Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.110 n.11-12 Pretoria Nov./Dec. 2014
http://dx.doi.org/10.1590/sajs.2014/20130342
RESEARCH ARTICLE
http://dx.doi.org/10.1590/sajs.2014/20130342
Early planting and hand sorting effectively controls seed-borne fungi in farm-retained bean seed
Ernest DubeI; Julia SibiyaII; Morris FanadzoIII
IAgricultural Research Council -Small Grain Institute, Bethlehem, South Africa
IIAfrican Centre for Crop Improvement, University of KwaZulu-Natal, Pietermaritzburg, South Africa
IIIDepartment of Agriculture, Cape Peninsula University of Technology, Wellington, South Africa
ABSTRACT
Home-saved bean (Phaseolus vulgaris L.) seed can be hand-sorted to remove discoloured seed, thereby reducing the level of contamination by certain seed-borne fungi and improving seed germination. In this study, the effect of planting date on the infection and discolouration of bean seed by seed-borne fungi was investigated in order to improve the quality of hand-sorted, farm-retained bean seeds used by resource poor smallholder farmers. The germination quality and level of seed-borne fungi in hand-sorted first-generation bean seed harvested from an early-, mid- and late-summer season planted crop was therefore assessed. The highest percentage of discoloured seed (68%) was obtained from the mid-summer season planting. Non-discoloured seed from early- and late-season plantings had significantly (o<0.001) higher normal germination (82% and 77%, respectively) than that from the mid-season planting date (58%). Irrespective of planting date, unsorted seed and discoloured seed had higher levels of infection by Fusarium spp. and Phaeoisariopsis spp. than the non-discoloured seed. Removal of discoloured seed by hand sorting eliminated Rhizoctonia spp. from all seed lots. Farmers can eliminate this pathogen by simply removing discoloured seed. Non-discoloured seed from the early-planted crop had the lowest level of infection by Fusarium spp. and Phaeoisariopsis spp. The results indicate that planting date is an important consideration in improving the quality of hand-sorted farm-retained bean seed.
Keywords: common bean; planting date; seed-borne fungi; seed quality; visual sorting
Introduction
Common bean (Phaseolus vulgaris L.) is the most important grain legume for direct human consumption and it provides a cheap source of dietary proteins for poor people in several countries.12 It is commonly consumed for its delicacy, high protein content and as a source of certain antioxidants, minerals and polyphenols.3 In addition to the superior quality of the protein, common bean is an excellent source of starch, dietary fibre, vitamins and minerals.4 The superior nutritional attributes of common bean make it a potential crop for improving the nutritional security of resource poor communities. However, insect pests and diseases, especially those caused by fungal pathogens, constitute the major constraint to bean production.56 Diseases may cause 80-100% yield loss for common beans on resource poor farms.7 Of all transmittable seed-borne diseases of beans, fungi cause the most damage.8 This damage includes shrunken seeds, seed rot, seed discolouration and, above all, diseases in emerging seedlings that may kill a certain proportion of the seedlings.89 These effects on seedlings lead to poor stands and reduced yield.
The use of certified bean seed minimises yield reduction from fungal seed-borne diseases. In order to reduce production costs, smallholder farmers prefer to retain their own bean seed,1011 which inevitably harbours more infected seed than certified treated seed. Seed-borne diseases are always more prevalent in farm-retained bean seed than in certified and treated seed.11,12
Seed-borne fungi may survive for 5 years in seeds that are air dried and stored at 4 0C.13 Several fungicides may be used to treat seed; however, these chemicals are expensive and they may pose a health hazard to smallholder farmers who lack technical expertise on their use. Chemical treatment of seed gives variable results because deep-seated infections may survive treatment. Avoidance of conditions favourable for seed infection during the growing period and prevention of the dissemination of the pathogen spores may be used as a management strategy for minimising seed-borne diseases. Consequently, it has often been recommended that farmers should select suitable planting dates and appropriate field sites in order to avoid infection.14
There are three types of seed discolouration caused by seed-borne fungi: (1) superficial necrotic lesions, (2) fungal coatings and (3) pigmentation.8 These characteristic symptoms are the basis of visual sorting of bean seed to reduce or exclude seed-borne disease from the seed.91516 The formal seed sector has invested large amounts of money into seed cleaning machinery and sophisticated electronic colour sorters to remove disease-stained seeds. Although these machines are efficient, they may not give a better result than hand sorting. However, nondiscoloured seeds may also harbour latent infections. The extent of seed discolouration caused by seed-borne fungi depends on the seed micro-flora, environmental conditions, host cultivar, physiology and genetics.17
The identification of seed-borne pathogens of importance to the quality of common bean seeds is mostly based on practical experience of crop damage. Some important seed-borne fungal pathogens that have been shown to cause significant bean yield reductions in smallholder farmer fields in the tropics include: Colletotrichum lindemuthianum (Saccardo and Magnus) Scribner, Phaeoisariopsis griseola (Sacc.) Ferr., Fusarium solani (Mart.) Sacc., Fusarium oxysporum (Schltdl.), Macrophomina phaseolina (Tassi) Goid. (charcoal rot fungus) and Rhizoctonia solani (A.B. Frank) Donk.11,14Phaeoisariopsis griseola (Sacc.) Ferr. is the causal agent of angular leaf spot on beans. It is also a necrotic fungus which causes reddish spots with typical angular shapes on the bean seed coat. These spots have been reported to be central to the hilum.18Colletotrichum lindemuthianum is the causal agent of bean anthracnose and infected seeds often have oily brown droplets that coalesce to cover the whole seed as the fungi grow old. Rhizoctonia solani, F. solani and F oxysporum are responsible for damping off, collar rots and wilt diseases in bean seedlings.
It was hypothesised in this study that planting time has an effect on the infection and extent of discolouration of common beans caused by these problematic seed-borne fungi. Planting time would thus affect the quality of hand-sorted farm-retained bean seed that is used by resource poor smallholder farmers. The objectives of the study were to determine the extent of seed-borne fungal infections and the germination quality of non-discoloured and discoloured farm retained seed harvested from crops planted in the early-, mid- and late-summer season.
Materials and methods
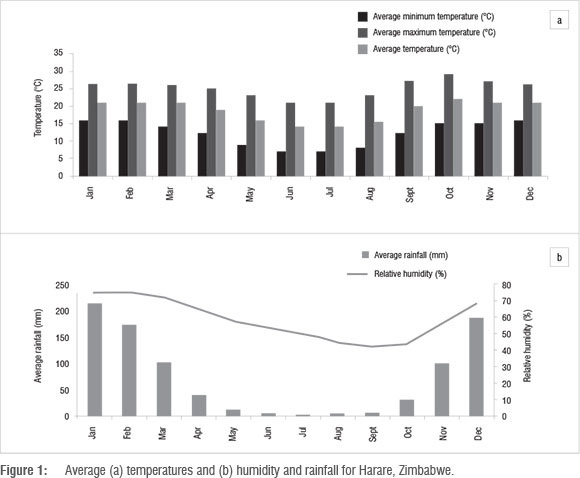
Field experiments were replicated on two sites in Harare, Zimbabwe. The sites were at the University of Zimbabwe Crop Science fields (170 51' S and 31010' E) and on the University of Zimbabwe farm (170 48' S and 310 00' E). The sites are approximately 20 km apart and have similar soil and climatic conditions. The Harare region has moderate annual rainfall averaging 750-1000 mm and mean annual temperatures of 21-27 0C. The average temperature, rainfall and humidity are presented in Figure 1. These conditions are conducive for high disease prevalence during summer. The soils are deep and red, belonging to the fersiallitic group.
Certified common bean seed (cultivar PAN 116, Pannar Seed [Pvt] Ltd, Zimbabwe) was planted. The cultivar has a determinate, bushy type growth habit and is commonly referred to as red speckled or 'sugar beans'. The three planting dates used were early summer (planting date: 01 October), mid-summer (planting date: 01 December) and late summer (planting date: 01 February). The experimental design of the field trials was a randomised complete block design with three replicates. Seeds were planted at a seed density of 30 seeds/m2 and a depth of approximately 30 mm using hand hoes. Seedlings were later thinned to 22 plants/m2 to give a population of 220 000 plants/ha. Plots were 6 m long and had 10 rows that were spaced 300 mm apart. To prevent spore movement from one plot to another, each bean plot was surrounded by one row of maize (Zea mays L.). Compound D fertiliser (8% N, 14% P 7% K) was applied as basal dressing to all plots at a rate of 300 kg/ha. Bait pesticide composed of a mixture of 100 g carbaryl 85 WP and 20 kg maize meal was applied per row to control seedling pests. Dimethoate 40 EC (1 mL/L water) was applied at 3 and 10 weeks after crop emergence (WACE) to control leaf and pod insects. Weeds were controlled through hand hoeing at 2 and 5 WACE. At physiological maturity, plants from the net plot (three central rows, 4 m long) of each plot were hand harvested. The plants were air dried, and the beans in their pods were stored in a cold room at 4 0C.
The major objective of the seed tests was to determine the seed-borne fungal infection level and germination quality of non-discoloured and discoloured bean seed harvested from the three different planting dates. Therefore, bean seed for similar treatments from the two sites was mixed in a 1:1 ratio before seed tests were carried out. The seed tests were conducted in a seed pathology laboratory, 2 months after harvesting seed from the previous planting date. Preliminary work was done to grade the seed from each planting date into three seed categories: non-discoloured (no visible blemishes, lacerations or necrotic spots on seed coat), discoloured and unsorted seeds. This grading was done whilst shelling the bean pods in order to prevent surface contamination from other seeds. An experiment was then carried out to determine the effect of planting date on the germination quality and seed-borne fungal infection levels of the non-discoloured seed, the discoloured seed and the unsorted seed. Three planting dates were tested - early summer (October planting), mid-summer (December planting) and late summer (February planting). The design of the experiment was a split-plot, with planting date as the main plot factor and seed category as the sub-plot factor to give nine treatment combinations.
The experiment comprised a germination test and a blotter test for fungal pathogens. Each treatment was replicated four times and the replicates were randomly assigned to the different blocks according to incubator shelf level. For each replicate, 50 bean seeds were used, to give a total of 200 seeds per treatment and to meet the requirement of the International Seed Testing Association (ISTA).19 The 50 seeds of each replicate were plated between square wet blotter sheets 500 mm x 500 mm in size and placed according to their respective blocks in an incubator room under full light and temperature of 24-28 0C. The germination test was evaluated 9 days after incubation. Germination results were recorded according to the rules and regulations of ISTA.19 The test results were therefore categorised as percentage normal, abnormal and nongerminated seeds.
The blotter technique was used to isolate the following seed-borne pathogens in all nine treatments: Colletotrichum spp., Phaeoisariopsis spp., Fusarium spp., Macrophomina spp. and Rhizoctonia spp. Each treatment had 200 seeds19 and was represented by replicates of 50 seeds each. From the working samples, 10 seeds were counted randomly and plated in Petri dishes equidistantly as rings. The Petri dishes were lined with three layers of filter paper that was soaked in distilled water. Five such Petri dishes were prepared to represent a replicate of each treatment. The Petri dishes were incubated for 7 days at 22 0C, and subjected to alternating 12-h darkness and 12-h near ultraviolet light. Filter papers in the Petri dishes were rehydrated after 4 days of incubation. At the end of the incubation period, each seed was examined thoroughly under different magnifications of a stereomicroscope (model-S209, Motic Deutschland GmbH, Wetzlar, Germany) for growth of the fungi. Slides were prepared to observe any fruiting structures under higher magnifications (x 40 - Hund H 500 series, Hund Wetzlar, North Rhine-Westphalia, Germany) as a way of confirming the identifications. The fungal species were identified according to various descriptions.1820-22 Whenever identifiable growth of a fungus was seen on a seed, the respective seed was considered infected even if only one fructification was observed. Disease incidence was quantified by calculating the percentage of seeds infected by a particular pathogen.
Analysis of variance (ANOVA) was performed on germination and infection percentage data with significance at p< 0.05 using 5% least significant difference. The ANOVA was carried out using GenStat Release 12.1 statistical software.23 Data were tested for conformity with the assumptions underlying ANOVA, including homogeneity of variance tests, before being subjected to ANOVA. Seed quality improvement was calculated as the difference in normal germination or seed-borne fungi between non-discoloured and unsorted seed, expressed as a percentage of the value obtained for unsorted seed.
Results and discussion
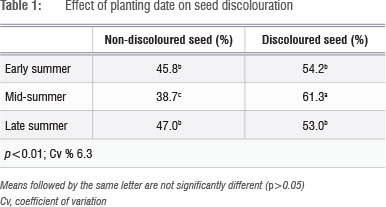
Early and late planting produced the most non-discoloured seed (Table 1). The highest percentage of discoloured seed was obtained from the mid-summer season planting (Table 1).
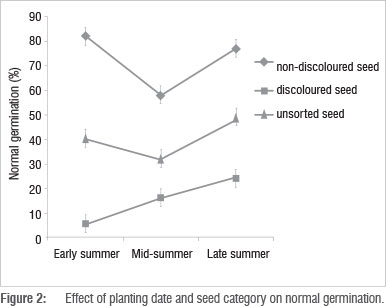
The interaction between planting date and seed category was significant (p<0.001). Non-discoloured seed from the early- and late-season planting dates had a significantly higher germination percentage than that from the mid-season planting date (Figure 2). For unsorted seed, the lowest normal germination was obtained from the mid-season planting date. However, the difference in germination between non-discoloured and unsorted seed was highest in the early-season planted seed. This finding implies that seed sorting based on discolourations for improving germination quality is more effective on seed from the early-season planting date than other planting dates. By proper choice of planting date and hand sorting alone, farmers may obtain bean seed which is nearly equal to certified seed in germination quality (80% or higher).
The planting date × seed category interaction was significant for Colletotrichum spp., Fusarium spp., Macrophomina spp. and Rhizoctonia spp. (Table 2). Non-discoloured seed from all the planting dates had equally low levels of infection by Colletotrichum spp. (Figure 3a). However, unsorted seed from the early- and mid-season planting dates had higher levels of infection than that from the late-season planting date. For Fusarium spp., the interaction between planting date and seed category showed that all planting dates had equal levels of infection in unsorted seed, but for non-discoloured seed, the early-summer planting date had the lowest level of infection (Figure 3b). This result implies that the visible symptoms of this pathogen (based on discolouration) were expressed the most in early-planted seed.
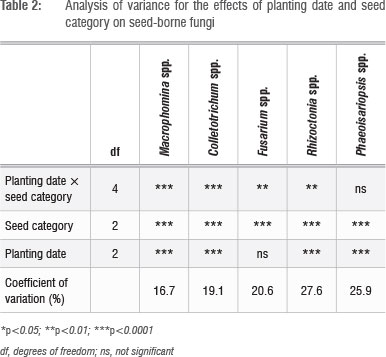
For Macrophomina spp., non-discoloured seed from the early- and midsummer planting date had a lower level of infection than the late season planting (Figure 3c). Rhizoctonia spp. were absent from non-discoloured seed of all planting dates (Figure 3d). However, the highest infection levels for this pathogen in unsorted and discoloured seed were obtained in seed from the early-planted crop (Figure 3d).
The interaction between planting date and seed category was not significantly different for Phaeoisariopsis spp. Seed category (subplot) and planting date (main plot) effects were, however, significant (Table 3). These results suggest that unsorted seed was more infected by Phaeoisariopsis spp. The early-season planted seed was significantly less infected by this pathogen when compared with seed from the mid-and late-season planting dates (Table 3).
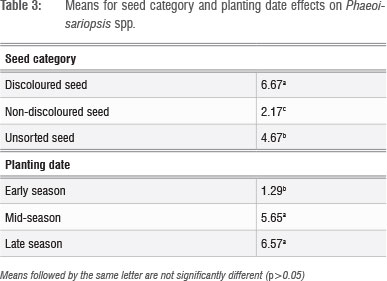
The greatest improvement in the quality of bean seed from different planting dates through hand sorting based on seed discolouration was obtained from early-planted seed (Table 4). As shown in Figure 1, in the early-summer season (October), less rainfall was received compared to mid and late summer (December to February). In addition, the highest temperature and lowest relative humidity were also observed in early summer. Most fungal pathogens are known to prefer wet and warmer conditions for infection and growth.14 It is therefore expected that the early season planting date would be the least conducive for proliferation of the fungal pathogens during the crop's early growth. It also appears that Rhizoctonia spp. can be eliminated from seed lots by hand sorting based on discolourations (Table 4). It is therefore recommended that farmers can eliminate this pathogen simply by removing discoloured seed from the seed lot. Rhizoctonia spp. are necrotic fungi that rapidly kill host cells, living saprophytically on the dead tissues.13 Severe infection of the bean seed by necrotrophic fungi can result in extensive seed discolouration.
Results also indicated that Phaeoisariopsis spp. could be reduced from early-planted seed based on discolourations (Table 4). Reduction of this pathogen was 96% (Table 4). The reduction of infection levels by hand sorting needs to be confirmed with field trials. There is currently no information on thresholds or economic injury level for these seed-borne fungal pathogens in bean seed.
The Spearman's rank correlation matrix (Table 5) showed that Colletotrichum spp. were the chief pathogen affecting normal germination of unsorted seed (rs = -0.856). Colletotrichum lindemuthianum is the causal agent of b ean anthracnose, a disease that causes serious crop loss in developing countries.24 It is one of the predominant seed-borne pathogens recorded in smallholder farming areas of Zimbabwe.12 Bioassays showed that 55% of the farm-retained seed was infected by the pathogen and, in severe cases, it caused up to 100% yield loss.
Fusarium spp. were another dominant pathogen affecting normal germination of non-discoloured seed (rs = -0.829) (Table 5). This may be explained by the fact that Fusarium is a biotrophic fungus.25 Deep penetration by biotrophic fungi results in hyphae being located in tissues of the embryo and cotyledons, and, in this case, the fungal pathogens may present no visible macroscopic symptoms. Biotrophic seed-borne fungi can also be found as intercellular mycelia and beneath the epidermal cell layers inside the bean seed coat, and in this case, they still present no visible symptoms. This implies that some infested seed may not display macroscopic symptoms, thereby making visual inspection and hand sorting of seeds less effective.9 An additional test would, therefore, be required in such cases to check for the presence of seed-borne infections, especially in seed production. It can also be noted that the red speckled bean seed discolourations can occur as a result of other pathogens such as bacteria and viruses. Further research is needed to investigate the effectiveness of visual inspection of seed from different sources on reduction of these other pathogens. DNA sequencing could be useful as a rapid data collection tool for these studies.
Conclusions and recommendations
Planting date affects the extent of bean seed discolouration by seed-borne fungal pathogens such as Colletotrichum spp., Macrophomina spp., Phaeoisariopsis spp. and Fusarium spp. It appears that the visual symptoms for these pathogens are generally better expressed in early-planted seed, resulting in better seed quality from this planting date after visual inspection and hand sorting. Rhizoctonia spp., however, can be totally eliminated based on seed discolouration, regardless of planting date.
Acknowledgements
We thank the University of Zimbabwe Crop Science Department technical staff for assisting in the management of field trials. The Rockefeller Foundation is also acknowledged for funding the 'bean diseases project', under which this study was conducted.
Authors' contributions
E.D. performed the experiments, and collected and analysed the data. J.S. was the project leader and made conceptual contributions. M.F. assisted in the data analyses and interpretation of the results. All authors contributed equally to the writing of the manuscript.
References
1. Piergiovanni, AR, Taranto G, Losavio FP Pignone D. Common bean (Phaseolus vulgaris L.) landraces from Abruzzo and Lazio regions (Central Italy). Genet Resour Crop Evol. 2006;53:313-322. http://dx.doi.org/10.1007/s10722-004-6144-7 [ Links ]
2. Jackson J, Kinabo J, Mamiro P Mamiro D, Jideani V. Utilisation of dry beans and pulses in Africa. In: Siddiq M, Uebersax MA, editors. Dry beans and pulses production, processing and nutrition. Oxford: Blackwell; 2012. p. 261-282. http://dx.doi.org/10.1002/9781118448298.ch11 [ Links ]
3. Boateng J, Verghese M, Walker LT, Ogutu S. Effect of processing on antioxidant contents in selected dry beans (Phaseolus spp. L.). LWT-Food Science and Technology. 2008;41 (9):1541 -1547. http://dx.doi.org/10.1016/jJwt.2007.11.025 [ Links ]
4. Audu SS, Aremu MO. Effect of processing on chemical composition of red kidney bean (Phaseolus vulgaris L.) flour. Pak J Nutr. 2011;110(11):1069-1075. Available from: http://docsdrive.com/pdfs/ansinet/pjn/2011/1069-1075.pdf [ Links ]
5. Schwartz HF, Pastor-Corrales MA. Bean production problems in the tropics. 2nd edn. Cali, Colombia: Centro Internacional de Agricultura Tropical (CIAT); 1989. [ Links ]
6. Wortmann CS, Kirkby RA, Eledu CA, Allen DJ. Atlas of common bean (Phaseolus vulgaris L.) production in Africa. Cali, Colombia: Centro Internacional de Agricultura Tropical (CIAT); 1998. [ Links ]
7. Beebe S, Rao IM, Mukankusi C, Buruchara R. Improving resource use efficiency and reducing risk of common bean production in Africa, Latin America and the Caribbean. In: Hershey C, editor. Issues in tropical agriculture. I. Eco-efficiency: From vision to reality. Cali, Colombia: Centro Internacional de Agricultura Tropical (CIAT); 2012. p. 117-134. [ Links ]
8. Neergard P Seed pathology. Vol. 1-2. London: Macmillan; 1977. [ Links ]
9. Walcott RR. Detection of seed borne pathogens. HortTechnology. 2003;13:40-47. Available from: http://horttech.ashspublications.org/content/13/1/40.full.pdf [ Links ]
10. Chiduza C. Farm data handbook. Harare, Zimbabwe: FAO Agricultural Services Division; 1994. [ Links ]
11. Icishahayo D, Sibiya J, Dimbi S, Madakadze IC, Manyangarirwa W, Chipindu B. Assessment of quality and health of field bean seeds home-saved by smallholder farmers. African Crop Science Conference Proceedings. 2009;9:609-615. Available from: http://www.cabi.org/cabdirect/FullTextPDF/2013/20133232505.pdf [ Links ]
12. Kutywayo V. Identification and survival of bean (Phaseolus vulgaris L.) pathogens and the effects of fungicide seed dressing and cultural practices on the incidence of the diseases they cause under smallholder conditions in Zimbabwe [MSc thesis]. Harare: University of Zimbabwe; 2000. [ Links ]
13. Maude RB. Seed-borne diseases and their control: Principles and practice. Wallingford, UK: CAB International; 1997. [ Links ]
14. Agrios GN. Plant pathology. 5th ed. Burlington, MA: Elsevier Academic Press; 2005. [ Links ]
15. Paulsen MR. Using machine vision to inspect oil seeds. INFORM. 1990;1:50-55. [ Links ]
16. Walcott RR, McGee DC, Misra MK. Detection of asymptomatic fungal infections of soya bean seeds by ultrasound analysis. Plant Dis. 1998;82:584-589. http://dx.doi.org/10.1094/PDIS.1998.82.5.584 [ Links ]
17. Shetty SA. Biology and control of some seed-borne pathogens in rice [PhD thesis]. Karnataka, India: University of Mysore; 1986. [ Links ]
18. Mathur SB, Kongsdal O. Common laboratory seed health testing methods for detecting field fungi. Copenhagen: Danish Government Institute for Seed Pathology for Developing Countries; 2001. [ Links ]
19. ISTA. International rules for seed testing. Seed Sci Technol. 1996;24:1-335. [ Links ]
20. Commonwealth Mycological Institute (CMI). CMI descriptions of pathogenic fungi and bacteria: No. 275, 316, 462 and 847. Kew: CMI;1986. [ Links ]
21. Kulshrestha DD, Mathur DB, Neegard P Identification of seed-borne species of Colletotricum spp. Copenhagen: Danish Government Institute of Seed Pathology for Developing Countries; 1976. [ Links ]
22. Nath R, Neergard P Mathur SB. Identification of Fusarium species on seeds as they occur in blotter test. Proceedings of the International Seed Testing Association. 1970;35:121-144. [ Links ]
23. Lawes Agricultural Trust. GenStat release 12.1. Harpenden, Hertfordshire, UK: Rothamsted Experimental Station; 2009. [ Links ]
24. Wortman CS, Allen DJ. African bean production environments: Their definition, characteristics and constraints. Network on bean research in Africa. Occasional Paper Series 11. Dar es Salaam, Tanzania: International Centre of Tropical Agriculture; 1994. [ Links ]
25. Nelson PE, Dignani MC, Anaisse EJ. Taxonomy, biology and clinical aspects of Fusarium species. Clin Microbiol Rev. 1994;7:479-504. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC358338/pdf/cmr00033-0067.pdf [ Links ]
 Correspondence:
Correspondence:
Morris Fanadzo
Department of Agriculture
Cape Peninsula University of Technology
Private Bag X8
Wellington 7654
South Africa
EMAIL: fanadzom@cput.ac.za
Received: 04 Nov. 2013
Revised: 07 Feb. 2014
Accepted: 26 Mar. 2014














