Hypoperfusion following major injury is associated with an increased expression of thrombomodulin, which binds thrombin, resulting in increased activation of protein C that subsequently inhibits plasminogen activator inhibitor, leading to increased fibrinolysis.[3]
The coagulopathy of trauma is associated with an increased mortality rate,[4] and the key to improving survival lies in identifying and correcting early potentially reversible risk factors. Although traditionally hypoperfusion has been diagnosed and classified on the basis of clinical vital signs, occult hypoperfusion may occur in the presence of normal haemodynamic parameters. The measurement of lactate, the product of anaerobic metabolism, and the surrogate base deficit (BD) are well-recognised markers of cellular hypoperfusion and the severity of shock,[5] although BD may also be elevated under aerobic conditions such as acute kidney injury or hyperchloraemia, both of which commonly occur after major trauma as a result of renal hypoperfusion and excessive saline-containing resuscitation fluids. Both lactate and BD have been extensively investigated in trauma patients as markers of injury severity, end-points of resuscitation and predictors of outcome.
Although indicating the severity of hypoperfusion, measurement of lactate alone may not portray the extent of the metabolic acidosis. BD quantifies the extent of both anaerobic and aerobic acidoses and may be a better indicator of the risk of a coagulation disturbance. This study was undertaken to identify the correlation between BD, lactate and injury severity with the development of early-onset coagulopathy and mortality.
Methods
The study was approved by the Biomedical Research Ethics Committee (BE207/09) of the University of KwaZulu-Natal, Durban, South Africa. A retrospective chart analysis of 75 trauma patients admitted directly from the scene of injury to the level I trauma unit at Inkosi Albert Luthuli Central Hospital, Durban, from 2007 to 2008 was performed and data pertaining to patients with evidence of hypoperfusion were extracted. Hypoperfusion was defined as a BD >-2 (for the purpose of this study the symbol 'greater than' describes a more negative BD) and coagulopathy as an International Normalized Ratio (INR) of >1.2. All blood results were those taken on admission to the resuscitation room via an arterial sample. The BD was obtained from arterial blood gas analysis. Arterial lactate, chloride ion concentration and INR results were extracted from the Medicom electronic hospital laboratory database. Tympanic membrane temperature was recorded on admission to the resuscitation room. The Injury Severity Score (ISS) was computed after all injuries had been identified using the Abbreviated Injury Scale (AIS) 90 reference book. Death or survival was ascertained from the electronic hospital discharge notes. Complete data were available for 28 patients and were included in the analysis.
Student's t-test and Fisher's exact test were used for continuous and categorical variables, respectively. Correlation curves were used to determine the degree of association between the variables BD, lactate and ISS with respect to the INR. A p-value of <0.05 was considered statistically significant.
Results
Of the 28 patients, 18 were males (64.3%) and 10 females (35.7%). The mean age was 31 years (range 1 - 75 years, median 30 years). The mechanism of injury was penetrating trauma in 5 cases (17.9%) and blunt trauma in 23 (82.1%). The median ISS was 24 (range 4 - 59). In 16 patients (57.1%) the INR was within normal limits, but in 12 (42.9%) it was >1.2. The differences in mean BD, lactate, INR, ISS, systolic blood pressure and temperature between these two groups are shown in Table 1.
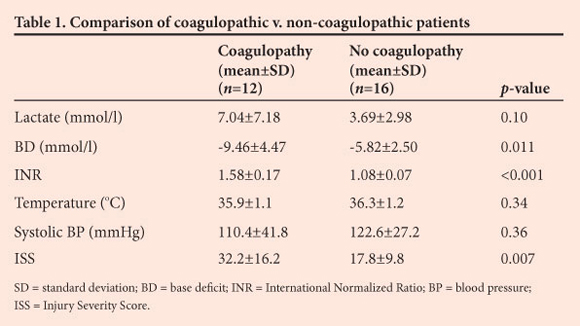
Lactate and BD showed a strong and significant correlation (r=0.739; p<0.001). There was a moderate and significant correlation between BD and INR (r=0.393; p=0.019), and the ISS demonstrated a stronger significant association (r=0.565; p<0.001). Lactate showed a weak and non-significant association with the INR (r=0.232; p=0.18). Chloride was above the upper limit of normal in 17 patients (60.7%), although there was no significant difference between the coagulopathic and non-coagulopathic patients.
There were a total of 12 deaths (42.8%) in this cohort of patients with biochemical evidence of hypoperfusion. In the group with coagulopathy 9/12 (75.0%) died, compared with only 3/16 (18.7%) of those with a normal INR (p=0.006).
Discussion
Metabolic acidosis has been incriminated as one of the instigators of coagulation disturbances following injury. The BD is defined as the amount of milli-equivalents of base required to titrate a litre of blood to a pH of 7.4 at an arterial partial pressure of carbon dioxide (PaCO2) of 40 mmHg.[6]
This measurement is unaffected by acute changes in PaCO2, is universally elevated in the presence of all pathological changes that induce a metabolic acidosis, and as such is a more reliable marker of the severity of the underlying metabolic state than pH, which will be altered by respiratory compensation. In the trauma setting, increased lactate production arises from anaerobic metabolism as a consequence of hypoperfusion, most commonly due to haemorrhage, but will not be affected by a metabolic acidosis that arises under aerobic conditions. Despite the fact that the BD not only reflects the degree of lactate accumulation but also provides a more composite view of a patient's metabolic status,[6,7] lactate clearance has been more popular than BD as a predictor of mortality in the trauma population.[8] Davis[9] showed a strong correlation between BD and lactate in a porcine haemorrhagic shock model and concluded that the BD was as accurate a marker of tissue hypoperfusion as lactate.The admission BD has also been shown to correlate well with the ISS, development of multiple organ failure, length of intensive care unit and hospital stay, need for blood transfusions and mortality[10] and a BD of ≥-6 has been shown to be a marker of injury severity and mortality in the trauma population, irrespective of age.[3,10,11]
Although hypoperfusion and the subsequent lactic acidosis play a key role in the pathogenesis of the early coagulopathy of trauma, the addition of another mechanism for metabolic acidosis will accentuate the risk. Our results show a moderate but significant correlation between an elevated BD and coagulopathy as measured by the INR, but we could not demonstrate a similar association with lactate despite the BD and lactate showing a strong correlation. As mentioned above, the reason may lie in the fact that lactate only reflects an acidosis arising from anaerobic metabolism, whereas the BD is a global marker of both anaerobic and aerobic acidoses. In addition to (and as a consequence of) hypoperfusion, acute kidney injury may contribute to the acidosis of injury over and above that from anaerobic metabolism. Hyperchloraemia is a common consequence of kidney injury, and although previously thought innocuous has a detrimental effect on outcome when combined with another acidosis.[12] Furthermore, hyperchloraemia may not only contribute to an acidotic state during renal insufficiency, but aggravate renal damage further by inducing renal vasoconstriction[13,14] Given the incidence of hyperchloraemia in our patient population, which in addition to hypoperfusion may contribute to metabolic acidosis, acute kidney injury and coagulopathy, the BD may be a more useful predictor of the risk of coagulation disturbances and an indicator for the early use of plasma rather than clear fluids for resuscitation.
The ISS showed the strongest association with an elevated INR. Although hypoperfusion is undoubtedly a contributing factor in the coagulopathy of trauma, it is not a prerequisite. Tissue factor exposure has been incriminated as a major trigger mechanism[2,15] and it may be assumed that a higher ISS would be associated with more severe tissue damage, an increase in coagulation disturbances and a higher mortality rate. The presence of coagulopathy at admission is associated with increased injury severity, length of hospital stay and number of organ failures and mortality,[16] and we have shown a statistically significant increase in mortality in our cohort of patients with evidence of hypoperfusion and who were coagulopathic on admission.
In the acute resuscitation phase, acquiring an INR result may take up to an hour. In the emergency setting, thromboelastometry has been shown to be a quick, useful method in identifying patients with coagulopathy and guiding transfusion requirements. A strong correlation has been shown between thromboelastometry and derangements in conventional clotting parameters, results are available within 10 minutes, and this simple device has now gained acceptance as standard of care in the severely injured.[17]
It is imperative to identify patients with acute coagulopathy early as this may influence outcome and resuscitation efforts. The BD can be obtained within minutes using a conventional blood gas analyser, which is available in most emergency departments, and has a significant association with acute coagulation disturbances. This finding could have far-reaching clinical applications. In addressing hypoperfusion in the resuscitation bay, the conventional use of crystalloids or colloids could be replaced by the immediate use of freeze-dried plasma that would lead to early replenishment of clotting factors in addition to sustained volume expansion. The recent war in Iraq has given rise to similar opinions, suggesting that fresh-frozen plasma be used as the resuscitation fluid of choice in the acute combat setting to decrease additional bleeding and replenish clotting factor deficiencies[18]
There are several limitations to our study. The sample may be too small in size and not fully representative of the trauma population. There are major discrepancies in the literature with regard to defining coagulopathy, and the INR value of >1.2 that we used may be too sensitive and overestimate the presence of coagulopathy in the study group. In a review by Brohi et al., four major studies used four different indices to define coagulopathy.[15] An estimate of pre-hospital fluid administration was lacking from our database and was not included in our assessment. Even considering these shortfalls, there is a growing body of evidence indicating that in order to improve survival in the seriously injured we need to address the possibility of a coagulation disturbance much earlier than was previously thought. The simple combination of BD and an estimate of the ISS will help to identify those at risk.
Conclusion
Our findings suggest that a significant correlation exists between BD, ISS and INR and that BD is superior to lactate in predicting coagulation disturbances. The combination of hypoperfusion and a coagulopathy at admission is associated with a significant increase in mortality.
REFERENCES
1. Tieu BH, Holcomb JB, Schreiber MA. Coagulopathy: Its pathophysiology and treatment in the injured patient. World J Surg 2007;31(5):1055-1064. [http://dx.doi.org/10j007/s00268-006-0653-9] [ Links ]
2. Ho AM, Karmakar MK, Dion PW. Are we giving enough coagulation factors during major trauma resuscitation? Am J Surg 2005;190(3):479-484. [http://dx.doi.org/10.1016/j.amjsurg.2005.03.034] [ Links ]
3. Brohi K, Cohen MJ, Ganter MT, et al. Acute coagulopathy of trauma: Hypoperfusion induces systemic anticoagulation and hyperfibrinolysis. J Trauma 2008;64(5):1211-1217. [http://dx.doi.org/10.1097/TA0b013e318169cd3c] [ Links ]
4. Brohi K, Singh J, Heron M, Coats T. Acute traumatic coagulopathy. J Trauma 2003;54(6):1127-1130. [http://dx.doi.org/10.1097/01.TA0000069184.06] [ Links ]
5. Paladino L, Sinert R, Wallace D, Anderson T, Yadav K, Zehtabchi S. The utility of base deficit and arterial lactate in differentiating major from minor injury in trauma patients with normal vital signs. Resuscitation 2008;77(1):363-368. [http://dx.doi.org/10.1016/j.resuscitation.2008.01.022] [ Links ]
6. Juern J, Khatri V, Weigelt J. Base excess: A review. J Trauma Acute Care Surg 2012;73(1):27-31. [http://dx.doi.org/10.1097/TA.0b013e318256999d] [ Links ]
7. FitzSullivan E, Salim A, Demetriades D, Arseno J, Martin MJ. Serum bicarbonate may replace the arterial base deficit in the trauma intensive care unit. Am J Surg 2005;190(6):961-967. [http://dx.doi.org/10.1016/j.amsurg.2005.08.024] [ Links ]
8. Abramson D, Scalea TM, Hitchcock R, Trooskin SZ, Henry SM, Greenspan J. Lactate clearance and survival following injury. J Trauma 1993;35(4):584-589. [http://dx.doi.org/10.1097/00005373-199310000-00014] [ Links ]
9. Davis JW. The relationship of base deficit to lactate in porcine hemorrhagic shock and resuscitation. J Trauma 1994;36(2):168-172. [http://dx.doi.org/10.1097/00005373-199402000-00002] [ Links ]
10. Davis JW, Parks SN, Kaups K, Gladen H, O'Donnel-Nichol SRN. Admission base deficit predicts transfusion requirements and risk of complications. J Trauma 1996;41(5):769-774. [http://dx.doi.org/10.1097/00005373-199611000-00001] [ Links ]
11. Rutherford EJ, Morris Ja, Reed GW, Hall KS. Base deficit stratifies mortality and determines therapy. J Trauma 1992;33(3):417-423. [http://dx.doi.org/10.1097/00005373-199209000-00014] [ Links ]
12. Shaw AD, Bagshaw SM, Goldstein SL, et al. Major complications, mortality, and resource utilization after open abdominal surgery. Ann Surg 2012;255(5):821-829. [http://dx.doi.org/10.1097/SLA.0b013e31825074f5] [ Links ]
13. Yunous NM, Bellomo R, Story D, Kellum J. Bench-to-bedside review: Chloride in critical illness. Crit Care 2010;14(4):226-235. [http://dx.doi.org/10.1186/cc9052] [ Links ]
14. Yunous NM, Bellomo R, Hegarty C, et al. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA 2012;308(15):1556-1572. [http://dx.doi.org/10.1001/jama.2012.13356] [ Links ]
15. Brohi K, Cohen MJ, Davenport RA. Acute coagulopathy of trauma: Mechanism, identification and effect. Current Opinion in Critical Care 2007;13(6):680-685. [http://dx.doi.org/10.1097/MCC.0b013e3282f1e78f] [ Links ]
16. Maegele M, Lefering R, Yucel N, et al. Early coagulopathy in multiple injury: An analysis from the German Trauma Registry on 8724 patients. Injury 2007;38(3):298-304. [http://dx.doi.org/10.1016/j.injury.2006.10.003] [ Links ]
17. Rugeri L, Levrat A, David JS, et al. Diagnosis of early coagulation abnormalities in trauma patients by rotation thrombelastography. J Thromb Haemost 2007;5(2):289-295. [http://dx.doi.org/10.1111/j.1538-7836.2007.02319.x] [ Links ]
18. Sebesta MAJ. Special lessons learned from Iraq. Surg Clin North Am 2006;86(3):711-772. [http://dx.doi.org/10.1016/j.suc.2006.03.002] [ Links ]
 Correspondence:
Correspondence:
D J J Muckart
(davidmuc@ialch.co.za)














