Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.111 n.9-10 Pretoria Sep./Oct. 2015
http://dx.doi.org/10.17159/SAJS.2015/20140401
RESEARCH ARTICLE
An LC-MS/MS based survey of contaminants of emerging concern in drinking water in South Africa
Christiaan OdendaalI; Maitland T. SeamanI; Gabre KempII; Huibreght E. PattertonII; Hugh-George PattertonII
ICentre for Environmental Management, University of the Free State, Bloemfontein, South Africa
IIDepartment of Microbial, Biochemical and Food Biotechnology, University of the Free State, Bloemfontein, South Africa
ABSTRACT
Advances in many analytical techniques allow the detection of compounds in water at very low concentrations (ng/L), which has facilitated the identification of many compounds in drinking water that went previously undetected. Some of these compounds are contaminants of emerging concern (CECs), which is broadly defined as any chemical or microorganism that is not currently being routinely monitored but has recently been identified as being present in the environment, and that may pose health or ecological risks. CECs can include pharmaceuticals, personal health care products and pesticides. Some CECs can act as endocrine disruptors, interfering with the normal functioning of the human endocrine system, potentially influencing foetal and child development. Although the level of many of these compounds are orders of magnitude below known acute toxicity levels, the health impact of long term exposure at low levels is mostly unknown. In this study, we present the results of a national survey over four seasons of potential CECs in the drinking water of major South African cities. The contaminants most often detected were the related herbicides atrazine and terbuthylazine, and the anticonvulsant and mood-stabilising drug, carbamazepine. The levels of these CECs were well below maximum levels proposed by the World Health Organization and the US Environmental Protection Agency. However, the range of CECs detected in drinking water, and seasonal and geographic variability in CECs levels, warrant a more frequent screening programme.
Keywords: pharmaceuticals; personal care products; mass spectrometry; multiple reaction monitoring; ToxNet
Introduction
Advances in analytic technologies allow the identification of chemical compounds at exceedingly low concentrations (10-9 g/L) in drinking water.1 This permits the identification of compounds which, until recently, were undetectable in water. These compounds fall into broad categories, including pesticides, pharmaceuticals and personal care products. Because we are only now becoming aware of the presence of these chemicals in drinking water, most of these compounds are not included in routine monitoring programmes. Although these compounds are generally present at concentrations several orders of magnitude below established acute toxicity levels, the effect of longterm exposure to very low concentrations of these compounds on human health and development is not known. This is particularly relevant to pharmaceutical contaminants, which are designed to be physiologically active at very low concentrations. Furthermore, some of these compounds interfere with the human endocrine system (endocrine disruptors), which may result in severe developmental defects with exposure of foetuses or infants during critical developmental windows. There is therefore a pressing need to investigate the potential health impacts of these compounds in drinking water, collectively known as contaminants of emerging concern (CECs).2,3
The US Geological Survey undertook several national reconnaissance studies, including a 1999/2000 programme, in which samples were analysed from 139 streams across 30 states in the USA.4 A wide range of chemicals present in residential, industrial, and agricultural wastewaters was found to occur at low concentrations in streams in the United States. The chemicals detected included human and veterinary drugs, natural and synthetic hormones, detergent metabolites, plasticisers, insecticides and fire retardants. One or more of these chemicals was found in 80% of the streams sampled. In a national groundwater study by the US Geological Survey, samples from 47 wells in 18 states were analysed for 65 chemicals.5 A profile of chemical pollutants similar to that observed in streams was found, although the contaminants were generally present at much lower levels. In another US Geological Survey study of untreated drinking water from 25 groundwater and 49 surface water sites in 25 states, pharmaceuticals, plasticisers and fire retardants were detected.6 Taken together, these studies provided valuable baseline information on the presence of CECs and other compounds in the US water system, and provides a valuable frame for further toxicity and public health impact studies.
The list of CECs is extensive, and includes sucralose, antimony, siloxanes, musks, nanomaterials, perfluorooctanoic acid, perfluorooctane sulphonate and other perfluorinated compounds, pharmaceuticals, hormones and hormone-active compounds, collectively known as endocrine disrupting compounds, drinking water disinfection by-products, sunscreens/UV filters, brominated flame retardants, benzotriazoles, naphthenic acids, cyanobacterial toxins, perchlorate, dioxane, pesticides and pesticide degradation products, and microorganisms, including viruses.7
Generally, organisations involved in water health and safety monitor CECs based on available technologies, known occurrence and health impacts.8,9 A technique that is currently widely used to monitor CECs is high performance liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS).7
Here we report the first national survey of CECs in the drinking water of major South African cities. The survey includes a qualitative screen for approximately 700 compounds, as well as the quantitation of three critical compounds identified in the qualitative screen, atrazine, terbuthylazine and carbamazepine. Atrazine is a herbicide used for the control of broadleaf weeds in the maize, sorghum and sugar cane agricultural industries. Epidemiological studies showed a correspondence between elevated atrazine levels in drinking water and low sperm volume and motility10, foetal growth defects, including restriction11, small-for-gestational-age12 and intrauterine growth retardation13, foetal gastroschisis14 and increases in limb reductions (upper and lower), hypospadias and epispadias, cryptorchidism, and spina bifida15. Terbuthylazine is a general, broad-spectrum pre- or post-emergence herbicide used in agriculture. Terbuthylazine was shown to cause an increase in DNA damage in cultured mammalian cells at concentrations equivalent to the occupational exposure limits.16 Carbamazepine is a therapeutic used as an anticonvulsant and a mood-stabilising drug. While it was reported that epilepsy patients who receive carbamazepine therapy during pregnancy delivered babies with an increased rate of congenital anomalies such as neural tube defects, and cardiovascular and urinary tract anomalies17, no epidemiological studies on the presence of carbamazepine at low concentrations in drinking water have been published to date.
Materials and methods
Reagents and materials
High purity (>98%) chemical standards for atrazine and carbamazepine were purchased from Sigma Aldrich (St. Louis, MO, USA), while terbuthylazine and deuterated-atrazine were purchased from Dr Ehrenstorfer (Augsburg, Germany). Stock solutions for each standard were prepared in methanol (1 ^g/L). High-performance liquid chromatography (HPLC) grade methanol (MeOH), acetonitrile (ACN), formic acid and ammonium hydroxide were purchased from Sigma Aldrich.
Ultra-pure water (18 mQ) was prepared with a Milli-Q purification device (Millipore, Billerica, MA, USA) and used in all experiments.
Method development and validation
The quantitative method was developed according to the Food and Drug Agency guidelines for method validation.18
Sampling
Samples (1 L) were collected in amber glass bottles from water treatment plants (WTPs) in Cape Town, Port Elizabeth, Durban, Pietermaritzburg, Johannesburg, Pretoria and Bloemfontein during months in each of the four seasons (February, April, July and October 2012), as well as from residential taps in Bloemfontein south and Bloemfontein north, supplied by two different reservoirs. Confidentiality agreements were entered into with the WTPs to not disclose the identity of the individual plants. Samples were collected and stored at 4 °C until analysis, usually within 24^8 h.
Solid phase extraction
Sample preparation involved compound extraction and reconstitution in 1 mL of H2O / 0.1% formic acid. Solid phase extraction is still the preferred approach of extraction, because it produces higher yields than liquid/liquid extraction, can be automated and significantly reduces preparation time.7,19 Milli-Q water fortified with CEC standards was used to optimise solid phase extraction parameters. Different solid phase extraction cartridges with varying sorbent characteristics were analysed to identify the cartridge with the best recovery.
Before extraction, cartridges were equilibrated with 6 mL pure MeOH. After equilibration, samples were loaded at a flow rate of approximately 6 mL/min. After samples were loaded, cartridges were washed with 6 mL of ultrapure water. Extracts were eluted into 6 mL tubes using 2 mL of MeOH and 2 mL of acetonitrile. Eluates were evaporated using a Savant SC 210A Speedvac concentrator with a Thermo RVT 4104 refrigerated vapour trap. Extracts were reconstituted in 1 mL of H2O / 0.1% formic acid and suspended using a vortex (Velp Scientifica, Italy) as well as by sonication (Branson, USA).
LC-MS/MS analysis
The analysis was performed on an HPLC (Agilent 1200) linked to a 3200 QTRAP hybrid triple quadrupole mass spectrometer (AB SciEx, Framingham, MA, USA). The HPLC was fitted with a 3-um Gemini-NX-C18 110-Â (150 x 2 mm) column (Phenomenex, CA, Torrance, USA). Formic acid (0.1% v/v) in water (solvent A) and formic acid (0.1% v/v) in MeOH (solvent B) were used as elution solvents for positively charged analytes. Negatively charged analytes were separated in NH3OH (0.1% v/v) in water (solvent a) and NH3OH (0.1% v/v) in MeOH (solvent B).
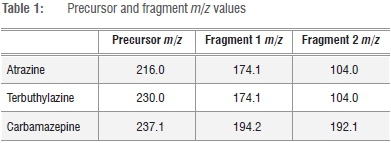
Analytes were detected and quantified using multiple reaction monitoring using precursor and two fragment transitions for each of the analytes.20,21 The m/z values used are shown in Table 1. Multiple reaction monitoring provides increased selectivity and reduces the likelihood of spectral interferences.
Results and discussion
Initial screening
We performed an initial LC-MS/MS analysis of drinking water from Bloemfontein and Johannesburg to obtain an insight into the range of CECs present in drinking water in South African cities. We made use of a MS/MS fragmentation library of approximately 700 compounds (see Supplementary table 1 online). The result of this initial screen is shown in Table 2.
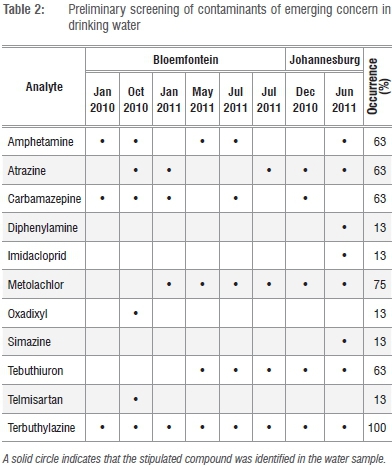
A review of the frequency of occurrence, coupled with toxicity data and community health impact from epidemiological studies,22 where available, suggested that atrazine, terbuthylazine and carbamazepine posed the highest public health risk to the South African water consumer. For this reason it was decided, apart from the general screening of drinking water for CECs, to also quantitate atrazine, terbuthylazine and carbamazepine in all collected water samples. In the absence of an established method, we needed to develop a robust protocol for the quantitation of these three CECs by LC-MS/MS. Method selectivity, accuracy and precision, as well as analyte recovery and stability are generally essential parameters to consider in method development and validation.18
Method validation Calibration curve
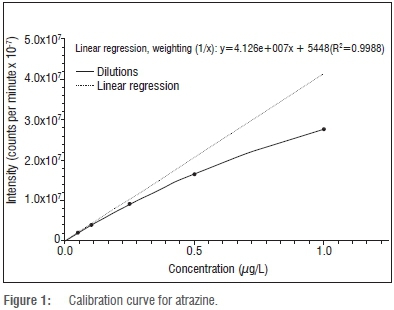
A calibration curve was determined by measuring the MS ion count over a concentration range of 5x10-5, 1x10-4, 5x10-4, 1x10-3, 5x10-3, 1x10-2, 5x10-2, and 1x10-1 μg/L for each of atrazine, terbuthylazine and carbamazepine. The representative calibration curve of atrazine is shown in Figure 1. Comparable results were obtained for terbuthylazine and carbamazepine (data not shown).
The limit of detection, lower limit of quantification and upper limit of quantification were determined for each of the three CECs using the MS spectra in the concentration range 5x10-5 - 1x10-1 µg/L. The limit of detection and lower limit of quantification were determined at signal-to-noise ratios of 3 and 10, respectively.23-25
The upper limits of quantification were defined as the highest concentration of analyte detectable with reasonable precision and accuracy.18,24,26 The lower limit of quantification, upper limit of quantification, recoveries, coefficient of variance and maximum contaminant levels are shown in Table 3. An internal standard, deuterated atrazine, was added at 1x10-1 µg/L before solid phase extraction. The same concentration of internal standard was injected into each of the vials (5x10-5 - 1x10-1 µg/L), and was used during quantification of atrazine and terbuthylazine.
Selectivity and crosstalk
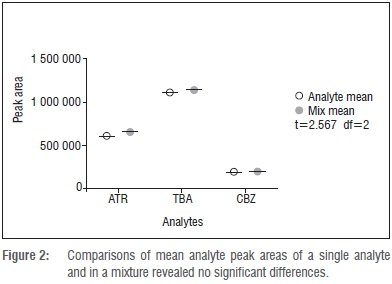
The selectivity of a method can be verified by establishing the absence of analyte peaks in a blank sample at the determined elution time for that analyte.18 The absence of crosstalk is shown by detecting comparable concentration for an analyte in a sample containing the single analyte compared to a sample containing a mixture of different, possibly interfering, analytes. To establish the selectivity and absence of crosstalk in our quantitation protocol, three vials were filled with 50 ng/L atrazine, terbuthylazine or carbamazepine, and a fourth vial was filled with a mixture that contained 50 ng/L of each of atrazine, terbuthylazine and carbamazepine. It was particularly important to demonstrate the absence of crosstalk for atrazine and terbuthylazine, because the m/z values of the two major fragments were identical (Table 1). The single analytes showed no significant difference compared to that of the mixture of three analytes in three independent repetitions of the experiment (paired f-test, confidence interval = 99%). Similarly, no analyte could be detected in sample blanks. The results are shown in Figure 2. Comparisons of mean analyte peak areas of a single analyte and in a mixture revealed no significant difference.
Accuracy and precision
The precision and accuracy of the quantitation protocol was demonstrated by determining the concentration of each of the three analytes in standard samples of 5x10-2μg/L, a concentration in the intermediate range between the lower limit of quantification and upper limit of quantification. In all cases, the coefficient of variance was less than 15% and the bias less than 20% (see Table 3), within the prescribed limits.18
Presence and seasonal variation of CECs in drinking wafer
Drinking water samples were taken at seven WTPs in major cities in South Africa at a point before the water entered the reticulation system. The samples were extracted on a solid phase cartridge, eluted, and analysed by LC-MS/MS. The precursor m/z as well as the m/z values of two major fragments were compared to a library of compounds (see Supplementary table 1 online). Compounds were identified where the precursor and well as both fragment m/z values could be matched to a library entry. The combined results of the screening of the seven drinking water samples are shown in Table 4. Atrazine, terbuthylzine and carbamazepine were detected in more than 60% of the drinking water samples. The seasonal distribution of atrazine fitted with its agricultural use as herbicide for summer crops. Carbamazepine, an anticonvulsant that is also prescribed for treatment of bipolar disorder, was present at a steady level in more than 70% of the samples. Cinchonidine, which is used in the chemical synthesis industries, was detected in almost 90% of the samples. Diphenylamine, which was present in about 40% of the samples, has wide application, including as an anti-scalding agent for fruit. The antifungal fluconazole and herbicides hexazinone and metolachlor were present in approximately 16% of the samples, with the latter present exclusively in the summer, most likely as a result of its agricultural application. Phenytoin, an anticonvulsant drug prescribed under the trademark 'Epanutin' in South Africa, was present in drinking water throughout the year. The antibacterial agent, sulphisomidine, was present in 18% of the samples. The herbicides, terbuthiuron and terbuthylazine, were consistently present in drinking water throughout the year. Interestingly, ephedrine, used as a decongestant and bronchodilator, was observed only in the winter, consistent with its expected increased medical use. Enilconazole, an antifungal agent widely used in the growing of citrus fruit, was observed only in autumn. Interestingly, we never detected any cyanobacterial microcystins, but had no information on the occurrence of upstream algal blooms.
Having established the frequency of occurrence of a range of pesticides and therapeutic compounds in metropolitan drinking water, it was decided to quantitate the levels of atrazine, terbuthylazine and carbamazepine, as these three compounds were present at very high frequency and were also associated with significant public health risks.
Quanfifafion of fhree crifical CECs in drinking wafer
The drinking water samples, treated as before, were separated by reverse phase HPLC and quantitated by multiple reaction monitoring on a hybrid triple quadrupole mass spectrometer using the developed method described above. This procedure involved the integration of the ion count during elution of a compound from the HPLC column, with concomitant confirmation of the identity of the compound by the presence of peaks at the correct precursor and major transition fragment m/z values. The peak area was used to deduce the concentration from the standard curve of each of the three compounds of interest. The concentrations are tabulated in Supplementary table 2 online.
The guideline value proposed by the World Health Organization (WHO) for atrazine is 100 mg/L27, whilst the maximum contaminant level stipulated by the US Environmental Protection Agency (EPA) is 3 mg/L8. Figure 3 indicates that the highest level of atrazine recorded during the one year survey was more than an order of magnitude below the maximum contaminant level set by the EPA. The level of atrazine was consistently high throughout the year in Johannesburg, compared to the average value recorded for all the samples. Interestingly, high atrazine values were also recorded in tap water in Bloemfontein in the autumn and spring, even though low levels were recorded at the WTP at the same times. This suggested that the concentration of atrazine may vary very sharply, and that a much higher sampling frequency is required to accurately determine its variation over time.
The guideline value proposed for terbuthylazine by the WHO is 7 mg/L.27 The EPA has no set maximum contaminant level for terbuthylazine.8 Referring to Figure 3, it is seen that the highest recorded concentration for terbuthylazine in drinking water (Pretoria, autumn) is at least an order of magnitude less that the WHO guideline value. Johannesburg, again, showed a consistently high level of terbuthylazine throughout the year, compared to the other WTPs.
The maximum contaminant level for the pharmaceutical carbamazepine was set at 12 mg/L.28 The highest level of carbamazepine detected in drinking water (see Figure 3) was significantly less than this level. Interestingly, the level of this anti-epileptic and mood-stabilising drug was consistently high throughout the year in Bloemfontein, compared to the average national level. Particularly high levels were recorded in the summer (Figure 3). We again observed a discordance between the carbamazepine concentrations recorded at the WTP and in tap water in Bloemfontein in the autumn. This result also suggests significant concentration spikes, indicating a need for a high sampling frequently to obtain a reliable insight into the level of this CEC in drinking water.
Conclusion
During this analysis, a method was developed to determine atrazine, terbuthylazine and carbamazepine quantities in drinking water. A qualitative analysis identified 29 potential CECs (Table 4). Importantly, the critical CECs identified during preliminary analyses were also part of the subsequent qualitative list of CECs. Quantification of atrazine, terbuthylazine and carbamazepine revealed no immediate health risks, since all concentrations were below the published thresholds.
Although the concentration levels were below published maximum contaminant level thresholds, the range of CECs routinely detected in drinking water, and the large geographical and seasonal variability that we observed, suggest that a qualitative survey and quantitation of select CECs should be performed more frequently to have a current view of the presence of levels of CECs in drinking water that may impact on human health. Also, with an increase in the pressure on water health as this resource in increasingly being utilised, the introduction of such a CEC monitoring programme becomes essential to ensure the production of healthy and safe drinking water for the consumer.
Acknowledgements
We thank the Water Management in Water-scarce Areas and the Advanced Biomolecular Research clusters at the University of the Free State for their contributions to this project. This study was supported by a grant from the Water Research Commission (to H.G.P.).
Authors' contributions
C.O. performed the sample preparation and LC-MS/MS analysis; M.T.S. performed data analysis and contributed to writing the manuscript; H.E.P performed data analysis and contributed to writing the manuscript; G.K. performed LC-MS/MS analysis, data analysis and contributed to writing the manuscript; H.G.P. managed the research project, performed data analysis and contributed to writing the manuscript.
References
1. Fatta-Kassinos D, Ledin A. Editorial. Environ Pollut. 2010;158:3016. http://dx.doi.org/10.1016/j.envpol.2010.07.008 [ Links ]
2. US Environmental Protection Agency. Contaminants of emerging concern [homepage on the Internet]. [ Links ] No date [cited 2015 Feb 15]. Available from: http://water.epa.gov/scitech/cec/
3. US Environmental Protection Agency. Basic information on CCL and regulatory determinations [homepage on the Internet]. [ Links ] No date [cited 2015 Feb 15]. Available from: http://www2.epa.gov/ccl/regulatory-determination-3
4. Koplin DW, Furlong ET, Meyer MT, Thurman EM, Zuagg SD, Barber LB, et al. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: A national reconnaissance. Environ Sci Technol. 2002;36:1202-1211. http://dx.doi.org/10.1021/es011055j [ Links ]
5. Barnes KK, Kolpin DW, Furlong ET, Zaugg SD, Meyer MT, Barber LB. A national reconnaissance of pharmaceuticals and other organic wastewater contaminants in the United States - I: Groundwater. Sci Total Environ. 2008;402:192-200. http://dx.doi.org/10.1016/j.scitotenv.2008.04.028 [ Links ]
6. Focazio MJ, Kolpin DW, Barnes KK, Furlong ET, Meyer MT, Zaugg SD, et al. A national reconnaissance for pharmaceuticals and other organic wastewater contaminants in the United States - II: Untreated drinking water sources. Sci Total Environ. 2008;402:201-216. http://dx.doi.org/10.1016/j.scitotenv.2008.02.021 [ Links ]
7. Richardson SD. Water analysis: Emerging contaminants and current issues. Anal Chem. 2009;81:4645-4677. http://dx.doi.org/10.1021/ac9008012 [ Links ]
8. Environmental Protection Agency. National primary drinking water regulations. Report EPA816-F-09-004. Washington, DC: Environmental Protection Agency; 2009. [ Links ]
9. World Health Organization. The WHO recommended classification of pesticides by hazard and guidelines to classification: 2009. Geneva: World Health Organization; 2010. [ Links ]
10. Swan SH. Semen quality in fertile US men in relation to geographical area and pesticide exposure. Int J Androl. 2006;29:62-68. http://dx.doi.org/10.1111/j.1365-2605.2005.00620.x [ Links ]
11. Chevrier C, Limon G, Monfort C, Rouget F, Garlantézec R, Petit C, et al. Urinary biomarkers of prenatal atrazine exposure and adverse birth outcomes in the Pelagie birth cohort. Environ Health Pers. 2011;119:1034-1041. http://dx.doi.org/10.1289/ehp.1002775 [ Links ]
12. Ochoa-Acuna H, Frankenberger J, Hahn L, Carbajo C. Drinking-water herbicide exposure in Indiana and prevalence of small-for-gestational-age and preterm delivery. Environ Health Persp. 2009;117:1619-1624. http://dx.doi.org/10.1289/ehp.0900784 [ Links ]
13. Munger R, Isacson P Hu S, Burns T Hanson J, Lynch CF et al. Intra-uterine growth retardation in Iowa communities with herbicide-contaminated drinking water supplies. Environ Health Persp. 1997;105:308-314. http://dx.doi.org/10.1289/ehp.97105308 [ Links ]
14. Waller SA, Paul K, Peterson SE, Hitti JE. Agricultural-related chemical exposures, season of conception, and risk of gastroschisis in Washington State. Am J Obstet Gynecol. 2010;202(3), Art. #241, 6 pages. http://dx.doi.org/10.1016/j.ajog.2010.01.023 [ Links ]
15. Davis JA. Environmental atrazine exposure and congenital malformations in New York State: An ecologic study. Birth Defects Res A Clin Mol Teratol. 2005;73:926. [ Links ]
16. Mladinic M, Zeljezic D, Shaposhnikov SA, Collins AR. The use of FISH-comet to detect c-Myc and TP 53 damage in extended-term lymphocyte cultures treated with terbuthylazine and carbofuran. Toxicol Lett. 2002;211:62-69. http://dx.doi.org/10.1016/j.toxlet.2012.03.001 [ Links ]
17. Matalon S, Schechtman S, Goldzweig G, Ornoy A. The teratogenic effect of carbamazepine: A meta-analysis of 1255 exposures. Reprod Toxicol. 2002;16:9-17. http://dx.doi.org/10.1016/S0890-6238(01)00199-X [ Links ]
18. Food and Drug Administration. Guidance for industry bioanalytical method validation. Silver Spring, MD: Food and Drug Administration; 2001. Available from: http://www.fda.gov/downloads/Drugs/Guidances/ucm070107.pdf. [ Links ]
19. Sigma-Aldrich. Guide to solid phase extraction. Bulletin 910. St. Louis: Sigma-Aldrich; 1998. Available from: http://www.sigmaaldrich.com/Graphics/Supelco/objects/4600/4538.pdf. [ Links ]
20. Cox DM, Zhong F, Du M, Duchoslav E, Sakuma T, Mcdermott JC. Multiple reaction monitoring as a method for identifying protein post-translational modifications. J Biomol Tech. 2005;16:83-90. [ Links ]
21. Botitsi HV Garbis SD, Economou A, Tsipi DF. Current mass spectrometry strategies for the analysis of pesticides and their metabolites in food and water matrices. Mass Spectrom Rev. 2010;30:907-939. http://dx.doi.org/10.1002/mas.20307 [ Links ]
22. National Library of Medicine. TOXNET: Toxicology data network. Washington, DC: National Library of Medicine; 2013. Available from: http://toxnet.nlm.nih.gov/. [ Links ]
23. European Union. Commission decision of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Brussels: European Union; 2002. Available from: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32002D0657 [ Links ]
24. Bennet G, Hale S, Scifres J, Wasko M. Laboratory operations and quality assurance manual. Atlanta, GA: Environmental Protection Agency; 2011. [ Links ]
25. Van Iterson RA. A guide to validation in HPLC. Emmen: Drenthe College; 2011. Available from: http://parasshah.weebly.com/uploads/9/1/3/5/9135355/hplc_validation_pe.pdf. [ Links ]
26. Shah VP Midha KK, Findlay JWA, Hill HM, Hulse JD, Mcgilveray IJ, et al. Bioanalytical method validation - A revisit with a decade of progress. Pharm Res. 2000;17:1551-1557. http://dx.doi.org/10.1023/A:1007669411738 [ Links ]
27. World Health Organization. Guidelines for drinking-water quality. Geneva: World Health Organization; 2011. [ Links ]
28. Washington Suburban Sanitary Commission. Emerging contaminants. Washington, DC: Washington Suburban Sanitary Commission; 2013. Available from: https://www.wsscwater.com/files/live/sites/wssc/files/watet%20quality/2013wqr.pdf [ Links ]
 Correspondence:
Correspondence:
Hugh-George Patterton
Institute for Wine Biotechnology
Stellenbosch University
Private Bag X1, Matieland 7602
South Africa
Email: hpatterton@sun.ac.za
Received: 14 Nov. 2014
Revised: 15 Feb. 2015
Accepted: 15 Feb. 2015
Note: This article is supplemented with online only material.














