Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.111 n.9-10 Pretoria Sep./Oct. 2015
http://dx.doi.org/10.17159/SAJS.2015/20140258
REVIEW ARTICLE
A review of carbon dioxide as a refrigerant in refrigeration technology
Paul MainaI, II; Zhongjie HuanI
IDepartment of Mechanical Engineering, Tshwane University of Technology, Pretoria, South Africa
IIDepartment of Mechanical Engineering, Moi University, Eldoret, Rift Valley, Kenya
ABSTRACT
Tough environmental laws and stringent government policies have revolutionised the refrigeration sector, especially concerning the cycle fluid known as the refrigerant. It has been observed that only natural refrigerants are environmentally benign. When other refrigerant qualities are considered, especially those relating to toxicity and flammability, carbon dioxide emerges as the best among the natural refrigerants. However, carbon dioxide based refrigerants are not without drawbacks. Even though the use of R744 -a carbon dioxide based refrigerant gas - has solved the direct effect of emissions on the environment, studies to investigate the indirect effects of these systems are needed. Improvement in existing technical solutions and the formulation of additional solutions to existing R744 refrigeration problems is paramount if this technology is to be accepted by all, especially in areas with warm climates. National policies geared to green technologies are important to clear the way and provide support for these technologies. It is clear that carbon dioxide is one of the best refrigerants and as environmental regulations become more intense, it will be the ultimate refrigerant of the future.
Keywords: environment; safety; heat pump; energy efficiency; transcritical
Introduction
Most refrigerators use a liquefiable vapour to transfer heat. This fluid is known as the refrigerant. Refrigerant selection is a key design decision that influences the mechanical design of the refrigeration equipment. Factors that must be considered in refrigerant selection include performance, safety, reliability, environmental acceptability and cost. However, the primary requirements are safety, reliability and, nowadays, environmental friendliness (in terms of ozone depletion and global warming potential). Table 1 summarises the properties of some refrigerants and indicates that no progress has been made in terms of global warming potential (GWP) when switching from hydrochlorofluorocarbons (HCFCs) to the hydrofluorocarbon (HFC) family. When securely contained in a properly operating system, refrigerants do not impact climate change; however, system leaks and improper recovery of refrigerants during repairs or at end of life result in these harmful gases entering the atmosphere. Furthermore, during production of refrigerants, toxic and harmful wastes are released into the environment, which cause air, water and land pollution in addition to releasing greenhouse gases. An alternative to HFCs is to apply naturally occurring and ecologically safe substances, the so-called natural working fluids. The most important substances in this category are hydrocarbons, ammonia and carbon dioxide, although when safety concerns are raised (toxicity and flammability), R744, a carbon dioxide based refrigerant gas, becomes the best substitute.
Carbon dioxide (CO2) is a clear gas (at atmospheric conditions) without a particular smell when the concentration is below suffocation level. When the concentration reaches toxic levels, it has a slightly pungent smell and somewhat acidic taste. It has a higher density than air, which has its own advantages with respect to refrigeration and disadvantages with respect to safety. CO2 is made both naturally and artificially - artificially through the burning of fuel and other industrial processes.1,3 Approximately 0.04% of atmospheric air is CO2, thus CO2 is at a concentration of approximately 380 parts per million (ppm) in air. Exhaled air from the body has a CO2 concentration of about 4%.
History of R744 as a refrigerant
Since the invention of the vapour-compression cycle by Evans and Perkins in 1834, R744 has been a candidate for a refrigerant. Documented studies state that Alexander Twining was the first to propose R744 refrigeration using a steam compression system in his British patent of 1850. However, Thaddeus Lowe was the first to actually build a refrigerator running on R744 for ice production in 1866 after discovering its potential while using it in military balloons. Carl Linde followed suit and built a better refrigerator running on R744 in 1881, just after Windhausen had built the first R744 compressor in 1880. In 1884, W Raydt built a R744 refrigeration system for making ice using a vapour compression mechanism while, at the same time, J Harrison was the first person to build a device for manufacturing R744 purely for refrigeration use. The British company J and E Hall built the first R744 marine refrigerator in 1890 using Windhausen's compressor designs, while in the USA, continuous production of these refrigerators was started in 1897, mainly by Kroeschell Bros. Ice Making Company. Owing to its safety aspects when compared to other refrigerants during this period, R744 refrigerators grew in number, especially in the marine sector. At the same time, its technology was improving. For example, in 1889, J and E Hall created a two-stage R744 compressor which was more efficient, and in 1905, Voorhees created a flash chamber which was very similar to a liquid-vapour separator.4-7
Calcium chloride solution was used in most refrigerators as a secondary fluid. The salt solution was cooled to around -10 °C (evaporation temperature of -15 °C). Originally, the evaporator and condensers used galvanised steel pipes, 32 mm in diameter for small refrigerators and 51 mm in diameter for large cold rooms. Tank and coil heat exchangers were the first to be used, before tube in tube (double pipe) technology was introduced in 1902. The shell and tube type were invented in the early 1930s and fin technology in the 1920s. Copper replaced steel pipes during this decade too, with pipe diameters being reduced to 13 mm because of the increased heat transfer offered by the fins and copper. Air circulating fans were introduced around this time for improved cooling, especially in cold rooms. R744 used to cost around 9 cents per kg but the price increased to 12 cents per kg in the late 1920s.

With the invention of compressors, vertical, cylinder-type compressors of up to 42 kW (325 rpm) were first used but were later replaced by horizontal compressors of up to 176 kW (120 rpm) in size. Both these constructions were similar to the steam engine design. R744 required heavy duty parts for valves, fittings, compressors and heat exchangers as a result of its associated pressure. Refrigerator size increased up to 704 kW by 1916.7
The use of R744 air conditioners for comfort cooling began in the 1900s. Because of the toxicity and/or flammability of NH3 and SO2, R744 gained in popularity, especially in food-related industries (food markets and eateries) and human comfort applications, e.g. in theatres, bars, hospitals, ships and hotels. In 1900, only 25% of all ships were using R744 as the refrigerant, but by the 1930s, this proportion had increased to 80%. Although the ships used old technology, R744 equipment still worked, albeit inefficiently, especially because they used a convectional subcritical refrigeration cycle. In addition, there were sealing and capacity loss problems related to R744 high pressures. These disadvantages encouraged a search for safe and efficient (especially at high discharge temperatures in warm climates) refrigerants which ended with the discovery of chlorofluorocarbons (CFCs) in the 1930s. The invention of CFCs, coupled with a lack of technological improvements from the R744 refrigeration industry, caused the decline in the use of R744. The last large R744 refrigeration system was installed in 1935 for Commonwealth Edison Company headquarters. The system was replaced by CFC refrigerators 15 years later. CFCs eliminated the problems encountered when using R744, such as the need for high pressure sealing, capacity and efficiency loss and the high cost of components. Eventually by the 1950s, R744 refrigeration was completely phased out.4-7
After the discovery of the adverse effects of synthetic refrigerants in the late 1980s, there was a renewed interest in R744. Professor Gustav Lorentzen was the pioneer of the revival of R744 refrigeration in the early 1990s, with many studies and ideas dedicated to its improvement. He suggested that, because of the properties of R744, motor vehicle air-conditioning systems (the leading sector in refrigerant leakage -60% of all leakages8) and water heat pumps are best suited for R744 refrigeration.9 His idea was positively acknowledged with many motor vehicle and water heat pump companies investing in R744 research. Leading car manufacturers, such as Nissan, Bavarian Motor Works AG (BMW) and DaimlerChrysler, have installed R744 air-conditioning systems in new cars.4,10 Many R744 domestic heat pumps are manufactured and marketed in Asia and Europe.11-13 Recently, there has been a keen interest in using R744 in supermarkets and other commercial refrigeration applications.14,15 Leading beverage companies like CocaCola and PepsiCo have embarked on converting their vending machines to use R744 as the refrigerant of choice.12 Also, R744 refrigeration has been applied in residential and commercial buildings' air-conditioning systems with great promise.16 In short, there are currently many application prospects for R744 refrigeration under investigation, most of which are quite promising.
R744 properties
General introduction
As indicated in Table 1, R744 is a non-toxic, non-flammable natural refrigerant with an ozone depletion potential of zero and a GWP of 1. Furthermore, it is widely and cheaply available as a constituent of the atmosphere and as a result of industrial processes, especially the ones involving fuel. Its critical temperature, however, is 31.1 °C. Low critical temperature means that R744 cannot be used effectively in a convectional (subcritical) refrigeration cycle simply because the condenser will not transfer heat above the critical temperature. Therefore, the condenser will be ineffective and many losses may occur. Furthermore, at temperatures that are near the critical temperature but less than it, there is a drastic reduction on the vaporisation enthalpy which leads to a reduction in heating capacity and reduced system performance. Therefore, as Lorentzen has suggested, R744 can be effectively used only in a transcritical cycle.9
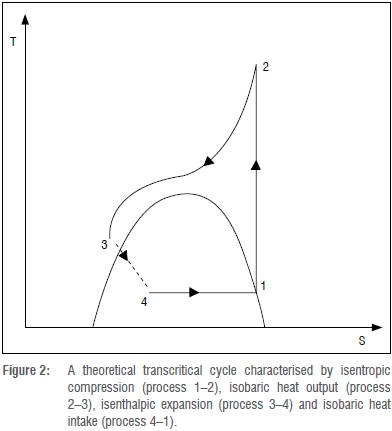
Transcritical cycle
A transcritical cycle (Figure 2) is not limited by the critical temperature because heat output is through a temperature glide. The condenser in a transcritical cycle is replaced by a gas cooler because there is no condensation taking place, but rather a gas cooling process. This temperature glide is advantageous especially in applications such as water heating and air heating (e.g. drying processes) because of the associated efficiencies. The temperature range in which R744 refrigeration can operate in transcritical operation is the highest when compared to other convectional refrigerants, i.e. -50 °C to 120 °C.4 The only drawback with the transcritical cycle for R744 is the high pressure. The critical pressure for R744 is 73.7 bars. If R744 is operated through a transcritical cycle, then its high pressure will be above 73.7 bar, which is quite high. This necessitates an equipment design that can handle such a high pressure. The high pressure has its own advantages (e.g. compact equipment and design) and disadvantages (costly equipment and safety issues). However, with current technological advances, this pressure is not a big concern.
Thermophysical properties
The high latent heat of vaporisation and volumetric heat transfer caused by the high pressures involved means that R744 equipment components can be designed in smaller size. Coupled with the fact that R744 has very low viscosity, Reynolds number is reached even with a low flow rate, which means that most flows are turbulent in nature and the heat transfer rate is high. Apart from the flow nature, R744 has a small liquid to vapour density ratio (especially near the critical point) which leads to uniform and homogeneous distribution of the refrigerant in channels. This homogeneity also adds to the high heat transfer rate. Furthermore, proximity to the critical point and less pressure loss, especially in the gas cooler, contributes to improved convective heat transfer. When the heat transfer rate is high, the size of the heat exchangers can be drastically reduced for the same amount of heat transfer to occur.6
The compressor size can also be reduced because of the associated compression ratio involved. Even though the R744 transcritical cycle deals with high operational pressures, the ratio between the high pressure and low pressure is low when compared to other refrigerants. The ratio is also closely associated with the high adiabatic index of R744 (which is approximately 1.3). These factors lead to a compact, smaller and more efficient compressor, ideal for applications for which space is limited, e.g. in mobile air conditioners in cars.3 The R744 compressor efficiency increase is also brought about by the low effect of valve pressure drops and the re-expansion ratio it experiences. Furthermore, internal leakages and piston blow-by losses are negligible when compared to those of other compressors. Even though the compressor walls must be thicker because of the pressures involved, R744 volumetric capacity dictates small parts in the compressor and therefore the overall size of the compressor is smaller when compared to compressors of convectional refrigerants of the same capacity.5,6
Properties of R744 at a supercritical state are always in between those at liquid state and those at gaseous state. The critical point, defined as the point at which no liquefaction occurs above the critical pressure and no gas is formed above the critical temperature, is peculiar in nature because, near the critical point, there is always a sudden variation in the properties. Specific heat, thermal conductivity, enthalpy, entropy, density and viscosity undergo a major change as the critical point is approached.17 Figure 3 shows the variation of specific heat at constant pressure (cp) against temperature for R744 at several pressures.18 As can be seen from Figure 3, the highest value of cp occurs at a pseudo-critical temperature for that pressure. This property can be incorporated into the design of gas coolers to maximise on their output.
R744's surface tension is smaller than that of other refrigerants. The significance of this observation is that surface tension affects the wetting characteristics, flow characteristics and evaporation characteristics of a fluid. Low surface tension might be positive in that the temperature required to initiate and maintain nucleate boiling is reduced, but it can also be negative because of drop formation and entrainment, especially when there is reduced surface stability. On the other hand, the thermal conductivity of R744 is considerably higher than that of most synthetic refrigerants, while its viscosity is lower. A high thermal conductivity means a higher heat transfer rate, while viscosity affects flow properties, which in turn affects heat transfer and pressure drop. Therefore, the thermophysical characteristics of R744 are favourable and encourage its use as a refrigerant.5 Still, being relatively inert, R744 is compatible with most lubricants and equipment materials, as documented in numerous studies (even ones not related to refrigeration).
High (gas cooler) pressure
At supercritical state, the temperature and pressure of R744 are independent of each other and thus can be regulated independently to optimise output. Still, compressor input power is proportional to the high pressure. As shown in Figure 2, if the gas cooling process occurs at a constant pressure (process 2-3), the magnitude of the pressure will affect the specific enthalpy. This pressure is not controlled by the cooling fluid conditions (temperature and flow rates) as is the case with convectional refrigerants; it is mostly controlled by the amount of refrigerant charge present.8 As the pressure increases, there will be an initial increase in heat output with a moderate increase in compressor power input, thus there will be an overall increase in system efficiency. As the pressure is increased further, it will reach a point at which the additional work input is more than the additional heat output, and thus the efficiency of the
refrigerator will begin to decrease. This behaviour is attributed to the shape of isotherms (which affect the heat output) and isentropes (which affect the compressor power) at pressures above the critical point. There is always an optimum high pressure value which corresponds to certain operational conditions. The optimum pressure depends on the gas cooler outlet temperature, the evaporation temperature, the compressor isentropic efficiency and the amount of refrigerant.19,20 Therefore, it is paramount that there is a means of controlling the high pressure. The optimum pressure can approximately be given by:
Popt= 2.6Texit+ 8 Equation1
where P is expressed in bars and Texit is the gas cooler exit temperature in °C.
Another important characteristic of R744 is the relationship between its pressure and temperature. The vapour pressure of R744, apart from being higher than other refrigerants (Table 2), also has a greater variation per unit temperature change, especially near the critical point. The slope of change in vapour pressure to change in temperature is much steeper for R744 than for other refrigerants. This means that for every unit change in pressure, there is a lesser change in temperature with R744 as compared to other refrigerants. Therefore, the effect of pressure loss is less severe and more tolerable in R744 than in other refrigerants. The high vapour pressure also causes high vapour density (Clausiu-Clapeyron's relation). Because volumetric heat transfer is the product of vapour density and latent heat of evaporation, a high vapour density results in a high volumetric heat transfer.5 Furthermore, a high vapour density results in a low velocity of R744 in pipes, thus resulting in less of a pressure drop. In addition to high thermal conductivity of R744 (Table 2), high vapour density also allows the use of small components, e.g. tubes, which results in lower radiation losses.21 Phase separation characteristics between vapour and liquid phases are directly controlled by phase density differences. R744 has a low density ratio, which is necessary for a homogenous two-phase flow.4
Even though R744 has very favourable properties, especially when used as a heat pump, its cycle is still affected by many losses. Given an evaporation temperature and minimum heat rejection temperature, the transcritical cycle is affected by higher throttling losses when compared to the convectional subcritical cycle. These losses increase the theoretical work done on the transcritical cycle. The throttling losses in refrigeration are normally caused by the temperature difference in the throttle device and the refrigerant properties. With the temperature being set at a specific value, R744's unique properties, especially near the critical point (i.e. high liquid specific heat and low evaporation enthalpy), increase the throttling losses and thus increase the compressor power required. Therefore, even though compressor losses are lower in R744 machines, other factors tend to increase power consumption. Thus, it is paramount to reduce the losses as much as possible if R744 technology is to be fully embraced.5
Safety
Finally, R744 is considered non-toxic, although at concentrations above 2% (about 5 kg of R744 in a 120-m3 room) it can start to become harmful. It is colourless and odourless so it cannot easily be detected; therefore, in installations for which a high amount of charge is used (25 kg by European standards) and the ventilation is poor, R744 detectors should be installed. Furthermore, having a higher density than air (R744 relative density is about 1.53 at atmospheric conditions), it will tend to occupy lower areas so can prove to be more deadly when spilled in a non-ventilated room. Therefore, the detector should be installed near the floor. Table 3 gives the effects of R744 at various concentrations in the air. To prevent an accumulation of R744 in an enclosed space with a big installation:
- sufficient ventilation must be provided in the equipment room
- R744 containers must always be in an upright position, especially when charging the system
In the event of a large R744 release, the equipment room should be avoided until the concentration in the room is within allowable limits (measured either through sensors or other means). In case of exposure, the person should be taken into open air immediately.
Apart from toxicity issues, explosions as a result of the operational pressures experienced in R744 equipment is also a safety concern. The shock and flying fragments caused by a blast might cause injury and harm. Apart from explosions caused by the pressure in R744 systems, there is another severe type of explosion known as boiling liquid expanding vapour explosion (or BLEVE), which usually occurs when a vessel containing pressurised saturated liquid is rapidly depressurised as a result of a crack or rupture and can cause a blast which is more severe than the pressure blast. The rapid depressurisation leads to explosive vaporisation and a sudden overpressure in the tank which might blow the vessel.21 To prevent these catastrophes, the safety design of the R744 equipment should include the following5:
- The equipment should have an over-pressure release valve on both the high pressure side and low pressure side.
- Components should be pressure tested at twice (or more) the amount of normal operation pressure and temperature.
- The pressure test should incorporate the other operational effects like fatigue due to pressure cycles, creep, vibration and corrosion (especially if there is water present).
- All components should be designed with the highest pressure and temperature (including a safety factor) the system may encounter even in standstill mode.
When considering the size of the system versus the pressures involved, the relative explosion energy in R744 systems is approximately the same as in convectional systems. This is mainly because R744 systems tend to have a smaller system size and less charge for the same capacity.8
R744 applications
Apart from being a refrigerant, R744 is used in many processes and applications. However, in refrigeration, there are numerous applications for R744, some of which have been commercialised.
Water heat pumps
Production of hot water is the best application for R744 heating refrigerators (heat pumps) as the temperature slide in the transcritical cycle suits the thermodynamic properties of water well.5 Very efficient heat transfer and very high water temperatures are achieved with water heating applications, especially when using a counterflow gas cooler.13 With heat pumps being the preferred water heating method when compared to electricity or fuel-fired systems, governments and other energy efficiency conscious bodies are encouraging their use. This promotion comes after it was realised that water heat pumps with an average coefficient of performance (COP) of 3 can reduce energy usage by 67% when compared to electric heating, and by even more when compared to fuel heating. The percentage energy saving of a heat pump (ΔΕ) when compared to another heating system with an efficiency of η is given by21:

where η for electric heaters is approximately 1 while η for fuel-fired heaters varies from 0.5 to 0.95, depending on the equipment and the type of fuel.
In addition, environmentalists are encouraging the use of heat pumps which are environmentally friendly, such as R744 heat pumps.22 This is one of the applications that the 'father' of R744 refrigeration re-invention, Prof. Lorentzen, suggested for R744 as a refrigerant.9 In fact, it is this sector that has seen major commercialisation of R744 refrigerators. Since 2001, the production of commercial R744 heat pump units has taken place under the general name of ECO CUTE in Japan (Figure 4). These units, which are marketed both in Asia and Europe, have now surpassed an annual production rate of 1 million, a growth encouraged both by their high efficiency and by incentives from government and environmental bodies.3,11,23 Other manufacturers have introduced similar systems for residential, commercial and industrial use. The possibility of efficiently producing hot water at 90 °C and above is encouraging the industrial and commercial use of R744 heat pumps, especially in hotels, hospitals and the food industry.5
With water heating being one of the highest energy uses, especially in the residential sector (approximately 20%), coupled by the environmental and energy efficiency advantages, R744 heat pump technology has great future prospects.22 Lorentzen has described the possibility of using R744 heat pumps for both heating and cooling applications simultaneously. These systems have high overall system efficiencies and can find application in places where both refrigeration and hot water are needed, for example, in hospitals, supermarkets, hotels, and the food processing and chemical industries.9
Mobile air conditioning
Mobile air conditioning is the second application that Lorentzen envisioned for R744 refrigeration systems. Compact R744 systems coupled with good heat transfer characteristics between air and R744 encourage the use of these systems in a sector in which equipment space and weight is limited while energy efficiency is paramount.9 Mobile air conditioning is the largest consumer of refrigerants in the world, followed by commercial refrigeration. Mobile air conditioning consumes 31% of the world's refrigerant, which adds up to more than 150 000 t/year.15,24 On the same note, mobile air conditioning has the highest leakage rate.21 This means a complete change to R744 will be highly advantageous. Recent studies demonstrated the superiority of R744 systems when compared to commonly used HFC systems.25 With the phasing out of HFC in progress, especially in European countries, R744 is the best alternative for mobile air conditioning. R744 air conditioning will work especially well with fuel-efficient hybrid or electrical cars with little waste heat available. With electrical cars specifically, if the airconditioning system is efficient enough, more energy will be used to drive the car and thus more travelling distance will be covered before the electricity runs out.8
The greatest disadvantage of R744 in mobile air-conditioning applications is its high heat rejection temperature. However, in the new technologically advanced vehicles, in which there is little or no excess heat generation, this high temperature heat output can be effectively used, especially in cold season, not only for human comfort but also for heating essential vehicle parts like engine fluids. Common HFCs used in mobile air conditioning do not perform well as heat pumps because of their thermophysical properties.
As mobile air conditioning systems are prone to more maintenance problems than stationary systems, logistical concerns arise as R744 systems are fairly new and are different from convectional systems. There is a need for qualified technicians to handle the new technology, which is made more complex in vehicles because of the limited weight and space requirements coupled with integration of the air-conditioning system and other electrical and mechanical systems of the car. These, among other minor economic issues, need to be addressed before R744 mobile air conditioning is fully embraced. Furthermore, frost accumulation on evaporators presents a complication which has not yet been solved effectively, especially in a mobile application. Despite these challenges, R744 is proving to be an ideal refrigerant, capable of providing high temperature heat instantly while requiring less air to convey the heat. This makes it a hot research topic for complex mobile environment control with improved efficiency.5
Commercial refrigeration
Commercial refrigeration is the equipment used by retail outlets to display, hold or prepare food and beverages that customers purchase. This equipment includes refrigerated display counters in supermarkets, refrigerated vending machines, water coolers/heaters and ice generating machines. Commercial refrigeration consumes about 28% of worldwide refrigerants, thus is the second largest user of refrigerants.15,24 This makes it one of the largest emitters of refrigerants into the environment and accounts for approximately 37% of worldwide emissions. In 2002, commercial refrigeration was responsible for more than 185 000 t of leaked refrigerant into the atmosphere.26 Furthermore, the energy utilisation in this sector is usually very high, necessitating a need for efficient refrigeration systems.
Ironically, until the year 2000, R744 applications in commercial refrigeration were not considered viable. The perception has since changed with its use either as a heat transfer fluid, in a cascade system, or on its own in either a transcritical cycle or a subcritical cycle, depending on the environmental temperature. External factors like safety requirements, extra tax on HFC systems and limitations on the maximum amount of HFC charge that can be used on a single system were the main reasons for R744 acceptability in commercial refrigeration. It was first purely used indirectly as a heat transfer fluid, then in cascade systems in conjunction with HFC at a reduced charge or with hydrocarbons (HCs). With time, more skills and knowledge were acquired and the cascade systems were replaced with fully transcritical R744 systems. With the possibility of heat recovery (for space heating or tap water heating), R744 commercial refrigeration has a great potential. As the world accepts the use of R744 in supermarkets, studies show that its associated costs and energy consumption are comparable to other convectional refrigeration systems. Actually, it has been suggested that R744 systems are the dominant technology of the future because of their good thermophysical and safety properties.8
R744 technology is even being utilised in stand-alone bottle coolers and both hot and cold vending machines. Major investment in the light commercial sector is directed to R744 technology, with more than 85 000 units in operation worldwide.8 Most of these vending machines utilise the transcritical cycle.
Other applications Residential air conditioning
R744 air in air refrigerators has been the focus of investigations by both research institutions and industry because of the high demand for such equipment and the requirement for HFC alternatives. The annual demand for residential air-conditioning units is more than 40 million units and further market growth is expected.21 Air conditioning is the second largest consumer of energy after water heating in most residential areas. Environmental concerns in this application are more focused on the indirect impacts of emissions due to energy use, than on the direct impacts of refrigerant leaking. Therefore, energy efficiency is paramount.5
There have been promising results with the application of R744 in stationary air-conditioning systems. Units with a one-way refrigerant circuit are working reliably and are more efficient than those with complicated switching devices for redirecting refrigerant flow in different seasons. Redirecting refrigerant flow involves additional valves and fittings which increase the capital costs and efficiency losses through leakages and pressure loss. One-way refrigerant flows (with the air flow redirected in different seasons) are simple, flexible, compact and cost efficient.
Heat pump dryers
Compared to convectional dryers, heat pumps may reduce energy consumption of food dryers by up to 80%. Other products which need drying include wood, laundry and sewage sludge. The product quality can be optimised more easily by using a heat pump because of the availability of greater control options for different drying chamber conditions. Water vapour from the products is absorbed by warm air, which is heated by the gas cooler prior to the drying chamber. As this air passes through the evaporator, it is dehumidified and cooled down before returning to the gas cooler to be reheated. This closed air cycle provides remarkable energy conservation, contributes tremendously high energy efficiency (by participating in the heat transfer in both the gas cooler and evaporator) and reduces environmental contaminants and the unpleasant odour experienced in some drying processes, e.g. drying of sewage sludge.21 Owing to the gliding temperature and better temperature adaptation of heat exchangers, R744 heat pumps can achieve substantial energy savings when used as a dryer. Higher air temperatures can be easily and efficiently achieved in these systems, thus enhancing the moisture extraction rate.27 As more efficient R744 equipment parts are produced (e.g. compressors and heat exchangers), commercial heat pump dryers using R744 as the refrigerant are becoming a possibility.21
Transport refrigeration
The efficient, reliable and compact characteristics of R744 equipment encourage their application in the transport sector.8 R744 refrigeration systems are considered as a replacement for HFC refrigeration systems, both in public and goods (especially perishable) transport. These transport modes include perishable goods trucks, public and goods trains and ships. The relatively high density and capacity of R744 is an added advantage in this sector. Furthermore, because of the global nature of transport refrigeration, an environmentally benign alternative is required which is available everywhere (even in rural areas where the refrigerated trucks operate) and acceptable to all (some countries in which the refrigerated ships dock have stringent environmental rules which need to be strictly adhered to5).
Environment control units
Military operations usually require space conditioning for their temporary shelters, command modules and vehicles, which should be able to withstand the unique operational environment. The compactness of R744 equipment in addition to its availability globally has led to an increased interest in R744 space-conditioning systems for the military.5,28
Future applications for R744
The potential of R744 refrigeration is far wider than the applications discussed above. Experts assert that R744 as a refrigerant shows promise of capturing more markets, even outside the refrigeration industry, although currently, it is the natural refrigerant with the widest range of use.21 Some future potential uses of R744 as a refrigerant are information technology (IT) equipment cooling, industrial heat pumps and industrial waste heat recovery.
Information technology equipment cooling
The necessity of high performance data centres is increasing as the need for information to be made available at anytime from anywhere to anyone grows. For this to be possible, high-density data storage and processing environments are required, and must be coupled with an efficient heat absorption system which ensures that working environments are not overheated by processes and that a smooth operation and flow of information can exist. A high-efficiency cooling system with high COP and automatic control is therefore paramount for both energy-saving purposes and smooth operation of the data centre. R744 cooling systems can provide the heat absorption process efficiently and reliably without interfering with the IT equipment because R744 is electrically benign when compared to traditional water-based systems. Another advantage of R744 cooling systems is their compactness, which eases their integration with the IT equipment and surrounding structures. Furthermore, it is a form of waste heat recovery with the possibility of simultaneous heating and cooling.21
Industrial heat pumps
R744 can be used for recovery of useful waste heat while at the same time providing low temperature heat (up to 130 °C) for industrial processes. The application of R744 heat pumps in this sector therefore has potential while providing energy savings. Heat pump drying is an example of such an application already introduced into the market. Other sectors with future potential are21:
- Washing processes: Warm to hot water is required in some industrial processes, for example, textile washing, washing of food and cosmetics production facilities. This hot water can easily and efficiently be provided by R744 heat pumps.
- Process water: Warm process water is required in certain industries, for example, the production of starch and other viscous chemicals.
- Process air: Some industries use warm to hot air instead of water, for example, in the production of flake boards and some plastic containers.
- Steaming processes: Steam is required in most manufacturing processes as a heating media or just for cleansing purposes. A good example where R744's simultaneous heating and cooling can be effectively applied is in the regeneration of activated carbon filters in order to recover solvents. In this process, steam generated by the R744 gas cooler vaporises the activated carbons which are loaded with solvents. Subsequently, the steam absorbs the solvent, which is condensed in the evaporator and extracted. Even though it might be difficult to produce steam at the required temperature and pressure with the current R744 heat pump technology, studies are being conducted to investigate this possibility. Still, R744 can be used to preheat feed water to the boilers, thus reducing the amount of energy used in the boiler while improving overall system efficiency.
Industrial waste heat recovery
Many industrial processes produce heat that is released to the environment as waste because it cannot be reused effectively. This waste heat can be used efficiently by a R744 heat pump to produce useful heat, either for air conditioning or for tap water applications. This waste heat recovery will reduce the energy costs of generating hot tap water and/or building air. In addition, the industry will reduce its waste product treatment by meeting temperature regulations concerning waste products released into the environment and will reduce expenditure on waste products while providing useful heat.
R744 applications in South Africa
South Africa is ranked 16th in the world in terms of total primary energy consumption. This makes it the highest consumer of commercial energy per capita in Africa and thus a relatively energy intensive country. However, in terms of energy efficiency, South Africa performs poorly. In fact, it was ranked among the bottom 50 of the 150 countries compared in a study.29 Therefore, energy costs form a large part of total production costs because the efficiency of utilisation is low. South Africa produces about 2% of the world's carbon emissions and thus requires a check in its carbon emissions.30 With the threat of less energy created than the current energy demand, an energy crisis is looming in South Africa and energy efficiency is essential through incentives from the government and carbon tax adoption. South Africa committed itself at the Copenhagen Accord in 2009 that by 2020 it would reduce its greenhouse gas emissions to 34% below its projected emission value and by 2025 by 42%. By the look of its energy, environmental and industrial policies, South Africa is trying to achieve this commitment even though it is not legally binding, simply because of the numerous economic and resource advantages that can be achieved by being a low carbon economy.31
Refrigeration equipment (i.e. industrial, commercial and residential equipment) accounts for a sizeable chunk of national energy consumption. Unfortunately, in addition to the indirect effect of this equipment on the environment, most of the equipment in South Africa, and Africa in general, also still requires synthetic refrigerants. Ozone depleting and global warming refrigerants like R22 are still common in most refrigerators, while new equipment uses high GWP refrigerants like R134a and R404a.32 Between 2005 and 2009 in South Africa, HCFCs such as R22 had the highest consumption of 25 759 t (81.4%), HFCs such as R134a had a consumption of 3.439 t (10.9%), HFC blends such as R404a of 1089 t (3.4%), methyl bromide of 747 t (2.4%) and bromochloromethane of 624 t (2%).33 With high refrigerant leakage rates reported in the literature (between 10% and 15%),26,34 it can be assumed that system leaks are also relatively high in South Africa. Therefore, direct emission effects are high too. South Africa, being a signatory to the Montreal Protocol, needs to reduce its HCFC consumption to 90% of baseline (2010 amount) by 2015, to 65% by 2020, to 32.5% by 2025, to 2.5% by 2030 and be completely phased out by 2040, while its methyl bromide consumption is supposed to be completely phased out by 2015.33 To reduce the environmental effects due to refrigerant leakages, it is paramount that these synthetic refrigerants are replaced by more efficient natural and environmentally friendly ones. While these environmentally friendly refrigerants are getting the required attention and acceptance in Europe and Asia, application is still at the infancy stage in South Africa and Africa in general.32
Currently in South Africa, there are approximately 30 industrial and commercial installations of R744 refrigeration equipment and a negligible number of R744 residential and transport refrigeration installations. In a country where industrial and commercial refrigeration installations exceed 2000 and there are millions of residential and commercial refrigerators, it is clear how far behind we are in terms of green technology. Still, compared to the rest of Africa, South Africa leads with this green technology, therefore emphasising how the continent is lagging behind. The country's main power generating company (Eskom) has also encouraged the adoption of heat pumps and environmentally friendly refrigerators. This was brought about by its agreement to the carbon tax regime of reducing carbon emissions by 20% by 2025. The Department of Energy of South Africa forecast that for this target to be easily and smoothly achieved, alternative and efficient technologies need to be adopted.35 To achieve carbon emission targets, Eskom has encouraged all stakeholders to reduce electricity usage by 40% by 2015 while for ozone depletion substances, the government introduced a policy of eliminating the use of R22 in all new commercial and industrial refrigerators.36
As per Eskom estimates, for every 1 kWh of electricity produced at the power station, 1.4 L of water and 530 g of coal are consumed. The pollutants emitted from the generation of 1 kWh of electricity include: 7.75 g of SO2, 4.18 g of NOx, 990 g of CO2 and 157 g of ash. These estimates do not include the pollutants emitted while mining the coal, i.e. directly from the mine (methane which has a GWP of 20 is normally released during mining), indirectly from the mining equipment, from transporting the coal to the power station, and from establishing and maintaining the power station and mine infrastructure. By Eskom estimates, if a single household converts from electrical geysers to a normal water heat pump with a COP of about 3, approximately 355 kWh of electricity will be saved in 1 month. If the above pollutant estimates are used, it is possible to protect the environment from a large amount of pollutants. This becomes clearer if the estimated 5.4 million households which use electric geysers in South Africa all convert to heat pumps. Still, this is a conservative estimate of pollutants saved because it only considers power station production. In addition, if heat pumps with higher efficiencies are used, fewer pollutants will be released.37
In addition to being environmentally friendlier, the efficiency of operation of R744 refrigerators is also comparable (if not better) to conventional systems, thus they are also competitive in terms of indirect emissions, as reported in the literature.32 In the first R744 refrigeration supermarket in South Africa (Woolworths), a 35% reduction of electricity consumption was achieved, resulting in much fewer pollutants being released.36 The only setback is that most studies concerning R744 refrigeration and other environmentally benign alternatives have been done in countries with cold climates. Studies in warm tropical climates like in South Africa and Africa in general are scarce, especially in the open literature. It is therefore paramount that more studies are conducted in this field so as to ascertain the advantages of these systems in warm regions. With the existing R744 installations, the commercial and industrial owners have reported satisfactory performance to date and are motivated to install more of these refrigerators. Still, because of the perceived low efficiencies of R744 refrigerators in warm climates owing to their low critical point, additional studies are required in order to further improve the performance of these refrigerators and make them even more attractive.
Therefore, even though the use of R744 has solved the direct effect of emissions on the environment, if there are no studies to investigate the indirect effects of these systems, we might end up with inefficient systems consuming much energy, thus still affecting the environment.38 Improvement of existing technical solutions and the formulation of more solutions to existing R744 refrigeration problems is vital if this technology is to be accepted by all, especially in areas with warm climates. Their installation and operating costs should be lower than that for conventional systems. Theoretical and experimental studies should be conducted on existing and new R744 systems in order to perfect this technology. System optimisation and modification are paramount if this technology is to completely replace conventional synthetic refrigerants. Also, national policies geared to encourage R744 refrigeration and other green technologies are important so as to clear the way and provide support for these technologies. In addition to research and industrial input, other stakeholders like the government and other policy organisations are important in facilitating the widespread use of these technologies.
Conclusion
Carbon dioxide as a refrigerant was explored from its historical background to specific properties which affect its performance in the refrigeration industry. As a result of its superior properties, especially concerning refrigeration, we believe R744 will be a dominant refrigerant in many applications of the refrigeration technology in the future.
Acknowledgement
We acknowledge the financial and logistical assistance of Eskom and the Tshwane University of Technology.
Authors' contributions
PM. conducted the literature review under the guidance of Z.H. who was also the project leader. Both authors wrote the paper.
References
1. Bensafi A, Thonon B. Transcritical R744 (CO2) heat pumps. Report no. 2414173. Villeurbanne: Centre Technique Des Industries; 2007. [ Links ]
2. Mohanraj M, Jayaraj S, Muraleedharan C. Environment friendly alternatives to halogenated refrigerants - A review. Int J Greenh Gas Con. 2009;3(1):108-119. http://dx.doi.org/10.1016/j.ijggc.2008.07.003 [ Links ]
3. Hua T, Zhao Y MinXia L, YiTai M. Research and application of CO2 refrigeration and heat pump cycle. Sci China Ser E-Technol Sci. 2009;52(6):1563-1575. http://dx.doi.org/10.1007/s11431-009-0175-4 [ Links ]
4. Sarkar J. Transcritical carbon dioxide heat pumps for simultaneous cooling and heating. Kharagpur: Indian Institute of Technology; 2005. [ Links ]
5. Kim MH, Pettersen J, Bullard CW. Fundamental process and system design issues in CO2 vapor compression systems. Prog Energ Combust. 2004;30(2):119-174. http://dx.doi.org/10.1016/j.pecs.2003.09.002 [ Links ]
6. Ma Y, Liu Z, Tian H. A review of transcritical carbon dioxide heat pump and refrigeration cycles. Energy. 2013;55:156-172. http://dx.doi.org/10.1016/j.energy.2013.03.030 [ Links ]
7. Bodinus WS. The rise and fall of carbon dioxide systems. ASHRAE J. 1999;41(4):37-42. [ Links ]
8. Neksa P Walnum HT, Hafner A. CO2 - A refrigerant from the past with prospects of being one of the main refrigerants in the future. Paper presented at: The 9th IIR Gustav Lorentzen Conference; 2010 Apr 2-14; Sydney, Australia. [ Links ]
9. Lorentzen G. Revival of carbon dioxide as a refrigerant. Int J Refrig. 1994;17(5):292-301. http://dx.doi.org/10.1016/0140-7007(94)90059-0 [ Links ]
10. Hoffmann G, Plehn W. Natural refrigerants for mobile air conditioning in passenger cars. Dessau: German Federal Environment Agency, Office GFEAP; 2010. [ Links ]
11. Hashimoto K. Technology and market development of CO2 heat pump water heaters (ECO CUTE) in Japan. Boras, Sweden: IEA Heat Pump Centre; 2006. [ Links ]
12. Kolke GV. Natural refrigerants: Sustainable ozone- and climate-friendly alternatives to HCFCs. Eschborn: Deutsche Gesellschaft für Technische Zusammenarbeit GmbH (GTZ); 2008. [ Links ]
13. Neksa P Rekstad H, Zakeri GR, Schiefloe PA. CO2 heat pump water heater: Characteristics, system design and experimental results. Int J Refrig. 1998;21(3):172-179. http://dx.doi.org/10.1016/S0140-7007(98)00017-6 [ Links ]
14. Girotto S, Minetto S, Neksa P. Commercial refrigeration system using CO2 as the refrigerant. Int J Refrig. 2004;27(7):717-723. http://dx.doi.org/10.1016/j.ijrefrig.2004.07.004 [ Links ]
15. Sawalha S. Carbon dioxide in supermarket refrigeration. Stockholm: Royal Institute of Technology; 2008. [ Links ]
16. Stene J. Residential CO2 heat pump system for combined space heating and hot water heating. Trondheim: Norwegian University of Science and Technology; 2004. [ Links ]
17. Hwang Y Radermacher R. Theoretical evaluation of carbon dioxide refrigeration cycle. HVAC&R Res. 1998;4(3):245-263. http://dx.doi.org/10.1080/10789669.1998.10391403 [ Links ]
18. NIST. REFPROP V.6.0. NIST thermodynamic and transport properties of refrigerants and refrigerant mixtures database [database on the Internet]. [ Links ] No date [cited 2015 Aug 31]. Available from: http://www.boulder.nist.gov/div838/theory/refprop.htm.
19. Stene J. Integrated CO2 heat pump systems for space heating and hot water heating in low-energy houses and passive houses. International Energy Agency (IEA) Heat Pump Programme - Annex 32 - Workshop; 2007 Dec 06; Kyoto, Japan. Paris: International Energy Agency; 2007. p. 1-14. [ Links ]
20. Liao SM, Zhao TS, Jakobsen A. A correlation of optimal heat rejection pressures in transcritical carbon dioxide cycles. Appl Therm Eng. 2000;20(9):831-841. http://dx.doi.org/10.1016/S1359-4311(99)00070-8 [ Links ]
21. Reulens W. Natural refrigerant CO2. Diepenbeek: Katholieke Hogeschool Limburg, Diepenbeek C; 2009. [ Links ]
22. Neksa P CO2 heat pump systems. Int J Refrig. 2002;25(4):421-427. http://dx.doi.org/10.1016/S0140-7007(01)00033-0 [ Links ]
23. Shecco. 50 examples of natural refrigerant stories in article 5 countries [document on the Internet]. [ Links ] No date [cited 2015 Aug 31]. Available from: http://conf.montreal-protocol.org/meeting/oewg/31oewg/ngo-publications/Observer%20Publications/50%20Examples-Part1-of%20Natural%20Refrigerants%20Stories%20in%20Article%205%20Countries.pdf
24. Kullheim J. Field measurements and evaluation of CO2 refrigeration systems for supermarkets. Stockholm: KTH School of Industrial Engineering and Management; 2011. [ Links ]
25. Hafner A, Neksa P, editors. Global environmental and economic benefits of introducing R744 mobile air conditioning. Paper presented at: The 2nd International Workshop on Mobile Air Conditioning and Auxiliary Systems; 2007 Nov 29-30; Orbassano, Italy. [ Links ]
26. Freléchox D. Field measurements and simulations of supermarkets with CO2 refrigeration systems [MSc dissertation]. Stockholm: KTH Royal Institute of Technology; 2009. [ Links ]
27. Steimle F. CO2 drying heat pumps. Essen: Institut fuer Angewandte Thermodynamik und Klimatechnik, Universitaet Esse; 1998. [ Links ]
28. Manzione JA, Neksa P Halozan H. Development of carbon dioxide environmental control unit for the US Army. Paris: Institut International du Froid; 1998. [ Links ]
29. Department of Environmental Affairs and Tourism (DEAT). How energy generation causes environmental change in South Africa. Pretoria: DEAT; 2001. [ Links ]
30. Fawkes H. Energy efficiency in South African industry. J Energy South Afr. 2005;16(4):18-25. [ Links ]
31. Covary T. Development of 1st draft of a national energy efficiency action plan (NEEAP) for the Republic of South Africa. Johannesburg: Unlimited Energy; 2013. [ Links ]
32. Siegele B. Conversion of supermarket refrigeration systems from F-gases to natural refrigerants. Eschborn: Federal Ministry for the Environment, Nature Conservation and Nuclear Safety, Proklima P; 2008. [ Links ]
33. United Nations. Goal 7: Ensure environmental sustainability. In: The Millenium Development Goals report. New York: United Nations; 2010. p. 52-64. Available from: http://www.un.org/millenniumgoals/pdf/MDG%20Report%202010%20En%20r15%20-low%20res%2020100615%20-.pdf [ Links ]
34. Likitthammanit M. Experimental investigations of NH3/CO2 cascade and transcritical CO2 refrigeration systems in supermarkets. Stockholm: KTH School of Energy and Environmental Technology; 2007. [ Links ]
35. Blackharvest Trading. Energy efficiency CO2 systems [document on the Internet]. [ Links ] c2010 [cited 2015 Aug 31]. Available from: http://www.doe-irp.co.za/hearing1/BLACKHARVEST_Carbon_Efficiency.pdf
36. Smith J. Good business journey: Retail sector. African Utility Week: Delivering Beyond Tomorrow 2013. [ Links ]
37. Eskom. Annual report. Johannesburg: Eskom; 2011. [ Links ]
38. CottineauV. Calculation and comparison of different supermarket refrigeration systems. Stockholm: KTH School of Industrial Engineering and Management; 2010. [ Links ]
39. Riffat SB, Afonso CF, Oliveira AC, Reay DA. Natural refrigerants for refrigeration and air-conditioning systems. Appl Therm Eng. 1997;17(1):33-42. http://dx.doi.org/10.1016/1359-4311(96)00030-0 [ Links ]
40. Heaney C, Swinard R, Pang A, West S. Natural refrigerants case studies. Melbourne: Australian Institute of Refrigeration, Airconditioning and Heating; 2007. [ Links ]
41. Kristensen AM. Natural refrigerants for new applications. Oslo: Nordic Chemicals Group (NKG), Kuldebrukere FF; 2008. [ Links ]
42. Mate J, Papathanasopoulos C, Latif S. Cool technologies: Working without HFCs. Amsterdam: Greenpeace; 2012. [ Links ]
 Correspondence:
Correspondence:
Paul Maina
Department of Mechanical
Engineering, Tshwane University
of Technology, Private Bag X680
Pretoria 0001
South Africa
Email: mainap@tut.ac.za
Received: 31 July 2014
Revised: 13 Nov. 2014
Accepted: 19 Jan. 2015














