Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.109 no.5-6 Pretoria Jan. 2013
RESEARCH ARTICLE
Effect of selenium on cadmium-induced oxidative stress and esterase activity in rat organs
Kevin DzoboI; Yogeshkumar S. NaikI, II
IDepartment of Biochemistry, Faculty of Science, University of Zimbabwe, Harare, Zimbabwe
IIDepartment of Environmental Science and Health, National University of Science and Technology, Bulawayo, Zimbabwe
ABSTRACT
Metal toxicity is a threat mainly in the industrialised world where industry discharges many toxic metals into the environment. We investigated the effects of two metals - cadmium and selenium - on the cytosolic antioxidant enzymes and esterases in the liver, kidneys and testes of rats. Male Sprague-Dawley rats( N =28) were divided equally into four groups: control, cadmium, selenium and cadmium/selenium. Salts of the metals were administered intraperitoneally for 15 days. In the liver, cadmium treatment (1.67 mg/kg per day) resulted in a decrease in catalase activity and an increase superoxide dismutase (SOD) activity. Selenium treatment (0.23 mg/kg per day) resulted in increases in glutathione s-transferase, catalase and DT-diaphorase activities. Treatment with both cadmium and selenium resulted in an increase in glutathione peroxidase (GPx) activity. Esterase activities were significantly lower in the presence of cadmium. In the kidney, cadmium treatment caused a decrease in catalase, DT-diaphorase, and SOD activities and selenium supplementation reversed the cadmium-induced decrease in these enzyme activities. Selenium treatment increased catalase and SOD activities in the kidney. In the testis, cadmium treatment decreased GPx and SOD activities, but at the same time increased catalase and DT-diaphorase activities. Esterase activities increased in the presence of selenium in both the kidney and testis. These results suggest that selenium might be toxic to the liver while at the same time play a protective role against cadmium-induced oxidative stress and toxicity in the kidney and testis.
Keywords: selenium; cadmium; antioxidant; oxidative stress; rat organs
Introduction
Heavy metals are known to pose a serious threat to human health.1 Among the heavy metals, cadmium (Cd) is known to be present in water, air, food and even cigarette smoke.2 It is an industrial pollutant from battery, plastic and fertiliser industries.3 Animal studies have shown that Cd can stimulate formation of reactive oxygen species (ROS) and induce damage to various tissues.4-6 Cd is known to enhance lipid peroxidation and DNA damage and can induce abnormal expression of the main antioxidant molecules in cells.3,7 It has been listed as a chemical substance that is potentially dangerous on a global level and exposure to it must be minimised.8
Protection of cells from Cd-induced oxidative processes caused by ROS and free radicals is in the form of both enzymatic and non-enzymatic defence mechanisms present in the cell.9,10 Antioxidants include the enzymes superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx) and esterases,10,11 molecules such as glutathione (GSH)12 and trace metals such as selenium (Se). Trace elements such as Se have been known to have beneficial effects on Cd-induced oxidative stress.13 Se is an important co-factor of antioxidant enzymes such as GPx and thioredoxin.14 The protective effect of Se is known to vary from organ to organ.15 However, high levels of Se can be toxic to animal cells.16
The liver and the kidney are two critical organs with respect to Cd toxicity.2,17 Cd accumulates mainly in the liver and kidneys, where it causes metabolic changes and abnormal gene expression.18,19 Elaborate morphological changes have been observed in both organs in cases of Cd poisoning.20 The mechanism of Cd-induced hepatotoxicity and renal toxicity is poorly understood. Cd toxicity in testes has been studied in many animal models and its pathogenicity includes necrosis, atrophy and severe haemorrhage. Reduction in sperm count and motility and decreased testosterone levels are some of the effects of Cd-induced testicular damage.6,21.
Although studies have been done on Cd-induced toxicity in the liver, kidneys and testes,15,22 it is not surprising that conflicting results have been reported on the activities of antioxidant enzymes in oxidative stress induced by Cd in various organs.23,24 These discrepancies are mainly because studies focus on one organ at a time or exclude an important group of enzymes involved in the metabolism of organophosphates, namely the esterases. The effects of Cd on the antioxidant enzymes and esterases of the liver, kidneys and testes have not yet been studied simultaneously. Thus the aim of the present study was to simultaneously investigate the effect of Cd on the antioxidant enzymes and esterases of the liver, kidneys and testes. The possible protective role of Se was also investigated.
Materials and methods
Chemicals
Cadmium chloride (CdCl2) was obtained from Merck (Darmstadt, Germany). Reduced (GSH) and oxidised (GSSG) glutathione, nicotinamide adenine dinucleotide phosphate (reduced form; NADPH), 1-chloro-2,4-dinitrobenzene (CDNB), glutathione reductase, tert-butylhydroperoxide (tBOOH), bovine serum albumin (BSA), ascorbic acid (vitamin C), sodium carbonate (Na2CO3), nitroblue tetrazolium (NBT), butylated hydroxytoluene (BHT), thiobar-bituric acid (TBA) and 2,4-dinitrophenylhydrazine were purchased from Sigma Chemical Co. (St. Louis, MO, USA). All other chemicals were of analytical grade and were purchased from commercial suppliers.
Animals and treatment
The experiments were carried out using 8-week-old male Sprague-Dawley rats weighing approximately 240 g at the start of experiments. The rats were housed in individual cages at 21±2 °C, 40-60% relative humidity and exposed- to a 12-h light-dark cycle, with the light cycle coinciding with daylight hours. The rats were allowed to feed and drink tap water ad libitum. After 2 weeks of acclimation, rats were allocated randomly into four groups of seven animals each and received daily intraperitoneal injections over a 15-day period of either: (1) saline solution (control group), (2) CdCl2 in saline solution at a dose of 1.67 mg/kg body weight (cadmium-treated group), (3) sodium selenite solution at a dose of 0.23 mg/kg body weight (selenium-treated group) or (4) CdCl2 in saline solution at a dose of 1.67 mg/kg body weight and sodium selenite solution at a dose of 0.23 mg/kg body weight (cadmium/selenium-treated group). These doses have been used before and were administered either orally or peritoneally.4,5,6,13 At the end of treatment, rats were fasted overnight before being sacrificed by cervical dislocation. All animals were killed between 8:00 and 10:00 to avoid any possible diurnal variations in antioxidant enzyme level. Organ tissues were isolated, cleaned of adhering matters and washed with saline solution. The livers, kidneys and testes were quickly excised, rinsed in ice-cold saline to clear them of blood, weighed, finely minced in the same solution and homogenised (approximately 30% w/v) in a Corning Potter Elvehjem homogeniser with a Teflon pestle. Organ homogenates were used for thiobarbituric acid reactive substance (TBARS) determination. The homogenates were centrifuged (Beckman, Indianapolis, IN, USA) at 100 000 g for 30 min at 4 °C . The resulting cytosolic fractions were stored at -80 °C until required for experiments and post-mitochondrial supernatants were used for enzyme assays.
International rules and regulations guiding the handling and care of animals were followed throughout the study. This study was approved by the Ethics and Animals Welfare Committee of the University of Zimbabwe and was conducted in accordance with international guidelines such as the NIH guidelines for the treatment and care of laboratory animals.
Biochemical assays Enzyme assays
Glutathione s-transferase (GST) activity was determined using the method of Habig et al.25 This method is based on the reaction of 1-chloro-2,4-dinitrobenzene (CDNB) with the -SH group of glutathione catalysed by GST contained in the samples. The reaction proceeded in the presence of 1 mM GSH in phosphate buffer at a pH of 6.5 and a temperature of 37 °C. The CDNB-glutathione produced absorbs light at 340 nm, and the activity of GST was thus determined by measuring absorbance at this wavelength. The GST units (U/mg protein) were defined as nmol GSH used/min/mg protein.
Catalase activity was assayed according to the method of Clairborne26 and was expressed as ^mol hydrogen peroxide (H2O2) consumed/min/ mg protein. This method is based on the rate of H2O2 degradation by the action of catalase contained in the examined samples, which is observed spectrophotometrically at 230 nm. In this procedure, 1 M Tris-HCl and 5 mM ethylinediamine tetraacetic acid at a pH of 8 were used as a buffer and 10 mM H2O2 was used as a substrate.
Glutathione peroxidase (GPx) activity was measured according to the method of Flohe and Gunzler27. The GPx activity (U/mg protein) was expressed as nmol of GSH oxidised/min/mg protein. DT-diaphorase activity was determined using the method of Ernster28. The reduction of 2,6 dCpIP was observed at 600 nm with NADH as the electron donor. Total SOD was assayed using the method of Marklund and Marklund29. Pyrogallol was used as a substrate and SOD activity was expressed as units/mg protein.
Lipid peroxidation assay
The concentration of malondialdehyde (MDA) in tissues, an index of lipid peroxidation, was determined spectrophotometrically according to the method of Draper and Hadley30. A 0.5-mL aliquot of kidney extract supernatant was mixed with 1 mL of trichloroacetic acid solution and centrifuged at 2500 g for 10 min. Thereafter, 1 mL of a solution containing 0.67% thiobarbituric acid (TBA) and 0.5 mL supernatant was incubated for 15 min at 100 °C and then cooled. Absorbance of the TBA-MDA complex was determined spectrophotometrically at 532 nm excitation and 555 nm emission. Lipid peroxidation was expressed as nmol of TBARS, using 1,1,3,3-tetra-ethoxypropane as a standard.
Esterase activity
General esterase activity was measured according to the method of Mackness and Walker31 using para-nitrophenyl acetate and α-naphthyl as substrates. The hydrolysis of α-naphthyl acetate releases α-naphthol and acetate. The α-naphthol reacts with Fast Blue salt to produce a colour which is measured spectrophotometrically at 605 nm. The hydrolysis of para-nitrophenyl acetate releases p-nitrophenol and acetate. The rate of appearance of p-nitrophenol is measured at 400 nm and is proportional to the amount of enzyme present. Arylesterase activity was measured using the end point method of Lorentz et al.32 using phenyl acetate as a substrate. The principle in this method is that phenol generation by the action of the enzyme produces a stable indophenol dye with 4-aminoantipyrine and potassium ferricyanide. The enzyme assay is based on the formation of 4-(p-benzoquinone monoimino)-phenazone which absorbs maximally between 492 nm and 510 nm. Esterase activities were initially expressed as Aabsorbance units/min and normalised by tissue total protein content.
Protein
Protein content was determined by the method of Lowry et al.33 using bovine serum albumin as a reference.
Statistical analysis
Statistical analysis was performed using GraphPad Prism version 4.01. All data were expressed as mean±SD. The paired Student's t-test was used to evaluate the statistically significant differences between control and experimental groups. Values of p<0.05 were considered significant. All experiments were repeated at least three times.
Results
Evaluation of body and absolute organ weights
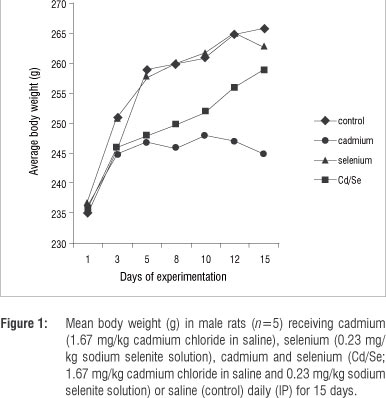
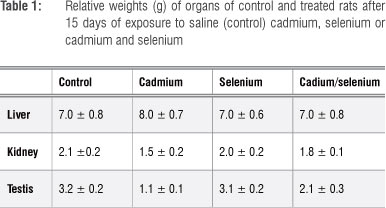
No deaths occurred during the experimental period. The results presented in Figure 1 indicate that the average body weights of the Se-treated group were similar to those of the control rats, while the body weights of the Cd-treated and the Cd/Se-treated groups were significantly lower than those of the control rats from day 3 of the experimental period (p<0.05). Organ weights shown in Table 1 indicate that the liver weights of treated groups were similar to those of control rats. Kidney and testis weights of the Cd-treated group were significantly lower than those of the control group (p<0.05) and a slight decrease was observed in the kidney and testis weights in the Cd/Se-treated group.
Antioxidant enzyme activities
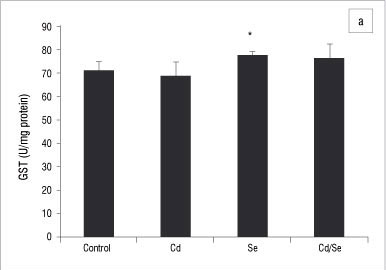
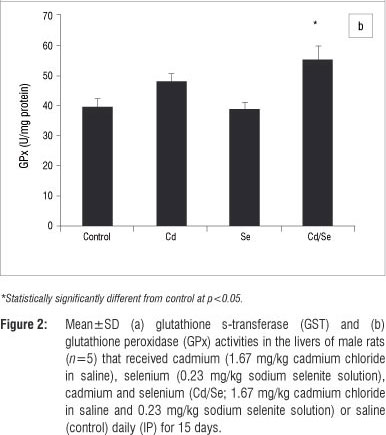
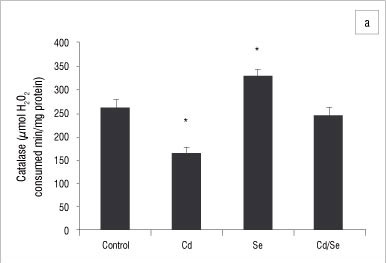
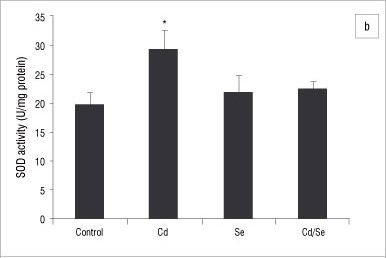
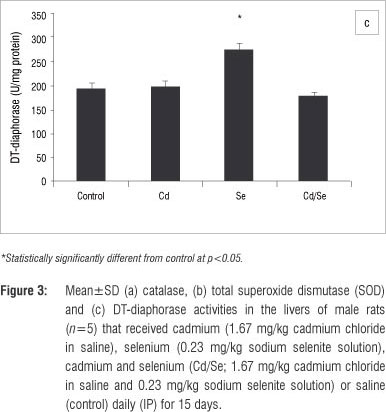
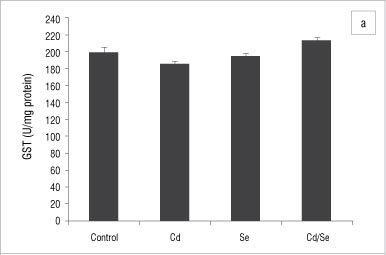
In the liver, GST activity was significantly increased only in the Se-treated group compared with the control group (p<0.05; Figure 2a). GPx activity was significantly increased in the Cd/Se-treated group compared with the control group (p<0.05; Figure 2b). Catalase activity decreased significantly in the Cd-treated group while supplementation of Se returned catalase activity to the levels of the control group (p<0.05; Figure 3a). Se on its own induced a significant increase in catalase activity (p<0.05). SOD activity was significantly higher in Cd-treated rats than in control rats (p<0.05) but was restored to normal levels in rats treated with Se (Figure 3b). DT-diaphorase activity was significantly increased only in the Se-treated rats (p<0.05; Figure 3c).
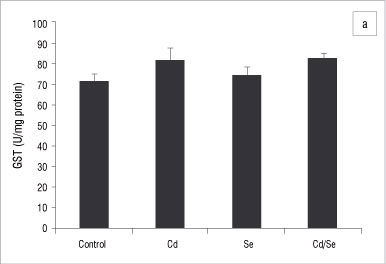
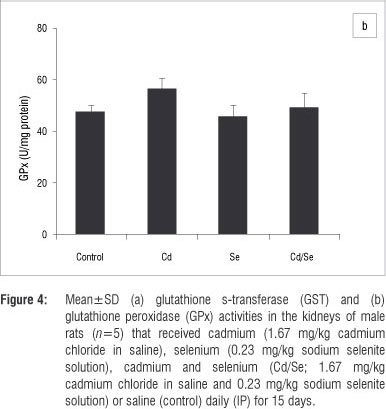
In the kidneys, no major changes occurred in GST and GPx activities in treated groups compared with the control group (Figure 4a and 4b). Catalase activity decreased significantly in the Cd-treated group compared with the controls while Se supplementation increased catalase activity significantly (p<0.05; Figure 5a). Treatment with Se only, also induced catalase activity. SOD activity was significantly lower in Cd-treated rats compared with controls whilst Se-treated rats had a higher SOD activity compared with controls (p<0.05; Figure 5b). The co-administration of Cd and Se resulted in normal levels of SOD activity. DT-diaphorase activity was significantly reduced in Cd-treated rats compared with controls (p<0.05), whilst Se-treated and Cd/Se-treated rats had normal levels of DT-diaphorase (Figure 5c).
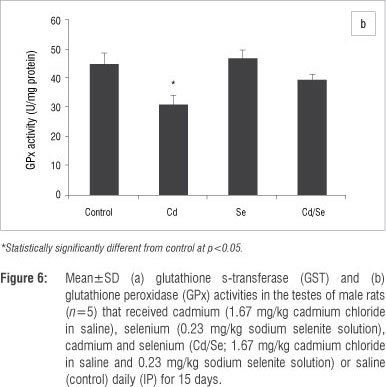
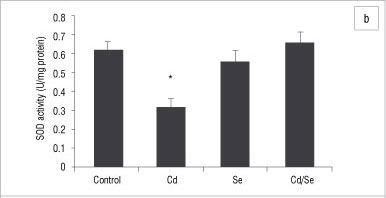
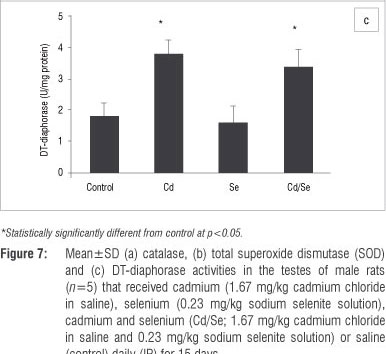
In the testes, the GST activity of treated groups was similar to those of control rats, while GPx activity was significantly reduced in the Cd-treated rats (p<0.05; Figure 6a and 6b). The Se-treated and Cd/Se-treated groups had similar levels of GPx activities as those of the control group. Catalase activity increased significantly in the Cd-treated group compared with the control group (p<0.05) whilst the Se-treated group had similar catalase activities to the controls. The Cd/Se-treated group had increased catalase activity compared with the control group (p< 0.05; Figure 7a). SOD activity was significantly lower in the Cd-treated rats compared with control rats (p<0.05) whilst both Se-treated and Cd/Se-treated rats had similar SOD activities to those of the control group (Figure 7b). DT-diaphorase activity was significantly increased in Cd-treated rats compared with the control group (p<0.05). In the Se-treated group, DT-diaphorase activity was similar to the control rats whilst in the Cd/Se-treated group it was significantly increased (Figure 7c).
SOD/GPx activity
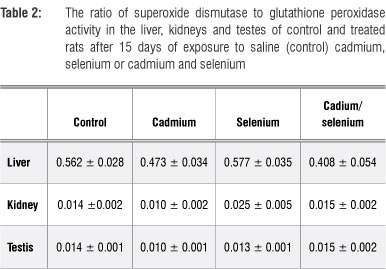
In the liver, the ratio of SOD to GPx activity was significantly lower in Cd-treated and Cd/Se-treated groups than in the control group (p<0.05; Table 2). In the Se-treated group, the ratio was similar to the control group. In the kidney, the ratio of SOD to GPx activity was significantly higher in the Cd-treated group than in the controls (p<0.05) and with Se supplementation, the ratio recovered to levels comparable with those of the control group. In the testis, the ratio of SOD to GPx activity was significantly higher in the Cd-treated group than in the controls (p<0.05); when Se was supplied there was a recovery to levels comparable with those of the control group.
Esterase activities
In the liver, esterase activities were significantly lower in the Cd-treated group compared with the control group (p<0.05). Se on its own induced an increase in esterases activities (p<0.05; Table 3). The co-administration of Cd and Se resulted in significantly higher esterase activities compared with the control group (p<0.05). In the kidneys, there was a significant increase in esterase activities after treatment with Se, for both Se-treated and Cd/Se-treated groups (p<0.05; Table 3). The Cd-treated group had similar esterase activities to the control group. In the testis, there was a general increase in esterase activities after treatment with Se (p<0.05; Table 3). There was a decrease in para-nitrophenyl acetate activity in the Cd-treated group and Se supplementation resulted in similar activities to those of the control group. Both α-naphthyl acetate and phenyl acetate activities were increased significantly after treatment with Se, for both Se-treated and Cd/Se-treated groups (p<0.05).
MDA levels
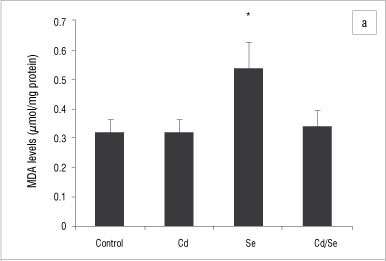
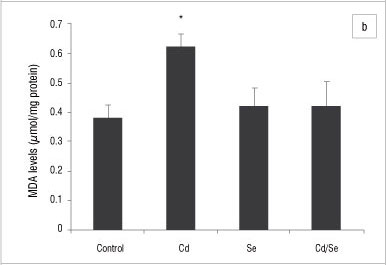
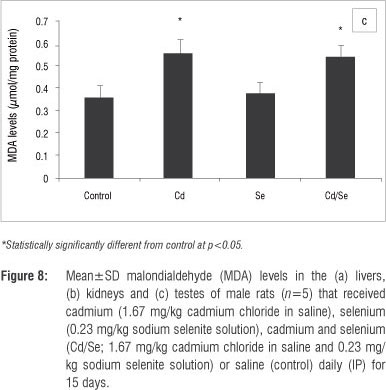
MDA levels indicate the intoxication and generation of oxidative stress in organs and were determined in the liver, kidney and testis homogenates. In the liver, the MDA levels in the Cd-treated and Cd/Se-treated groups were similar to those of the control group (Figure 8a). There was a significant increase in MDA levels in the Se-treated group compared with the control group (p<0.05). In the kidney, the Cd-treated group had increased MDA levels compared with the control group (p<0.05) whilst the Se-treated and Cd/Se-treated groups had similar MDA levels to those of the control group (Figure 8b). In the testis, there was a significant increase in the MDA levels in the Cd-treated and Cd/Se-treated groups compared with the control group (p<0.05), whilst the Se-treated group had MDA levels similar to those of the control group (Figure 8c).
Discussion
Cadmium is a potent toxic metal that is very harmful to the environment as well as to humans because it can accumulate in tissues, causing metabolic, histological and pathological changes. The objective of the current study was to concurrently investigate the protective effect of Se on Cd-induced oxidative stress in three organs - liver, kidney and testis. To this end, the activities of antioxidant enzymes such as catalase, GST GPx, DT-diaphorase and the SOD were determined in these organs. The SOD/GPx activity ratio was also determined. The ratio of SOD to GPx is an important indicator of the balance between the first and second step of the enzymatic antioxidant pathway. MDA concentrations, which are a good indicator of lipid peroxidation, were also determined.
Previous studies have shown that Cd induces oxidative stress through ROS and that antioxidant enzyme activities are altered in the presence of Cd.34 These antioxidant enzymes include catalase, SOD and GPx - enzymes that are important in the elimination of free radicals. Cd is also known to generate lipid peroxidation and thus cause damage to cell membranes. In our study, exposure to Cd led to a decrease in catalase activity and an increase in total SOD activity in the liver. There was no change in MDA concentration. The decrease in catalase activity in the livers of rats exposed to Cd might be a result of metal deficiency. It is known that Cd decreases the levels of iron (Fe) in the liver35 and because Fe is a component of the active site of catalase, a decrease in Fe might result in a decrease in catalase activity. A Cd-induced increase in lipid peroxidation could be why SOD activity increased, as SOD would have to remove excess ROS. Exposure to Cd led to a significant decrease in the SOD to GPx activity ratio in the liver.
Kidneys are vulnerable to damage as a result of perfusion and the increased concentrations of excreted compounds that occur in renal tubular cells. Exposure to Cd led to a distinct decrease in the activities of total SOD and catalase and an increase in MDA levels. SOD activity is known to be inhibited by Cd as Cd can replace Zn on the SOD molecule therefore resulting in an inactive enzyme.36 The decrease in catalase activity could be a result of Fe deficiency. At the same time, Cd-induced reduction in hepatic and renal catalase activities may reflect a reduced capacity to eliminate H2O2 in response to Cd in the mitochondria and microsomes of both organs. Exposure to Cd led to a significant increase in the SOD to GPx activity ratio in the kidneys. An increase in this ratio has been suggested to reflect a high production of radicals such as H2O2. These results demonstrate that Cd accumulates in the kidneys of rats, and this accumulation results in marked alterations in enzymatic activities. Indeed, it has been shown that in rats the kidney is the organ that accumulates the highest levels of Cd.37
In the testis, exposure to Cd led to decreased GPx and total SOD activities and resulted in increased MDA levels. Se is a component of GPx and a reduction in GPx activity in the Cd/Se-treated group might be as a result of depletion of Se.38 Increases in catalase and DT-diaphorase activities could be induced by the high levels of radicals present. In agreement with this hypothesis is the increase in MDA levels, an indicator of lipid peroxidation. Lipid peroxidation is the product of oxidative damage and plays an important role in the toxicity of Cd. These results confirm that Cd causes a significant increase in lipid peroxidation in the testes of rats. As in the kidney, the ratio of total SOD to GPx increased in the presence of Cd. Se is an essential element and is a well-established antioxidant.39 It is also well documented that Se can decrease the effects of Cd on the antioxidant system in different tissues. This conclusion was, however, made based on studies that examined the effects of Cd on different organs individually. To our knowledge, our study is the first to examine the protective effect of Se on Cd-induced stress on antioxidant enzymes in these three organs (liver, kidney and testis) simultaneously. It is not surprising therefore that some of the results obtained in this study differ from those published, as we took a whole-animal approach in determining the effects of these two metals. In this study, Se treatment resulted in an increase in MDA concentration in the liver. Co-administration of Se and Cd resulted in similar MDA levels to those of the control group in both the liver and the kidneys, with an increase in MDA levels in the testes. These results imply that Cd has a protective effect or that the concentration of Se used could be harmful to cells in the liver. In the kidney, however, Se- had a protective effect on Cd-induced toxicity. This result is in contrast with results reported by others.40 It is highly improbable that the dose of Se used in this study is toxic to the liver. Similar or even higher doses of Se have been used before and were found to be protective against Cd- and chromium-induced nephrotoxicity and hepatotoxicity.41 On the other hand, in the testis, Cd induced an increase in MDA levels and Se on its own resulted in MDA concentrations similar to controls. The observed decrease of MDA levels in the kidney and testis in the presence of Se is supported by other investigators.40 This finding illustrates the importance of Se in preventing lipid peroxidation and in the protection of the integrity and functioning of tissues.40 It has, however, been found that Se sometimes does not provide the expected hepatic and renal protection from Cd.40
In fact, it has been shown before that Se can be harmful at certain concentrations and to certain organs whilst at the same time being protective to others. Se was reported to provide protection to the liver but not the reproductive organs in an atrazine model of experimental toxicity.42 Co-administration of Se and Cd resulted in an increase in GPx activity with MDA levels in the liver remaining at the levels of control animals. This response might be an adaptation to free radical damage to the liver. In the kidney, co-administration of Se and Cd resulted in increases in GST and catalase activities. These results might explain an induction of these enzymes in response to an increase in lipid peroxidation. In the testis, the co-administration of Se and Cd resulted in increases in catalase and DT-diaphorase activities and in MDA levels. Again, this induction of catalase and DT-diaphorase activities may be in response to an increase in free radical levels. Se concentration has been found to be higher in the testis than in the liver and other organs.41
Lastly, Cd administration caused significant decreases in liver esterase activity for the three substrates used. Esterases are enzymes that principally catalyse the hydrolysis of esters and they are widely distributed in the animal kingdom. The classification of esterases is not absolute as there is overlapping substrate specificities.43 The trend in the liver antioxidant activities, including the esterases, is not very clear, with some enzyme activities increased whilst others decreased. Thus a person simultaneously exposed to Cd and organophosphates will not be able to metabolise the organophosphates with the same efficiency as in an individual not exposed to Cd. Se administration appears to reverse the effect of Cd on liver, kidney and testis esterase activities. Based on these results of esterase activity, Se appears to offer protection from Cd-induced toxicity in the kidney and testis. Thus, whether Se has a protective effect on Cd-induced oxidative stress or not, depends on the organ. According to our results, the concentration of Se used does not appear to protect the liver against Cd-induced oxidative stress, but does alleviate the oxidative stress induced by Cd in the kidney and the testis.
Conclusion
We have demonstrated that Cd-induces oxidative stress and depletes antioxidant enzymes in rat kidneys and testes. We have also shown that Se appears to have protective effects against Cd-induced oxidative damage in the kidneys and the testes of rats. However, the protective effect of Se on Cd-induced oxidative stress in other organs like the liver, although possible, is not completely clear and deserves further investigation.
Acknowledgements
We are indebted to Mr Lazarus Chirombe for technical assistance and Dr Nomsa Ndiweni for assistance with the glutathione peroxidase assay. This work was supported by grants from the University of Zimbabwe and the International Programme in Chemical Sciences, Uppsala University, Sweden.
Authors' contributions
K.D. performed all the experiments and calculations and prepared all the samples. Y.S.N. was the project leader. Both authors were responsible for the experimental and project design and wrote the manuscript.
References
1. Nordberg M, Winblad B, Basun H. Cadmium concentration in blood in an elderly urban population. Biometals. 2000;13(4):311-317. http://dx.doi.org/10.1023/A:1009268123320
2. Friberg L, Elinder CG, Kjellstrõm T, editors. Cadmium: Environmental health criteria (EHC) 134. Geneva: World Health Organization; 1992. p. 1-25. [ Links ]
3. Stohs SJ, Bagchi D. Oxidative mechanisms in the toxicity of metal ions. Free Radical Biol Med. 1995;18(2):321-336. http://dx.doi.org/10.1016/0891-5849(94)00159-H [ Links ]
4. Thijssen S, Maringwa J, Faes C, Lambrichts I, Van Kerkhove E. Chronic exposure of mice to environmentally relevant, low cadmium leads to early renal damage, not predicted by blood or urine cadmium levels. Toxicology. 2007;229(1-2):145-156. http://dx.doi.org/10.1016/j.tox.2006.10.011 [ Links ]
5. Alvarez SM, Gómez NN, Scardapane L, Fornés MW Giménez MS. Effects of chronic exposure to cadmium on prostate lipids and morphology. Biometals. 2007;20(5):727-741. http://dx.doi.org/10.1007/s10534-006-9036-9 [ Links ]
6. Thompson J, Bannigan J. Cadmium: Toxic effects on the reproductive system and the embryo. Reprod Toxicol. 2008;25(3):304-315. http://dx.doi.org/10.1016/j.reprotox.2008.02.001
7. Sugiyama M. Role of physiological antioxidants in chromium VI-induced cellular injury. Free Radical Biol Med. 1992;12(5):397-407. http://dx.doi.org/10.1016/0891-5849(92)90089-Y
8. IARC. International agency for research on cancer monographs on the evaluation of carcinogenic risks to humans vol. 58: Beryllium, cadmium, mercury and exposures in the glass manufacturing industry. Scand J Work Environ Health. 1993;19(5):360-363. http://dx.doi.org/10.5271/sjweh.1461 [ Links ]
9. Droge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002;82(1):47-95.
10. Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. 4th ed. New York: Oxford University Press; 2007. [ Links ]
11. Matés M. Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology. 2000;153(1-3):83-104. http://dx.doi.org/10.1016/S0300-483X(00)00306-1
12. Griffith OW. Biological and pharmacologic regulation of mammalian glutathione synthesis. Free Radical Biol Med. 1999;27(9-10):922-935. http://dx.doi.org/10.1016/S0891-5849(99)00176-8
13. El-Sharaky AS, Newairy AA, Badreldeen MM, Eweda SM, Sheweita SA. Protective role of selenium against renal toxicity induced by cadmium in rats. Toxicology. 2007;235(3):185-193. http://dx.doi.org/10.1016/j.tox.2007.03.014 [ Links ]
14. Perottoni J, Rodrigues OE, Paixao MW, Zeni G, Lobato LP Braga AL, et al. Renal and hepatic ALA-D activity and selected oxidative stress parameters of rats exposed to inorganic mercury and organoselenium compounds. Food Chem Toxicol. 2004;42(1):17-28. http://dx.doi.org/10.1016/j.fct.2003.08.002 [ Links ]
15. Shamberger RJ. Biochemistry of selenium. 2nd ed. New York: Plenum Press; 1983. http://dx.doi.org/10.1007/978-1-4684-4313-4 [ Links ]
16. Chen C, Hedstrom O, Whanger PD. Effect of vitamin B12 on performance and tissue selenium content in rats fed sub-toxic levels of selenite. Toxicology. 1993;85(2-3):101-115. http://dx.doi.org/10.1016/0300-483X(93)90035-Q
17. Kopp SJ, Perry M, Perry EF, Erlanger M. Cardiac physiologic and tissue metabolic changes following chronic low-level cadmium and cadmium plus lead ingestion in the rat. Toxicol Appl Pharmacol. 1983;69(1):149-160. http://dx.doi.org/10.1016/0041-008X(83)90131-X [ Links ]
18. Stohs SJ, Bagchi D, Hassoun E, Bagchi M. Oxidative mechanisms in the toxicity of chromium and cadmium ions. J Environ Pathol Toxicol Oncol. 2001;20(2):77-88. http://dx.doi.org/10.1615/JEnvironPatholToxicolOncol.v20.i2.10 [ Links ]
19. Waisberg M, Joseph P, Hale B, Beyersmann D. Molecular and cellular mechanisms of cadmium carcinogenesis: A review. Toxicology. 2003;192(2-3):95-117. http://dx.doi.org/10.1016/S0300-483X(03)00305-6 [ Links ]
20. Koyu A, Gokcimen A, Ozguner F, Bayram DS, Kocak A. Evaluation of the effects of cadmium on rat liver. Mol Cell Biochem. 2006;284(1-2):81-85. http://dx.doi.org/10.1007/s11010-005-9017-2 [ Links ]
21. Santos FW, Graca DL, Zeni G, Rocha JBT, Weis SN, Favero AM, et al. Sub-chronic administration of diphenyl diselenide potentiates cadmium-induced testicular damage in mice. Reprod Toxicol. 2006;22(3):546-550. http://dx.doi.org/10.1016/j.reprotox.2005.12.009 [ Links ]
22. Fotakis G, Timbrell JA. Role of trace elements in cadmium chloride uptake in hepatoma cell lines. Toxicol Lett. 2006;164(2):97-103. http://dx.doi.org/10.1016/j.toxlet.2005.11.016
23. Shukla GS, Hussain T, Srivastava RS, Chandra SV. Glutathione peroxidase and catalase in liver, kidney, testis and brain regions of rats following cadmium exposure and subsequent withdrawal. Ind Health. 1989;27(2):59-69. http://dx.doi.org/10.2486/indhealth.27.59
24. Sarkar S, Yadav P Trivedi R, Bansal AK, Bhatnagar D. Cadmium-induced lipid peroxidation and the status of the antioxidant system in rat tissues. J Trace Elem Med Biol. 1995;9(3):144-149. http://dx.doi.org/10.1016/S0946-672X(11)80038-6 [ Links ]
25. Habig WA, Pabist MJ, Jacoby WB. GST, the first step of mercapturic acid formation. J Chem. 1974;249(22):7130-7139.
26. Clairborne A. Catalase activity. CRC handbook of methods for radical research. Boca Raton, FL: CRC Press; 1985. p. 283-284. [ Links ]
27. Flohe L, Gunzler WA. Assays of glutathione peroxidise. Methods Enzymol. 1984;105:114-121. http://dx.doi.org/10.1016/S0076-6879(84)05015-1
28. Ernster L. DT-diaphorase. Methods Enzymol. 1967;10:309-317. http://dx.doi.org/10.1016/0076-6879(67)10059-1
29. Marklund S, Marklund G. Involvement of the superoxide anion radical in the auto-oxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974;47(3):469-474. http://dx.doi.org/10.1111/j.1432-1033.1974.tb03714.x
30. Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Ezymol. 1990;86:421-431. http://dx.doi.org/10.1016/0076-6879(90)86135-I
31. Mackness MI, Walker CH. Multiple forms of sheep serum A-esterase activity associated with the high-density lipoprotein. Biochem J. 1988;250(2):539-545.
32. Lorentz JK, Flatter B, Augustin E. Arylesterase in serum. Clin Chem. 1979;25(10):1714-1720.
33. Lowry OH, Rosebrough NJ, Farr AL, Randal RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265-267.
34. Liu J, Qian SY Guo Q, Jiang J, Waalkes MP Mason RP Kadiiska MB. Cadmium generates reactive oxygen- and carbon-centered radical species in rats: Insights from in vivo spin-trapping studies. Free Radical Biol Med. 2008;45(4):475-481. http://dx.doi.org/10.1016/j.freeradbiomed.2008.04.041 [ Links ]
35. Jurczuk M, Brzóska MM, Moniuszko-Jakoniuk J, Gãazyn-Sidorczuk M, Kulikowska-Karpinska E. Antioxidant enzymes activity and lipid peroxidation in liver and kidney of rats exposed to cadmium and ethanol. Food Chem Toxicol. 2004;42(3):429-438. http://dx.doi.org/10.1016/j.fct.2003.10.005 [ Links ]
36. Bauer R, Demeter I, Hasemann V, Johansen JT. Structural properties of the zinc site in Cu, Zn-superoxide dismutase: Perturbed angular correlation of gamma ray spectroscopy on the Cu, 111Cd-superoxide dismutase derivative. Biochem Biophys Res Commun. 1980;94(4):1296-1302. http://dx.doi.org/10.1016/0006-291X(80)90560-4
37. Zabulyte D, Paltanaviciene A, Uleckiene S, Kalibatas J, Juozulyna A, Jascaniniene N, et al. Cadmium accumulation in the kidney and its excretion with urine, and possible influence of PAH's on these processes in rats. Bull Vet Inst Pulawy. 2007;51:257-260.
38. Lazarus M, Orct T, Blanusa M, Kostial K, Pirsljin J, Beer-Ljubic B. Effect of selenium pre-treatment on antioxidative enzymes and lipid peroxidation in Cd-exposed suckling rats. Biol Trace Elem Res. 2011;142(3):611-622. http://dx.doi.org/10.1007/s12011-010-8775-1
39. Klotz LO, Krõncke KD, Buchczyk DP Sies H. Role of copper, zinc, selenium and tellurium in the cellular defense against oxidative and nitrosative stress. J Nutr. 2003;133(5 suppl 1):1448S-1451S.
40. Ognjanovic BI, Markovic SD, Pavlovic SZ, Zikic RV Stajn AS, Saicic ZS. Effect of chronic cadmium exposure on antioxidant defense system in some tissues of rats: Protective effect of selenium. Physiol Res. 2008;57(3):403-411. [ Links ]
41. Brandao R, Santos FW, Oliveira R, Roman SS, Nogueira CW. Involvement of non-enzymatic antioxidant defenses in the protective effect of diphenyl diselenide on testicular damage induced by cadmium in mice. J Trace Elem Med Biol. 2009;23(4):324-333. http://dx.doi.org/10.1016/j.jtemb.2009.06.006
42. Adesiyan AC, Oyejola TTO, Abarikwu SO, Oyeyemi MO, Farombi EO. Selenium provides protection to the liver but not the reproductive organs in an atrazine-model of experimental toxicity. Exp Toxicol Pathol. 2011;63(3):201-207. http://dx.doi.org/10.1016/j.etp.2009.11.008
43. Juhl M, Holmstrup P. Epithelial-naphthyl acetate esterases in the green vervet monkey gingiva before and after periodontal surgery and during tooth eruption. Scand J Dental Res. 1993;101(2):92-97.
 Correspondence:
Correspondence:
Kevin Dzobo
International Centre for Genetic Engineering & Biotechnology and Division of Medical Biochemistry
Faculty of Health Sciences, University of Cape Town, Wernher and Beit Building (South)
UCT Medical Campus, Anzio Road, Observatory 7925, South Africa
Email: kdzobosnr@yahoo.com
Received: 13 Oct. 2011
Revised: 15 Oct. 2012
Accepted: 16 Oct. 2012