Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.116 n.7-8 Pretoria Jul./Aug. 2020
http://dx.doi.org/10.17159/sajs.2020/8531
COMMENTARY
'Preventing the next pandemic' - A 2020 UNEP Frontiers Series Report on zoonotic diseases with reflections for South Africa
Bianca WerneckeI, II; Danielle A. MillarIII; Michele WaltersIV; Andre GanswindtV; Luthando DzibaVI, VII; Caradee Y. WrightIII, VIII
IEnvironment and Health Research Unit, South African Medical Research Council, Johannesburg, South Africa
IIDepartment of Environmental Health, University of Johannesburg, Johannesburg, South Africa
IIIEnvironment and Health Research Unit, South African Medical Research Council, Pretoria, South Africa
IVBiodiversity and Ecosystem Services, Council for Scientific and Industrial Research, Pretoria, South Africa
VMammal Research Institute, Department of Zoology and Entomology, University of Pretoria, Pretoria, South Africa
VIConservation Services Division, South African National Parks, Pretoria, South Africa
VIIDepartment of Zoology, Nelson Mandela University, Port Elizabeth, South Africa
VIIIDepartment of Geography, Geoinformatics and Informatics, University of Pretoria, South Africa
Keywords: human health, zoonoses diseases, environmental health, One Health, Planetary Health
Zoonoses account for about 25% of the infectious disease burden in low-income countries.1 Poverty might increase the risk for zoonotic disease where the active human-livestock and human-wildlife interfaces can increase the likelihood of disease transmission.1 A combined disease burden exists for people in areas such as tropical and subtropical Africa, where there is likelihood of co-infection with zoonotic diseases and other pathogenic or infectious diseases, such as malaria, tuberculosis and HIV.1 Many endemic zoonoses remain widely neglected in such settings, undetected and underreported, because their impacts are borne largely by impoverished and marginalised communities.2 Due to these unique contexts, the prevention and management of emerging and endemic zoonotic diseases in many African countries is a complex undertaking needing evidence-based guidance.1,3
In early 2020, the United Nations Environment Programme (UNEP) and the International Livestock Research Institute (ILRI) took on the urgent task to provide an up-to-date, rapid scientific assessment on zoonotic diseases as part of the UNEP's Frontiers Report Series.4-6 The goal of the report is to provide relevant information for policymakers on how to 'prevent the next pandemic' by interrogating what is known about zoonotic diseases and how best one can break the chain of transmission. As the world presently faces the SARS-CoV-2 pandemic, this timely report helps decision-makers with evidence-based actions, not only to flatten the curve of COVID-19 incidence, but to answer questions about zoonoses in general and plan for the future. In this Commentary, we give a brief overview of UNEP's latest report7 and then relate some of the key messages and recommendations for policymakers to a South African context.
An overview of zoonotic diseases
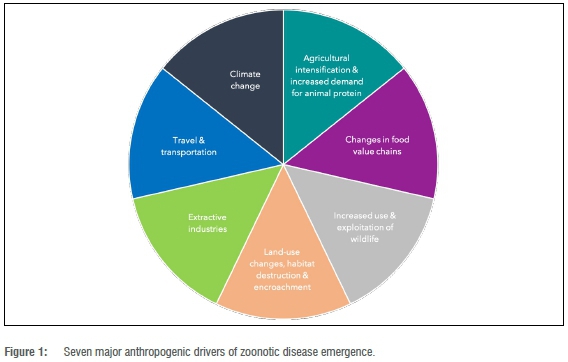
Many microorganisms are shared between people and other animals, but only some of these microorganisms, known as pathogens, cause disease.7 Most current zoonotic infections in people are transmitted from domesticated wildlife, livestock, and pets - true wildlife zoonoses are rarer but can be significant and catastrophic. Coronavirus disease 2019 (COVID-19) is ongoing; however, in recent decades there have also been zoonotic influenza (Bird Flu), pandemic human influenza (H1N1), Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS) - most of which have a suspected or proven domestic or peri-domestic wildlife reservoir. The three types of zoonotic diseases - i.e. emerging (e.g. Zika virus 2015-2016 in the Americas), outbreak or epidemic (e.g. Rift Valley fever 2015-2016 in Mauritania), and neglected zoonotic diseases (e.g. pig tapeworm) - can (and have) all contribute(d) to significant public health and related societal impacts. While seven major anthropogenic drivers for zoonotic disease emergence have been identified (Figure 1), it is not necessarily a simple task to determine which driver(s) lead to a zoonotic occurrence.
Between January 2017 and July 2018, South Africa experienced the largest listeriosis outbreak in the world to date.8,9 The National Institute for Communicable Diseases (NICD) detected a total of more than 1000 laboratory-confirmed cases and 200 fatalities,10,11 all of which were linked to eating contaminated polony (a type of bologna sausage). Polony was confirmed to contain Listeria monocytogenes after undergoing genome sequencing.10,11 In response to the outbreak, a national multisectoral task force coordinated investigations and response activities. Consequently, national authorities took measures to limit further infections, including issuance of safety recall notices and compliance notices, and communication about risks with vulnerable groups. In December 2017, listeriosis was made a notifiable medical condition in South Africa.12 Listeriosis, like many foodborne diseases, is a zoonosis.
Coronaviruses, common elements and origins of coronavirus pandemics
'Corona' refers to the crown-like arrangement of spike-shaped proteins on the surface of membranes of coronaviruses. In an infographic, the UNEP report gives an insightful timeline of various coronaviruses and other zoonotic disease occurrences from 1931 to the present day.7 Among some of these coronaviruses, such as MERS and porcine epidemic diarrhoea (PED) virus in 1971, there are common elements and origins: epidemics are driven by agricultural intensification/increased demand for animal protein; they have high economic costs, particularly due to social and economic disruptions; it seems that bats are natural reservoir hosts; and transmission is often associated with traditional markets or value chains. These elements are often interlinked, as described next.
Linkages between habitat loss and zoonoses
Human population increase has led to greater encroachment of humans into natural habitats, thereby proportionately increasing the risk of animal-to-human disease transmission.7 Changes in biodiversity and landscape conversion lead to changes in natural food cycles, often with an increase in vectors and pests and a decrease in predators. This causes a change in the species composition found in certain areas. Animals such as rodents are often present and considered opportunistic animals which frequently can represent vectors for disease or pathogen reservoirs. The 'dilution effect' suggests that the greater the species diversity, the fewer the opportunistic animals and pathogen reservoirs, translating into decreased disease transmission.7 However, this theory is dependent on animal and human population density and the transmission mode of the pathogen. The coevolution theory suggests that the change in landscape causes an adaptive change of migratory and resident animals, including pathogens, which increases the risk of disease spill-over and transmission. While there is a large variety of zoonotic pathogens, the greatest risk factor for transmission spill-over remains increased exposure to animals.
Linkages between trade and use of wildlife and zoonoses
Wildlife harvesting, trade and consumption are age-old activities, with wild animals being hunted and captured for human subsistence, for recreation, and for sale of animal body parts and/or their derivatives.7 Today, many households in Africa, Asia and Latin America harvest and consume wild meat, such as herbivores, rodents and reptiles. Although less documented, the harvesting and consumption of aquatic mammals (e.g. manatees and whales) and turtles is increasing too. The consumption of wild meat is driven by, among other factors, an increasing human population and an increased demand for protein-rich food and income. Not everyone may have access to, or be able to satisfy these needs with traditional resources, such as land, labour and livestock.
Much wild meat and many live wild animals are sold in traditional markets, which lack adequate biosafety measures, thus increasing the risk for zoonotic disease emergence and transmission. While legal ventures of wild meat production are increasing, there is also an increase in illegal wild meat production - an industry that employs many people and contributes to food and job security.7 Wild animals that are subject to trade often live in captivity and close quarters. As such, viruses spread between the diverse species and have higher pandemic potential as the viruses are considered to have greater 'host plasticity', which increases the possibility that they can spread to humans. Wildlife use, trade and consumption brings zoonotic risks. For example, zoonoses can be introduced at any point during the transfer of animal meat between hunting and handling. There is also the risk of transmission when meat is opportunistically harvested, such as in the case of the 2013-2016 Ebola outbreak in Guinea, Liberia and Sierra Leone.
Reflections for South Africa
Here, we reflect upon the Ten Key Messages put forward by the UNEP 'Preventing the next pandemic' report (Table 1) and how they may be discussed and applied in South Africa. In terms of knowledge to understand current systems and particularly those dealing with host populations, we need continued research in South Africa. As we put greater pressure on ecological systems, the way these systems respond changes, meaning that what was true yesterday can be different today.13 If we are to keep a 'finger on the pulse' of future disease outbreaks, we need research (especially baseline data) and monitoring of zoonotic diseases which takes these changes into account. Monitoring systems form the basis for tools to help respond to future pandemics. Data, including spatial data on drivers of disease, need to be made freely available across research fields such as wildlife ecology, human health, agricultural health and environmental health. Such information would prepare us better to respond with urgency to regional outbreaks.
In South Africa, while a scientific assessment as mentioned in the UNEP/ILRI report is necessary to explore the role of wildlife and domesticated animals in disease transmission, it is also necessary to look into respective 'animal groups' from a regional economic (and maybe spiritual) value perspective, as well as from a species diversity and conservation point of view. A case in point of the latter are countries where most of a species resides, like the southern white rhino in South Africa. Perhaps it is timeous to discuss global financial support mechanisms for those regions (or countries) in the world that by default must look after certain ecological hotspots or flagstone species.
To determine a holistic scope of the problem of zoonotic diseases and how to prevent the next outbreak, South Africa may need to discuss and prioritise next steps. We need a clear understanding of the 'food system': what people are eating, the sources of food, and how food is processed, stored and distributed, with a clear distinction between in-country consumption and/or export. In South Africa's present situation, the impacts of lockdown (and poverty in general) may force a reliance on wild meat, particularly for those who were dependent on the tourism industry to sustain a livelihood, which has largely closed. Increasing unemployment drives people towards harvesting of wildlife. Recent action by government to pass new laws on what constitutes domesticated animals14 and what is being considered under the Meat Safety Act15 means we will be brought closer to or be in greater contact with more species of animals. If we are to intensify pressures on wildlife in this way and start incorporating more wildlife into our diets, stringent testing for infection in these species should occur and potential disease and species-specific protocols should be put in place. Culture is also an important driver in South Africa. Countries reliant on traditional medicines and foods (also wild meat harvests) and those with an expanding demand for animal protein, including insects, for human and animal consumption may be at risk.16
Ecological disruption caused by an ever-growing human population, and its demand for and pressure on resources, may allow us to predict that future disease outbreaks will occur. In ecological systems in general, however, we are becoming less able to predict patterns and processes particularly in 'food webs' or 'assembly rules' and future projections (in this case around communities and food webs) are becoming increasingly messy.13 This has obvious consequences for our ability to monitor or understand zoonotic disease flows. In South Africa, these flows are complicated by issues of poverty, health inequity, landscape alterations, waste management and pollution, and the quadruple burden of disease. In terms of connectivity and complexity, the problem is intricate indeed but also constantly changing. As the predictability of biological and ecological processes decline, so too does our understanding of the connectivity and complexity at any point in time, particularly into the future. Hence, the need to mainstream and implement a 'One Health' approach.
An approach for the future
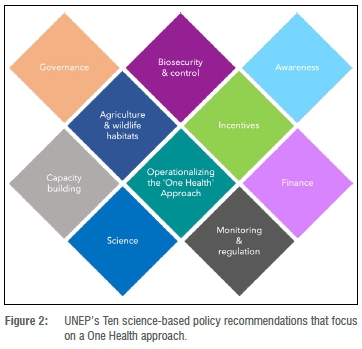
The latest UNEP report adopts 'One Health' as a collaborative, multisectoral and transdisciplinary approach - working at local, regional, national and global levels - to achieve optimal health and well-being outcomes, recognising the interconnections between people, animals, plants and their shared environment (Figure 2).17 Other approaches, such as 'Planetary Health'18 and 'One Welfare'19 also exist; however, UNEP considered 'One Health' as an easily understood umbrella term.
South Africa's 'One Health' Programme, led by the US Centers for Disease Control and Prevention with partners, as well as other initiatives such as AHEAD (Animal & Human Health for the Environment and Development)20 have done work to apply the approach in South Africa. UNEP's support of the 'One Health' approach now gives new impetus to finding ways in which South Africa can holistically embrace the philosophy. For example, a 'One Health' approach in policy development is critical for the future and has been sorely lacking. South Africa needs to adopt greater interdepartmental cooperation and joint decision-making when it comes to policy development. It is also essential that 'One Health' provides a suitable inclusive approach to assess the environmental context due to our country's unique, interconnected multi-layer diversity (e.g. ethnic, cultural, religious, social, economic and ecological diversity).
Promisingly, several actions and impacts of 'One Health' implementation in Africa do exist. Collaborative projects implementing joint livestock and child vaccination campaigns in pastoralist communities in Chad resulted in economic savings for the Chadian public health and animal health ministries and improved vaccination coverage of children and women who would otherwise have no access to health care.21 Shared human and veterinary laboratories to diagnose brucellosis in febrile patients in Mali resulted in brucellosis being considered as a differential diagnosis for febrile illness (along with malaria and typhoid fever) in an area where raw milk consumption is still prevalent.21 Lastly, Uganda's environmental health practitioners embody 'One Health' for healthy people, animals and environment.22 They investigate zoonotic disease outbreaks, ensure disease vector control, and provide communities with health education on vaccination of pets and food safety. These are encouraging success stories, from which South Africa should learn.
It is imperative that South Africa uses the rich interconnected multi-layer diversity in a call to all environmental health practitioners, public health professionals, doctors, veterinarians, conservationists, people living in communities, industries and small businesses selling animal products, and others to discuss, debate, and embrace 'One Health' as promoted by the UNEP report to prevent future epidemics and pandemics, locally and internationally. A solid evidence-based policy foundation is needed, as are human and financial capital, technological advances and advocacy, to facilitate creation of the tools and platforms for effective response and crisis mitigation.
Acknowledgements
Two SAMRC environmental health researchers (B.W. and C.Y.W.) gave direct input to the UNEP/ILRI 2020 report and thank UNEP for this opportunity.
References
1.Asante J, Noreddin A, El Zowalaty ME. Systematic review of important bacterial zoonoses in Africa in the last decade in light of the 'One Health' concept. Pathogens. 2019;8(50):1-29. https://doi.org/10.3390/pathogens8020050 [ Links ]
2.Halliday JEB, Allan KJ, Ekwem D, Cleaveland S, Kazwala RR, Crump JA. Endemic zoonoses in the tropics: A public health problem hiding in plain sight. Vet Rec. 2015;176(9):220-225. http://dx.doi.org/10.1136/vr.h798 [ Links ]
3.Kemunto N, Mogoa E, Osoro E, Bitek A, Kariuki Njenga M, Thumbi SM. Zoonotic disease research in East Africa. BMC Infect Dis. 2018;18(1):545-554. https://doi.org/10.1186/s12879-018-3443-8 [ Links ]
4.United Nations Environment Programme (UNEP). UNEP Frontiers 2018/19 report: Emerging issues of environmental concern. Nairobi: UNEP; 2019. Available from: https://wedocs.unep.org/bitstream/handle/20.500.11822/27538/Frontiers1819.pdf?sequence=1&isAllowed=y [ Links ]
5.United Nations Environment Programme (UNEP). UNEP Frontiers 2017 report: Emerging issues of environmental concern. Nairobi: UNEP; 2017. Available from: https://wedocs.unep.org/bitstream/handle/20.500.11822/22255/Frontiers_2017_EN.pdf?sequence=1&isAllowed=y [ Links ]
6.United Nations Environment Programme (UNEP). UNEP Frontiers 2016 report: Emerging issues of environmental concern. Nairobi: UNEP; 2017. https://doi.org/10.18356/4392feb8-en [ Links ]
7.United Nations Environment Programme (UNEP) and International Livestock Research Institute (ILRI). Preventing the next pandemic: Zoonotic diseases and how to break the chain of transmission. Nairobi: UNEP and ILRI; 2020. Available from: https://wedocs.unep.org/bitstream/handle/20.500.11822/32316/ZP.pdf?sequence=1&isAllowed=y [ Links ]
8.Kaptchouang Tchatchouang C-D, Fri J, De Santi M, Brandi G, Fiorella Schiavano G, Amagliani G, et al. Listeriosis outbreak in South Africa: A comparative analysis with previously reported cases worldwide. Microorganisms. 2020;8(1):135. https://doi.org/10.3390/microorganisms8010135 [ Links ]
9.Thomas J, Govender N, McCarthy KM, Erasmus LK. Outbreak of listeriosis in South Africa associated with processed meat. N Engl J Med. 2020;382(7):632-643. https://doi.org/10.1056/NEJMoa1907462 [ Links ]
10.South African National Institute for Communicable Diseases (NICD). Situation report on listeriosis outbreak, South Africa, 2017 [document on the Internet]. [ Links ] c2017 [cited 2020 Jun 20]. Available from: https://www.nicd.ac.za/wp-content/uploads/2017/12/NICD-Situation-report-on-listeriosis-outbreak-South-Africa_18-December-2017.pdf
11.South African National Listeria Incident Management Team. Listeriosis outbreak situation report [document on the Internet]. [ Links ] c2018 [cited 2020 Jun 20]. Available from: https://www.nicd.ac.za/wp-content/uploads/2018/07/Listeriosis-outbreak-situation-report-_26July2018_fordistribution.pdf
12.World Health Organization (WHO). Listeriosis - South Africa. Disease Outbreak News [document on the Internet]. [ Links ] c2018 [cited 2020 Jun 20]. Available from: http://www.who.int/csr/don/28-march-2018-listeriosis-south-africa/en/
13.Berger J, Wangchuk T, Briceno C, Vila A, Lambert, JE. Disassembled food webs and messy projections: Modern ungulate communities in the face of unabating human population growth. Front Ecol Evol. 2020;8(1):128. https://doi.org/10.3389/fevo.2020.00128 [ Links ]
14.Somers MJ, Walters M, Measey J, Strauss WM, Turner AA, Venter JA, et al. The implications of the reclassification of South African wildlife species as farm animals. S Afr J Sci. 2020;116(1/2), Art. #7724. https://doi.org/10.17159/sajs.2020/7724 [ Links ]
15.Alden C, Harvey R. South African proposal to breed wildlife for slaughter courts disaster. The Conversation. 2020 Jun 14. Available from: http://theconversation.com/south-african-proposal-to-breed-wildlife-for-slaughter-courts-disaster-140399 [ Links ]
16.Mishyna M, Chen J, Benjamin O. Sensory attributes of edible insects and insect-based foods - Future outlooks for enhancing consumer appeal. Trends Food Sci Technol. 2020;95:141-148. https://doi.org/10.1016/j.tifs.2019.11.016 [ Links ]
17.One Health Commission. Why One Health? What is One Health? [webpage on the Internet]. [ Links ] c2020 [cited 2020 Jun 20]. Available from: https://www.onehealthcommission.org/en/why_one_health/what_is_one_health/
18.Whitmee S, Haines A, Beyrer C, Boltz F, Capon AG, de Souza Dias BF, et al. Safeguarding human health in the Anthropocene epoch: Report of the Rockefeller Foundation-Lancet Commission on planetary health. The Lancet. 2015;386:1973-2028. https://www.doi.org/10.1016/S0140-6736(15)60901-1 [ Links ]
19.Bourque T. One Welfare. Can Vet J. 2017;58(3):217-218. [ Links ]
20.Osofsky S. Times evoke: Preventing the next pandemic - We can make this a 'never again' moment. India News. The Times of India. 2020 April 07. Available from: https://timesofindia.indiatimes.com/preventing-the-next-pandemic-we-can-make-this-a-never-again-moment/articleshow/74974789.cms [ Links ]
21.Kamani TM, Kazwala R, Mfinanga S, Haydon D, Keyyu J, Lankester F, et al. Viewpoint: One Health: A concept led by Africa, with global benefits. Vet Rec. 2015;176(19):496-497. https://doi.org/10.1136/vr.h2461 [ Links ]
22.Musoke D, Ndejjo R, Atusingwize E, Halage AA. The role of environmental health in One Health: A Uganda perspective. One Health. 2016;2(1):157-160. https://doi.org/10.1016/j.onehlt.2016.10.003 [ Links ]
 Correspondence:
Correspondence:
Caradee Wright
Email: cwright@mrc.ac.za
Published: 29 July 2020














