Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.110 n.11-12 Pretoria Nov./Dec. 2014
http://dx.doi.org/10.1590/sajs.2014/20130293
RESEARCH ARTICLE
http://dx.doi.org/10.1590/sajs.2014/20130293
Antibacterial, antioxidant activities and cytotoxicity of plants against Propionibacterium acnes
Richa Sharma; Namrita Lall
Department of Plant Science, University of Pretoria, Pretoria, South Africa
ABSTRACT
The use of plants to treat skin ailments has strong support in the current trend of drug discovery. Propionibacterium acnes, an anaerobic pathogen, plays an important role in the occurrence of acne. The present study was conducted to evaluate the antimicrobial and antioxidant activities against P acnes and cytotoxic effects of 48 medicinal plants grown in South Africa. The broth dilution and DPPH radical scavenging methods were used to determine antibacterial and antioxidant activities, respectively. Cytotoxicity was determined on mouse melanocytes (B16-F10). The ethanolic bark extract of Acacia galpinii Burtt Davy. (Leguminosae) exhibited the lowest minimum inhibitory concentration of 62.5 µg/mL. Excellent antioxidant activity was shown by Aspalathus linearis (Burm.f.) R.Dahlgren (Leguminosae), Combretum apiculatum Sond. (Combretaceae), Harpephyllum caffrum Bernh. ex Krauss (Anacardiaceae) and Sclerocarya birrea Hochst. (Anacardiaceae), with 50% radical scavenging activity (EC50) at concentrations ranging from 1.6 µg/mL to 3.5 µg/mL. Greyia sutherlandii Hook. & Harv. (Greyiaceae) also exhibited good antioxidant activity with an EC50 value of 7.9±0.23 µg/mL. A. linearis, G. sutherlandii and S. birrea showed low toxicity with 50% viability of cells (EC50) at concentrations of 125.09±0.71 pg/mL, 107.85±1.53 pg/mL and 92.07±0.09 pg/mL, respectively. The extracts of A. linearis, G. sutherlandii and S. birrea showed good antibacterial and antioxidant activities and low toxicity. Therefore, these plants can be considered as possible anti-acne agents and warrant further investigation.
Keywords: anti-acne; mouse melanocytes; medicinal plants; 2,2-diphenyl-1 picrylhydrazal; ethanolic extract
Introduction
Acne, one of the most common disorders of the skin, is a polymorphic disease with non-inflammatory (blackhead or whitehead) and inflammatory (papules, pustules, or nodules) aspects and a wide spectrum of severity. Acne can have a significant impact on the psychosocial and physical aspects of life. It affects up to 85% of adolescents to some extent but is less common among infants. Its prevalence has been estimated to be 95-100% in male adolescents and 83-85% in female adolescents.1-4
Propionibacterium acnes, a Gram-positive anaerobic bacterium, is a normal component of the microbiota of human skin. P acnes causes an increase in the secretion of sebum from sebaceous glands, which is accompanied by the thickening of the epidermis at the outlet to the pilosebaceous follicles. As a result, there is an obstruction to the flow of sebum outwards, and a comedone develops. Colonisation of the follicles with P acnes and the host's inflammatory response play a pivotal role in the development of typical inflammatory papulopustular lesions.5 In an anaerobic environment, the bacteria secretes nucleases, nuraminidases, hyaluronidases, acid phosphatises, lecithinases and other lipases. As a result of the action of these enzymes, the sebum content changes and reactive oxygen species may be released from the damaged follicular walls. Reactive oxygen species may also be the reason for the progression of inflammation in the pathogenesis of disease.6
Conventional drugs commonly used in acne treatment - such as tetracycline, erythromycin, mynocycline and metronidazole - act as antioxidants and antibacterials. Benzoyl peroxide, a topical agent for the treatment of acne, shows the ability to induce an inflammatory reaction mediated by reactive oxygen species in addition to its antibacterial activity.6 These drugs also have various known side effects. The topical antibiotics can lead to dryness, redness and irritation of the skin, as well as hypopigmentation while oral antibiotics have age restrictions, can cause gastrointestinal disorders and increase the risk of venous thromboembolism.5
Herbal medicines are an important part of African tradition and also have very deep roots in the treatment of dermatological ailments. Ethnobotanical studies have documented the use of plants by traditional healers for the treatment of various skin ailments.7 Different plant parts commonly used as cosmetics or face masks, known as umemezis, are widely used in southern Africa for skin problems like inflammation, wounds, burns, eczema and puberty acne.8
Because many skin disorders like atopic dermatitis and acne are associated with inflammation and the release of free radicals, which lead to oxidative and cellular damage and bacterial infections such as P acnes, the presence of antioxidant and antimicrobial agents can explain the effectiveness of plants in the treatment of skin infections. In order to develop the therapeutic and drug potential of these plants, it is important to know whether they have any cytotoxic effects. Therefore, ethanol extracts of selected plants were evaluated for their antibacterial and antioxidant activities and cytotoxicity.
Limitations in the usage of some drugs and the prevailing side effects of the various chemically derived compounds have led to the search for alternative herbal agents to treat acne. The aim of this study was to test the effect of selected plant extracts on the pathogenic bacteria P acnes, and to identify which plant extracts could be considered as possible anti-acne agents.
Methods
Materials
Tetracycline, vitamin C, p-iodonitrotetrazolium salt and 2,2-diphenyl-1 -picrylhydrazyl (DPPH) were obtained from Sigma-Aldrich (Johannesburg, South Africa). Nutrient agar and nutrient broth were obtained from Merck SA (Pty) Ltd (Johannesburg, South Africa). P acnes (ATCC 11827) was purchased from Anatech Company South Africa (Johannesburg, South Africa). The cell culture reagents and the equipment were purchased from Highveld Biological (Johannesburg, South Africa), Labotech (Johannesburg, South Africa) and The Scientific Group (Johannesburg, South Africa). The B16-F10 mouse melanocyte cell line was obtained from Highveld Biological.
Preparation of plant extracts
Different plant parts (leaves, roots, bark and twigs) were collected from the Botanical Garden of the University of Pretoria (Pretoria, South Africa). The plants were identified by a taxonomist, Professor A.E. (Braam) van Wyk, at the H.G.W.J. Schweickerdt Herbarium of the University of Pretoria. The shade-dried plant material (80 g) was ground with a mechanical grinder, then soaked in 300 mL of ethanol and left on a shaker for 3 days. The plant material was then filtered and the solvent was evaporated under vacuum (Buchi Rotavapor, Labotech, Switzerland) to yield dry extracts. The plants were selected based on their medicinal usage as summarised in Table 1.
Antibacterial bioassay
The minimal inhibitory concentration (MIC) of the ethanolic extracts of the 48 selected plants was determined by a microdilution assay. This assay was done using the method described by Mapunya et al.36, with slight modifications. For this purpose, P acnes (ATCC 11827) was cultured from a Kwik-Stick on nutrient agar and incubated at 37 0C for 72 h under anaerobic conditions. The ethanolic extracts were dissolved in 10% dimethyl sulphoxide (DMSO) to obtain a stock solution of 2 mg/mL. The positive control (tetracycline) was dissolved in sterile distilled water to obtain a stock solution of 0.2 mg/mL. The 96-well plates were prepared by dispensing 100 μL of the nutrient broth into each well; 100 μL of the plant stock samples and positive control were added to the first row of wells in triplicate. Twofold serial dilutions were made in broth over a range to give concentrations of 3.9-500 μg/mL and 0.3-50 μg/mL for the plant extracts and positive control, respectively. The 72-h culture of bacteria was dissolved in nutrient broth and the suspensions were adjusted to 0.5 McFarland standard turbidity at 550 nm. Then 100 μΙ. of this bacterial inoculum with 1 05-106 CFU/mL was added to all the wells. The wells with 2.5% DMSO and bacterial suspension without samples served as the solvent and negative controls, respectively. The plates were then incubated at 37 0C for 72 h under anaerobic conditions. The MIC (defined as the lowest concentration that showed no bacterial growth) was determined by observing the colour change in the wells after the addition of p-iodonitrotetrazolium salt.
Antioxidant assay
The antioxidant activity of selected plant extracts was investigated using the DPPH radical scavenging method as previously described by Du Toit et al.37, with slight modifications. DPPH is a free radical, which is stable at room temperature and produces a violet solution in ethanol. When reduced in the presence of an antioxidant molecule, it gives rise to a colourless solution. DPPH was dissolved in ethanol to obtain a solution of 0.04% w/v.
The selected plant samples and the positive control (vitamin C) stock solutions (2 mg/mL) were serially diluted to final concentrations ranging from 0.78 μg/mL to 100 μg/mL. Ethanol and DPPH without any plant material were used as blanks while plant samples diluted in distilled water were used as controls. DPPH solution (90 μg/mL) was then added to all the wells except for the controls and allowed to react at room temperature. After 30 min, the absorbance values were measured at 515 nm using a Biotek Power-wave XS multiwell reader (A.D.P, Johannesburg, South Africa). The values were converted into the percentage antioxidant activity (AA) using the formula given below. The 50% inhibitory concentration (EC50) values were then calculated by linear regression of the plots using GraphPad Prism version 4.
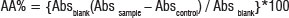
Mouse melanocyte cytotoxicity assay
The cytotoxicity of selected plant extracts was determined following a previously described method.36 Briefly, mouse melanocyte (B16-F10) cells were plated in complete Roswell Park Memorial Institute medium (10% foetal bovine serum and 1% gentamycin) directly in the wells of a 96-well plate (105 cells per well). After an overnight incubation at 37 0C in 5% CO2 and a humidified atmosphere, extract samples and the positive control (actinomycin D) were added to the cells to give the final concentrations of plant extract and positive control of 3.13-400 μg/mL and 0.03x10-2-0.05 μg/mL, respectively. Plates were incubated at 37 °C in 5% CO2 in a humidified atmosphere for 3 days. The toxicity effects of the extracts on the B16-F10 cells were assayed using the sodium 3'-[1-(phenyl amino-carbonyl)-3,4-tetrazolium]-bis-[4-methoxy-6-nitrobenzene sulphonic acid hydrate (XTT) cytotoxicity assay. Thereafter, 50 μL of XTT reagent (1 mg/mL XTT with 0.383 mg/ mL penazine methosulphate) was added to the wells and incubated for 1 h. The optical densities of the wells were measured at 450 nm with background subtraction at 690 nm. Cell survival was assessed by comparison with the controls (medium with DMSO). The EC50 value, which represents the concentration of plant extract that causes death in 50% of the cells, was analysed using GraphPad Prism version 4.
Statistical analysis
All the assays were performed in triplicate with three independent studies for each assay. EC50 values for antioxidant and cytotoxicity tests were derived from a non-linear regression model (curve fit) based on a sigmoidal dose response curve (variable) and computed using GraphPad Prism version 4 (GraphPad, San Diego, CA, USA).
Results
Antibacterial activity of ethanolic extracts
The antibacterial activity of the selected plants against P. acnes is summarised in Table 2. After the addition of p-iodonitrotetrazolium salt, the MIC value of the positive drug control (tetracycline) was determined to be 3.1 μg/mL. Most of the plants exhibited antibacterial activity at MIC values ranging from 62.5 μg/mL to 500 μg/mL. The ethanolic bark extract of Acacia galpinii Burtt Davy. (Leguminosae) exhibited the lowest MIC value of 62.5 μg/mL. Some of the plant extracts -Aspalathus linearis (Burm.f.) R.Dahlgren (Leguminosae), Combretum apiculatum Sond. (Combretaceae), Combretum molle Engl. & Diels (Combretaceae), Galenia africana L. (Aizoaceae), Greyia sutherlandii Hook. & Harv. (Greyiacece), Harpephyllum caffrum Bernh. ex Krauss (Anacardiaceae), Ranunculus repens L. (Ranunculaceae), Sclerocarya birrea Hochst. (Anacardiaceae) and Warburgia salutaris (G. Bertol.) Chiov. (Cancellaceae) - exhibited MIC values of 125 μg/mL. Another 28 extracts inhibited the growth of bacteria at MIC values ranging from 250 μg/mL to 500 μg/mL, whereas the remaining 10 extracts did not show any antibacterial activity, even at the highest concentration (500 μg/mL) tested. A threshold MIC value of ~100 μg/mL is suggested for rating plant extracts as having significant antimicrobial activity.38 Therefore, the plant extracts exhibiting MIC values ranging from 62.5 μ/mL to 125 μg/mL were selected for the evaluation of antioxidant activity.
Antioxidant activity of selected extracts
Vitamin C, a widely used antioxidant compound, was used as the positive control (EC50=1.98 ±0.005 μg/mL). The plant extracts which demonstrated excellent radical scavenging activity, comparable to vitamin C, were A. linearis (EC50 of 3.5±0.5 μ/ml), C. apiculatum (EC50 of 1.6±10.02 μ/ml), H. caffrum (EC50 of 2.610.21 μg/mL) and S. birrea (EC50 of 2.06±0.03 μg/mL) (Figure 1). The plant extracts of C. molle and G. sutherlandii also showed good antioxidant activity with EC50 values of 9.8310.8 μg/mL and 7.9±0.23 μg/mL, respectively (Figure 1). A. galpinii and R. repens exhibited comparatively higher antioxidant activity with EC50 values of 16.05±2.25 μg/mL and 24.7±2.05 μg/mL, respectively. The extracts of G. africana and W. salutaris exhibited the lowest radical scavenging activity with the highest EC50 values of 90.92±1.2 μg/mL and 111±2.5 μg/mL, respectively.
Cytotoxicity of selected extracts
Cytotoxicity was assessed on the plant extracts which demonstrated EC50 values of <10 μg/mL for radical scavenging activity. The plant extracts; of A. linearis, G. sutherlandii and S. birrea showed low toxicity with 50% viability of cells (EC50) at concentrations of 125.09±0.71 μg/mL, 107.85±1.53μg/mL and 92.07±0.09 μg/mL, respectively (Figure 2). During a previous study by our research group, the leaf extract of H. caffrum showed toxicity to B16-F10 cells at a concentration of 100 μg/mL.39 The plant extract of C. molle showed moderate toxicity with an EC50 value of 48.83±0.21 μg/mL, whereas C. apiculatum was found to be the most toxic with an EC50 value of 12.15±0.03 μg/mL and was found to be lethal to almost all cells at the highest concentration of 400 μg/mL. Actinomycin D, the positive control, showed an EC50 value of 4.5x10-3 ± 0.5x10-3 μg/mL (Figure 2).
Discussions
Plant extracts were explored for antibacterial activity against P acnes. Similar to our findings, the ethanolic extract of Coscinium fenestratum (Gaertn.) Colebr. (Menispermaceae) inhibited the growth of P acnes at an MIC value of 46 μg/mL.40 According to Tsai et al.41, methanolic extracts of Rosa damascena Mill (Rosaceae), Eucommia ulmoides Oliv. (Eucommiaceae) and Ilex paraguariensis A. St.-Hil. (Aquifoliaceae) inhibited the growth of P acnes at MIC values of 2000 μg/mL, 500 μg/mL and 1000 μg/mL, respectively. To the best of our knowledge, the present study is the first scientific report of the antibacterial activity of all the selected plants against P acnes. However, some of the plants used in this study have been previously reported to be active against other pathogens. In another study, leaf extracts of A. linearis showed zones of inhibition against Bacillus cereus, Micrococcus luteus and Candida albicans of 7.0 mm, 6.4 mm and 8.5 mm, respectively.42
The antibacterial activity of C. apiculatum against Staphylococcus aureus, Pseudomonas aeruginosa and Escherichia coli was reported by Serage43. The acetone extract of the stem bark of C. molle showed antimicrobial activity against E. coli and Shigella spp. at an MIC of 50 000 μg/mL. The extract also showed inhibitory effects on the fungus C. albicans with complete inhibition at a concentration of 400 μg/mL.44 In a study done by Lining et al.45, the crude methanolic extract of Diospyros lycioides Desf. (Ebenaceae) showed activity against Streptococcus mutans and Prevotella intermedia at an MIC of 1250 μg/mL. In contrast, our results showed no activity of the ethanolic extract of D. lycioides against P acnes. In another study conducted by Mativandlela et al.46, the ethanolic extract of G. africana showed antimycobacterial activity against Mycobacterium tuberculosis and Mycobacterium smegmatis at MIC values of 780 μg/mL and 1200 μg/mL, respectively. The ethanolic extract of H. caffrum was reported to be active against four bacterial species, namely Bacillus subtilis, E. coli, Klebsiella pneumoniae and S. aureus, while an aqueous extract showed activity against C. albicans.47The acetone extracts of the bark and leaves of S. birrea were reported to be active against S. aureus, P. aeruginosa, E. coli and Enterococcus faecalis at MIC values ranging from 150 μg/mL to 3000 μg/mL.48 In a study done by Motsei et al.49, the leaf extracts of W. salutaris inhibited growth of C. albicans at MIC values ranging from 12 500 μg/mL to 25 000 μg/mL and the bark extracts showed growth of inhibition against S. aureus, Staphylococcus epidermis, B. subtilis and E. coli.50 No reports regarding the antimicrobial activity of G. sutherlandii and R. repens were found in the literature. However, in the present study, both of these plants showed growth inhibitory activity against P acnes at an MIC of 125 μg/mL. In a study conducted by Eloff and Katerere51, the acetone and chloroform leaf extracts of A. galpinii inhibited the growth of S. aureus and E. coli. Similar to our findings, the ethanol bark extract of A. galpinii exhibited good inhibitory effect on P acnes (MIC 62.5 μg/ml). However, no reports on the antimicrobial activity of bark extracts of A. galpinii were found in the literature.
Acne is associated with the production of free radicals along with the infection of P acnes. Reactive oxygen species are produced as a result of the action of hydrolytic enzymes released from bacteria on the follicular walls of pilosebaceous units. Therefore, the plant extracts were evaluated for antioxidant activity along with antibacterial activity. In our study, the ethanol extracts of A. linearis, C. apiculatum, H. caffrum, S. birrea, C. molle and G. sutherlandii exhibited significant antioxidant activity with EC50 values of <10 μg/mL. Our results are in agreement with other researchers. During a previous study by Joubert et al.52, the DPPH radical scavenging activity of A. linearis and its constituents were confirmed. The polar fractions of C. apiculatum showed antioxidant activity with an EC50 value of 3.91 μg/mL.53 The DPPH radical scavenging activity of H. caffrum and S. birrea was confirmed by Moyo et al.54 with EC50 values of 6.8 μg/mL and 5.02 μg/mL, respectively. In another study, acetone and dichloromethane extracts of C. molle displayed antioxidant activity after spraying with DPPH.55 It has been reported that DPPH free radicals abstract the phenolic hydrogen of the electron-donating molecule, which could be the general mechanism for the scavenging action of flavonoids.56 Based on the mechanism of reduction of the DPPH molecule that is correlated with the presence of hydroxyl groups on the antioxidant molecule, the antioxidant activity of the polar plant extracts in the present study can be explained as a result of the presence of their phytoconstituents (phenolics or flavonoids) which are radical scavengers with an available hydroxyl group and are known to occur abundantly in plant species.
In order to evaluate the therapeutic potential of the plants, the cytotoxicity of selected samples was tested on B16-F10 cells. To the best of our knowledge, the cytotoxicity of the extracts described in the present study is reported for the first time. However, previous researchers have documented similar cytotoxic effects on different cell lines. In a study by McGaw et al.57, A. linearis showed low toxicity on vero cells and brine shrimp larvae with LD50 values of >1000 μg/mL. S. birrea showed low cytotoxicity on vero cells with an IC50 value of 361.24 μg/mL.58 According to previous studies by Fyhrquist et al.59 on the cytotoxicity of C. molle, the extract showed IC50 values of 27.7 μg/mL, 72.6 μg/mL and 42.6 μg/ mL on T24 (bladder carcinoma), HeLa (cervical carcinoma) and MCF-7 (breast carcinoma) cells, respectively, while the C. apiculatum extract showed IC50 values of 65.0 μg/mL and 40.1 μg/mL for T24 and MCF-7 cells, respectively. No records of cell cytotoxicity for G. sutherlandii were found in the literature.
The results shown in this study prove the capability of medicinal plants as anti-acne agents, although the mode of action and in vivo studies are required to give conclusive results.
Conclusions
Based on the results obtained, it can be concluded that the ethanol bark extract of A. galpinii demonstrated the best activity against P acnes with acceptable antioxidant activity. This plant might have other attributes that were not investigated in the present study which could be useful in the treatment of P acnes. Although the plant extracts of H. caffrum, C. apiculatum and C. molle showed good antibacterial and excellent antioxidant activity, these samples also showed moderate toxicity to mouse melanocyte cells. The plant extracts of A. linearis, S. birrea and G. sutherlandii also exhibited good antibacterial and antioxidant activity but had low toxicity to the mouse melanocytes; these extracts therefore have potential as anti-acne agents, either alone or in combination.
Acknowledgements
We thank the University of Pretoria and the National Research Foundation (South Africa) for financial grants.
Authors' contributions
R.S. conducted the experiments and drafted the manuscript; N.L. supervised the work and edited the manuscript.
References
1. Bloch B. Metabolism, endocrine glands and skin disease, with special reference to acne vulgaris and xanthoma. Brit J Dermatol.1931;43:61-87. http://dx.doi.org/10.1111/j.1365-2133.1931.tb09468.x [ Links ]
2. Munro-Ashman D. Acne vulgaris in public schools. Trans St John's Hosp Dermatol Soc. 1963;49:144-148. [ Links ]
3. Burton JL, Cunliffe WJ, Stafford I, Shuster S. The prevalence of acne vulgaris in adolescence. Brit J Dermatol. 1971;85:119-126. http://dx.doi.org/10.1111/j.1365-2133.1971.tb07195.x [ Links ]
4. Rademaker M, Garioch JJ, Simpson NB. Acne in school children: No longer a concern for dermatologists. Brit Med J. 1989;298:1217-1219. http://dx.doi.org/10.1136/bmj.298.6682.1217 [ Links ]
5. Shaw L, Kennedy C. The treatment of acne. Paediatr Child Health. 2007;17(10):385-389. http://dx.doi.org/10.1016/j.paed.2007.07.005 [ Links ]
6. Arican O, Kurutas EB, Sasmaz S. Oxidative stress in patients with acne vulgaris. Mediat Inflamm. 2005;6:380-384. http://dx.doi.org/10.1155/MI.2005.380 [ Links ]
7. Hutchings A, Scott AH, Lewis G, Cunningham AB. Zulu medicinal plants. An inventory. Pietermaritzburg: University of Natal Press; 1996. [ Links ]
8. Van Wyk BE, Gerick N. People's plants. A guide to useful plants of southern Africa. Pretoria: Briza Publications; 2006. [ Links ]
9. Kokwaro O. Medicinal plant of East Africa. Kampala/Nairobi/Dar es Salaam: East African Literature Bureau; 1976. [ Links ]
10. Davis RH, Rosental KY Cesario LR, Rouw GA. Processed Aloe vera administered topically inhibits inflammation. J Am Podiat Med. 1989;79:395-397. http://dx.doi.org/10.7547/87507315-79-8-395 [ Links ]
11. Lee KY Weintraub ST, Yu BP. Isolation and identification of a phenolic antioxidant from Aloe barbadensis. Free Radical Bio Med. 2000;28(2):261-265. http://dx.doi.org/10.1016/S0891-5849(99)00235-X [ Links ]
12. Van Wyk BE, Van Oudtshoorn B, Gericke N. Medicinal Plants of South Africa. Pretoria: Briza Publications; 2007. [ Links ]
13. Wink M, Van Wyk BE. Mind-altering and poisonous plants of the world. Pretoria: Briza Publications; 2008. [ Links ]
14. Agroforestry Tree Database [database on the Internet]. [ Links ] No date [cited 2010 Nov 27]. Available from: http://www.worldagroforestry.org/treedb/AFTPDFS/Annona_senegalensis.pdf
15. Ziyyat A, Legssyer A, Mekhfi H, Dassouli A, Serhrouchni M, Benjelloun W. Phytotherapy of hypertension and diabetes in oriental Morocco. J Ethnopharmacol. 1997;58:45-54. http://dx.doi.org/10.1016/S0378-8741(97)00077-9 [ Links ]
16. Ziyyat A, Boussairi EH. Cardiovascular effects of Arbutus unedo L. in spontaneously hypertensive rats. Phytother Res. 1998;12:110-113. http://dx.doi.org/10.1002/(SICI)1099-1573(199803)12:2<110::AID-PTR199>3.0.CO;2-5 [ Links ]
17. Kivcak B, Mert T. Quantitative determination of tocopherol in Arbutus unedo by TLC-densitometry and colorimetry. Fitoterapia. 2001;72:656-661. http://dx.doi.org/10.1016/S0367-326X(01)00305-7 [ Links ]
18. Pabuccuoglu A, Kivcak B, Bas M, Mert T. Antioxidant activity of Arbutus unedo leaves. Fitoterapia. 2003;74:597-599. http://dx.doi.org/10.1016/S0367-326X(03)00110-2 [ Links ]
19. Stephen AO. Micro propagation and medicinal properties of Barleria greenii and Huernia hystrix [PhD thesis]. Pietermaritzburg: University of KwaZulu-Natal; 2010. [ Links ]
20. Whistler WA, Elevitch CR. Broussonetia papyrifera (paper mulberry). Species profiles for Pacific Island Agroforestry [document on the Internet]. [ Links ] c2006 [cited 2010 Dec 02]. Available from: http://www.agroforestry.net/tti/Broussonetia-papermulb.pdf.
21. Kivack B, Mert T, Ozturk HT. Antimicrobial and cytotoxic activities of Ceratonia siliqua L. extracts. Turk J Biol. 2002;26:197-200. [ Links ]
22. Mbambezeli G. Cryptocarya wodii Engl. [ Links ] [homepage on the Internet]. c2005 [cited 2010 Nov 27]. Available from: http://www.plantzafrica.com/plantcd/crytocarwood.htm
23. Roberts M. Dahlia. Edible and medicinal flowers. Cape Town: Spearhead Press; 2007. [ Links ]
24. Vries FA, Bitar HEL, Green IR, Klassen JA, Mabusela WT, Bodo B, et al. An antifungal extract from the aerial parts of Galenia africana. In: Proceedings of the 11th NAPRECA Symposium; 2005 August 9-12; Antananarivo, Madagascar. Nairobi: NApReCa; 2005. p. 123-131. Available from: http://napreca.net/publications/11symposium/ [ Links ]
25. Mbambezeli G. Greyia flanaganii Bolus [homepage on the Internet]. [ Links ] c2002 [cited 2010 Nov 27]. Available from: http://www.plantzafrica.com/plantefg/greyiaflan.htm.
26. Martinez M. Las plantas medicinales de Mexico [Medicinal plants of Mexico]. [ Links ] 4th ed. Mexico City: Edicinoes Botas; 1959. p. 343-347. Spanish.
27. Mellado V Chavez SMA, Lozoya X. Pharmacological screening of the aqueous extracts of Magnolia grandiflora L. Arch Med Res. 1980;11 (3):335-346. [ Links ]
28. Beentje HJ. Kenya trees, shrubs and lianas. Nairobi: National Museums of Kenya; 1994. [ Links ]
29. Mabogo DEN. The ethnobotany of Vhavenda [MSc thesis]. Pretoria: University of Pretoria; 1990. [ Links ]
30. Maharaj V, Glen HF. Parinari curatifolia [homepage on the Internet]. [ Links ] c2008 [cited 2010 Nov 27]. Available from: http://www.plantzafrica.com/plantnop/parinaricurat.htm.
31. Moreman D. Native American ethnobotany. Portland, OR: Timber Press; 1998. [ Links ]
32. Gundidza M, Gweru N, Mmbengwa V Ramalivhana NJ, Magwa Z, Samie A. Phytoconstituents and biological activities of essential oil from Rhus lancea L.F. Afr J Biotechnol. 2008;7(16):2787-2789. [ Links ]
33. Bosman F. Sideroxylon inerme L. [ Links ] [homepage on the Internet]. c2006 [cited 2010 Nov 27]. Available from: http://www.plantzafrica.com/plantqrs/sideroxinerm.htm
34. Buchman DD. Herbal medicine. New York: Gramercy Books/Random House Value Publishing; 1988. p. 101-171. [ Links ]
35. Smith A. A contribution to South African Materia Medica, chiefly from plants in use among the natives. Cape Town: Juta; 1985. [ Links ]
36. Mapunya MB, Hussein AA, Rodriguez B, Lall N. Tyrosinase activity of Greyia flanaganii Bolus constituents. Phytomedicine. 2011;18:1006-1012. http://dx.doi.org/10.1016/j.phymed.2011.03.013 [ Links ]
37. Du Toit R, Volsteedt Y Apostolides Z. Comparison of the antioxidant content of the fruits, vegetables and teas measured as vitamin C equivalents. Toxicology. 2001;166:63-69. http://dx.doi.org/10.1016/S0300-483X(01)00446-2 [ Links ]
38. Kuete V. Potential of Cameroonian plants and derived-products against microbial infections. Planta Med. 2010;76:1479-1491. http://dx.doi.org/10.1055/s-0030-1250027 [ Links ]
39. Mapunya MB, Nikolova RV, Lall N. Melanogenesis and antityrosinase activity of selected South African plants. Evid Based Complement Alternat Med. 2012, Article ID 374017, 6 pages. http://dx.doi.org/10.1155/2012/374017 [ Links ]
40. Kumar GS, Jayaveera KN, Kumar CK, Sanjay UP Swamy BM, Kumar DV. Antimicrobial effects of Indian medicinal plants against acne-inducing bacteria. Trop J Pharm Res. 2007;6(2):717-723. http://dx.doi.org/10.4314/tjpr.v6i2.14651 [ Links ]
41. Tsai T, Tsai T, Wu W, Tseng JT, Tsai P In vitro antimicrobial and antiinflammatory effects of herbs against Propionibacterium acnes. Food Chem. 2010;119(3):964-968. http://dx.doi.org/10.1016/j.foodchem.2009.07.062 [ Links ]
42. Almajano MP Carbo R, Jimrnez JAL, Gordon MH. Antioxidant and antimicrobial activities of tea infusions. Food Chem. 2008;108(1 ):55-63. http://dx.doi.org/10.1016/j.foodchem.2007.10.040 [ Links ]
43. Serage A. Isolation and characterization of antibacterial compounds present in Combretum apiculatum subsp. apiculatum [MSc thesis]. Pretoria: Programme for Phytomedicine, University of Pretoria; 2008. [ Links ]
44. Asres K, Mazumder A, Bucar F. Antibacterial and antifungal activities of Combretum molle. Ethop Med J. 2006;44:269-277. [ Links ]
45. Lining C, Guo-Xian W, Van der Bijl PWu CD. Namibian chewing stick, Diospyros lycioides, contains antibacterial compounds against oral pathogens. J Agri Food Chem. 2000;48(3):909-914. http://dx.doi.org/10.1021/jf9909914 [ Links ]
46. Mativandlela SPN, Meyer JJM, Hussein AA, Houghton PJ, Hamilton CJ, Lall N. Activity against Mycobacterium smegmatis and M. tuberculosis by extracts of South African medicinal plants. Phytother Res. 2008;22(6):841-845. http://dx.doi.org/10.1002/ptr.2378 [ Links ]
47. Buwa LV Van Staden J. Effects of collection time on the antimicrobial activities of Harpephyllum caffrum bark. S Afr J Bot. 2007;73(2):242-247. http://dx.doi.org/10.1016/j.sajb.2006.09.006 [ Links ]
48. Eloff JN. Antibacterial activity of Marula (Sclerocarya birrea (A. Rich.) Hochst. subsp. caffra (Sond.) Kokwaro, (Anacardiaceae) bark and leaves. J Ethnopharmacol. 2001;76(3):305-308. http://dx.doi.org/10.1016/S0378-8741(01)00260-4 [ Links ]
49. Motsei ML, Lindsey KL, Van Staden J, Jager AK. Screening of traditionally used South African plants for antifungal activity against Candida albicans. J Ethnopharmacol. 2003;86(2):235-241. http://dx.doi.org/10.1016/S0378-8741(03)00082-5 [ Links ]
50. Rabe T, Van Staden J. Antibacterial activity of South African plants used for medicinal purposes. J Ethnopharmacol. 1997;56(1 ):81 -87. http://dx.doi.org/10.1016/S0378-8741(96)01515-2 [ Links ]
51. Eloff JN, Katerere DR. Variation in chemical composition, antibacterial and antioxidant activity of fresh and dried Acacia leaf extracts. S Afr J Bot. 2004;70(2):303-305. [ Links ]
52. Joubert E, Winterton P Britz TJ, Ferreira D. Superoxide anion and α,α-diphenyl-β-picrylhydrazyl radical scavenging capacity of rooibos (Aspalathus linearis) aqueous extracts, crude phenolic fractions, tannin and flavonoids. Food Res Int. 2004;37(2):133-138. http://dx.doi.org/10.1016/j.foodres.2003.09.011
53. Aderogba MA, Kgatle DT, McGaw LJ, Eloff JN. Isolation of antioxidant constituents from Combretum apiculatum subsp. apiculatum. S Afr J Bot. 2012;79:125-131. http://dx.doi.org/10.1016/j.sajb.2011.10.004 [ Links ]
54. Mbyo M, Ndhlala AR, Finnie JF, Van Staden J. Phenolic composition, antioxidant and acetylcholinesterase inhibitory activities of Sclerocarya birrea and Harpephyllum caffrum (Anacardiaceae) extracts. Food Chem. 2010;123(1):69-76. http://dx.doi.org/10.1016/j.foodchem.2010.03.130 [ Links ]
55. Masoko P Eloff JN. Screening of twenty-four South African Combretum and six Terminalia species (Combretaceae) for antioxidant activities. Afr J Tradit Complement Altern Med. 2007;4(2):231-239. [ Links ]
56. Ratty AK, Sunamoto J, Das NP Interaction of flavonols with 1,1 - diphenyl- 2-picrylhydrazyl free radical liposomal membranes and soyabean lipoxygenase- 1. Biochem Pharmacol. 1998;37:989-995. http://dx.doi.org/10.1016/0006-2952(88)90499-6 [ Links ]
57. McGaw LJ, Steenkamp V Eloff JN. Evaluation of Athrixia bush tea for cytotoxicity, antioxidant activity, caffeine content and presence of pyrrolizidine alkaloids. J Ethnopharmacol. 2007;110(1):16-22. http://dx.doi.org/10.1016/j.jep.2006.08.029 [ Links ]
58. Gathirwa JW, Rukunga GM, Njagi ENM, Omar SA, Mwitari PG, Guantai AN, et al. The in vitro anti-plasmodial and in vivo anti-malarial efficacy of combinations of some medicinal plants used traditionally for treatment of malaria by the Meru community in Kenya. J Ethnopharmacol. 2008;115(2):223-231. http://dx.doi.org/10.1016/j.jep.2007.09.021 [ Links ]
59. Fyhrquist P Mwasumbi L, Vuorela P Vuorela H, Hiltunen R, Murphy C, et al. Preliminary antiproliferative effects of some species of Terminalia, Combretum, and Pteleopsis collected in Tanzania on some human cancer cell lines. Fitoterapia. 2006;77(5):358-366. http://dx.doi.org/10.1016/j.fitote.2006.05.017 [ Links ]
 Correspondence:
Correspondence:
Namrita Lall
Department of Plant Science
University of Pretoria
Private Bag X20
Hatfield 0028
South Africa
EMAIL: namrita.lall@up.ac.za
Received: 16 Sep. 2013
Revised: 03 Jan. 2014
Accepted: 23 Mar. 2014














