Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Surgery
versão On-line ISSN 2078-5151
versão impressa ISSN 0038-2361
S. Afr. j. surg. vol.51 no.2 Cape Town Jan. 2013
GENERAL SURGERY
Pseudomonas aeruginosa burn wound infection in a dedicated paediatric burns unit
E CoetzeeI; H RodeII; D KahnIII
IMB ChB, FCS (SA), MMed (Surg), Cert Gastrol Surg (SA). Department of Surgery, Faculty of Medicine, University of Cape Town, South Africa
IIMB ChB, MMed (Surg), FCS (SA), FRCS (Edin). Department of Surgery, Faculty of Medicine, University of Cape Town, South Africa
IIIMB ChB, FCS (SA), ChM. Department of Surgery, Faculty of Medicine, University of Cape Town, South Africa
ABSTRACT
BACKGROUND: Pseudomonas aeruginosa infection is a major cause of morbidity in burns patients. There is a paucity of publications dealing with this infection in the paediatric population. We describe the incidence, microbiology and impact of P. aeruginosa infection in a dedicated paediatric burns unit.
METHODS: A retrospective review of patients with clinically significant P. aeruginosa infection between April 2007 and January 2010 in the burns unit at Red Cross War Memorial Children's Hospital in Cape Town, South Africa, was performed.
RESULTS: During the 36-month study period, 2 632 patients were admitted. Of 2 791 bacteriology samples sent for microscopy, culture and sensitivity, 406 (14.5%) were positive for P. aeruginosa. Thirty-four patients had clinically significant P. aeruginosa wound infection, giving an incidence of 1.3%. Three patients had loss of Biobrane or allografts, and 23 cases of skin graft loss occurred in 18 patients. An average of 12 dressing days was needed to obtain negative swabs. All isolates were sensitive to chlorhexidine, whereas 92.5% were resistant to povidone-iodine. Piperacillin-tazobactam was the systemic antimicrobial to which there was most resistance (36.1%), and tobramycin had least resistance (3.3%).
CONCLUSIONS: The incidence of clinically significant burn wound infection is low in our unit, yet the morbidity due to debridement and re-grafting is significant. We observed very high resistance to topical povidone-iodine. Resistance to systemic antimicrobials is lower than that reported from other burns units.
Burn wound infection due to Pseudomonas aeruginosa poses a significant challenge in terms of systemic sepsis, graft loss, prolonged hospital stay, and even increased mortality.[1-12] Armour et al. reported on 48 adult patients with gentamicin-resistant P. aeruginosa colonisation.[1] Compared with case-matched controls, this cohort required a twofold increase in grafting procedures as well as an average of 15 days' longer hospital stay.[1]-Geyik and colleagues reported the only specific series to detail the effect of P. aeruginosa in children.[2] They found a 65% frequency of wound colonisation, with systemic sepsis in 7.2% which, when associated with bacteraemia, was associated with an 80% mortality.
Red Cross War Memorial Children's Hospital (RCH) Cape Town, has the only dedicated paediatric burns unit in the Western Cape province of South Africa. It serves a population of over 5.8 million and annually admits an average of 1 100 burns patients. The frequency, systemic and local therapy, and morbidity of P. aeruginosa infection in the burns unit of RCH have not been documented previously. This study aims to address this deficit.
Methods
The cohort for analysis was accrued from all admissions to the burns unit at RCH between April 2007 and January 2010. The unit admits children with partial-thickness burns greater than 10% total body surface area (TBSA), and all patients with full-thickness burns, circumferential limb involvement, facial burns, perineal burns and burns to hands or feet. The patients are treated according to standard management protocols. The standard of care during the study period was to treat wounds infected with P. aeruginosa (even if clinically suspected and not yet proven with a positive swab) with daily soaks with an unbuffered sodium hypochlorite solution,[13] after which the wounds were dressed with Flamazine dressings. Chlorhexidine was substituted for Flamazine if an organism resistant to Flamazine was isolated.
Numbers of P. aeruginosa wound isolates and isolates from other sites were noted for the whole cohort. For patients with clinically significant P. aeruginosa burn wound infection (defined as the combination of a positive isolate from a wound swab and the wound characteristics of excessive slough, erythema, green discoloration and the characteristic smell), the following information was recorded from the case records: patient demographics, percentage of TBSA of the burn, time to acquisition of infection, Biobrane, allograft and autologous skin graft loss, number of theatre visits, wound dressings and their frequency of application, use of antimicrobial agents, time to eradication of infection, delay to definitive skin grafting, and overall hospital stay. Microbiological data recorded were the resistance patterns of P. aeruginosa isolates to topical and systemic antimicrobial agents.
Results
Patient demographics
During the 36-month study period, 2 632 patients were admitted to the burns unit at RCH, 2 791 bacteriology samples were sent- for microscopy, culture and sensitivity and 906 had an organism isolated; of these samples, 406 (14.5% of the total) were positive for P. aeruginosa.
Thirty-four patients were recorded to have clinically significant Pseudomonas burn wound infection, yielding a frequency of 1.3%. Three patient records had been lost and could not be reviewed. Thirty-one patients were therefore analysed.
Twenty of the 31 patients were male and 11 were female. The mean age of those who developed P. aeruginosa infection was 3 years (range 4 months - 10 years). The mean TSBA involved was 27% (range 8 - 65%).
The mean time between admission and diagnosis of Pseudomonas infection was 10 days (range 1 - 34 days), that between a positive culture and initiation of appropriate management 2 days (range 0 - 9 days), and that between diagnosis and negative culture 13 days (range 2 - 32 days).
The diagnosis of clinically significant P. aeruginosa burn wound infection was confirmed with a positive wound swab in all patients. Seven patients also had positive blood cultures and 1 had a positive urine culture. Thirteen of the 31 patients received systemic antibiotics. Systemic antibiotics were administered only to patients with systemic signs of sepsis, i.e. fever, tachycardia and leucocytosis; they included all those with positive blood or urine cultures.
Bacteriology
In all patients with clinically significant P. aeruginosa burn wound infection, the resistance patterns of the isolates to antimicrobials were analysed: 61 isolates of P. aeruginosa were tested against systemic antimicrobials and 53 were tested against topical antimicrobials. The sensitivities of the 61 isolates tested against systemic antimicrobials are shown in Table 1. Apart from piperacillin-tazobactam (63.9%), ciprofloxacin and ceftazidine (80.3%) and cefepime (82.0%), the antimicrobial agents had >90% sensitivity.
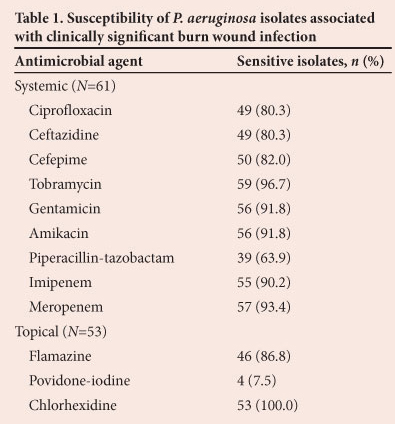
A multiresistant P. aeruginosa, defined as an organism resistant to three classes of antipseudomonal antimicrobials, was isolated in only 1 patient. This patient had no signs of systemic sepsis and did not receive systemic antimicrobials. In 22 patients the first isolate was sensitive to all systemic antipseudomonal antibiotics. Only 3 were treated with systemic antibiotics. Isolates with resistance to systemic antimicrobials were cultured from 9 patients as the first positive wound swab. Four of these 9 patients received systemic antibiotics according to the sensitivities of the cultured isolates.
Table 1 also details the sensitivities of 53 isolates to topical antimicrobial agents. Notably, only 4 were sensitive to povidone-iodine, but all were sensitive to chlorhexidine.
Wound dressings and management
Table 2 summarises the dressings used for the management of the 31 patients with clinically significant P. aeruginosa wound infection. A total of 368 dressing days were needed until negative wound swabs were obtained, with an average of 12 dressing days per child (range 2 - 30 days). Sodium hypochlorite soaks followed by Flamazine dressings accounted for the vast majority of the dressings, with chlorhexidine being used when an organism resistant to Flamazine was isolated. Other dressings were used in 7 patients for specific indications as decided by the senior surgeon in the unit.
Eleven children needed surgical debridement of wounds to help control P. aeruginosa infection, and of these 3 needed additional debridements. Fifty skin grafts were performed on 26 of the patients, with an average of 2 skin grafts per patient (range 1 - 4).
There was no relapse of P. aeruginosa infection after successful treatment of the wound infection.
Complications
Three patients had loss of Biobrane or allografts. Twenty-three cases of autologous skin graft loss occurred in 18 patients, with 5 needing two graft attempts. None of these patients had concomitant beta-haemolytic streptococcal wound infection. There were 45 theatre visits among 18 patients (mean 2.5 theatre visits per patient, range 1 - 5) for debridement of wounds after the diagnosis of P. aeruginosa infection was made. A cumulative total of 68 weeks were spent in hospital for management of the wound infections, at an average of 2 weeks per child (range 1 - 4 weeks).
Discussion
P. aeruginosa infection in burns patients is responsible for significant morbidity.[1,2,8] In this study, we documented the incidence of P. aeruginosa burn wound infection at RCH and its impact on burn wound management.
The average TBSA of burn wounds in the children with P. aeruginosa infection in our study was 27%, and the average time spent in hospital before diagnosis of P. aeruginosa infection was 10 days. This is in keeping with the findings of other authors (7 - 15 days).[5,8,14,15]
In this study the rate of P. aeruginosa wound colonisation was 14.5%. This is at the lower end of the spectrum reported in the reviewed literature (11 - 74%).[2-5,7,10,11,14-16] It is unclear from the definitions and methods used why there should be such a variation in the incidence of wound colonisation between different burns units. Possible explanations include different patient loads, nurse/ patient ratios, sampling protocols, unit architecture, and cleaning and hygiene practices.
All but 4 of the isolates of P. aeruginosa cultured from our patients were resistant to povidone-iodine, whereas all were sensitive to chlorhexidine. P. aeruginosa resistant to Flamazine was cultured from 6 patients. No other publications on the sensitivities of P. aeruginosa to topical antimicrobials in other burns units were found. The high incidence of resistance to povidone-iodine has resulted in a change of practice in our unit. Before this study, non-infected burn wounds were dressed with povidone-iodine-impregnated paraffin gauze swab dressings (Inadine). We now use chlorhexidine-impregnated paraffin gauze swab dressings (Bactigras).
The proportion of patients developing clinically significant P. aeruginosa burn wound infection in our unit during the 36-month study period was 1.3%. This is lower than the 7.2% and 13% reported in the literature.12,121 In the study by Rogers et al.,[12]-the average TSBA burned was 15%, compared with 27% in our review. We believe that this reflects on the efficacy of the standard management protocols and infection control practices in our unit, as well as the dedication of our nurses.
Compared with the reported susceptibility of P. aeruginosa to antimicrobials in the burns literature reviewed, we found a low prevalence of resistance of the organism to systemic antibiotics (Table 3).[7,10,11,15,16] This may be due to the infrequent use of systemic antibiotics in our unit. Systemic antibiotics are only prescribed for patients with systemic signs of sepsis.
One of the most dreaded non-lethal complications for a burn patient is graft loss. P. aeruginosa is a major cause of graft loss, and occurred in 23 of our 31 patients with clinically significant infection. Nine patients lost more than one attempted graft. In all patients who lost skin grafts, the diagnosis of P. aeruginosa infection was made after the initial skin grafting. Re-grafting was only done after negative wound swabs were obtained. There was no relapse of P. aeruginosa infection after successful treatment of the burn wound infection.
Conclusion
The incidence of clinically significant P. aeruginosa burn wound infection is low in our unit, attesting to the use of well-monitored infection control protocols. Despite effective wound therapy for P. aeruginosa, the morbidity stemming from graft loss and re-grafting is significant. We observed very high resistance to topical povidone-iodine, which has led to a change of practice. Resistance to systemic antimicrobials is lower than reported from other burns units, and this may be because their use as an adjunct to wound care is restricted to patients with systemic sepsis. Vigilance on the part of the clinical team treating a child with thermal injuries is necessary in order to diagnose P. aeruginosa burn wound infection early. Management protocols should be based on regular local audits of the resistance patterns of this troublesome organism.
REFERENCES
1. Armour A, Shankowsky H, Swanson T, Lee J, Tredget E. The impact of nosocomially-acquired resistant Pseudomonas aeruginosa infection in a burn unit. J Trauma 2007;63(1):164-171. [http://dx.doi.org/10.1097/01.ta.0000240175.18189.af] [ Links ]
2. Geyik MF, Aldemir M, Hosoglu S, Tacyildiz HI. Epidemiology of burn unit infections in children. Am J Infect Control 2003;31(6):342-346. [http://dx.doi.org/10.1016/S0196-6553(02)48226-0] [ Links ]
3. Lari AR, Alaghehbandan R. Nosocomial infections in an Iranian burn care center. Burns 2000;26(8):737-740. [http://dx.doi.org/10.1016/S0305-4179(00)00048-6] [ Links ]
4. Mousa HA. Aerobic, anaerobic and fungal burn wound infections. J Hosp Infect 1997;37(4):317-323. [http://dx.doi.org/10.1016/S0195-6701(97)90148-1] [ Links ]
5. Nasser S, Mabrouk A, Maher A. Colonization of burn wounds in Ain Shams University Burn Unit. Burns 2003;29(3):229-233. [http://dx.doi.org/10.1016/S0305-4179(02)00285-1] [ Links ]
6. Oncul O, Yuksel F, Altunay H, Acikel C, Celikoz B, Cavuslu S. The evaluation of nosocomial infection during 1-year-period in the burn unit of a training hospital in Istanbul, Turkey. Burns 2002;28(8):738-744. [http://dx.doi.org/10.1016/S0305-4179(02)00106-7] [ Links ]
7. Oncul O, Ulkur E, Acar A, et al. Prospective analysis of nosocomial infections in a burn care unit, Turkey. Indian J Med Res 2009;130(6):758-764. [ Links ]
8. Rastegar Lari A, Bahrami Honar H, Alaghehbandan R. Pseudomonas infections in Tohid Burn Center, Iran. Burns 1998;24(7):637-641. [http://dx.doi.org/10.1016/S0305-4179(98)00090-4] [ Links ]
9. Shahid M, Malik A. Resistance due to aminoglycoside modifying enzymes in Pseudomonas aeruginosa isolates from burns patients. Indian J Med Res 2005;122(4):324-329. [ Links ]
10. Singh NP, Goyal R, Manchanda V, Das S, Kaur I, Talwar V. Changing trends in bacteriology of burns in the Burns Unit, Delhi, India. Burns 2003;29(2):129-132. [http://dx.doi.org/10.1016/S0305-4179(02)00249-8] [ Links ]
11. Song W, Lee KM, Kang HJ, Shin DH, Kim DK. Microbiologic aspects of predominant bacteria isolated from the burn patients in Korea. Burns 2001;27(2):136-139. [http://dx.doi.org/10.1016/S0305-4179(00)00086-3] [ Links ]
12. Rodgers GL, Mortensen J, Fisher MC, Lo A, Cresswell A, Long SS. Predictors of infectious complications after burn injuries in children. Pediatr Infect Dis J 2000;19(10):990-995. [http://dx.doi.org/10.1097/00006454-200010000-00010] [ Links ]
13. Coetzee E, Whitelaw A, Kahn D, Rode H. The use of topical, un-buffered sodium hypochlorite in the management of burn wound infection. Burns 2012;38(4):529-533. [http://dx.doi.org/10.1016/j.burns.2011.10.008] [ Links ]
14. Erol S, Altoparlak U, Akcay MN, Celebi F, Parlak M. Changes of microbial flora and wound colonization in burned patients. Burns 2004;30(4):357-361. [http://dx.doi.org/10.1016/j.burns.2003.12.013] [ Links ]
15. de Macedo JL, Santos JB. Bacterial and fungal colonization of burn wounds. Mem Inst Oswaldo Cruz 2005(5);100:535-539. [http://dx.doi.org/10.1590/S0074-02762005000500014] [ Links ]
16. Revathi G, Puri J, Jain BK. Bacteriology of burns. Burns 1998;24(4):347-349. [http://dx.doi.org/10.1016/S0305-4179(98)00009-6] [ Links ]
 Corresponding author:
Corresponding author:
E Coetzee (edt.coetzee@gmail.com)














