Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.112 no.1-2 Pretoria Jan./Fev. 2016
http://dx.doi.org/10.17159/sajs.2016/20150139
RESEARCH ARTICLE
Radiological and genetic analysis of a Late Iron Age mummy from the Tuli Block, Botswana
Frank J. RühliI; Maryna SteynII, III; Morongwa N. MosothwaneII, IV; Lena ÖhrströmI; Molebogeng K. BodibaII; Abigail BouwmanI
I'Institute of Evolutionary Medicine, University of Zurich, Zurich, Switzerland
IIForensic Anthropology Research Centre, Department of Anatomy, University of Pretoria, Pretoria, South Africa
IIISchool of Anatomical Sciences, University of the Witwatersrand, Johannesburg, South Africa
IVArchaeology Unit, University of Botswana, Gaborone, Botswana
ABSTRACT
Mummified human remains are valuable sources of information on past populations. Here we report on the radiological and molecular findings of a partially mummified individual found in northern Botswana. This desiccated mummy from the Tuli region is the first to have been reported from this region. The remains were those of an older male adult of African origin. He was interred in a tightly flexed position and wrapped in an animal skin. Computerised tomography (CT) scanning revealed that none of the internal organs was preserved. Multiple post-mortem alterations are seen, but apart from some degenerative changes of the lower vertebral column, the axial skeleton has remained intact. The advanced osteophytosis suggests an older age than what was previously estimated. The aDNA analysis confirms Sotho-Tswana and possibly Khoesan genetic relatedness, as could be expected from individuals from that region. These results represent one of the first CT scans of a mummified individual from southern Africa, and also the first successful aDNA extraction from such remains.
Keywords: aDNA; computerised tomography; mummified remains; population relationships; southern Africa
Introduction
Mummified remains are uncommon finds, especially in southern Africa. They are valuable sources of information on past populations1-5, and they also tend to evoke considerable emotion as they provide a very vivid view of life in the past6. Such remains in southern Africa are usually preserved as a result of desiccation, and they are thus found in caves, rock shelters or other areas where they are protected from water. Many other factors also contribute to mummification, such as level of humidity, temperature, and covering or clothing. For example, mummified remains have been recovered from a rock shelter in the Kouga region7, the Historic Cave in the Makapan Valley6 and Eland Cave8.
Recently, Mosothwane9 described the discovery of a partially mummified individual from the Tuli region of Botswana - the first such remains found in Botswana (Figure 1). The site is located in a privately owned game lodge, the Northern Tuli Game Reserve, approximately 20 km west of the Shashe-Limpopo confluence. Bone collagen from the mummy was radiocarbon dated to 140±30 BP or AD 1675-1735 (2-sigma calibrated) or alternatively from AD 1800 to post-1950 (Beta Analytic; 400348).

The remains were found in a shallow grave at the base of a cliff. The cliff provided a roof directly above the grave which then limited the amount of rain water falling directly on the grave. The cliff also provided full-day shade coverage above the grave. The body was tightly flexed at the hips and elbows and wrapped with a cow skin with the furry part against the human remains. It was then tied with a rope made of plant fibre.
The remains were nearly complete, except for parts of the right femur and some phalanges which may have been carried off by scavenging animals. Skin, tendons, hair and nails were preserved. Based on the facial hair and skeletal characteristics that were visible, the remains were assessed to be that of a male adult aged between 40 and 55 years. When the animal skin was removed as part of the detailed study described below, soft tissues of the genital region could be visualised, further confirming the sex of the individual. The initial publication by Mosothwane9 only provided a basic morphological description of the remains, with no detailed or specialised analyses.
Specialised medical analysis on mummified remains including diagnostic imaging technologies such as X-ray or computerised tomography (CT)10, isotopic analyses11,12, DNA extraction13-15 and histological analyses16 have become the standard for the assessment of mummies across the world and to a limited extent also in southern Africa6,7. Such studies allow us to answer more questions on the origins, diseases, and reasons for preservation of the remains.
The aim of this paper is to report on the outcomes of specialised analyses of the Tuli mummy, including CT scanning and aDNA analysis.
Materials and methods
The Tuli mummy is currently housed at the Botswana National Museum in Gaborone, Botswana. The mummified remains were radiographically and macroscopically analysed. In addition, various samples were extracted. These samples included those for aDNA extraction from soft tissue and teeth and radiocarbon dating from bone (collagen). aDNA comparative samples were obtained from the GenBank database. Overall the macroscopic sample preservation was extremely good as indicated by the results of the radiographic and osteometric analysis. Likewise, bone collagen preservation was good and yielded reliable radiocarbon dates.
The remains of the Tuli individual were examined using a clinical CT scanner (Toshiba) at the Bokamoso Private Hospital in Gaborone in August 2O11. Imaging parameters were as follows: 512x512 matrix; 5 mm slice thickness, 80 mA tube current, 120 kV tube voltage and 1.087x1.087 pixel spacing. In total, three series were obtained: axial (194 images), coronal (38 images) and sagittal (30 images). The data sets were processed with OsiriX-64 bit (version 5.8.5) software, including multiplanar reconstructions and three-dimensional volume rendering.
After the remains were scanned, the cow hide was carefully removed and several samples were collected at the archaeology laboratory of the University of Botswana. This sampling was done under controlled circumstances, with investigators wearing masks and gloves so as to avoid contaminating the remains. However, it should be taken into account that the remains were not excavated under such controlled conditions, but it can be assumed that the cow hide would have to some extent protected the remains from contamination. These samples were exported and DNA extraction was undertaken at dedicated aDNA laboratories in Zurich (Switzerland) at the Institute for Evolutionary Medicine. Other samples were sent to Beta Analytic Inc. (Miami, FL, USA) for radiocarbon dating.
Both bone and soft tissue were used to extract DNA and this extraction was performed under strict, sterile conditions. A modified phenolchloroform method17,18 was used, in which elution of the DNA was done by using a column tube with a chaotropic agent. DNA concentration was measured using a digital spectrophotometer (Qubit). This method was followed by standard polymerase chain reaction (PCR) amplification of the SRY gene and real-time PCR for the AMELX and AMELY genes to determine genetic sex. Standard PCR amplification of ancestry-specific mtDNA markers (HVRI, HVRII, COII) was performed using a HotStart polymerase and PCR primers that targeted overlapping gene regions. Table 1 is a summary of the mtDNA primers used (with forward and reverse sequences), the optimal annealing temperature for each primer pair, as well as the expected size of the PCR product after amplification. The PCR temperature conditions were as follows: 98 °C, 3 s, (98 °C, 10 s, Tm, 20 s, 72 °C, 15 s) x 46 cycles, 72 °C, 5 min, 10°C, ∞.

PCR was followed by cloning in Escherichia coli bacteria, purification and automated Sanger sequencing. CLCBio (www.clcbio.org) was used to view and edit the sequences and to create contigs. Then the MEGA6 platform19 was used to create alignments with matching reference sequences from GenBank; the algorithm SplitsTree4 was used to construct an allele network.
Results
Radiocarbon dating
Two possible dates were obtained, based on where the samples fell on the calibration curve: 140±30 BP or AD 1675-1735 (2-sigma calibrated) and AD 1800 to post-1950. The earlier date seems more likely based on the few available potsherds, although the cultural material does not help to refine the age of the burial. Further dating, perhaps of the wooden posts, may provide more clarity. Broadly speaking it can be concluded that the age of this burial fell into the Late Iron Age.20
CT scanning
The quality of the CT scans (slice thicknesses of several millimetres) did not allow a definite anatomical-pathological assessment of this individual. Any diagnosis thus has to be taken as provisional.
Several bones and bone parts were missing, including the proximal part of the right femur, some carpals, metacarpals and phalanges of the left hand and some phalanges of the right hand. These parts probably were lost as a result of wild animals burrowing into the grave.9 The mummy was in a foetal position, with the arms and legs flexed and the head inclined forward. The left hand was under the head, close to the angle of the jaw, and the right hand was found in front of the mouth area. The chest and abdomen were heavily compressed, with the entire left side of the chest and abdomen severely damaged. The distal part of the right femur was out of position, in front of the body but rotated. The rotation was a result of post-mortem damage, most probably caused by animals burrowing into the grave. Figure 2 shows a volume-rendered image of the remains, in which the reversed position of the distal femur can be seen. The rest of the femur had been broken off post-mortem but was recovered next to the burial. Skin lesions and damage were found at multiple parts of the body.
As far as the skull specifically was concerned, the facial bones were found to be intact but no remnants of the brain could be identified (Figure 3). The lamina cribrosa as well as the foramen magnum itself were intact, which mostly excludes artificial excerebration, as seen in, for example, Egyptian mummies. The cranial cavity was almost completely filled with radiodense material (rangingfrom -177 to +344 HU); only the right cranial cavity showed a 20-mm wide empty region. The infill was probably soil, although some of the debris potentially included brain or dura remnants. The fact that the soil had accumulated on the left side of the cranial cavity with empty space on the right, indicates that the remains had been buried lying on the right side from the start (i.e. it is unlikely that the body fell over after initially having been positioned in a sitting position, for example). The scalp is mostly intact.
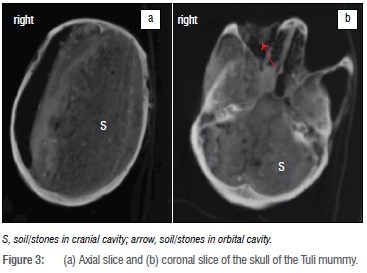
All sinuses as well as the mastoids were well pneumatised. The contents of both orbits were not preserved; while the left orbit was empty the right orbit was filled with soil. The mouth was slightly open and empty, but the nasal cavity was filled with soil.
Because of the relatively large slice thicknesses of the CT scans, assessment of the oral structures and dental pathologies was difficult.
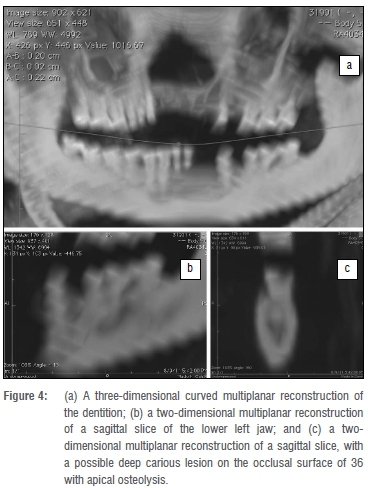
Several teeth were lost, probably post-mortem (lower left incisors, upper right incisors, upper left incisors and canine, upper left second premolar, lower left first premolar) (Figure 4). These teeth were not found elsewhere in the wrapping and it is assumed that they may have fallen out and been lost. The alveolar bone of the maxilla and mandible seemed to be lost around the incisival area. It seemed that there was heavy abrasion or wear on the molars, and possibly a deep carious lesion on the occlusal surface of the left lower first molar, with possible ante-mortem loss of the second molar.
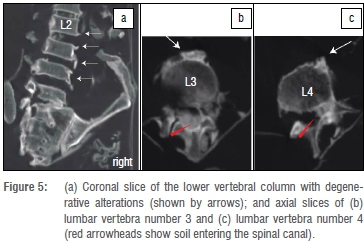
The vertebral canal was filled with soil and possibly some remains of dura. The dens of the axis was intact but slightly moved forward within the atlanto-axial joint. The disc spaces from L2 to L5 were widened and an anterolisthesis L4 to L5 (10 mm) was seen (post-mortem alterations). Multiple osteophytes could be observed on the lumbar spine (L2-L5; very pronounced at L3 and L4) (Figure 5), mainly at the right frontal side of the vertebral bodies. No further degenerative changes were seen on the vertebral column.
The pelvic girdle was compressed. While the right hip joint was damaged, the left hip was intact with the head of the femur positioned inside the acetabulum. The joint space was filled with soil. The sacroiliac joints were intact, and some soil entered in the joint space on the left side.
The chest was heavily compressed, mainly on the left side (Figure 6). The left shoulder girdle was moved dorsally whereas the right side was displaced more anteriorly. Both clavicles were rotated dorsally. While the costo-vertebral joints remained intact the ribs were luxated forward in many places and showed multiple fractures. These distortions and fractures are all the result of post-mortem/perimortem damage. The left thoracic cavity was filled with soil; the right thoracic cavity was empty. No internal organs nor the mediastinum could be differentiated. Diverse large skin lesions (most likely post-mortem as a result of handling and taphonomic alterations) were found in the upper chest and neck area.
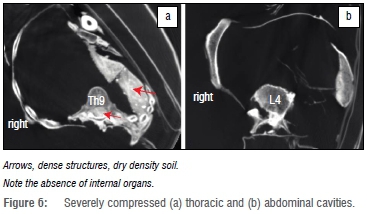
While the left part of the abdomen was compressed, the right was mainly intact (Figure 6b). No internal organs could be identified. Again, the radiodense material (soil) was seen in the lesser pelvis.
Both arms were in a flexed position, with the elbow joints intact and showing no degenerative changes. Both knee joints were fully luxated. However, the patello-tibial as well as the proximal tibio-fibular joints remained intact and the right patellar ligament was visible. Both feet were in eversion, with subluxed ankle joints. This position may be the result of tightly flexing the body while wrapping it in the animal skin. Radiodense material was visible in the distal right femur; most likely surrounding soil had entered into the open long bone, but it might also have been liquid material which thickened postmortally.
Molecular analyses
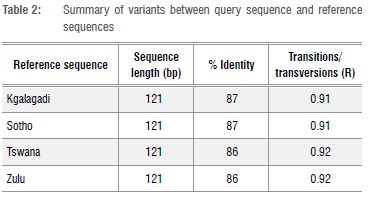
Some minimal contamination of the PCR reactions was noted, namely by microbial genes such as Candida, Mycobacter and Gluconacetobacter, which was verified through comparison with GenBank sequences. Contaminated samples were not used in further analyses. The DNA sequences obtained were not all usable, and owing to the degraded nature of aDNA, only the HVRI sequences could be used. Sequences were then queried against the GenBank database and apart from the above-mentioned contaminated samples, all shared some sequence similarities with human mtDNA sequences. Among the matches were mtDNA sequences from a Kgalagadi individual, Sotho, Khoi, Tswana and Zulu individuals. The names of these sequences, named after cultural affinities, were based on self-identification of the donors. Table 2 shows a summary of the alignments between the Tuli mummy and each of the above-mentioned reference sequences. Figure 7 depicts these alignments.
When compared to the revised Cambridge Reference Sequence, the mtDNA sequence obtained from the Tuli mummy was assigned to haplogroup L0, without any further resolution as to which sub-haplogroup it may belong to, owing to the poor quality of the sequences. Therefore, in order to better understand the likely genetic heritage of the Tuli mummy, the sequences were used to create an alignment that also included mtDNA control region sequences from other African sequences, accessed via GenBank.21 Most of these were southern African and were added for better comparison. This alignment was used to create a haplotype network, using the program SplitsTree422, which is presented in Figure 8. The abbreviations to the sequence labels denote countries of origin or group affinities as reported by anonymous DNA donors.
Discussion
Huffman20 describes burial practices in Zimbabwe, in which burials of sacred leaders were said to receive special attention. After such a leader died, his body was not immediately interred but instead was mummified, usually by slowly drying it over a low fire. This preserved body would then be wrapped in a cloth or bull hide, and buried at the same time that the successor came into power. It seems possible that a similar process was followed here, and that the body may have been cured or smoked over a low fire which lead to its preservation. What is not clear is whether the internal organs were removed before this process. In the Tuli mummy, no evidence of the presence of internal organs was found. It is possible that the organs have decayed completely, but the possibility that they were removed prior to desiccation should be considered. The extreme flexion of the body is interesting, as is the fact that this level of flexion could be achieved possibly such a long time after death; although wrapping the body in an animal skin and then tying it together with a rope assisted in the process. On the other hand, the fact that other biological material (e.g. buried wooden poles, the skin wrapping the body and plant fibre used to tie the body) was found in good preservation raises the possibility that desiccation of the remains may have occurred naturally.
The individual shows multiple post-mortem alterations, including a compressed thorax/abdomen as well as missing bone parts. However, apart from some degenerative changes of the lower vertebral column, the axial skeleton remained intact. The presence of these multiple degenerative changes warrants a revision of the initial age estimation, indicating that the individual was probably older than what was initially thought. This revision is supported by the observation of advanced dental wear. An age estimate of over 50 years is thus deemed to be more accurate. The Tuli mummy could not be fully evaluated based only on the available CT scans. Difficulties were caused by the slice thickness and partially incongruent scan series, as well as the soil, which entered at diverse regions. Unfortunately, limited facilities are available in Gaborone for this purpose and specialised analyses remain difficult.
The aDNA alignments (Figure 7) show that generally the query sequence, when compared to the reference sequences, presented an 11 nucleotide gap at the query sequence positions 45 to 55, as well as deletions at positions 72 and 105. Only 122 positions could be aligned with relative significance and the transition/transversion bias of 0.91 denotes a higher number of transitions over transversions; however, this ratio is not significant because of the lengths of these alignments and also because of the 11 nucleotide gap.
It is also evident from the median joining network presented in Figure 8 that there is some genetic relatedness between the Tuli mummy and the present day Sotho/Tswana and Khoesan. It also appears that the one Kgalagadi sequence represents an ancestral node that is common to the Tuli sequence as well as that of the Sotho, Tswana and Khoesan. This association is not surprising. The DNA analysis does not imply that the Tuli individual was indeed Khoesan or Sotho/Tswana, but does show at least that there are some mtDNA sequences shared between the Tuli mummy and the present day Khoesan and Sotho/Tswana. It is therefore not impossible that this individual shared maternal genetic heritage with either (or both) of the above-mentioned groups. The similarity shared between this individual and the Zulu is also noteworthy as it could suggest shared ancestry as a result of the migration of different groups (both foragers and farmers), that may have resulted in population interaction and genetic exchange. The assignment of this individual to haplogroup L0 only indicates that he was of sub-Saharan African origin, but this is the extent of the secure interpretations because of the poor quality of the sequence obtained. The morphological analyses support this suggestion. Even so, these data will contribute to what will hopefully be a growing database of ancient DNA results from this region - a field which is still in its infancy in southern Africa.
Southern African mummies are rare finds, and detailed analyses even rarer. We have reported the first successful extraction of aDNA from such mummified remains, and included detailed CT scanning results. These may serve as reference for future such studies, which may include other such remains, for example the Kouga mummy.
Authors' contributions
F.J.R. is a specialist on mummies and took the samples for aDNA analysis, assisted with the CT scanning and the interpretation thereof, contributed to the writing, and provided funding in Switzerland; M.S. coordinated the project, took the lead in the writing of the manuscript and provided funding in South Africa and Botswana; M.N.M. discovered the mummy, interpreted the dating and archaeology, organised permission in Botswana and coordinated the project in Botswana; L.O. provided expert advice on the CT scans; M.K.B. performed the aDNA analysis (as part of her postgraduate studies) and A.B. oversaw and assisted with the aDNA analysis.
Acknowledgements
We acknowledge SANPAD and the Mäxi Foundation (Switzerland) for funding parts of the project. We also thank the radiologists at the Bokamoso Private Hospital for their assistance. The research of M.S. is funded by the National Research Foundation (NRF) of South Africa. Any opinions, findings and conclusions expressed are those of the authors and therefore the NRF does not accept any liability in regard thereto. The permit to analyse the mummy was granted by the Botswana Government through the Botswana National Museum.
References
1. Aufderheide AC, Rodríguez-Martín C. The Cambridge encyclopedia of human paleopathology. Cambridge: Cambridge University Press; 1998. [ Links ]
2. Ortner DJ. Identification of pathological conditions in human skeletal remains. 2nd ed. San Diego, CA: Elsevier Academic Press; 2003. http://dx.doi.org/10.1016/B978-012528628-2/50058-2 [ Links ]
3. Rühli FJ, Chhem RK, Böni T. Diagnostic paleoradiology of mummified tissues: Interpretation and pitfalls. Can Assoc Radiol J. 2004;55:218-227. [ Links ]
4. Asingh P, Lynnerup N. Grauballe Man: An Iron Age bog body revisited. Aarhus: Jutland Archaeological Society Publications; 2007. p. 49. [ Links ]
5. Rühli F, Böni T, Perlo J, Casanova F, Baias M, Egarter Vigl E, et al. Non-invasive spatial tissue discrimination in ancient mummies and bones in situ by portable nuclear magnetic resonance. J Cult Her. 2007;8:257-263. http://dx.doi.org/10.1016/j.culher.2007.03.002 [ Links ]
6. Esterhuysen AB, Sanders VM, Smith JM. Human skeletal and mummified remains from AD 1854 siege of Mugobane, Limpopo, South Africa. J Archaeol Sci. 2009;36:1038-1049. http://dx.doi.org/10.1016/j.jas.2008.12.006 [ Links ]
7. Steyn M, Binneman J, Loots M. The Kouga mummified human remains. S Afr Archaeol Bull. 2007;62:3-8. [ Links ]
8. Sealy J, Pfeiffer S, Yates R, Willmore K, Manhire A, Maggs T, et al. Hunter-gatherer child burials from the Pakhuis mountains, Western Cape: Growth, diet and burial practices in the Late Holocene. S Afr Archaeol Bull. 2000;55:32-43. http://dx.doi.org/10.2307/3888890 [ Links ]
9. Mosothwane MN. Tuli mummy: A preliminary report from northeastern Botswana. S Afr Archaeol Bull. 2011;66:157-160. [ Links ]
10. Lynnerup N. Methods in mummy research. Anthropol Anz. 2009;67:357-384. http://dx.doi.org/10.1127/0003-5548/2009/0028 [ Links ]
11. Knudson KJ, Tung TA, Nystrom KC, Price TD, Fullagar PD. The origin of the Juch'uypampa Cave mummies: Strontium isotope analysis of archaeological human remains from Bolivia. J Archaeol Sci. 2005;32(6):903-913. http://dx.doi.org/10.1016/j.jas.2005.01.007 [ Links ]
12. Finucane BC. Mummies, maize and manure: Multi-tissue stable isotope analysis of late prehistoric human remains from the Ayacucho Valley, Perú. J Archaeol Sci. 2007;34(12):2115-2124. http://dx.doi.org/10.1016/j.jas.2007.02.006 [ Links ]
13. Handt O, Krings M, Ward RH, Pääbo S. The retrieval of ancient human DNA sequences. Am J Hum Gen. 1993;59(2):368-376. [ Links ]
14. Burger J, Hummel S, Hermann B, Henke W. DNA preservation: A microsatellite-DNA study on ancient human skeletal remains. Electrophoresis. 1999;20:1722-1728. http://dx.doi.org/10.1002/(SICI)1522-2683(19990101)20:8<1722::AID-ELPS1722>3.0.CO;2-4 [ Links ]
15. Guhl F, Jaramillo C, Vallejo GA, Yockteng R, Cárdenas-Arroyo F, Fornaciari G, et al. Isolation of Trypanosoma cruzi DNA in 4,000-year-old mummified human tissue from northern Chile. Am J Phys Anthropol. 1999;108:401-407. http://dx.doi.org/10.1002/(SICI)1096-8644(199904)108:4<401::AID-AJPA2>3.0.CO;2-P [ Links ]
16. Shin DH, Youn M, Chang BS. Histological analysis on the medieval mummy in Korea. Forensic Sci Int. 2003;137(2-3):172-182. http://dx.doi.org/10.1016/S0379-0738(03)00335-9 [ Links ]
17. Sambrook J, MacCallum P Russell D. The condensed protocols from molecular cloning: A laboratory manual. New York: Cold Spring Harbor Press; 2006. [ Links ]
18. Suzuki T, Udo K, Adachi M, Sanada H, Tanaka K, Mizuta C, et al. Evolution of the diverse array of phosphagen systems present in annelids. Comp Biochem Physiol B Biochem Mol Biol. 2009;152(1):60-66. http://dx.doi.org/10.1016/j.cbpb.2008.09.087 [ Links ]
19. Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28(10):2731-2739. http://dx.doi.org/10.1093/molbev/msr121 [ Links ]
20. Huffman TN. Handbook to the Iron Age: The archaeology of pre-colonial farming societies in southern Africa. Pietermaritzburg: University of KwaZulu-Natal Press; 2007. [ Links ]
21. Benson DA, Cavanaugh M, Clark K. GenBank. Nucleic Acids Res. 2013;41:36-42. http://dx.doi.org/10.1093/nar/gks1195 [ Links ]
22. Huson DH, Bryant D. Application of phylogenetic networks in evolutionary studies. Mol Biol Evol. 2006;23(2):254-267. http://dx.doi.org/10.1093/molbev/msj030 [ Links ]
 Correspondence:
Correspondence:
Maryna Steyn
School of Anatomical Sciences, 7 York Road, Parktown 2193, South Africa
maryna.steyn@wits.ac.za
Received: 13 Apr. 2015
Revised: 14 July 2015
Accepted: 24 July 2015














