Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.115 n.7-8 Pretoria Jul./Aug. 2019
http://dx.doi.org/10.17159/sajs.2019/4653
REVIEW ARTICLES
Cardioprotection conferred by rooibos (Aspalathus linearis): A mini review to highlight a potential mechanism of action
Gerald J. Maarman
Cardiovascular Research Group, Division of Medical Physiology, Department of Biomedical Sciences, Stellenbosch University, Stellenbosch, South Africa
ABSTRACT
A number of cardioprotective interventions have been identified throughout the years, and these include the use of natural antioxidants in sources like rooibos (Aspalathus linearis) tea. Recent studies have demonstrated that rooibos (either its isolated components or the crude rooibos extract/tea) confers cardioprotection in diabetic cardiomyopathy and myocardial ischaemic injury. In addition, a clinical study has shown that regular rooibos consumption reduces the risk for cardiovascular disease in adults. However, rooibos is currently not considered an official treatment against cardiac disease, mainly because the underlying mechanisms for rooibos-induced cardioprotection are not fully elucidated. Physiological actions of rooibos must be well investigated before rooibos can be used in a clinical setting as adjunct treatment for patients with heart disease. Thus, research to delineate the underlying mechanisms of rooibos-induced cardioprotection is key. In the light of the aforementioned, the available literature on rooibos-induced cardioprotection is reviewed here, highlighting the fact that rooibos preserves and maintains cardiac energy homeostasis. It is postulated that rooibos activates an AMPK-GLUT-4 glucose oxidation (cardiac energy-shortage sensing) pathway to shift cardiac energy usage, thereby conferring cardioprotection.
SIGNIFICANCE:
•It is hypothesised that rooibos may alter the way in which the human heart uses energy and oxygen, in order to protect the heart against disease. The heart's mitochondria are responsible for the heart's energy processes, and therefore are most likely involved in rooibos-induced cardioprotection.
•Cardioprotection conferred by rooibos is likely via an AMPK-GLUT-4 glucose oxidation pathway.
•The mechanism of cardioprotection is important for future studies investigating how rooibos alters cardiac mitochondria.
•The more information gathered about the underlying mechanisms of rooibos, the easier it will be to recommend rooibos as an official cardioprotective intervention in patients with heart disease.
Keywords: South Africa; medicinal plants; novel heart drugs; heart research
Introduction
Cardioprotection is the manipulation of cellular processes with various therapeutic interventions to protect the heart before, during or after disease.1-3 These interventions include pre- and post-conditioning4-6, remote ischaemic conditioning4-6, and intake of antioxidants like melatonin7,8 and rooibos (Aspalathus linearis)9,10. Rooibos is an endemic South African fynbos plant species that grows exclusively in the Cederberg mountain region.11,12 Research investigating rooibos mainly describes the multiple biological effects of the two forms of rooibos leaf extracts: unfermented green and fermented rooibos.13 Bioactive components within rooibos extracts/tea are categorised as polyphenols and flavonoids14,15 and are responsible for the anti-inflammatory, anti-cancer16,17 and antioxidant properties13,18, of rooibos. Considering that polyphenols and flavonoids have a well-documented role in cardioprotection19,20, rooibos extract is a possible candidate for cardioprotection21,22.
Cumulative evidence suggests that either the individual rooibos components9,23,24 or the crude rooibos extract/tea confers cardioprotection against heart disease in diabetes10 and heart attacks25. In one recent study, rooibos conferred cardioprotection in rats that were on antiretroviral therapy and displayed myocardial ischaemic damage.26 However, in all these aforementioned studies, the mechanisms underpinning the rooibos-induced cardioprotection were not fully understood. Considering that rooibos is reported as a cardioprotective strategy for patients with heart disease21,22, the underlying mechanisms of rooibos-induced cardioprotection should be better understood. Without this understanding, in addition to a lack of evidence on the active ingredient(s) and safety/toxicity for patients taking heart disease medication27, rooibos may not pass clinical standards as a cardioprotective intervention for heart disease patients. If more information were gathered about the physiological mechanisms of rooibos, there would be a solid scientific foundation for the recommendation of rooibos as an effective cardioprotective intervention in patients with heart disease.
Considering the variety of bioactive components present in rooibos tea, it is likely that more than one component of rooibos contributes to cardioprotection. Additionally, these different components may activate different, yet synergistic, molecular pathways to bring about cardioprotection. This likelihood complicates the understanding of the underlying mechanisms of rooibos-induced cardioprotection. However, over the years, multiple studies have highlighted isolated mechanistic aspects related to rooibos-induced cardioprotection. To this end, a review paper is necessary to provide a novel perspective on current evidence, and to highlight a possible underlying mechanism for rooibos-induced cardioprotection. This elucidation of possible mechanisms might be useful for the design of future studies.
Overview of cardiac energy substrate utilisation in cardioprotection
Cardiac energy substrate utilisation plays a key role in cardiomyopathies28,29 and cardioprotection30,31. In diabetes32,33, high blood glucose (hyperglycaemia) as well as high glucose uptake without simultaneous glucose utilisation/oxidation, induces toxic effects on the heart34,35. It induces this toxicity by activating programmed cell death (apoptosis of cardiomyocytes)36 and thereby contributes to diabetic cardiomyopathy37. In this case, a sensible approach is to increase glucose oxidation, a pathway that can reduce the amount of glucose entering cardiomyocytes.38 This approach limits cardiomyocyte apoptosis and confers cardioprotection.39,40 Against this background, several therapeutic interventions modulate cardiac energy substrate utilisation in favour of glucose oxidation39,40, in order to confer cardioprotection.
Another dimension to the negative impact of hyperglycaemia on the heart, is that it increases the production of toxic molecules called reactive oxygen species41 that contribute to oxidative damage in the heart during hyperglycaemia. Therefore, in theory, a therapeutic intervention that can (1) increase myocardial glucose oxidation and (2) reduce reactive oxygen species, would accelerate glucose breakdown and thus limit the toxic effects of hyperglycaemia on the heart (Figure 1, green side).
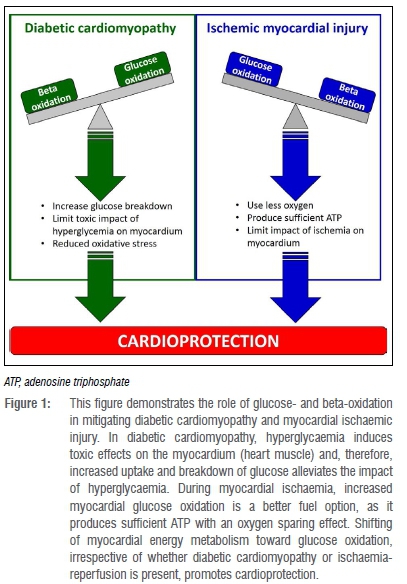
In the case of a heart attack (myocardial ischaemia), oxygen availability to the myocardium is markedly reduced.2 The heart normally generates energy through both glucose oxidation and fatty acid oxidation, while the latter uses less oxygen for the production of a similar amount of energy (adenosine triphosphate, ATP).42 Thus, if fatty acid (beta) oxidation is elevated during a heart attack and not altered, the already limited oxygen will be used to produce ATP, which will produce more severe ischaemic effects on the heart.29,43 Therefore, increased glucose oxidation during myocardial ischaemia is considered a better pathway as it produces sufficient ATP with an oxygen sparing effect.29,43 In keeping with this logic, previous reports have demonstrated that in myocardial ischaemia, pharmacological modulation of cardiac energy metabolism (how the heart uses glucose or fatty acids as energy) confers cardioprotection mainly because it increases glucose oxidation.31,44,45 Furthermore, other reports have demonstrated that a shift in cardiac energy substrate utilisation toward glucose oxidation (irrespective of whether cardiomyopathy is from hyperglycaemia or ischaemia) contributes to cardioprotection43,46 (Figure 1, blue side).
Evidence that rooibos shifts cardiac energy substrate utilisation
A number of studies have demonstrated a link between rooibos and changes in energy substrate utilisation in the liver47 and skeletal muscle48. A recent study showed that rooibos reduced hepatic insulin resistance by modulating adenosine 5′ monophosphate activated protein kinase (AMPK) pathways.49 One research group was able to demonstrate that rooibos increases liver activity of adenosine AMPK.48 Similarly, rooibos is able to increase AMPK activity, glucose oxidation48, mitochondrial activity and ATP production in muscle50. AMPK is also the main sensor for cardiac energy status51, and thus this effect of rooibos on AMPK was also recently demonstrated in the heart9,10,52. Taken together, these findings demonstrate the ability of rooibos to modulate cardiac energy substrate utilisation.
Rooibos also preserves and maintains cardiomyocyte ATP production9 and improves cardiac functional recovery25 by enhancing glucose oxidation9. From these data, it can be inferred that rooibos alters AMPK activity to enhance myocardial glucose uptake via glucose transport-4 (GLUT-4)48 in order to promote myocardial glucose oxidation (Figure 2).
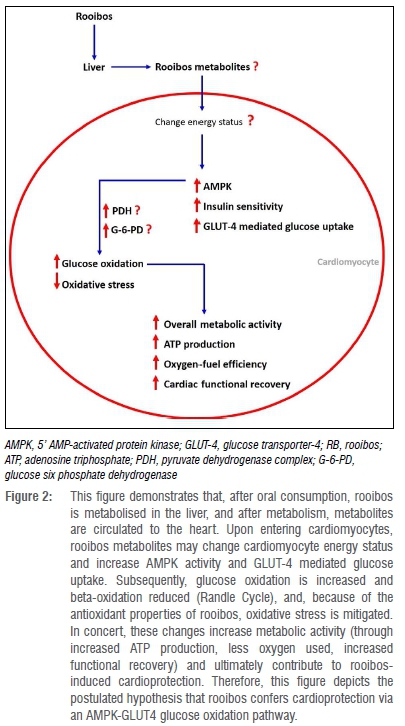
As a consequence of the feedback inhibition of increased glucose oxidation on beta-oxidation (also referred to as the Randle relationship)37, rooibos may reduce fatty acid uptake/oxidation9. In concert, these processes allow the myocardium to have a reasonably high cardiac mitochondrial activity, and consume less oxygen in order to produce sufficient ATP production (Figure 2). Although not tested, this hypothesis could explain the enhanced myocardial functional recovery after ischaemia-reperfusion25, and the preserved cardiomyocyte ATP production9 as previously reported. Therefore, in Figure 2, a hypothesis is depicted contending that rooibos may alter the overall cardiomyocyte energy status, and act via an AMPK-GLUT-4 glucose oxidation pathway to confer cardioprotection. Future studies could investigate this hypothesis, and test whether rooibos extracts are able to activate this pathway to execute its favourable impact on cardiac energy substrate utilisation, and confer cardioprotection.
Furthermore, considering that pyruvate dehydrogenase53,54 and glucose-6-phosphate dehydrogenase55 are regulators of glucose oxidation, it should also be investigated whether rooibos modulates these enzymes in order to activate this AMPK-GLUT-4 glucose oxidation pathway. Merely enhancing myocardial glucose oxidation may not solely account for rooibos-induced cardioprotection - other factors, such as reduced apoptosis signalling9,10,23,25 mitochondrial connexin-4356 and cardiolipin57, G-protein-coupled receptor kinase-258 and the reduction of reactive oxygen species21,59 may also contribute. There is a need for therapeutic interventions that confer cardioprotection, particularly those that can be consumed through dietary intake of rooibos. However, the cardioprotective properties of rooibos are not well described and the exact underlying mechanisms remain to be delineated.60 Therefore, ongoing research is necessary to aid in the understanding of how rooibos confers cardioprotection and studies investigating the underlying mechanisms should be a priority. The identification of these underlying mechanisms would contribute to the body of knowledge on rooibos-induced cardioprotection, and possibly facilitate the use of rooibos as an accepted cardioprotective intervention in the clinical setting.
Conclusions
Studies on rooibos-induced cardioprotection, particularly those studies using models of diabetic cardiomyopathy and myocardial ischaemic injury, were reviewed. Based on the evidence presented and discussed in this review, it is hypothesised that rooibos may alter myocardial energy status, acting via an AMPK-GLUT-4 glucose oxidation pathway to confer cardioprotection. Future studies could further investigate this hypothesis and the activation of the proposed pathway in a suitable experimental model of cardiomyopathy.
Acknowledgement
I thank the South African Rooibos Council (SARC), South African Medical Research Council (SA-MRC), and the Faculty of Medicine and Health Sciences, Stellenbosch University, for financial support.
Disclosure statement
G.J.M. discloses that he was awarded a research grant from the South African Rooibos Council.
References
1.Gerczuk PZ, Kloner RA. An update on cardioprotection: A review of the latest adjunctive therapies to limit myocardial infarction size in clinical trials. J Am Coll Cardiol. 2012;59(11):969-978. [ Links ]
2.Habertheuer A, Kocher A, Laufer G, Andreas M, Szeto WY, Petzelbauer P, et al. Cardioprotection: A review of current practice in global ischemia and future translational perspective. Biomed Res Int. 2014;2014, Art. #325725, 11 pages. https://doi.org/10.1155/2014/325725 [ Links ]
3.Shaikh F, Dupuis LL, Alexander S, Gupta A, Mertens L, Nathan PC. Cardioprotection and second malignant neoplasms associated with dexrazoxane in children receiving anthracycline chemotherapy: A systematic review and meta-analysis. J Natl Cancer Inst. 2016;108(4), djv357, 11 pages. https://doi.org/10.1093/jnci/djv357 [ Links ]
4.Spath NB, Mills NL, Cruden NL. Novel cardioprotective and regenerative therapies in acute myocardial infarction: A review of recent and ongoing clinical trials. Future Cardiol. 2016;12(6):655-672. https://doi.org/10.2217/fca-2016-0044 [ Links ]
5.Hausenloy DJ, Barrabes JA, Botker HE, Davidson SM, Di Lisa F, Downey J, et al. Ischaemic conditioning and targeting reperfusion injury: A 30 year voyage of discovery. Basic Res Cardiol. 2016;111(6), Art. #70, 24 pages. https://doi.org/10.1007/s00395-016-0588-8 [ Links ]
6.Hausenloy DJ, Yellon DM. Ischaemic conditioning and reperfusion injury. Nat Rev Cardiol. 2016;13(4):193-209. https://doi.org/10.1038/nrcardio.2016.5 [ Links ]
7.Nduhirabandi F, Lamont K, Albertyn Z, Opie LH, Lecour S. Role of toll-like receptor 4 in melatonin-induced cardioprotection. J Pineal Res. 2016;60(1):39-47. https://doi.org/10.1111/jpi.12286 [ Links ]
8.Maarman G, Blackhurst D, Thienemann F, Blauwet L, Butrous G, Davies N, et al. Melatonin as a preventive and curative therapy against pulmonary hypertension. J Pineal Res. 2015;59(3):343-353. https://doi.org/10.1111/jpi.12263 [ Links ]
9.Johnson R, Dludla P, Joubert E, February F, Mazibuko S, Ghoor S, et al. Aspalathin, a dihydrochalcone C-glucoside, protects H9c2 cardiomyocytes against high glucose induced shifts in substrate preference and apoptosis. Mol Nutr Food Res. 2016;60(4):922-934. https://doi.org/10.1002/mnfr.201500656 [ Links ]
10.Dludla PV, Muller CJ, Louw J, Joubert E, Salie R, Opoku AR, et al. The cardioprotective effect of an aqueous extract of fermented rooibos (Aspalathus linearis) on cultured cardiomyocytes derived from diabetic rats. Phytomedicine. 2014;21(5):595-601. https://doi.org/10.1016/j.phymed.2013.10.029 [ Links ]
11.Joubert E, De Beer D. Rooibos (Aspalathus linearis) beyond the farm gate: From herbal tea to potential phytopharmaceutical. S Afr J Bot. 2011;77(4):869-886. https://doi.org/10.1016/j.sajb.2011.07.004 [ Links ]
12.Joubert E, Schulz H. Production and quality aspects of rooibos tea and related products. A review. J Appl Bot Food Qual. 2006;80(2):138-144. [ Links ]
13.Joubert E, Winterton P, Britz TJ, Gelderblom WC. Antioxidant and pro-oxidant activities of aqueous extracts and crude polyphenolic fractions of rooibos (Aspalathus linearis). J Agric Food Chem. 2005;53(26):10260-10267. https://doi.org/10.1021/jf051355a [ Links ]
14.Smith C, Swart AC. Rooibos (Aspalathus linearis) facilitates an anti-inflammatory state, modulating IL-6 and IL-10 while not inhibiting the acute glucocorticoid response to a mild novel stressor in vivo. J Funct Foods. 2016;27:42-54. https://doi.org/10.1016/j.jff.2016.08.055 [ Links ]
15.McKay DL, Blumberg JB. A review of the bioactivity of South African herbal teas: Rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia). Phytother Res. 2007;21(1):1-16. https://doi.org/10.1002/ptr.1992 [ Links ]
16.Marnewick JL, Van der Westhuizen FH, Joubert E, Swanevelder S, Swart P, Gelderblom WCA. Chemoprotective properties of rooibos (Aspalathus linearis), honeybush (Cyclopia intermedia) herbal and green and black (Camellia sinensis) teas against cancer promotion induced by fumonisin B-1 in rat liver. Food Chem Toxicol. 2009;47(1):220-229. https://doi.org/10.1016/j.fct.2008.11.004 [ Links ]
17.Marnewick J, Joubert E, Joseph S, Swanevelder S, Swart P, Gelderblom W. Inhibition of tumour promotion in mouse skin by extracts of rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia), unique South African herbal teas. Cancer Lett. 2005;224(2):193-202. https://doi.org/10.1016/j.canlet.2004.11.014 [ Links ]
18.Snijman PW, Joubert E, Ferreira D, Li XC, Ding YQ, Green IR, et al. Antioxidant activity of the dihydrochalcones aspalathin and nothofagin and their corresponding flavones in relation to other rooibos (Aspalathus linearis) flavonoids, epigallocatechin gallate, and trolox. J Agric Food Chem. 2009;57(15):6678-6684. https://doi.org/10.1021/jf901417k [ Links ]
19.Tangney CC, Rasmussen HE. Polyphenols, inflammation, and cardiovascular disease. Curr Atheroscler Rep. 2013;15(5):324. https://doi.org/10.1007/s11883-013-0324-x [ Links ]
20.Geleijnse JM, Hollman PCH. Flavonoids and cardiovascular health: Which compounds, what mechanisms? Am J Clin Nutr. 2008;88(1):12-13. https://doi.org/10.1093/ajcn/88.1.12 [ Links ]
21.Marnewick JL, Rautenbach F, Venter I, Neethling H, Blackhurst DM, Wolmarans P, et al. Effects of rooibos (Aspalathus linearis) on oxidative stress and biochemical parameters in adults at risk for cardiovascular disease. J Ethnopharmacol. 2011;133(1):46-52. https://doi.org/10.1016/j.jep.2010.08.061 [ Links ]
22.Persson IAL, Persson K, Hagg S, Andersson RGG. Effects of green tea, black tea and rooibos tea on angiotensin-converting enzyme and nitric oxide in healthy volunteers. Public Health Nutr. 2010;13(5):730-737. https://doi.org/10.1016/j.jep.2010.08.061 [ Links ]
23.Dludla PV, Muller CJ, Joubert E, Louw J, Gabuza KB, Huisamen B, et al. Phenylpyruvic acid-2-O-beta-D-glucoside attenuates high glucose-induced apoptosis in h9c2 cardiomyocytes. Planta Med. 2016;82(17):1468-1474. https://doi.org/10.1055/s-0042-110856 [ Links ]
24.Krishna KM, Annapurna A, Gopal GS, Chalam CRV, Madan K, Kumar VK, et al. Partial reversal by rutin and quercetin of impaired cardiac function in streptozotocin-induced diabetic rats. Can J Physiol Pharmacol. 2005;83(4):343-355. https://doi.org/10.1139/y05-009 [ Links ]
25.Pantsi WG, Marnewick JL, Esterhuyse AJ, Rautenbach F, Van Rooyen J. Rooibos (Aspalathus linearis) offers cardiac protection against ischaemia/reperfusion in the isolated perfused rat heart. Phytomedicine. 2011;18(14):1220-1228. https://doi.org/10.1016/j.phymed.2011.09.069 [ Links ]
26.Webster I, Imperial EG, Westcott C, Strijdom H. The cardiovascular effects of Aspalathus linearis supplementation in male Wistar rats receiving fixed-dose combination first-line antiretroviral therapy. Cardiovasc J Afr. 2019;30:1-8. https://doi.org/10.5830/cvja-2018-075 [ Links ]
27.Liperoti R, Vetrano DL, Bernabei R, Onder G. Herbal medications in cardiovascular medicine. J Am Coll Cardiol. 2017;69(9):1188-1199. https://doi.org/10.1016/j.jacc.2016.11.078 [ Links ]
28.Harvey PA, Leinwand LA. The cell biology of disease: Cellular mechanisms of cardiomyopathy. J Cell Biol. 2011;194(3):355-365. https://doi.org/10.1083/jcb.201101100 [ Links ]
29.Boudina S, Abel ED. Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord. 2010;11(1):31-39. https://doi.org/10.1007/s11154-010-9131-7 [ Links ]
30.Aravindhan K, Bao WK, Harpel MR, Willette RN, Lepore JJ, Jucker BM. Cardioprotection resulting from glucagon-like peptide-1 administration involves shifting metabolic substrate utilization to increase energy efficiency in the rat heart. PLoS ONE. 2015;10(6), e0130894, 18 pages. https://doi.org/10.1371/journal.pone.0130894 [ Links ]
31.Jaswal JS, Keung W, Wang W, Ussher JR, Lopaschuk GD. Targeting fatty acid and carbohydrate oxidation - A novel therapeutic intervention in the ischemic and failing heart. Biochim Biophys Acta Mol Cell Res. 2011;1813(7):1333-1350. https://doi.org/10.1016/j.bbamcr.2011.01.015 [ Links ]
32.Gray S, Kim JK. New insights into insulin resistance in the diabetic heart. Trends Endocr Metab. 2011;22(10):394-403. https://doi.org/10.1016/j.tem.2011.05.001 [ Links ]
33.Abel ED. Myocardial insulin resistance and cardiac complications of diabetes. Curr Drug Targets Immune Endocr Metabol Disord. 2005;5(2):219-226. https://doi.org/10.2174/1568008054064869 [ Links ]
34.Liang JL, Xiao DZ, Liu XY, Lin QX, Shan ZX, Zhu JN, et al. High glucose induces apoptosis in AC16 human cardiomyocytes via macrophage migration inhibitory factor and c-Jun N-terminal kinase. Clin Exp Pharmacol Physiol. 2010;37(10):969-973. https://doi.org/10.1111/j.1440-1681.2010.05420.x [ Links ]
35.Kawahito S, Kitahata H, Oshita S. Problems associated with glucose toxicity: Role of hyperglycemia-induced oxidative stress. World J Gastroenterol. 2009;15(33):4137-4142. https://doi.org/10.3748/wjg.15.4137 [ Links ]
36.Singh AB, Guleria RS, Nizamutdinova IT, Baker KM, Pan J. High glucose-induced repression of RAR/RXR in cardiomyocytes is mediated through oxidative stress/JNK signaling. J Cell Physiol. 2012;227(6):2632-2644. https://doi.org/10.1002/jcp.23005 [ Links ]
37.Hue L, Taegtmeyer H. The Randle Cycle revisited: A new head for an old hat. Am J Physiol Endocr Metab. 2009;297(3):E578-E591. https://doi.org/10.1152/ajpendo.00093.2009 [ Links ]
38.Ceriello A, Esposito K, Giugliano D. Glucose metabolism and hyperglycemia. Am J Clin Nutr. 2008;87(1):217S-222S. https://doi.org/10.1093/ajcn/87.1.217s [ Links ]
39.Lloyd S, Brocks C, Chatham JC. Differential modulation of glucose, lactate, and pyruvate oxidation by insulin and dichloroacetate in the rat heart. Am J Physiol Heart Circ Physiol. 2003;285(1):H163-H172. https://doi.org/10.1152/ajpheart.01117.2002 [ Links ]
40.Bhashyam S, Fields AV, Patterson B, Testani JM, Chen L, Shen YT, et al. Glucagon-like peptide-1 increases myocardial glucose uptake via p38alpha MAP kinase-mediated, nitric oxide-dependent mechanisms in conscious dogs with dilated cardiomyopathy. Circ Heart Fail. 2010;3(4):512-521. https://doi.org/10.1161/circheartfailure.109.900282 [ Links ]
41.Ansley DM, Wang BH. Oxidative stress and myocardial injury in the diabetic heart. J Pathol. 2013;229(2):232-241. [ Links ]
42.Wolff AA, Rotmensch HH, Stanley WC, Ferrari R. Metabolic approaches to the treatment of ischemic heart disease: The clinicians' perspective. Heart Fail Rev. 2002;7(2):187-203. https://doi.org/10.1023/a:1015384710373 [ Links ]
43.Fillmore N, Mori J, Lopaschuk GD. Mitochondrial fatty acid oxidation alterations in heart failure, ischaemic heart disease and diabetic cardiomyopathy. Br J Pharmacol. 2014;171(8):2080-2090. https://doi.org/10.1111/bph.12475 [ Links ]
44.Maarman G, Marais E, Lochner A, Du Toit EF. Effect of chronic CPT-1 inhibition on myocardial ischemia-reperfusion injury (I/R) in a model of diet-induced obesity. Cardiovasc Drugs Ther. 2012;26(3):205-216. https://doi.org/10.1007/s10557-012-6377-1 [ Links ]
45.Bhandari B, Subramanian L. Ranolazine, a partial fatty acid oxidation inhibitor, its potential benefit in angina and other cardiovascular disorders. Recent Pat Cardiovasc Drug Discov. 2007;2(1):35-39. https://doi.org/10.2174/157489007779606095 [ Links ]
46.Lopaschuk GD. AMP-activated protein kinase control of energy metabolism in the ischemic heart. Int J Obes. 2008;32:S29-S35. https://doi.org/10.1038/ijo.2008.120 [ Links ]
47.Beltran-Debon R, Rull A, Rodriguez-Sanabria F, Iswaldi I, Herranz-Lopez M, Aragones G, et al. Continuous administration of polyphenols from aqueous rooibos (Aspalathus linearis) extract ameliorates dietary-induced metabolic disturbances in hyperlipidemic mice. Phytomedicine. 2011;18(5):414-424. https://doi.org/10.1016/j.phymed.2010.11.008 [ Links ]
48.Mazibuko SE, Muller CJ, Joubert E, De Beer D, Johnson R, Opoku AR, et al. Amelioration of palmitate-induced insulin resistance in C(2)C(1)(2) muscle cells by rooibos (Aspalathus linearis). Phytomedicine. 2013;20(10):813-819. https://doi.org/10.1016/j.phymed.2013.03.018 [ Links ]
49.Mazibuko-Mbeje SE, Dludla PV, Roux C, Johnson R, Ghoor S, Joubert E, et al. Aspalathin-enriched green rooibos extract reduces hepatic insulin resistance by modulating PI3K/AKT and AMPK pathways. Int J Mol Sci. 2019;20(3), Art. #633, 16 pages. https://doi.org/10.3390/ijms20030633 [ Links ]
50.Ulicna O, Vancova O, Kucharska J, Janega P, Waczulikova I. Rooibos tea (Aspalathus linearis) ameliorates the CCl4-induced injury to mitochondrial respiratory function and energy production in rat liver. Gen Physiol Biophys. 2019;38(1):15-25. https://doi.org/10.4149/gpb_2018037 [ Links ]
51.Qi D, Young LH. AMPK: Energy sensor and survival mechanism in the ischemic heart. Trends Endocr Metab TEM. 2015;26(8):422-429. https://doi.org/10.1016/j.tem.2015.05.010 [ Links ]
52.Kawano A, Nakamura H, Hata SI, Minakawa M, Miura Y, Yagasaki K. Hypoglycemic effect of aspalathin, a rooibos tea component from Aspalathus linearis, in type 2 diabetic model db/db mice. Phytomedicine. 2009;16(5):437-443. https://doi.org/10.1016/j.phymed.2008.11.009 [ Links ]
53.Lydell CP, Chan A, Wambolt RB, Sambandam N, Parsons H, Bondy GP, et al. Pyruvate dehydrogenase and the regulation of glucose oxidation in hypertrophied rat hearts. Cardiovasc Res. 2002;53(4):841-851. https://doi.org/10.1016/s0008-6363(01)00560-0 [ Links ]
54.Zhang S, Hulver MW, McMillan RP, Cline MA, Gilbert ER. The pivotal role of pyruvate dehydrogenase kinases in metabolic flexibility. Nutr Metab (Lond). 2014;11(1), Art. #10, 9 pages. https://doi.org/10.1186/1743-7075-11-10 [ Links ]
55.Van de Werve G, Lange A, Newgard C, Mechin MC, Li Y, Berteloot A. New lessons in the regulation of glucose metabolism taught by the glucose 6-phosphatase system. Eur J Biochem. 2000;267(6):1533-1549. https://doi.org/10.1046/j.1432-1327.2000.01160.x [ Links ]
56.Rodriguez-Sinovas A, Ruiz-Meana M, Denuc A, Garcia-Dorado D. Mitochondrial Cx43, an important component of cardiac preconditioning. Biochim Biophys Acta Biomembr. 2018;1860(1):174-181. https://doi.org/10.1016/j.bbamem.2017.06.011 [ Links ]
57.Paradies G, Paradies V, Ruggiero FM, Petrosillo G. Mitochondrial bioenergetics and cardiolipin alterations in myocardial ischemia-reperfusion injury: Implications for pharmacological cardioprotection. Am J Physiol Heart Circ Physiol. 2018;315(5):H1341-H1352. https://doi.org/10.1152/ajpheart.00028.2018 [ Links ]
58.Sato PY, Chuprun JK, Grisanti LA, Woodall MC, Brown BR, Roy R, et al. Restricting mitochondrial GRK2 post-ischemia confers cardioprotection by reducing myocyte death and maintaining glucose oxidation. Sci Signal. 2018;11(560), eaau0144. https://doi.org/10.1126/scisignal.aau0144 [ Links ]
59.Villano D, Pecorari M, Testa MF, Raguzzini A, Stalmach A, Crozier A, et al. Unfermented and fermented rooibos teas (Aspalathus linearis) increase plasma total antioxidant capacity in healthy humans. Food Chem. 2010;123(3):679-683. https://doi.org/10.1016/j.foodchem.2010.05.032 [ Links ]
60.Smith C, Swart A. Aspalathus linearis (rooibos) - a functional food targeting cardiovascular disease. Food Funct. 2018;9(10):5041-5058. https://doi.org/10.1039/c8fo01010b [ Links ]
 Correspondence:
Correspondence:
Gerald Maarman
Email: gmaarman@sun.ac.za
Received: 08 Mar. 2018
Revised: 10 Apr. 2019
Accepted: 12 Apr. 2019
Published: 30 July 2019
EDITOR: John Butler-Adam
FUNDING: South African Rooibos Council; South African Medical Research Council; Stellenbosch University














