Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.110 n.9-10 Pretoria Oct. 2014
http://dx.doi.org/10.1590/sajs.2014/20140011
RESEARCH ARTICLE
Tufa stromatolite ecosystems on the South African south coast
Renzo PerissinottoI; Tommy G. BornmanII, III; Paul-Pierre SteynIII; Nelson A.F. MirandaI; Rosemary A. DorringtonIV; Gwynneth F. MatcherIV; Nadine StrydomV; Nasreen PeerI
IDST/NRF Research Chair in Shallow Water Ecosystems, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa
IISouth African Environmental Observation Network, Elwandle Node, Grahamstown, South Africa
IIICoastal & Marine Research Unit, Department of Botany, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa
IVDST/NRF Research Chair in Marine Natural Products, Department of Biochemistry, Microbiology and Biotechnology, Rhodes University, Grahamstown, South Africa
VDepartment of Zoology, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa
ABSTRACT
Following the first description of living marine stromatolites along the South African east coast, new investigations along the south coast have revealed the occurrence of extensive fields of actively calcifying stromatolites. These stromatolites have been recorded at regular distances along a 200-km stretch of coastline, from Cape Recife in the east to the Storms River mouth in the west, with the highest density found between Schoenmakerskop and the Maitland River mouth. All active stromatolites are associated with freshwater seepage streams flowing from the dune cordon, which form rimstone dams and other accretions capable of retaining water in the supratidal platform. Resulting pools can reach a maximum depth of about 1 m and constitute a unique ecosystem in which freshwater and marine organisms alternate their dominance in response to vertical mixing and the balance between freshwater versus marine inflow. Although the factors controlling stromatolite growth are yet to be determined, nitrogen appears to be supplied mainly via the dune seeps. The epibenthic algal community within stromatolite pools is generally co-dominated by cyanobacteria and chlorophytes, with minimal diatom contribution.
Keywords: tufa stromatolites; microbialites; South Africa; barrage pools; waterfall deposits.
Introduction
The first extant marine tufa stromatolites along the southern African coast were described in the early 2000s from Cape Morgan12 and later investigated in some detail from a geochemical and geomorphological point of view3. Located on a dolerite headland shaped into a wave-cut platform, these stromatolites consist of continuous, extensive laminar growths or discrete accretions bridging gaps between separate boulders. In either case, the formation results in enclosed rock pools capable of trapping carbonate-rich groundwater seeps. Although other rare, isolated examples of similar formations have been reported to occur from Port Elizabeth to Tofu in Mozambique,3 the recent discovery of numerous and closely spaced living stromatolites on the coastline south of Port Elizabeth appears to be extraordinary. While the Cape Morgan headland includes about 50 stromatolite colonies, each of about 3 m2 on average,3 the formations mapped thus far to the west of Cape Recife include 540 colonies, ranging in cover from <1 m2 to >10 m2.
Stromatolites are important because they are regarded as the oldest type of calcified formations in which cyanobacteria play a major role in the deposition of calcite crystals, either directly on the cell surface or, more commonly, through inclusion within the mucilaginous sheath that surrounds the cell.4,5 The process requires CaCO3 supersaturation of the water, which only occurs in today's marine environment in a few special circumstances, e.g. under states of hypersalinity, excess evaporation or mixing of extremely different water types.3,6,7 As stromatolites date back in the fossil record at least 2.7-3.5 billion years, the study of the few extant colonies still remaining in the marine environment may be instrumental in understanding the hydrospheric conditions that prevailed at the onset of life on earth.
Southern African tufa stromatolites are regarded as unique in their nature, because they typically occur at the interface between freshwater seepage points and the marine penetration.3 The closest type to those discovered on the southeast coast of South Africa are the tufa deposits recently reported from the southwest coast of Western Australia8 and from the Giant's Causeway of Northern Ireland9. In Australia, most tufa formations are associated with inland spring discharges, whereas the South African types are all upper intertidal to supratidal in position, with strong and regular marine influence. By comparison, the Northern Ireland formations have a very limited thickness and are seldom inundated by marine waters.9
Here we report a preliminary account of the nature and extent of the newly discovered tufa stromatolites on the Eastern Cape south coast, including basic observations on their location, extent, structure and ecological functioning. More comprehensive investigations will be undertaken during the next few years within a project recently initiated at the Nelson Mandela Metropolitan University, in collaboration with the South African Environmental Observation Network and Rhodes University.
Materials and methods
Three different tufa pools of the 'barrage' type were selected for the preliminary study. These pools were located at representative sites along the coastline, from south of Cape Recife (Pool A, 34°02'42.13''S, 25°34'07.50"E) to Schoenmakerskop (Pool B, 34°02'28.23''S, 25°32'18.60''E) and on the eastern outskirts of Seaview (Pool C, 34°01'03.16"S, 25°21'56.48"E) (Figure 1). Pool A was located far from any residential disturbance, while Pool B was just below the village of Schoenmakerskop and likely affected by anthropogenic effluents. Pool C was situated just below the eastern residential extension of the village of Seaview and was, therefore, potentially most affected by leaching of anthropogenic effluents.
Continuous recording of temperature and salinity at the three selected pools was initiated in April 2013, using Star Oddi DST CT (http://www.star-oddi.com/) probes deployed in crevices at the bottom of each pool. Data were downloaded after 6 months. A full survey of the three pools was undertaken during 13-15 October 2013, when a range of physicochemical parameters was measured using a YSI 6600-V2 multiprobe system (YSI Incorporated, Yellow Springs, OH, USA). These parameters include depth, temperature, salinity, conductivity, pH, dissolved oxygen and turbidity. Samples for nutrients and chlorophyll-a analyses were also collected from the water column, along with epibenthic macrofauna, macrophytes, algae and cores for microphytobenthic chlorophyll-a analysis.
Percentage composition of algal classes was estimated both in the water column and at the sediment surface using a bbe Moldaenke FluoroProbe (bbe Moldaenke, Schwentinental, Germany) fitted with a dual bentho-pelagic switch system. FluoroProbe is an in-situ fluorometer that uses the spectral fluorescence approach to discriminate among major algal classes on the basis of selective excitation of accessory pigments (which differ between the major taxonomic groups of algae).10,11 Sediment cores to a standard depth of 50 mm were collected using a stainless-steel corer (17 mm internal diameter) pushed through the tufa deposits with a hammer. Cores were fractionated into 5 x 10 mm units and again extracted in 30 mL of 90% acetone solution for 48 h. Fluorescence readings were taken using a Turner 10-AU (Turner Designs, Sunnyvale, CA, USA) narrow-band system. Dissolved inorganic nitrogen and phosphorus were measured by determining the concentration of ammonium, nitrate and nitrite, and orthophosphate, respectively, using standard colorimetric methods.12 Qualitative samples of benthic macrofauna, fish, micro- and macroalgae as well as macrophytes were collected at each pool, to identify key constituents of barrage pool communities.
For microbial diversity studies, approximately 0.8 g of stromatolite material from an upper and lower elevation of Pool B (Figure 1) was crushed using a sterile pestle and mortar in liquid nitrogen. Genomic DNA was extracted from crushed material using the PowerSoil DNA Extraction kit (MoBio Laboratories Inc., Carlsbad, CA, USA) and variable regions 4 and 5 of the bacterial 16S rRNA gene were amplified through a polymerase chain reaction (PCR) using fusion primers including a multiplex identifier tag as well as a nucleotide sequence required for the sequencing reaction. PCR products were analysed by 454-pyrosequencing (454 sequencing platform, Roche Products, Johannesburg, South Africa). Primer tags, sequence reads shorter than 200 bp, reads containing ambiguous nucleotides as well as reads with homopolymeric runs longer than seven nucleotides, were culled from the data set using Mothur13. The remaining 5733 sequences were classified using the Naïve Bayesian rRNA classifier algorithm14,15 against the Ribosomal Database Project (http://rdp.cme.msu.edu/) and visualised in Xcel or MEGAN v416. Sequence data were submitted to the Sequence Read Archives hosted at the National Center for Biotechnology Information, with accession number SRX478035.
Results and discussion
Range and extent of tufa stromatolites
Reconnaissance surveys have established that the newly discovered stromatolite fields of the southern Cape coast are spread across approximately 200 km of coastline from east of the Storms River mouth (34T12.14"S, 23°54'10.66''E) to west of Cape Recife (25°42'6.40''S, 34°1 '45.15"E) (Figure 1). Thus they are among the most extensive tufa deposits currently occurring along any coastline (for comparison, Western Australia's deposits extend to 90 km8 and those in Northern Ireland to 25 km9). A preliminary survey of a 10-km section of the coastline between Cape Recife and Maitlands revealed 341 freshwater seeps (a density of one in every 25 m), 90% of which exhibited stromatolite deposits. Of the 346 freshwater seeps (a density of one in every 41 m) in a 14-km section between Cape St Francis and Oyster Bay, about 58% formed tufa deposits of some kind. All active deposits are supratidal to upper intertidal in position and receive regular inflow of seawater, either as wave overtopping at high spring tide or wave splash during storm surges.
Structure of stromatolite formations
Both 'barrage pools' with downstream rimstone dams (Figure 2a) and 'waterfall deposits' (sensu Forbes et al.8) (Figure 2b) occur at regular intervals along the coastline, with the former type generally dominating both in terms of frequency and areal extent. Additionally, 'shell conglomerates' form under higher flow conditions, when fast-flowing streams with a high CaCO3 content end up directly onto a beach made up of shell grit, pebbles and sand, with calcification eventually cementing everything together. Barrage pools seem restricted to flat coastline areas, with very few developing along steep areas or where a longer distance between seep and the shore is involved. Conversely, waterfall deposits are most abundant on steep slopes, where freshwater seeps among rocks, from waterfalls or the occasional pump house (Figure 2b).
All three mesofabric structures recognised earlier in the Cape Morgan fields23 can be observed in the south coast formations. These structures include 'colloform growths', most often found underwater in the deeper parts of the barrage pools. They exhibit a typical multiconvex lamination with irregular margins probably imparted by bioerosion.17 'Laminar/ columnar deposits' occur mainly at pool margins, as boundstone connecting bedrock outcrops to form rimstone dams with a thickness not unusually in excess of 1 m (Figure 2a). 'Pustular formations' are partially emergent and occur in shallow water around pool rims or as waterfall deposits (Figure 2b). Their morphology is irregular with disrupted lamination probably imparted by wave or current action. In all formations, the typical laminar structure observed previously in the Morgan Bay stromatolites is also found, with a micritic network composed mainly of radial laminae separated by thinner, longitudinal climax laminae.23
Stromatolite ecosystem dynamics
The barrage pools are regularly inundated with seawater (Figure 3), with frequency depending on the elevation of the formation. An increase in marine overtopping events was evident in winter, coinciding with frontal storm events. The variability in salinity is further increased by the volume of freshwater input. Pool A, although subjected to almost daily marine overtopping events in winter, also receives a high flow of fresh water that quickly replaces the seawater. The less frequent marine overtopping events in Pool B have a longer residence time as a result of lower freshwater inflow, thereby creating more stable conditions for biota (Figure 3).
Haloclines and stratification develop in calm conditions between the upper freshwater lid and the heavy saline water at the bottom (Table 1). From upstream seepage pools through to the barrage pools and the downstream pools a progressive trend of pH increase, from about 7.2 to about 9.1, can be observed, indicating CO2 degassing and CaCO3 precipitation.3 Inside barrage pools, diurnal oxygen levels can reach extremes in excess of 345% or 27.87 mg/L supersaturation at the interface between sediment and water, as a result of photosynthesis by the benthic algal community (Table 1).
In the three pools sampled during the study, dissolved inorganic nitrogen was dominated overwhelmingly by the nitrate + nitrite fraction, accounting for 99% of the total nitrogen. A comparison of nutrient contents in barrage pools versus upstream seepage pools and downstream marine adjacent pools, shows that there was a high dissolved inorganic nitrogen content in the fresher waters, while no differences were observed in the distribution of dissolved inorganic phosphorus. There was, however, a gradient of increasing concentration from Pool A (Cape Recife) to Pool C (Seaview) (Figure 4), which appears to be correlated with the intensity of residential settlements in proximity of the pools. The nitrogen is in the most oxidised form and highest concentration in the upper pools, hence the nutrient source fuelling stromatolite productivity appears to be in the seepage water.
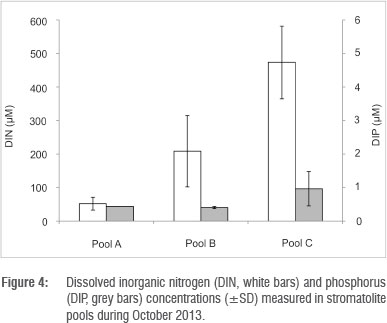
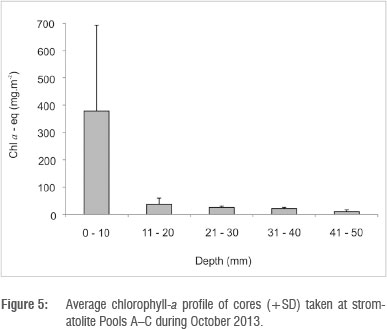
Total core (50 mm thick) benthic microalgal biomass in stromatolite tufa formations was in the range of 197-853 mg (chla-eq)/m2. Of this biomass, 80.5% was found in the upper 10 mm, decreasing sharply with depth to 7.6% at 20 mm, 5.5% at 30 mm, 4.4% at 40 mm and 2.2% at 50 mm (Figure 5). In terms of algal classes, in barrage pools benthic algae were dominated by chlorophytes with 51-55% of total epilithon, followed by cyanobacteria with 43-48% and diatoms with only 0-2.7% (Table 2). Chlorophyte activity appeared to peak immediately after marine overtopping events, subsiding during periods of no marine penetration. In the water column, phytoplankton classes were dominated by chlorophytes with 2-49% of the total, followed by cyanobacteria with 6-20%. 'Yellow substances', presumably from leaching dune vegetation, accounted for the bulk of the fluorescent signal (Table 2).
The bacterial assemblage associated with the upper and lower stromatolite formations sampled from Pool B was dominated by Cyanobacteria, Bacteroidetes and Proteobacteria (Figure 6a). Verrucomicrobia, Planctomycetes and Chloroflexi also occurred in significant numbers but were not as numerically dominant. The bacterial diversity profiles of stromatolites from Western Australia and the Bahamas, whilst containing large numbers of Bacteroidetes and Cyanobacteria, are dominated by Proteobacteria.18-21 By contrast, stromatolites from Spain contained higher abundances of Cyanobacteria followed by Bacteroidetes and Proteobacteria.22 Closer examination of the dominant cyanobacterial operational taxonomic units (OTUs) in the stromatolites from Pool B shows a single OTU, classified as Lyngbya, representing almost 60% of the cyanobacterial reads within the upper pool stromatolite (Figure 6b). By contrast, the cyanobacterial OTUs within the stromatolite from the lower pool are more evenly distributed with dominant genera of Lyngbya, Plectonema, and Leptolyngbya. With respect to the Proteobacteria found within the stromatolites in this study, the bulk of the reads was assigned to the Alphaproteobacteria (Figure 6b). Of interest is the occurrence of photosynthetic genera in the lower pool stromatolites (e.g. Rhodobacter and Erythrobacter), as well as the distinctive difference in distribution patterns of the proteobacterial genera between the two stromatolite samples. Stromatolite formation is hypothesised to be cyclical in nature, consisting of the formation of microbial mats dominated by Cyanobacteria, which leads to the rapid production of exopolymeric substances which bind sediment granules. This formation is then followed by a hiatus characterised by a decrease in sedimentation and an increase in mineral precipitation, during which heterotrophic bacteria increase in abundance.23-25 Both samples examined in this study had high abundances of Cyanobacteria and heterotrophic Bacteroidetes and Proteobacteria (Figure 6a). The lower pool sample exhibited higher relative abundances of Cyanobacteria and phototrophic purple bacteria, suggesting that it is still within the active sedimentation phase, whilst the increased heterotrophic component in the upper pool indicates a possible transition from sedimentation to hiatus phase.
Plants and macroalgae from the tufa stromatolite environments were sampled in the period between overtopping events, during which the systems were highly stratified. The resulting salinity gradient allowed for the occurrence of eight different macroalgal species, ranging from marine to typically freshwater species within the span of a single barrage pool complex (ca. 10-30 m). Spirogyra sp. was common near the inflow point to the barrage pools, while Ulva intestinalis and U. prolifera were particularly abundant near the outflow where the influence of the sea was greater. Overall, the macroalgal species recorded in the barrage pool systems show strong similarities to the macroalgae of Eastern Cape estuaries.26 Similarly, the macrophytes associated with the barrage pool systems included typical freshwater wetland species27,28, as well as plants normally associated with Cape Estuarine Saltmarsh vegetation29.
Based on the macrophyte species data, the tufa stromatolite environment appears to have more in common with estuarine systems than with freshwater wetlands.
With few exceptions, metazoan communities have generally been regarded as incompatible with the very existence of stromatolites.6,30 ,31 However, a diverse macrofauna community characterises the stromatolite pools of the Eastern Cape, with euryhaline species occurring throughout the system and typical freshwater and marine species alternating with each other for pool dominance, or even coexisting in different layers of each pool. A case in point is provided by the true crabs (brachyurans), which exhibit abundances of a typical freshwater species, Potamonautes perlatus (Figure 7a), next to a typical marine intertidal dweller, Cyclo-grapsus punctatus (Figure 7b). The two can be found occasionally in the same barrage pool, but at different depths, with the marine species generally under rocks in the deeper parts where the denser saline water sinks. Mass mortality of C. punctatus has been observed when pools become fresh water dominated as a result of an imbalance of flows from the two sources or when turbulent conditions force mixing and the breakdown of haloclines in the water column. Stromatolite pools appear to be colonised by a unique suite of common fish species, most of which are usually associated with estuaries. It is unknown to what extent these unique habitats serve as a fish nursery. In the fresher pools, there are often frogs and dense aggregations of tadpoles.
Most extant tufa stromatolite deposits around the world are already protected. Those occurring on the southwest coast of Western Australia, resembling most closely the South African type, are registered as 'Threatened Ecological Communities' with the status of endangered ecosystems.8 Special 'Ecological Water Requirements' have been allocated to them, in order to secure their long-term sustainability. Because of threats of residential encroachment, groundwater extraction, coastal development and invasion by alien species, it would be advisable for South Africa to follow the Australian example and place all tufa stromatolite formations under statutory protection.
Acknowledgements
This work is based on the research supported by the South African Research Chairs Initiative (SARChI) and the South African Environmental Observation Network (SAEON) of the Department of Science and Technology (DST) and the National Research Foundation (NRF) of South Africa. Any opinion, finding, conclusion or recommendation expressed in this material is that of the authors and the NRF does not accept any liability in this regard. Microbial diversity studies were funded by a grant from the Rhodes University Sandisa Imbewu Fund.
Authors' contributions
R.P, TB. and P-PS. were the project initiators and co-ordinators, and were responsible for the sampling design and the compilation of physicochemical and algal/macrophyte components; N.A.F.M. was responsible for the sampling design and sample collection and analysis and compiled the physico-chemical graphs; R.D. and G.M. were responsible for the sampling design and microbial analyses; N.S. was responsible for sample collection and fish analysis; and N.P was responsible for sample collection and invertebrate analysis.
References
1. Smith AM, Uken R. Living marine stromatolites at Kei River mouth. S Afr J Sci. 2003;99:200. [ Links ]
2. Smith AM, Uken R, Thackeray Z. Cape Morgan peritidal stromatolites: The origin of lamination. S Afr J Sci. 2005;101:107-108. [ Links ]
3. Smith AM, Andrews JE, Uken R, Thackeray Z, Perissinotto R, Leuci R, et al. Rock pool tufa stromatolites on a modern South African wave-cut platform: Partial analogues for Archaean stromatolites? Terra Nova. 2011;23(6):375-381. http://dx.doi.org/10.1111/j.1365-3121.2011.01022.x [ Links ]
4. Riding R. A hard life for cyanobacteria. Science. 2012;336:427-428. http://dx.doi.org/10.1126/science.1221055 [ Links ]
5. Bosak T, Knoll AH, Petroff AP. The meaning of stromatolites. Annu Rev Earth Planet Sci. 2013;41:21-44. http://dx.doi.org/10.1146/annurev-earth-042711-105327 [ Links ]
6. Riding R. Cyanobacterial calcification, carbon dioxide concentrating mechanisms, and Proterozoic-Cambrian changes in atmospheric composition. Geobiology. 2006;4:299-316. http://dx.doi.org/10.1111/j.1472-4669.2006.00087.x [ Links ]
7. Basso D, Bracchi VA, Favalli AN. Microbialite formation in southern Sinai (Egypt). Facies. 2013;59(1 ):7-18. http://dx.doi.org/10.1007/s10347-012-0330-4 [ Links ]
8. Forbes M, Vogwill R, Onton K. A characterization of the coastal tufa deposits of south-west Western Australia. Sed Geol. 2010;232:52-65. http://dx.doi.org/10.1016/j.sedgeo.2010.09.009 [ Links ]
9. Cooper JAG, Smith AM, Arnscheidt J. Contemporary stromatolite formations in high intertidal rock pools, Giant's Causeway, Northern Ireland: Preliminary observations. J Coast Res Spec Issue. 2013;65:1675-1680. [ Links ]
10. Beutler M, Wiltshire KH, Meyer B, Moldaenke C, Luring C, Meyerhöfer M, et al. A fluorometric method for the differentiation of algal populations in vivo and in situ. Photosynth Res. 2002;72:39-53. http://dx.doi.org/10.1023/A:1016026607048 [ Links ]
11. Catherine A, Escoffier N, Belhocine A, Nasri AB, Hamlaoui S, Yéprémian C, et al. On the use of the FluoroProbe, a phytoplankton quantification method based on fluorescence excitation spectra for large-scale surveys of lakes and reservoirs. Water Res. 2012;46:1771-1784. http://dx.doi.org/10.1016/j.watres.2011.12.056 [ Links ]
12. Parsons TR, Maita Y Lalli CM. A manual of chemical and biological methods for seawater analysis. Oxford: Pergamon Press; 1984. [ Links ]
13. Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, et al. Introducing Mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol. 2009;75:7537-7541. http://dx.doi.org/10.1128/AEM.01541-09 [ Links ]
14. Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ, et al. The Ribosomal Database Project: Improved alignments and new tools for rRNA analysis. Nucleic Acids Res. 2009;37(Database Issue):D141 -D145. http://dx.doi.org/10.1093/nar/gkn879 [ Links ]
15. Wang Q, Garrity GM, Tiedje JM, Cole JR. Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol. 2007;73:5261-5267. http://dx.doi.org/10.1128/AEM.00062-07 [ Links ]
16. Huson DH, Mitra S, Weber N, RuscheweyhH, Schuster SC. Integrative analysis of environmental sequences using MEGAN4. Genome Res. 2011;21:1552-1560. http://dx.doi.org/10.1101/gr.120618.111 [ Links ]
17. Walter MR, editor. Stromatolites. Developments in sedimentology 20. Amsterdam: Elsevier; 1976. [ Links ]
18. Goh F, Allen MA, Leuko S, Kawaguchi T, Decho AW, Burns BP et al. Determining the specific microbial population and their spatial distribution within the stromatolite ecosystem of Shark Bay. ISME J. 2009;3:383-396. http://dx.doi.org/10.1038/ismej.2008.114 [ Links ]
19. Papineau D, Walker JJ, Mojzsis SJ, Pace NR. Composition and structure of microbial communities from stromatolites of Hamelin Pool in Shark Bay Western Australia. Appl Environ Microbiol. 2005;71:4822-4832. http://dx.doi.org/10.1128/AEM.71.8.4822-4832.2005 [ Links ]
20. Havemann SA, Foster JS. Comparative characterization of the microbial diversities of an artificial microbialite model and a natural stromatolite. Appl Environ Microbiol. 2008;74:7410-7421. http://dx.doi.org/10.1128/AEM.01710-08 [ Links ]
21. Khodadad CLM, Foster JS. Metagenomic and metabolic profiling of nonlithifying and lithifying stromatolitic mats of Highborne Cay, the Bahamas. PLoS ONE. 2012;7:e38229. doi:10.1371/journal.pone.0038229. http://dx.doi.org/10.1371/journal.pone.0038229 [ Links ]
22. Santos F, Pena A, Nogales B, Soria-Soria E, Ma Ángeles García del Curac, González-Martín JA, et al. Bacterial diversity in dry modern freshwater stromatolites from Ruidera Pools Natural Park, Spain. Sys Appl Microbiol. 2010;33:209-221. http://dx.doi.org/10.1016/j.syapm.2010.02.006 [ Links ]
23. Stal LJ. Cyanobacterial mats and stromatolites. In: Whitton BA, Potts M, editors. The ecology of cyanobacteria: Their diversity in time and space. Dordrecht: Springer; 2002. p. 61-120. http://dx.doi.org/10.1007/0-306-46855-7_4 [ Links ]
24. Baumgartner LK, Spear JR, Buckley DH, Pace NR, Reid RP Dupraz C, et al. Microbial diversity in modern marine stromatolites, Highborne Cay, Bahamas. Environ Microbiol. 2009;11:2710-2719. http://dx.doi.org/10.1111/j.1462-2920.2009.01998.x [ Links ]
25. Reid RP, Visscher PT, Decho AW, Stolz JF, Bebout BM, Dupraz C, et al. The role of microbes in accretion lamination and the early lithification of modern marine stromatolites. Nature. 2000;406:989-992. http://dx.doi.org/10.1038/35023158 [ Links ]
26. Prinsloo S. The distribution and diversity of macroalgae in selected estuaries along the Eastern Cape coast of South Africa [MSc thesis]. Port Elizabeth: Nelson Mandela Metropolitan University; 2012. [ Links ]
27. Lubke RA. Freshwater plants. In: Lubke RA, De Moor I, editors. Field guide to the eastern & southern Cape coasts. Cape Town: Cape Town University Press; 1998. p. 235-248. [ Links ]
28. Cook C. Aquatic and wetland plants of southern Africa. Leiden: Backhuys Publishers; 2004. [ Links ]
29. Mucina L, Rutherford MC. The vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. Pretoria: South African National Biodiversity Institute; 2006. [ Links ]
30. Lee RE. Phycology. 3rd ed. Cambridge: Cambridge University Press; 1999. [ Links ]
31. Tarhan LG, Planavsky NJ, Laumer CE, Stolz JF, Reid RP Microbial mat controls on infaunal abundance and diversity in modern marine microbialites. Geobiology. 2013;11(5):485-497. http://dx.doi.org/10.1111/gbi.12049 [ Links ]
 Correspondence:
Correspondence:
Renzo Perissinotto
Department of Zoology
Nelson Mandela Metropolitan University
PO Box 77000, Port Elizabeth 6031, South Africa
EMAIL: renzo.perissinotto@nmmu.ac.za
Received: 09 Jan. 2014
Revised: 03 Mar. 2014
Accepted: 05 Mar. 2014


















