Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.110 n.7-8 Pretoria Aug. 2014
RESEARCH ARTICLE
Assessing leaf spectral properties of Phragmites australis impacted by acid mine drainage
Heidi van Deventer; Moses A. Cho
Ecosystems Earth Observation, Natural Resources and the Environment, Council for Scientific & Industrial Research, Pretoria, South Africa
ABSTRACT
The decanting of acid mine drainage (AMD) from the Western Basin on the Witwatersrand in late 2010 raised concerns about AMD risks in other gold, coal and copper mining areas of South Africa. Field spectroscopy and the use of vegetation indices could offer an affordable and easy means of monitoring the impact of mine water and/or AMD on vegetation. The impact of raw and treated mine water or contaminated soil on wetland vegetation often manifests in growth inhibition and reduction of foliar pigments and nutrient levels. Surveying the impact on wetland vegetation or underlying soils can be difficult and expensive considering the cost of laboratory analysis of samples. The potential of field spectroscopy for detecting the impact of mine water on wetland vegetation was examined by assessing (1) whether there was a significant difference in leaf spectra between sites receiving mine water and a non-impacted control site and (2) whether there was a gradation of vegetation condition downstream from the decanting site. Two vegetation indices were derived from portable field spectrometer-measured spectra of five green leaves of Phragmites australis - the chlorophyll red edge position (REP) and the normalised difference vegetation index (NDVI) - for two dormant (winter) and peak growth (summer) seasons in 2011-2012. Mean REP and NDVI values were significantly (p<0.05) lower for affected sites compared to the control site for both seasons and years. The range of REP values for young green leaves in winter for affected sites was 695-720 nm compared to the narrower range of 705-721 nm for the control site. The mean REP values for young green leaves in winter was 708 nm for the affected sites compared to 716 nm for the control site. The downstream gradation, however, fluctuated for REP and NDVI over the study period. We conclude that field spectroscopy shows potential to serve as a relatively quick and affordable means to assess the condition and health of vegetation affected by AMD.
Keywords: leaf spectra; mine water; chlorophyll red edge position; NDVI; field spectroscopy
Introduction
The decanting of acid mine drainage (AMD) from the Western Basin gold mining void in South Africa, in late August 2010, highlighted the environmental risks of AMD.1-3 AMD occurs through the exposure to oxygenated water of the naturally occurring mineral iron disulphide (FeS2).4,5 A number of chemical reactions result in the formation of ferrous iron, sulphate and acidity.4,5 The solubility of trace and heavy metals in the environment increases when exposed to AMD,2 which results in increased levels of these metals in water, sediments or vegetation. The exceptionally high rainfall since 2009 in the Witwatersrand region increased the groundwater discharge and enhanced the formation of AMD in the historical gold mining voids. Existing pump and treatment facilities could not accommodate the increased AMD volumes which decanted through historical incline and ventilation shafts.2,3 Other sources of AMD are open-cast coal and copper mines elsewhere in South Africa, as well as mine dumps, that contain iron disulphide and are exposed to oxygenated rainwater.1,3 AMD decants into natural wetland systems, and affects natural ecosystems and agricultural lands.3,6 The extent and magnitude of the impacts of AMD need to be determined and monitored to provide a basis for effective management.
Vegetation serves as the first trophic level linking the physical environment and the upper trophic levels of the food chain.7,8 Therefore, monitoring of the condition and health of vegetation impacted by AMD could assist in the identification of the extent and degree of affected areas in support of selecting appropriate management intervention measures. Metal uptake into plant parts is dependent on aspects such as species, bioavailability and state of the metal, preference of metals for certain vegetation species, metal speciation, soil organic matter, soil moisture, as well as water or soil pH levels.9-12 Plant species less tolerant of mine water conditions such as low pH, high sulphate levels and exposure to trace and/or heavy metals would show signs of toxicity, whereas some species have the ability to adapt to these conditions and even experience stimulated growth.11 At low concentrations, Al, Co and Ni have been reported to stimulate growth in some plant species.13,14 On the other hand, high concentrations of metals such as Al (at low pH), As, Cd, Cr, Cu, Hg, Mg, Ni and Pb are likely to show toxicity impacts on plants including significant changes in biochemical and physiological responses, and the modification of several metabolic processes.10,15 Chlorosis may result from the inhibition of essential macronutrients in the presence of some toxic trace elements in the water or substrate, or through the substitution of essential ions.8,16 For example, high levels of Cu can substitute the Mg2+ molecule in chlorophyll, affecting photosynthesis negatively.17 Several species have developed adaptive ways to tolerate metal toxicity, such as selective absorption, excretion, avoidance of translocation to aerial parts through storage in roots, and the development of enzymes or proteins as means to adapt to increased levels.8,16
Assessing the geographical extent and degree of impact of mine water on vegetation is time consuming and costly, considering the deep intricate root systems of macrophytes and trees. Often, contaminated soils are covered by vegetation and difficult to access.18,19 The cost of laboratory analysis of samples for various minerals or chemicals can hugely increase the cost of assessments and continuous monitoring. Monitoring techniques for assessing vegetation health should consider feasible, practical and affordable solutions, even if these are mere surrogates of vegetation health.
Remote sensing offers an alternative means of monitoring vegetation health in areas impacted by mine water. Research has highlighted specific absorption features in the electromagnetic spectrum that are highly correlated with foliar biochemicals, such as carotenoids, chlorophyll and nitrogen.20 An increase in foliar chlorophyll levels, for example, results in higher absorption levels in the red region (reduced reflection), whereas increases in biomass and leaf structure increase reflection in the near-infrared (NIR) region.21 Furthermore, high chlorophyll levels cause the broadening of the absorption pit in the red (660-680 nm) and a shift of the red edge reflectance slope (680-760 nm) and the point of maximum slope in the red edge known as the chlorophyll red edge position (REP) towards the longer wavelengths, referred to as a red shift.22,23 On the other hand, decreasing chlorophyll causes an increase in reflectance in the red region of the electromagnetic spectrum, and a shift of the red edge slope and REP towards the shorter wavelengths - a blue shift. A number of vegetation health indices have been developed in earth observation studies to assess both plant pigment and nutrient status, and variability in leaf structure or biomass using the red, REP and NIR bands.21
Trace and heavy metal contamination in the receiving water or substrate or adsorbed into the plants can cause varied responses in vegetation spectra.18,24,25 Zn was found to cause a traditional blue shift in the REP with a decrease in the NIR24, and similarly the radionuclides Cs and Sr at Chernobyl were highly negatively correlated to the REP green and NIR regions26. An opposite trend was observed in plants exposed to Cd24, Pb27, a combination of heavy metals (As, Cd, Cr, Cu, Pb and Zn)18 and radionuclides26, in which the REP decreased and the NIR increased with exposure. Chlorophyll response was found to correlate only partly with metal contamination18,19, in some instances estimated as merely 30%28. This finding may be attributed to the unique absorption patterns and storage location of heavy metals in plants12,29, hence spectral responses may also be unique to plant species25. The efficiency of remote sensing may require that relevant indices be investigated for each site, species and set of mine water conditions prior to use in monitoring.
Phragmites australis, Typha capensis and alien poplar trees (Populus x canescens) are known to tolerate conditions associated with mine water.19,30 These species are largely found in natural wetlands in South Africa. Phragmites australis, known to be tolerant to low pH levels, reduces the uptake of heavy metals through plaque formation at its roots30, yet still adsorbs a significant amount and releases a low amount of heavy metals, and so is considered appropriate for use in phytoremediation31-36. Metals are mostly stored in rhizomes, with lower concentrations in stems and leaves, although certain metals are exceptions.35,37-40 Compared to Phragmites australis, Typha capensis and poplars are less prevalent across both disturbed and pristine wetland types in South Africa, and therefore provide less opportunity for monitoring downstream from a decanting site. Poplars are also less suitable for monitoring because they often are targeted for invasive alien species control.
The aim of our research was to assess whether leaf-level spectroscopy could serve as a means of assessing and monitoring vegetation health impacted by AMD. Leaf reflectance of Phragmites australis sites affected by a combination of raw and treated mine water was compared to that of a control site to determine if there were differences in spectral indices of vegetation health. Green leaves in the dormant and peak growth seasons were sampled for a 2-year period. Two vegetation indices were derived from the leaf spectra: the chlorophyll REP41 and the normalised difference vegetation index (NDVI)42,43. The variation of these indices was also assessed per site to determine a gradation of vegetation condition with distance downstream from the mine water source.
Materials and methods
Study area and background
The study area was located along the Tweelopiespruit (around 26°06'S and 27°43'E) situated west of Johannesburg, South Africa (Figure 1). The word 'spruit' refers to a creek or small tributary stream. The Tweelopiespruit is the first drainage to receive mine water discharge from the West Rand Goldfield (i.e. the Western Basin), and joins its main stem - the Rietspruit - some 6 km to the north of the mine water source (Figure 1). The region experiences a mean annual rainfall of ~700 mm44 with characteristically wet summers and dry winters. The geohydrology of the study area is complex, comprising contiguous dolomitic karst aquifers and quartzitic fractured rock aquifers which produces complicated drainage patterns and varied groundwater recharge dynamics.2,45 Dolomitic springs along the reach of the Tweelopiespruit contribute ~2 ML/day to this drainage.
Mine water decants onto the surface commenced in August 2002 via boreholes and shafts (two ventilation shafts - Winze 17 and Winze 18) and an abandoned incline (the Black Reef Incline).45 A high density sludge (HDS) treatment plant with a capacity of ~12 ML/day was commissioned to manage this decant, and the neutralised product is then released into the Tweelopiespruit, generating a perennial flow of similar magnitude. Natural run-off during the rainfall season increased the stream discharge to ~24 ML/day for a mean annual flow of ~18 ML/day. These circumstances prevailed until early 2010, when recharge during an abnormally wet summer precipitated unmanageable quantities (>40 ML/day) of raw mine water discharge.46 A return to pre-2010 water chemistry and flow conditions was only achieved in mid-2012 as a result of a combination of factors, most notably a reduction in rainfall and the commissioning of a refurbished HDS treatment plant with a greater capacity of ~24 ML/day.
The circumstances described above gave rise to surface water chemistry discharges characteristic of AMD into the Tweelopiespruit. Prior to 2010, the dominant treated mine water chemistry was characterised by a circumneutral pH of 7, sulphate concentrations of ~2500 mg/L, and iron and manganese concentrations of <1 mg/L and <20 mg/L, respectively.46 In early 2010 to mid-2012, the dominant raw mine water chemistry was reflected in a pH value of ~3, electrical conductivity (EC) levels of ~300 mS/m, sulphate levels of ~3 000 mg/L, and iron and manganese concentrations of ~200 mg/L and ~60 mg/L, respectively.46
The hillslopes on the mine property are dominated by grassland, except for the narrow artificial wetlands in the stream channel, which are primarily dominated by Phragmites australis. Downstream of the mine property, the combination of treated and untreated mine water flows through artificial wetlands in the Krugersdorp Municipal Nature Reserve for a distance of about 5 km. Being a relatively small sub-catchment (38 km2), run-off and potential contamination from adjacent land uses are minimal. The wetlands are primarily dominated by Phragmites australis and large stands of Populus x canescens. The Tweelopiespruit exits the reserve prior to its confluence with the Rietspruit (Figure 1).
Our sampling sites were designated Sites 1 to 9, and two additional sites (Sites 10 and 11) were added as controls (Table 1; Figure 1). Control Site 10 was chosen for comparison of the water chemistry and trace element levels in the sediment and Phragmites australis as it had similar geology to the Tweelopiespruit. Control Site 11 was chosen for comparison of the leaf spectra results to the Tweelopiespruit. Control Site 11 (Figure 1, inset map 1) is located approximately 24.5 km north-northeast in the valley of the Skeerpoort River (25° 53' 40" S; 27° 45' 48" E), which flows through the John Nash Nature Reserve. This area receives a similar mean annual rainfall of ~700 mm.44 The Skeerpoort River, fed by dolomitic springs yielding a total discharge2 of 9.5 Mm3/annum, is considered to be in a near pristine condition. The site has shown no land use impact for the past 50 years, and, in particular, has not experienced any AMD impacts. Average water chemistry (measured since 1964) shows mean pH as above 7, EC at 32.4 mS/m (s.d.=2.58) and mean sulphate levels as 8 mg/L (s.d. = 3.8). The control Phragmites australis population occurs here on the edge of an artificial wetland.
Data collection
A study was conducted in 2009 to compare the AMD chemistry of the Tweelopiespruit sites with a non-AMD control site. Water, sediment and plant material were collected in the winter of 2009 from five sites along the Tweelopiespruit (Table 1, Sites 1-5) and at a control site (Site 10). At each site, sediment was collected from 100 mm below the water column, in close proximity to Phragmites australis. Specimens of the rhizomes, stems and leaves of Phragmites australis were also collected at each site and subsequently dried in an oven at 65° C for 3 days. Water samples were analysed for pH, EC and sulphates (SO4), while element concentrations in the water, sediment and plant material were assessed using inductively coupled plasma elemental analysis.
Green leaves of Phragmites australis were sampled in 2011-2012 to compare the physiological differences between the sites affected by AMD (Sites 6-9) and a site not impacted by AMD (control Site 11). Sites 6-9 are situated on the Tweelopiespruit (Figure 1) and control Site 11 in the unspoilt Skeerpoort River valley (Figure 1, inset map) in the John Nash Nature Reserve at the chalets. Phragmites australis occur here on the sides of an artificial wetland flanked on both sides by indigenous trees.
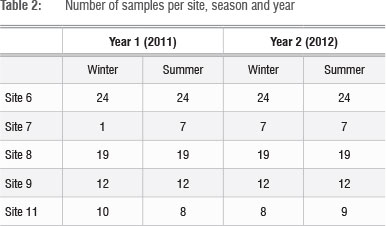
At each sampling site, sampling points were selected based on the availability and accessibility of green leaves during the dry period (June 2011). Sampling was carried out in the dormant (winter) and peak growth (summer) periods of 2011 and 2012 (Table 2). At each sampling point, five green leaves were collected for leaf level spectral reflectance measurements from five different stems of Phragmites australis within an area of 4 m2. In the winter, these leaves were primarily small green leaves of new growth, whereas in the summer they were large mature leaves.
Leaf level spectral reflectance measurements
An analytical spectral device, the FieldSpec Pro spectroradiometer (Analytical Spectral Devices Inc, Boulder, CO, USA), was used to record the leaf spectra of the five collected green leaves of Phragmites australis. The instrument has a spectral range of 350-2500 nm and a bandwidth of 3 nm re-sampled to 1 nm (http://www.asdi.com). A contact probe was used for spectral reflectance measurements to avoid external factors (such as light illumination differences). The contact probe enables calibration with a white reflectance panel, and has a dark panel for measuring reflectance. For each sampling point, the five leaf spectra were leaf averaged. The REP linear extrapolation method41 was derived from the spectral measurements using a three-step procedure:
Calculate maximum first derivative spectrum

Calculate the reflectance at the inflexion point

Calculate the red edge position
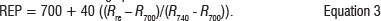
The NDVI was derived from the spectral measurements according to the formula

Data analyses
The raw and derivative reflectance spectra were used to explore the differences between sites and seasons. For each vegetation index, the mean and standard deviation values were derived for sites affected by mine water and for the control site for the two seasons and years. A one-way analysis of variance (ANOVA) and post-hoc Tukey honest significant difference (HSD) test were performed in Statistica (version 7, StatSoft Inc.) to assess the significance of differences between the sites affected by mine water and the control site.
In addition, the mean and standard deviation values were calculated for each site for the two seasons and years to investigate the variation in gradation from upstream to downstream. ANOVA and post-hoc Tukey HSD tests were repeated to assess differences between sites for the sampling periods.
Results
Element analysis of water, sediment and plant material
The pH and EC show an improvement downstream of the decant site (Table 3), yet no consistent trend is shown in sulphate levels downstream.
A high concentration of heavy metals (Table 3) was found in sediments and the rhizomes, stems and leaves of Phragmites australis. Sediment samples taken from the top 100 mm on the sides of the wetlands show that the highest values for As and Fe were recorded at the decanting site (Site 1) and Site 5, whereas Al was the highest at Sites 2 and 5. Cr, Pb and Zn measured highest at Site 4. Site 5 (or Site 9) show the highest measurements for Cu, Mn, Ni and U, although Cr, Pb and Zn also were high at Site 5. Metals accumulated primarily in the rhizomes of the reeds with lower concentrations in the aerial parts. No consistent trend could be noted for all the metals downstream from the decant site.
A tier 1 risk assessment, where site concentrations are rated in respect to regulatory standards, was done of the top 0.5 m of wetland sediment (below 1 m the water surface) at three sites in the Tweelopiespruit. This assessment also indicated high levels of Ni and U above acceptable standards of the European Union and the South African National Nuclear Regulator.47
Green leaf spectra of affected sites compared to the control site
Overall, the average reflectance spectra of green leaves for both winter and summer of the 2 years show normal vegetation curves peaking in the green, absorbing in the red and having high reflectance in the NIR region (Figure 2). The average reflectance spectra for the affected sites are higher than that of the control site in the visible region (VIS) over the two winter periods, although are very similar in the NIR. Both summer periods show higher average reflectance spectra in the VIS and NIR for the affected sites in comparison to the control site.
Both winter and summer seasons over the 2 years show a blue shift for the affected sites, compared to control Site 11 (Figure 3). The double peak feature41,48 and blue shift is particularly visible in the winter season of the first year (Figure 4), and less obvious in the second year.
The behaviour of the double peak feature of the first derivative in the red edge region between 660 nm and 780 nm was captured in the linear extrapolation REP model by Cho and Skidmore41. The dominance of the first peak (at around 700 nm) is associated with low chlorophyll and the dominance of the second peak is associated with high levels of chlorophyll.41 The control site shows a dominance of the second derivative peak (at about 725 nm) in the red edge region when compared to the affected site (Figure 6). Hence, the control site shows high levels of chlorophyll for all the sampling periods, compared to the affected sites. In the winter of both years the differences between control Site 11 and affected sites are particularly visible, with the affected sites showing medium levels of chlorophyll compared to the control site, which shows higher levels of chlorophyll (Winter Year 1 and Year 2 in Figure 4).
Green leaf spectra of individual sites compared to the control site
When comparing the average green leaf reflectance of each site over the two seasons and years, the control site seems to have lower reflectance in the VIS region than the other sites throughout the time periods (Figure 5). The control site shows more dominance of the second red edge derivative peak around 720 nm when compared to the affected site. In winter, the control site reflectance lies within the range of the other sites for the NIR region of the spectrum. In summer, however, it is lower in the NIR compared to the other sites. None of the affected sites show particular differences compared to the others in average reflectance spectra over the sampling period.
Most of the sites show a blue shift for the winter and summer seasons over the 2 years, compared to the control site (Figure 6). Leaves of the control site seem to have higher chlorophyll absorption levels compared to the affected sites in both winters (Figure 7). In the summer months, the affected sites appear to have slightly higher chlorophyll absorption levels compared to the control site. In the winter of Year 1, Site 7 shows the most stress. Only a single new green leaf was available at this site for sampling. The leaf structure is least developed (Figure 6, Year 1 Winter, Site 7) and chlorophyll levels are much lower compared to all the other sites. These observations support the circumstances that describe the period of greatest raw mine water discharge into the Tweelopiespruit.
The single leaf sampled in Year 1 Winter at Site 7 also is evident in Figure 7, where the second derivative peak shows a blue shift towards 690 nm and no double peak feature. There is a greater variation of chlorophyll absorption levels, the location of the second derivative peak and the double peak feature between sites during the winter season (Figure 7, see Year 1 and 2) compared to the variation of these features between sites in the summer seasons. In the winter, Site 6 shows a double peak feature in the second red edge derivatives, with the change in the first peak around 695 nm more prominent compared to the second peak near 720 nm. In the winter of Year 2, Site 9 follows a similar pattern. In contrast, Site 8 matches the slope of the control site more closely for both winters, with the peak near 720 nm.
In the summer seasons, the profile of the second red edge derivative peaks are closely matched for all sites. The affected sites tend to medium levels of chlorophyll (a double peak profile), compared to the higher chlorophyll levels of the control site (profile skewed towards second peak) for the summers of Years 1 and 2. The affected sites show more change in the second derivative profile around the first peak, near 690 nm, compared to the control site. For all the sites, the control shows the strongest dominance of the second derivative peak, indicating the highest levels of chlorophyll.
Variation of spectral indices between affected and control sites
REP and NDVI values between the affected sites and the control site for each season and year show significant differences (p<0.05). Sites affected by mine water had significantly lower REP values (p<0.05) over the two seasons and years compared to the control site (Table 4). Sites affected by mine water show a higher variability of REP values in winter compared to the summer seasons. The average REP value increased from winter to summer for the affected sites, and decreased slightly in the control site for the same period.
NDVI values also show a greater variation in the winter period for the affected sites compared to the control site (Table 4). In all instances, the ranges of NDVI values of the affected sites and control site overlap largely for both seasons and years. Average NDVI values in the winter were significantly lower (p<0.05) for affected sites than for the control site, whereas the average NDVI values in the summer were closer in value.
Downstream gradation of spectral indices
Generally, average REP and NDVI values in winter increase from Site 6 to Site 8 (downstream) and drop at Site 9 (Table 5), except for Site 2 in the winter of Year 1, where only one new green leaf was available. In summer, an opposite trend is noted for average REP values: in general, the average REP decreases downstream from Site 6 to Site 8 and then increases again at Site 9. Average NDVI in summer shows less distinct patterns downstream of Site 1. For the first three sites (6, 7 and 8), average REP in summer decreased from Year 1 to Year 2, whereas the average NDVI values decreased in winter and increased in summer. There appears to be a gradient of the REP values only in the winter of Year 1. On average, the control site values change slightly over the 2 years; however, affected sites (Sites 6-9) show more fluctuation, particularly in REP values.
When comparing all sites to one another (a combination of 10 pairs; Bonferroni-corrected p <0.005), Sites 6 and 11 are significantly different from one another in both seasons and years for both REP (Table 6) and NDVI (Table 7). Sites 6 and 8 also differ significantly for both seasons and years for REP. Few significant differences between sites are noted for NDVI.
Discussion and conclusions
Field spectroscopy shows potential as a means to assess the impact of mine water on wetland vegetation and to monitor impact over time. The REP and NDVI values for affected sites were significantly lower compared to the control site for both winter and summer seasons over the 2 years. The mean and standard deviation of REP and NDVI can therefore potentially serve as surrogates for establishing impact on wetland vegetation and monitoring vegetation condition over time in other areas of South Africa. It remains to be established whether the method is site and species specific, or whether it also applies to other sites regardless of mine water characteristics and environmental conditions.
Winter seasons presented more distinct differences in REP and NDVI values between affected sites and control Site 11. In addition, leaves of control Site 11 seemed to have higher chlorophyll absorption in the winter of both years, based on the dominance of the second derivative peak of the first derivative spectra of the REP region. The opposite trend of an increased reflectance in the NIR (i.e. biomass) and a reduction in chlorophyll (or REP values) was, on the other hand, particularly visible in the summer months, similarly to findings reported by others.24,27 The pattern was, however, less distinct in winter months, although leaf maturity may influence the responses in both the red and NIR regions in this instance.
Both REP and NDVI showed significant differences between the affected and control Site 11; however, in comparison, REP and NDVI values for individual sites yielded fewer significant differences between affected sites and control Site 11. The inconsistent responses of REP values of individual sites affected by AMD compared to control Site 11 are similar to those noted for Phragmites australis at two affected sites compared to a control site in Hungary.19 Regardless, the presence of high levels of, among others, Al, As, Cd, Cr, Cu, Fe, Mn, Ni, Pb, Zn and total U in the soils found in parts of the Tweelopiespruit47 may have a toxic effect on plants, hence at some sites the REP and NDVI indices show significant differences from control Site 11. Metal concentrations in sediments at particular sites were found to vary, not only as noted by Venter et al.47 at, for example, the Hippo Dam wetland, but also between the two studies at similar sampling points. Variation in metal concentrations may be attributed to the natural variation of water depth, flow and presence of vegetation in parts of a wetland. Variations in stream flow and pH during rainfall periods, when a larger volume of raw mine water and AMD floods through the spruit, would also cause changes in the bioavailability of metals from the stream bed and vegetation, contributing to further temporal and geographical variations in metal concentrations. In any event, the differences between sites are difficult to explain and can be attributed to a number of possible factors influencing the signatures. Further exploration of temporal and spatial variation of the metal concentrations and affected vegetation is essential to further the understanding of the impact of AMD in the Tweelopiespruit.
Acknowledgements
Funding for this work was provided by the CSIR as part of the Strategic Research Panel (SRP) acid mine drainage (AMD) project. Our gratitude is extended to the following individuals and institutions for providing access to their properties: The South African National Botanical Gardens, Rand Uranium / Gold 1, African Bush Adventures, Mr S. du Toit of Mogale City local municipality, Mr J. Rykaardt and Mr H. Rykaardt of Protea Eco-Adventures, and Mr H. Visser of John Nash Nature Reserve. We thank Dr J.F. Durand for his advice and fieldwork support. We are also grateful to CSIR colleagues and project participants for support and advice, including Dr B. Genthe, Dr P. Oberholzer, Mr P. Hobbs and Dr R. Mathieu. We are thankful to three anonymous reviewers whose suggestions assisted in improving the publication.
Authors' contributions
H.v.D. contributed to the experimental and project design, undertook the fieldwork and statistical analysis, and wrote and revised the manuscript. M.A.C. was responsible for the experimental and project design, and the paper structuring, editing and revision.
References
1. McCarthy TS. The impact of acid mine drainage in South Africa. S Afr J Sci. 2011;107(5/6):1-7.http://dx.doi.org/10.4102/sajs.v107i5/6.712 [ Links ]
2. Hobbs PJ. Situation assessment of the surface water and groundwater resource environments in the Cradle of Humankind World Heritage Site. Report prepared for the Management Authority. Pretoria: Economic Development Department; 2011. [ Links ]
3. South African Inter-Ministerial Committee: Expert Team. Mine water management in the Witwatersrand Gold Fields with special emphasis on acid mine drainage [document on the Internet]. c2010 [cited 2014 Jan 28]. Available from: http://www.dwaf.gov.za/Documents/ACIDReport.pdf. [ Links ]
4. Krige WG, Van Biljon M. The impacts of mining on the water resources and water-based ecosystems of the Cradle of Humankind World Heritage Site. In: The South African Karst Working Group, editor. The karst system of the Cradle of Humankind World Heritage Site. Report no KV241/10. Pretoria: Water Research Commission; 2010. p. 177-210. [ Links ]
5. Singer PC, Stumm W. Acidic mine drainage: The rate-determining step. Science. 1970;167(3921):1121-1123. http://dx.doi.org/10.1126/science.167.3921.1121 [ Links ]
6. Naicker K, Cukrowska E, McCarthy TS. Acid mine drainage arising from gold mining activity in Johannesburg, South Africa and environs. Environ Pollut. 2003;122:29-40.http://dx.doi.org/10.1016/S0269-7491(02)00281-6 [ Links ]
7. Verkleij JAC, Golan-Goldhirsh A, Antosiewisz DM, Schwitzguébel J, Schroder P. Dualities in plant tolerance to pollutants and their uptake and translocation to the upper plant parts. Environ Exp Bot. 2009;67(1):10-22. http://dx.doi.org/10.1016/j.envexpbot.2009.05.009 [ Links ]
8. Peralta-Videa JR, Lopez ML, Narayan M, Saupe G, Gardea-Torresdey J. The biochemistry of environmental heavy metal uptake by plants: Implications for the food chain. Int J Biochem Cell Biol. 2009;41(8-9):1665-1677. http://dx.doi.org/10.1016/j.biocel.2009.03.005 [ Links ]
9. Petrescu L, Bilal E. Plant availability of uranium in contaminated soil from Crucea Mine (Romania). Environ Geosci. 2003;10(3):123-135. http://dx.doi.org/10.1306/eg100302001 [ Links ]
10. Goetz A, Rock B, Rowan LC. Remote sensing for exploration: An overview. Econ Geol. 1983;78:573-590. http://dx.doi.org/10.2113/gsecongeo.78.4.573 [ Links ]
11. Smical A, Hotea V, Oros V, Juhasz J, Pop E. Studies on transfer and bioaccumulation of heavy metals from soil into lettuce. Environm Eng Manag J. 2008;7(5):609-615. [ Links ]
12. Deng H, Ye ZH, Wong MH. Accumulation of lead, zinc, copper and cadmium by 12 wetland plant species thriving in metal-contaminated sites in China. Environ Pollut. 2004;132(1):29-40. http://dx.doi.org/10.1016/j.envpol.2004.03.030 [ Links ]
13. Aller AJ, Bernal JL, Nozal MJD, Deban L. Effects of selected trace elements on plant growth. J Sci Food Agric. 1990;51(4):447-479. http://dx.doi.org/10.1002/jsfa.2740510404 [ Links ]
14. Osaki M, Watanabe T, Tadano T. Beneficial effect of aluminum on growth of plants adapted to low pH soils. Soil Sci Plant Nutr. 2013;43(3):551-563.http://dx.doi.org/10.1080/00380768.1997.10414782 [ Links ]
15. Macfarlane GR, Pulkownik A, Burchett MD. Accumulation and distribution of heavy metals in the grey mangrove, Avicennia marina (Forsk.) Vierh.: Biological indication potential. Environ Pollut. 2003;123:139-151. http://dx.doi.org/10.1016/S0269-7491(02)00342-1 [ Links ]
16. De Almeida AF, Valle RR, Mielke MS, Gomes FP Tolerance and prospection of phytoremediator woody species of Cd, Pb, Cu and Cr. Braz J Plant Physiol. 2007;19(2):83-98. [ Links ]
17. Liu J, Xiong Z, Li T, Huang H. Bioaccumulation and ecophysiological responses to copper stress in two populations of Rumex dentatus L. from Cu contaminated and non-contaminated sites. Environ Exp Bot. 2004;52:43-51. http://dx.doi.org/10.1016/j.envexpbot.2004.01.005 [ Links ]
18. Liu M, Liu X, Ding W, Wu L. Monitoring stress levels on rice with heavy metal pollution from hyperspectral reflectance data using wavelet-fractal analysis. Int J Appl Earth Obs. 2011;13(2):246-255. http://dx.doi.org/10.1016/j.jag.2010.12.006 [ Links ]
19. Hecker HJ. Investigation of the relationship between chlorophyll concentration and high spectral resolution data of Phragmites australis in heavy metal contaminated sites [MSc thesis]. Enschede: ITC; 2003. [ Links ]
20. Curran PJ. Remote sensing of foliar chemistry. Remote Sens Environ. 1989;30:271-278. http://dx.doi.org/10.1016/0034-4257(89)90069-2 [ Links ]
21. Rouse J, Haas R, Schell J, Deering D. Monitoring vegetation systems in the Great Plains with ERTS. Third Earth Resources Technology Satellite-1 Symposium - Volume 1. National Aeronautics and Space Administration Series Publication 351. 1973:309-317. [ Links ]
22. Horler DNH, Barber J, Darch JP, Ferns DC, Barringer AR. Approaches to detection of geochemical stress in vegetation. Adv Space Res. 1983;3(2):175-179. http://dx.doi.org/10.1016/0273-1177(83)90118-7 [ Links ]
23. Horler D, Dockray M, Barber J. The red edge of plant leaf reflectance. Int J Remote Sens. 1983;4(2):273-288. http://dx.doi.org/10.1080/01431168308948546 [ Links ]
24. Sridhar BBM, Han FX, Diehl SV, Monts DL, Su Y. Spectral reflectance and leaf internal structure changes of barley plants due to phytoextraction of zinc and cadmium. Int J Remote Sens. 2007;28(5):1041-1054. http://dx.doi.org/10.1080/01431160500075832 [ Links ]
25. Horler DNH, Barber J, Barringer AR. Effects of heavy metals on the absorbance and reflectance spectra of plants. Int J Remote Sens. 1980;1(2):121-136. http://dx.doi.org/10.1080/01431168008547550 [ Links ]
26. Davids C, Tyler AN. Detecting contamination-induced tree stress within the Chernobyl exclusion zone. Remote Sens Environ. 2003;85(1):30-38. http://dx.doi.org/10.1016/S0034-4257(02)00184-0 [ Links ]
27. Clevers JGPW, Kooistra L, Salas EAL. Study of heavy metal contamination in river floodplains using the red-edge position in spectroscopic data. Int J Remote Sens. 2004;25(19):3883-3895. http://dx.doi.org/10.1080/01431160310001654473 [ Links ]
28. Liu Y Chen H, Wu G, Wu X. Feasibility of estimating heavy metal concentrations in Phragmites australis using laboratory-based hyperspectral data - A case study along Le'an River, China. Int J Appl Earth Obs. 2010;12, Supplement 2:S166-S170. http://dx.doi.org/10.1016/j.jag.2010.01.003 [ Links ]
29. Liu J, Dong Y,Xu H, Wang D, Xu J. Accumulation of Cd, Pb and Zn by 19 wetland plant species in constructed wetland. J Hazard Mater. 2007;147(3):947-953. http://dx.doi.org/10.1016/j.jhazmat.2007.01.125 [ Links ]
30. Rufo L, Rodriguez N, de la Fuente V. Plant communities of extreme acidic waters: The Rio Tinto case. Aquat Bot. 2011;95(2):129-139. http://dx.doi.org/10.1016/j.aquabot.2011.05.001 [ Links ]
31. Bragato C, Schiavon M, Polese R, Ertani A, Pittarello M, Malagoli M. Seasonal variations of Cu, Zn, Ni and Cr concentration in Phragmites australis (Cav.) Trin ex steudel in a constructed wetland of North Italy. Desalination. 2009;246(1-3):35-44. http://dx.doi.org/10.1016/j.desal.2008.02.036 [ Links ]
32. Batty LC, Younger PL. The effect of pH on plant litter decomposition and metal cycling in wetland mesocosms supplied with mine drainage. Chemosphere. 2007;66(1):158-164. http://dx.doi.org/10.1016/j.chemosphere.2006.05.039 [ Links ]
33. Ait Ali N, Bernal MP, Ater M. Tolerance and bioaccumulation of cadmium by Phragmites australis grown in the presence of elevated concentrations of cadmium, copper, and zinc. Aquat Bot. 2004;80(3):163-176. http://dx.doi.org/10.1016/j.aquabot.2004.08.008 [ Links ]
34. Batty LC. Wetland plants - more than just a pretty face? Land Contam Reclam. 2003;11(2):173-180. http://dx.doi.org/10.2462/09670513.812 [ Links ]
35. Batty LC, Baker AJ, Wheeler BD. Aluminium and phosphate uptake by Phragmites australis: The role of Fe, Mn and Al root plaques. Ann Bot. 2002;89(4):443-449. http://dx.doi.org/10.1093/aob/mcf067 [ Links ]
36. Burke DJ, Weis JS, Weis P. Release of metals by the leaves of the salt marsh grasses Spartina alterniflora and Phragmites australis. Estuar Coast Shelf S. 2000;51:153-159. http://dx.doi.org/10.1006/ecss.2000.0673 [ Links ]
37. Wang H, Jia Y. Bioaccumulation of heavy metals by Phragmites australis cultivated in synthesized substrates. J Environ Sci. 2009;21(10):1409-1414. http://dx.doi.org/10.1016/S1001-0742(08)62433-X [ Links ]
38. Peverly JH, Surface JM, Wang T. Growth and trace metal absorption by Phragmites australis in wetlands constructed for landfill leachate treatment. Ecol Eng. 1995;5:21-35. http://dx.doi.org/10.1016/0925-8574(95)00018-E [ Links ]
39. Bragato C, Brix H, Malagoli M. Accumulation of nutrients and heavy metals in Phragmites australis (Cav.) Trin. ex Steudel and Bolboschoenus maritimus (L.) Palla in a constructed wetland of the Venice lagoon watershed. Environ Pollut. 2006;144(3):967-975. http://dx.doi.org/10.1016/j.envpol.2006.01.046 [ Links ]
40. Vymazal J, Svehla J, Krõpfelová L, Chrastný V. Trace metals in Phragmites australis and Phalaris arundinacea growing in constructed and natural wetlands. Sci Total Environ. 2007;380(1-3):154-162. http://dx.doi.org/10.1016/j.scitotenv.2007.01.057 [ Links ]
41. Cho MA, Skidmore AK. A new technique for extracting the red edge position from hyperspectral data: The linear extrapolation method. Remote Sens Environ. 2006;101(2):181-193. http://dx.doi.org/10.1016/j.rse.2005.12.011 [ Links ]
42. Deering DW. Rangeland reflectance characteristics measured by aircraft and spacecraft sensors [PhD thesis]. College Station, TX: Texas A&M University; 1978. [ Links ]
43. Tucker CJ. Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens Environ. 1979;8(2):127-150. http://dx.doi.org/10.1016/0034-4257(79)90013-0 [ Links ]
44. Middleton BJ, Bailey AK. Water resources of South Africa, 2005 study (WR2005) and book of maps. Research reports TT381/08 & TT382/08. Pretoria: Water Research Commission; 2008. [ Links ]
45. Hobbs PJ, Cobbing JE. The hydrogeology of the Krugersdorp Game Reserve area and implications for the management of mine water decant. CSIR report no. CSIR/NRE/WR/IR/2007/0116/C. Pretoria: Council for Scientific and Industrial Research; 2007. [ Links ]
46. Hobbs PJ. Pilot implementation of a surface water and groundwater resources monitoring programme for the Cradle of Humankind World Heritage Site. Status report for the period April to September 2012. CSIR/NRE/WR/ ER/0088/B. Pretoria: Council for Scientific and Industrial Research; 2012. [ Links ]
47. Venter J, Motlakeng T, Kotoane M, Coetzee H, Hobbs P. Section 7: Sediment chemistry. In: Hobbs PJ, editor. Situation assessment of the surface water and groundwater resource environments in the Cradle of Humankind World Heritage Site. Pretoria: Economic Development Department; 2011. p. 94-99. [ Links ]
48. Zarco-Tejada PJ, Pushnik JC, Doborwski S, Ustin SL. Steady-state chlorophyll a fluorescence detection from canopy derivative reflectance and double-peak red-edge effects. Remote Sens Environ. 2003;84(2):283-294. http://dx.doi.org/10.1016/S0034-4257(02)00113-X [ Links ]
 Correspondence:
Correspondence:
Heidi van Deventer
Ecosystems Earth Observation
Natural Resources and the Environment
Council for Scientific & Industrial Research
Building 33, PO Box 395, Pretoria 0001, South Africa
HvDeventer@csir.co.za
Received: 14 June 2013
Revised: 09 Jan. 2014
Accepted: 28 Jan. 2014














