Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.108 n.9-10 Pretoria Jan. 2012
REVIEW ARTICLE
Global challenges in the risk assessment of nanomaterials: relevance to South Africa
Mary GulumianI, II; Eileen D. KuempelIII; Kai SavolainenIV
INational Institute for Occupational Health, Johannesburg, South Africa
IIHaematology and Molecular Medicine, University of the Witwatersrand, Johannesburg, South Africa
IIINational Institute of Occupational Safety and Health, Cincinnati, OH, USA
IVNanosafety Research Centre, Finnish Institute of Occupational Health, Helsinki, Finland
ABSTRACT
Internationally, there are efforts to develop standardised toxicity testing and risk assessment methods for engineered nanomaterials (ENMs). To this end, health risk assessments need to be conducted on ENMs synthesised in South Africa. Country-specific risk characterisation requires specific exposure assessments for those ENMs for which the likelihood exists for occupational and environmental exposure in that country. A challenge in hazard identification and risk assessment related to ENMs, regardless of country of origin, is that data on toxicity, carcinogenicity, pharmacokinetics, and occupational or environmental exposure are generally not available for most ENMs. Although the mechanisms previously identified as important in the toxicity and carcinogenicity of particles and fibres may be applicable, the possibility exists that the unusual physicochemical properties of ENMs may give rise to unique, and as yet unidentified, adverse effects. Moreover, generalised exposure scenarios that consider the life cycle of the agent have not been developed and are needed for the complete risk characterisation of ENMs. As health risk assessment is both resource and labour intensive, it is imperative to identify the aims of such an exercise prior to embarking on large-scale projects, to ensure that the data most useful for public health decision-making is provided. Identifying priorities in South Africa, in coordination with international efforts, can facilitate the effective use of research efforts for risk assessment and risk management decision-making.
Background
Health risk assessments are required for new technologies and for new classes of materials introduced to the market for human use. Amongst the most extensive regulation in this area is the European Union Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) programme.1 Risk assessment strategies based on categories of nanomaterials have been proposed, with consideration given to their physicochemical properties and modes of action.2,3 These categories are: carbon-based (fullerenes and carbon nanotubes), metal-based (quantum dots, nanogold, nanosilver and metal oxides), dendrimers and composites.4,5
It is anticipated that workers and researchers handling these diverse engineered nanomaterials (ENMs), as well as the general public that use these products, will to some degree be exposed to, and therefore potentially at risk from, their adverse effects.6 Agencies and organisations in several countries have issued guidelines on good work and consumer practices for the handling and use of ENMs,2,7,8 and have formulated approaches for the evaluation of ENMs under existing health and safety regulations.5
A global effort to develop standardised testing procedures for risk assessment is led by the Organization for Economic Cooperation and Development (OECD). A list of priority ENMs has been developed and country sponsors have been identified9 (Table 1). South Africa is the sponsor for the most recent addition to this list, gold nanoparticles (AuNP); the USA is a co-sponsor, and the European Commission (EC) and Korea are contributors. The OECD standardised testing procedures include information on physicochemical characterisation, biotic systems, degradation and accumulation, and health effects.10 Phase I testing (introduced in 2007) includes the selection of the ENMs and the end points to be studied (Table 2). Phase II involves the evaluation of critical issues identified in Phase I or the performance of additional tests, including long-term tests and risk assessment. Data sharing of the tested ENMs will increase the efficiency of generating data needed for hazard and risk assessment.10
The Department of Science and Technology (DST) in South Africa has also emphasised the need for risk assessment of ENMs presently synthesised in the country.11 Without reliable data on effects and exposure, in-depth risk assessments of ENMs in South Africa and other countries may be limited. It is therefore essential to coordinate international efforts and develop standardised methods for in-vitro and in-vivo hazard identification to provide data for the risk assessment of representative
ENMs within major categories. The purposes of this review are to, (1) provide an overview of risk assessment history and practice, (2) describe coordinated international research efforts in which South Africa participates and (3) discuss challenges in health risk assessment and areas of prioritisation for South Africa.
Overview of health risk assessment of nanomaterials
The objective of human health risk assessment is to provide scientific information based on toxicology or epidemiology data (if available) to predict or estimate risk associated with exposure to potentially hazardous substances. Traditionally, health risk assessment involves four steps (proposed in 1983 by the US National Research Council)12: (1) hazard identification, (2) dose-response assessment, (3) exposure assessment and (4) risk characterisation (Figure 1). Throughout each step of this process, uncertainties involved in making estimates also need to be considered.4,13
Examples of the use of risk assessment for risk management decision-making include regulatory measures for establishing standards for water and air quality or requirements for environmental clean-up. Through the identification of potential sources and pathways of exposure for a population, and the determination of the health hazards associated with exposure, risk characterisation describes the potential risk of a substance, from those that present little or no concern, to those that are likely to cause adverse effects. Risk assessment supports evidence-based risk management decisions and facilitates societal evaluation of acceptable risk.14
Basically, it is anticipated that the risk assessment of ENMs will follow the traditional risk assessment paradigm used for conventional chemicals.4,12 Nanoparticle-specific data are also needed in applying the risk assessment process and to determine if there are any novel properties of nanomaterials that may result in unique toxicity. New strategies and methods may be needed to identify and consider any factors that may be specific to certain ENMs.15,16
Hazard identification
The first step in risk assessment is to evaluate the available hazard data, including that of in-vitro or in-vivo toxicity. The purpose of hazard identification is to identify the contaminants that may pose health hazards and to identify the conditions under which they could be toxic to humans.12
The potential hazards of a great number of ENMs have as yet not been identified. Numerous past investigations of inhaled particles and fibres have identified properties that determine toxicity as well as the mechanisms involved in this toxicity.
For example, sonre physicochemical properties identified are chemical composition, particle and fibre geometry and dimesions, biopersistence, surface activity and dose (as partice surface area, number, mass or volume). Mechanisms elucidated in particle and fibre toxicity include oxidative stress and persistent inflammation.6 It is therefore envisaged that similar mechanisms may be applicable to the potential toxicity of ENMs.
With the help of expert groups, a number of international agencies have proposed specific in-vitro and in-vivo tests to assess the toxicity of ENMs.10 In-vitro assays are considered useful for screening and prioritising substances for in-vivo studies. Such assays may also be useful in the elucidation of the mechanistic pathways involved in the toxicity of nanomaterials, which may need further validation with in-vivo studies. Some recent studies have shown progress in correlating acute in-vivo and in-vitro responses in hazard identification,6 which suggest that in-vitro assays could be used to reduce animal testing in the initial hazard evaluation of ENMs.
Internal dose estimation is important in the prediction of the biological effect at the target tissue (based on absorption, distribution, metabolism, and elimination processes). Although data still are limited, the role of physicochemical properties on the systemic distribution of some ENMs, including AuNP, has been studied. In-vivo studies have shown that the primary site of gold (and silver) accumulation is the liver,17 and that this process of accumulation is influenced by particle size, surface charge and route of exposure.18 For example, in a rat intratracheal instillation study of two sizes of AuNP, the pulmonary and systemic toxicity responses were similar for both particle sizes at the dose tested, and there was no clear relationship between toxicity and particle size or agglomeration state.19 A subchronic inhalation study of exposure to AuNP in rats showed no adverse effects in rats exposed to the two lower doses, but rats exposed to the highest dose showed reduced lung function and inflammatory changes in their lung tissue.20 Further studies are needed to assess the hazards of AuNP, although in-vivo and in-vitro studies suggest that particle size and charge can influence the fate of gold nanoparticles in the body. For other materials, particle shape also influences the ability of inhaled particles to translocate from the lungs and be retained in the pleural tissue. Such kinetic information can be used to identify the nanomaterial characteristics with the greatest potential to be hazardous and to design nanomaterials with safer properties (e.g. lower biopersistence).21
Dose-response assessment
The next step of the risk assessment process is the dose-response assessment, in which the quantitative relationship between the dose and a toxic response is determined. This assessment is typically performed using animal models, from which an equivalent human dose (often for a specific population, such as workers or the general public) is estimated.12
The aim of this step is to identify the doses at which certain toxicity end points (i.e. effect levels) occur. These end points can include acute and chronic toxicity, immunotoxicity, neurotoxicity, mutagenicity, reproductive or developmental toxicity, and carcinogenicity. Another aim of this step is to identify the effect level, such as the 'no observed adverse effect level' (NOAEL) or the 'lowest observed adverse effect level' (LOAEL). Non-cancer risk assessment using a NOAEL or LOAEL approach assumes a threshold mechanism such that exposures below the threshold are not expected to cause harmful effects. The most sensitive end point is typically selected and extrapolated to humans by adjusting for differences across species (e.g. body weight and metabolic rate). The human-equivalent effect level is then divided by a series of uncertainty factors to determine a reasonably safe exposure level. Uncertainty factors are typically those that account for a less than or equal to 10-fold difference caused by species differences, animal vs. human response, human inter-individual variability, use of a LOAEL instead of a NOAEL and use of subchronic rather than chronic dose-response data. Examples of regulatory levels derived using this approach include inhalation reference concentrations (RfC), oral reference doses (RfD) and acceptable daily intakes (ADI).22 Alternatively, risk-based effect levels can be estimated from statistical modelling of the dose-response data and low-dose extrapolation from the 95% lower confidence limit estimate (i.e. the benchmark dose limit, BMDL) of the benchmark dose (BMD). The BMDL is used to account for variability in the data and to provide confidence that the true BMD is greater than the BMDL. The US National Research Council risk assessment guidelines recommend a greater emphasis on risk-based approaches to decision-making, such as in the development of exposure limits and reference concentrations.13
There are two major issues that are of concern when establishing the dose-response relationship of ENMs. Firstly, challenges exist in the measurement of nanoparticles at target deposition sites and their biopersistence at those sites. A number of physicochemical properties, including their surface functional groups and dissolution of surface ions, may influence their distribution to these sites.23 Secondly, the establishment of the most biologically relevant dose metric for nanoparticles (mass, volume, number or surface area) poses a further challenge in the establishment of a dose-response relationship for ENMs.24 Particle volume or surface area, rather than mass, have been shown to better describe the overloading of lung clearance and the resulting inflammation response in rats.25,26 A recent study has concluded that no single dosimetric parameter of particles -mass, surface area or number - will be universally applicable to all nanomaterials. Hence, the selection of the best dose metric for toxicity testing and the risk assessment of specific ENMs will remain a challenge.27 Toxicity studies can help to reduce this challenge by providing standard measures of physicochemical properties at toxicity to facilitate hypothesis testing across groups of ENMs.
Exposure assessment
Obviously, there is no risk to health from a hazardous agent if there is no exposure to that agent. Exposure assessment identifies the exposure of a population to a contaminant, and includes the route, magnitude, duration and timing of the exposure.12
Exposure assessment may cover medical administration through intradermal, intraperitoneal and intravenous injections.28 Exposure may also be in the form of inhalation, ingestion and dermal absorption in occupational settings. In addition, exposure through the general environment, the fate and transport of the substance, as well as the points of entry into the environment or through the use of consumer items, should be considered.
Adequate exposure assessment cannot be conducted without sufficient information on the sources of exposure, the amount exposed to and the routes of exposure. Exposure via inhalation is possible for airborne nanomaterials; exposure via the gastrointestinal tract is possible because of the increasing number of applications of ENMs in food packaging and food products. Workers are at risk from dermal exposure to ENMs during the manufacture of ENMs. Workers and the general public are also at risk from dermal exposure to ENMs through the manufacture and use of textiles and cosmetics such as sunscreens. Because of lack of data on ENMs in the aquatic or terrestrial environment,29 it is even more challenging to estimate exposure of the general population or the environment to ENMs. Once again, the unique physicochemical properties of nanomaterials may determine their behaviour in different environments and therefore their frequency or patterns of exposure.
Finally, exposure varies on the basis of conditions such as the manner in which materials are handled in the workplace, the way in which ENMs partition to various phases (e.g. water and air), and the mobility and persistence of ENMs in each of these phases, and the magnitude of the sources (e.g. production volume or size of markets). Exposure scenarios for the identified ENMs should therefore be developed throughout their life cycles to address key questions and gaps in the risk assessments of the identified ENMs. It may be likely that when an ENM is embedded in polymers or in other materials, the potential for exposure is minimal,30 but this potential may change during the processing or recycling of the material, or as it enters a waste stream. In this instance, the life-cycle concept propagated by the Environmental Protection Agency should also be considered for the risk assessment of nanoparticles.31
Risk characterisation
The final step in risk assessment is risk characterisation, in which data obtained during hazard identification, dose-response assessment and exposure assessment are integrated, and the uncertainty in these estimates is evaluated.13
The paucity of data for many ENMs with regard to workplace exposure levels and hazard potential are key sources of uncertainty in the risk characterisation of these ENMs. There is also uncertainty about the potential novel or enhanced effects of certain ENMs as a result of their small size and their ability to interact with cells and cell organelles via mechanisms related to nanoscale properties. Additional uncertainties include the influence of the route of exposure (e.g. inhalation via air, ingestion via food or water or intravenous via medical treatment) on internal dose and toxicity; the role of particle size (e.g. agglomerated versus dispersed); the mechanisms involved in the uptake and fate of ENMs in the body; and the persistence or degradation of ENMs in the environment. A full life-cycle risk characterisation would include not only the risk assessment of workers at the site of production, but also a risk assessment along the life cycle of the ENMs, including their transport, use and disposal.31,32
The research efforts to address these questions are by nature multidisciplinary. Best work practice calls for greater exposure control and caution when there is increased uncertainty about potential adverse health effects.33 In the absence of information on specific ENMs, the scientific literature on substances with analogous physicochemical properties could be used to derive initial estimates of the hazard and the target level of exposure control.3
Examples of risk assessments and exposure limits for nanomaterials
The occupational exposure limits (OELs) currently proposed for ENMs are based on animal data, as human health effects data are not available for specific ENMs. The US National Institute of Occupational Safety and Health used quantitative risk assessment methods similar to those used for other airborne, poorly soluble particles34 as the basis for developing draft OELs for ultrafine (of nanometre diameter) and fine (of micrometre diameter) TiO2.35 Epidemiology studies of workers who handle TiO2 have generally shown no increase in the risk for lung cancer. Chronic inhalation studies in rats, however, have shown an increase in the risk for lung tumours; this risk is associated with the particle surface area dose of TiO2 and other poorly soluble particles in the lungs.35,36 Subchronic inhalation studies have shown that pulmonary inflammation is also associated with the particle surface area dose in the lungs of rats and mice. Because ultrafine TiO2 has a greater surface area per unit mass than fine TiO2, a lower mass dose of ultrafine TiO2 than that of fine TiO2 was associated with lung inflammation and tumours. These findings of adverse lung effects being associated with the particle size or surface area provided the health basis for the two OELs proposed for fine and ultrafine (or nano) TiO2: 0.3 mg/m3 for ultrafine TiO2 and 2.4 mg/m3 for fine TiO2 (8-h time-weighted average concentrations).35
Several research groups recently have proposed specific OELs for carbon nanotubes (CNTs): 50 μg/m3,37 30 μg/m3,38 7 μg/m3,39 1-2 μg/m3.40 These proposed OELs are 8-h time-weighted average concentrations and were derived from animal studies in which lung inflammation or fibrotic responses were exhibited after subchronic or shortterm exposure to different types of CNTs. Most of these risk assessments used data from subchronic inhalation studies of multiwalled CNTs in rats, in which 0.1 mg/m3 was the NOAEL in one study38 and a LOAEL in a study of a different type of multiwalled CNT.41 Another risk assessment used data from a 4-week inhalation study in rats, in which the LOAEL was 0.37 mg/m3.37
Although these examples show progress in the development of proposed OELs for ENMs, the number of different types of nanoparticles being developed and used in commercial products is outpacing efforts to develop OELs in the workplace. Reasons for this shortcoming include limited data on both the hazards of and the exposure to ENMs, for example, in workplaces.42 Challenges in the measurement of exposure include the current inability of the available online monitoring technologies to separate the ubiquitous background nanomaterials from the ENMs; limitations in the sensitivity of some analytical methods for detecting and quantifying exposures to nanoparticles in the workplace39; and the lack of consensus on the metrics and methods to be used for exposure measurement.4,16 Thus, new approaches are needed to better characterise potential hazards.
The concept of developing exposure limits for categories of ENMs with similar physicochemical properties - and well-characterised biological effects for representative 'benchmark' particles in each group - may be one approach to setting exposure limits for different types of nanomaterials.3 The British Standards Institute proposed four categories of ENMs and associated 'benchmark exposure levels', which were described as pragmatic guidance levels.2 A four-category approach was also adopted by the Institut für Arbeitsschutz43 in Germany. Standardised toxicity testing procedures and risk assessment methods are also needed for global harmonisation of health and safety practices for ENMs.10,32
Nanomaterials currently synthesised in South Africa
With the realisation of the importance of nanotechnology, and the foresight and leadership of the DST, centres dedicated to the research and development of nanotechnology have been established to research the synthesis and applications of nanomaterials.44 Tertiary institutions (the universities of the Witwatersrand, Cape Town, Tshwane, Johannesburg, Stellenbosch, Western Cape, Zululand, KwaZulu-Natal, Limpopo and Rhodes) and research centres (the Council for Scientific and Industrial Research, MINTEK and iThemba Laboratory for Accelerator Based Sciences) in South Africa are presently actively involved in the synthesis and application of nanomaterials. Nanomaterials that are currently being investigated at these centres range from metal and metal oxide particles to CNTs and nanocomposites, and thus represent some of the major categories of ENMs being developed and used worldwide (Table 3). A few examples of these activities, by no means an exhaustive list, are summarised below as an indication of the diversity of nanomaterials investigated in South Africa.
AuNPs are being investigated for use in catalysis,45 in the purification of air at room temperature46 and in molecular diagnostics.47 Almost every university and research centre is in some way involved with the synthesis or application of CNTs. The application of CNTs in catalysis48 and their use in the removal of hexavalent chromium from industrial wastes49 have been reported. These ENMs are also being studied for their potential to cheaply and efficiently treat water in order to meet drinking, industrial and environmental water quality standards.50
In South Africa, quantum dots and their application in diagnostics, security systems, biological probes and optics are being investigated.51 A great variety of nanocomposites is also being synthesised, including CNTs, polymers, quantum dots and metallic-based nanoparticles such as silicon and TiO2.52 Finally, the preparation of magnetite nanoparticles, using a variety of synthesis methodologies,53 with anticipated applications for effluent processing, therapeutic and diagnostic testing and densimetric separation, is being undertaken.
The complexity of understanding the potential exposure and toxicity of so many variations of ENMs is illustrated by the variety of compositions and applications. Determining which materials are most likely to result in exposure, prioritising materials for specific testing, and determining to what extent physicochemical properties can be used to infer hazard, are some of the challenges in the risk assessment of ENMs. Protecting researchers and workers who are producing and using these materials is paramount, starting from the research laboratory and production line to the use of these materials in various applications.
Challenges in health risk assessment: Relevance to South Africa
The general consensus is that considerable work remains to be done to generate the required data for ensuring appropriate risk evaluation of ENMs. These data may include the characterisation of the wide range of nanomaterials being produced and information on their complex behaviours in different media. Because of the diversity of ENMs, toxicity is specific to a tested nanomaterial and cannot be generalised or extrapolated, even within the same chemical family. Not much is known about the dose-response curves and toxicological modes of action of specific nanomaterials and how these materials might enter the body. Once inside the cell, the data needed for specific ENMs, such as toxicity and carcinogenicity, adsorption, distribution, metabolism and excretion, as well as occupational and environmental monitoring information, is largely unavailable. Moreover, longer-term effects of ENM need urgently to be addressed. Generalised exposure scenarios have also not been developed for ENMs for risk assessment along the life cycle of an ENM.4 Without reliable data on effects and exposure, in-depth risk assessments of ENMs for developing risk-based management strategies or regulations cannot be conducted.
There is no methodical data available on nanomaterial production levels, on exposure scenarios in working environments or research laboratories, or on exposures related to consumer products. Not much is known about emissions from nanomaterial production facilities and the fates of these emissions in the environment. In the absence of adequate data, extra precautions in controlling exposure to ENMs are recommended.33
The development of a risk assessment for ENMs in South Africa is fraught with similar challenges as those experienced internationally. ENMs synthesised in many countries, including South Africa, encompass a multitude of classes, which contain different subclasses and countless modified versions. Their diversity makes it unfeasible to conduct an ad-hoc risk assessment of every type of nanoparticle. Thus the development of risk assessment strategies based on categories of ENMs (e.g. based on mode of action) is needed.3 For ENMs to present a risk there must be both a potential for exposure and a hazard from such exposure. Prioritisation strategies for toxicity testing and risk assessment therefore consider which ENMs have the most commercial production and exposure potential54; these priority ENMs may also vary by country.
Exposure and response data obtained from animals or humans, as well as information on sources of exposure and the physicochemical properties of the ENMs along their life cycle are required for those ENMs identified as high priority for a comprehensive risk assessment. A tiered toxicity testing approach, that provides reliable and predictive evidence of ENM-related toxicity or safety would be extremely important to support the appropriate testing of safety and toxicity of these materials.4,10 Such an approach would allow one to separate materials of concern from those of lesser or no concern. Linkov and collaborators55 have developed a decision tree approach that incorporates tiered toxicity testing strategies. Another more comprehensive tiered testing strategy for ENMs has recently been proposed56 (Figure 2). The key issue in these approaches is well-defined decision points at which testing can be stopped or continued in a subsequent, more resource-intensive tier. Toxicity testing of ENMs should be initiated with a careful physicochemical characterisation of the materials (Figure 2), including the use of reference materials currently available as dispersions, especially for in-vitro studies.57
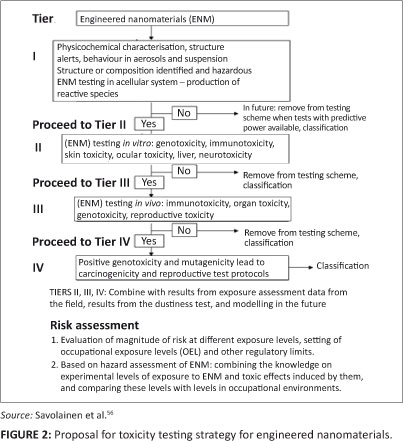
These well-characterised ENMs can then be further investigated in the next tier by first using acellular systems to explore the reactivity of the materials, before exploration at a subcellular level. It would then be possible to proceed to in-vitro cellular models that would support evidence-based testing processes.58 Such in-vitro testing methods should address carefully chosen and relevant end points shown to be predictive of human toxicity.59 Positive and consistent results from validated in-vitro tests with demonstrated predictive power would lead to higher tier testing procedures with experimental animals. The advantages of this tiered system is that these lower tier tests would be short-term studies, and although long-term studies with experimental animals would be required, these would be less frequently required than is the case today. A major challenge for the development of this kind of tiered testing procedure continues to be the validation of in-vitro tests with appropriate predictive power for in-vivo effects in whole organisms, although some progress has been shown for prediction of acute lung effects from in-vitro data.6,60
Summary and conclusion
Risk assessment is the process of determining whether exposure to a substance will lead to negative health effects. A comprehensive risk assessment of ENMs will therefore involve evaluation of the exposure potential, the hazardous properties and the dose-response relationship of ENMs. These findings could then be used to characterise the risk and to provide information for risk-based decision-making, such as establishing exposure limits and other risk management measures to protect human health and the environment - the ultimate goal of the risk assessment exercise.
For a resource-limited country such as South Africa, it is essential that best practice guidelines developed internationally be adopted and that research be conducted as a priority to provide the data needed for risk assessments specific to the situation in South Africa.
Acknowledgements
We acknowledge the support of the Department of Science and Technology, South Africa and its initiative towards research on the risk assessment of nanomaterials.
Competing interests
We declare that we have no financial or personal relationships which may have inappropriately influenced us in writing this paper.
Authors' contributions
M.G. was the project leader and wrote the first draft of the article. E.D.K. and K.S. contributed sections as per their expertise in risk assessment and assisted in finalising the article.
References
1. REACH. Nanomaterials in REACH. CA/59/2008 rev. Brussels: European Commission; 2008. [ Links ]
2. British Standards Institute. Nanotechnologies - Part 2: Guide to safe handling and disposal of manufactured nanomaterials. London: British Standards Institute; 2007. [ Links ]
3. Schulte PA, Murashov V, Zumwalde R, et al. Occupational exposure limits for nanomaterials: State of the art. J Nanopart Res. 2010;12:1971-1987. http://dx.doi.org/10.1007/s11051-010-0008-1 [ Links ]
4. U.S. Environmental Protection Agency. Nanotechnology White Paper [document on the Internet]. c2007 [cited 2012 June 06]. Available from: http://www.epa.gov/osa/pdfs/nanotech/epa-nanotechnology-whitepaper-0207.pdfReport. [ Links ]
5. Scientific Committee on Emerging and Newly Identified Health Risks. Scientific basis for the definition of the term 'nanomaterial' [document on the Internet]. c2010 [cited 2012 June 06]. Available from: http://ec.europa.eu/health/scientific_committees/emerging/docs/scenihr_o_032.pdf. [ Links ]
6. Donaldson K, Borm PJ, Oberdorster G, et al. Concordance between in vitro and in vivo dosimetry in the proinflammatory effects of low-toxicity, low-solubility particles: The key role of the proximal alveolar region. Inhal Toxicol. 2008;20(1):53-62. http://dx.doi.org/10.1080/08958370701758742 [ Links ]
7. Nanotechnologies Working Group 3 - Health, Safety and the Environment, Project Group 6. Guide to safe handling and disposal of manufactured nanomaterials. Draft report, NANO TC229 WG 3/PG 6 012-2009. Geneva: ISO; 2009. [ Links ]
8. NIOSH. Approaches to safe nanotechnology: Managing the health and safety concerns with engineered nanomaterials. Publication no. 2009-125 [document on the Internet]. c2009 [cited 2012 June 06]. Available from: www.cdc.gov/niosh/docs/2099-125/Report. [ Links ]
9. OECD: Environment, Health and Safety Division Environment Directorate. Work on the safety of manufactured nanomaterials [document on the Internet]. c2010 [cited 2012 June 06]. Available from: http://www.oecd.org/dataoecd/15/28/46371275.pdfLinks ] Arial, Helvetica, sans-serif" size="2">.
10. OECD. Guidance manual for the testing of manufactured nanomaterials ENV/JM/MONO(2009)20/REV [document on the Internet]. c2010 [cited 2012 June 06]. Available from: http://www.oecd.org/ officialdocuments/displaydocumentpdf?cote=env/jm/mono(2009)20/rev&doclanguage=enReport. [ Links ]
11. OECD. Current development/activities on the safety of manufactured nanomaterials - Tour De Table ENV/JM/MONO(2010)4 [document on the Internet]. c2010 [cited 2012 June 06]. Available from: http://www.oecd.org/dataoecd/49/49/44947758.pdfReport. [ Links ]
12. NRC. Risk assessment in the federal government: Managing the process. Washington DC: National Academy of Sciences; 1983. [ Links ]
13. NRC: Committee on Improving Risk Analysis Approaches Used by the U.S. EPA, Board on Environmental Studies and Toxicology, Division on Earth and Life Studies. Science and decisions: Advancing risk assessment. Washington DC: The National Academies Press; 2009. [ Links ]
14. Schierow L-J. 94036: The role of risk analysis and risk management in environmental protection [homepage on the Internet]. c1996 [cited 2012 June 06]. Available from: http://www.fas.org/spp/civil/crs/94-036.htm. [ Links ]
15. Kuempel ED, Geraci CL, Schulte PA. Risk assessment approaches and research needs for nanoparticles: An examination of data and information from current studies. In: Simeonova P, Opopol N, editors. Nanotechnology - Toxicological issues and environmental safety. New York: Springer-Verlag, 2007; p. 119-145. http://dx.doi.org/10.1007/978-1-4020-6076-2_8 [ Links ]
16. Scientific Committee on Emerging and Newly Identified Health Risks. Risk assessment of products of nanotechnologies [document on the Internet]. c2009 [cited year month day]. Available from: http://ec.europa.eu/health/ph_risk/committees/04_scenihr/docs/scenihr_o_023.pdfReport. [ Links ]
17. Johnston HJ, Hutchison G, Christensen FM, et al. A review of the in vivo and in vitro toxicity of silver and gold particulates: Particle attributes and biological mechanismsresponsiblefor the observedtoxicity. Crit RevToxicol. 2010;40(4):328-346. http://dx.doi.org/10.3109/10408440903453074 [ Links ]
18. Semmler-Behnke M, Kreyling WG, Lipka J, et al. Biodistribution of 1.4- and 18-nm gold particles in rats. Small. 2008;4(12):2108-2111. http://dx.doi.org/10.1002/smll.200800922 [ Links ]
19. Gosens I, Post JA, de la Fonteyne LJ, et al. Impact of agglomeration state of nano- and submicron sized gold particles on pulmonary inflammation. Part Fibre Toxicol. 2010;7(1):37. http://dx.doi.org/10.1186/1743-8977-7-37 [ Links ]
20. Sung JH, Ji JH, Park JD, et al. Subchronic inhalation toxicity of gold nanoparticles. Part Fibre Toxicol. 2011;8(1):16. http://dx.doi.org/10.1186/1743-8977-8-16 [ Links ]
21. Donaldson K, Murphy F, Schinwald A, Duffin R, Poland CA. Identifying the pulmonary hazard of high aspect ratio nanoparticles to enable their safety-by-design. Nanomedicine (Lond). 2011;6(1):143-156. http://dx.doi.org/10.2217/nnm.10.139 [ Links ]
22. Setzer RW, Kimmel CA. Use of NOAEL, benchmark dose, and other models for human risk assessment of hormonally active substances. Pure Appl Chem. 2003;75(11-12):2151-2158. http://dx.doi.org/10.1351/pac200375112151 [ Links ]
23. Hagens WI, Oomen AG, de Jong WH, et al. What do we (need to) know about the kinetic properties of nanoparticles in the body? Regul Toxicol Pharmacol. 2007;49(3):217-229. http://dx.doi.org/10.1016/j.yrtph.2007.07.006 [ Links ]
24. Oberdorster G, Oberdorster E, Oberdorster J. Concepts of nanoparticle dose metric and response metric. Environ Health Perspect. 2007;115(6):A290. http://dx.doi.org/10.1289/ehp.115-a290a [ Links ]
25. Pauluhn J. Poorly soluble particulates: Searching for a unifying denominator of nanoparticles and fine particles for DNEL estimation. Toxicology. 2011;279(1-3):176-188. http://dx.doi.org/10.1016/j.tox.2010.10.009 [ Links ]
26. Tran CL, Buchanan D, Cullen RT, et al. Inhalation of poorly soluble particles II. Influence of particle surface area on inflammation and clearance. Inhal Toxicol. 2000;12(12):1113-1126. http://dx.doi.org/10.1080/08958370050166796 [ Links ]
27. Maynard AD, Aitken RJ. Assessing exposure to airborne nanomaterials: Current abilities and future requirements. Nanotoxicology. 2007;1(1):26-41. http://dx.doi.org/10.1080/17435390701314720 [ Links ]
28. RIVM. Exposure to nanomaterials in consumer products. RIVM Letter Report 340370001/2009 [document on the Internet]. c2009 [cited 2012 June 06]. Available from: http://www.rivm.nl/bibliotheek/rapporten/340370001.pdf [ Links ]
29. Klaine SJ, Alvarez PJ, Batley GE, et al. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ Toxicol Chem. 2008;27(9):1825-1851. http://dx.doi.org/10.1897/08-090.1 [ Links ]
30. Kuhlbusch TA, Fissan H. Particle characteristics in the reactor and pelletizing areas of carbon black production. J Occup Environ Hyg. 2006;3(10):558-567. http://dx.doi.org/10.1080/15459620600912280 [ Links ]
31. Som C, Berges M, Chaudhry Q, et al. The importance of life cycle concepts for the development of safe nanoproducts. Toxicology. 2010;269(2-3):160-169. http://dx.doi.org/10.1016/j.tox.2009.12.012 [ Links ]
32. OECD. Report of the Workshop on Risk Assessment of Manufactured Nanomaterials in a Regulatory Context. Publications Series on the Safety of Manufactured Nanomaterials No. 21. ENV/JM/MONO(2010)10. Paris: OECD; 2010. [ Links ]
33. Schulte PA, Salamanca-Buentello F. Ethical and scientific issues of nanotechnology in the workplace. Environ Health Perspect. 2007;115(1):5-12. http://dx.doi.org/10.1289/ehp.9456 [ Links ]
34. Kuempel ED, Tran CL, Castranova V, et al. Lung dosimetry and risk assessment of nanoparticles: Evaluating and extending current models in rats and humans. Inhal Toxicol. 2006;18(10):717-724. http://dx.doi.org/10.1080/08958370600747887 [ Links ]
35. National Institute for Occupational Safety and Health, CDC. Occupational exposure to titanium dioxide. Publication no. 2011-160 [document on the Internet]. c2011 [cited 2012 June 06]. Available from: http://www.cdc.gov/niosh/docs/2011-160Links ] Arial, Helvetica, sans-serif" size="2">.
36. Dankovic D, Kuempel E, Wheeler M. An approach to risk assessment for TiO2. Inhal Toxicol. 2007;19(suppl. 1):205-212. http://dx.doi.org/10.1080/08958370701497754 [ Links ]
37. Pauluhn J. Multi-walled carbon nanotubes (Baytubes): Approach for derivation of occupational exposure limit. Regul Toxicol Pharmacol. 2010;57(1):78-89. http://dx.doi.org/10.1016/j.yrtph.2009.12.012 [ Links ]
38. Nakanishi J. Risk assessment of manufactured nanomaterials: "Approaches" - Overview of approaches and results. Final report. Tokyo: National Institute of Advanced Industrial Science and Technology; 2011. [ Links ]
39. National Institute for Occupational Safety and Health, CDC. Occupational exposure to carbon naotubes and nanofibers. External review draft [document on the Internet]. c2010 [cited 2012 June 06]. Available from: www.cdc.gov/niosh/docket/review/docket161A/Report. [ Links ]
40. Aschberger K, Johnston HJ, Stone V, et al. Review of carbon nanotubes toxicity and exposure - Appraisal of human health risk assessment based on open literature. Crit Rev Toxicol. 2010;40(9):759-790. http://dx.doi.org /10.3109/10408444.2010.506638 [ Links ]
41. Ma-Hock L, Treumann S, Strauss V, et al. Inhalation toxicity of multiwall carbon nanotubes in rats exposed for 3 months. Toxicol Sci. 2009;112(2):468-481. http://dx.doi.org/10.1093/toxsci/kfp146 [ Links ]
42. Brouwer D. Exposure to manufactured nanoparticles in different workplaces. Toxicology. 2010;269(2-3):120-127. http://dx.doi.org/10.1016/j.tox.2009.11.017 [ Links ]
43. IFA. Criteria for assessment of the effectiveness of protective measures. 2009. Available from: http://www.dguv.de/ifa/en/fac/nanopartikel/beurteilungsmassstaebe/index.jsp [ Links ]
44. Department of Science and Technology (DST). National nanotechnology strategy. Pretoria: DST; 2005. [ Links ]
45. Phala NS, Van Steen E. Intrinsic reactivity of gold nanoparticles: Classical, semi-empirical and DFT studies. Gold Bulletin. 2007;40(2):150-153. [ Links ]
46. McPherson JS, Thompson DT. Selectivity of gold catalysts for applications of commercial interest. Top Catal. 2009;52(6-7):743-750. http://dx.doi.org/10.1007/s11244-009-9205-5 [ Links ]
47. Tshikhudo TR, Demuru D, Wang Z, et al. Molecular recognition by calix[4] arene-modified gold nanoparticles in aqueous solution. Angew Chem Int Ed Engl. 2005;44:2913-2916. http://dx.doi.org/10.1002/anie.200462909 [ Links ]
48. Graham UM, Dozier A, Bahome MC, et al. Carbon nanotube docking stations - A new concept in catalysis. Catal Lett. 2009;129:39-45. http://dx.doi.org/10.1007/s10562-009-9866-5 [ Links ]
49. Pillay K, Cukrowska E, Coville NJ. Multi-walled carbon nanotubes as adsorbents for the removal of parts per billion levels of hexavalent chromium from aqueous solution. J Hazard Mater. 2009;166:1067-1075. http://dx.doi.org/10.1016/j.jhazmat.2008.12.011 [ Links ]
50. Krause RWM, Mamba BB, Dlamini LN, et al. Fe-Ni nanoparticles supported on carbon nanotube-co-cyclodextrin polyurethanes for the removal of trichloroethylene in water. Journal Nanopart Res. 2010;12(2):1-8. [ Links ]
51. Moloto N, Revaprasadu N, Musetha PL, et al. The effect of precursor concentration, temperature and capping group on the morphology of CdS nanoparticles. J Nanosci Nanotechnol. 2009;9(8):4760-4766. http://dx.doi.org/10.1166/jnn.2009.219 [ Links ]
52. Makhatha ME, Ray SS, Hato J, et al. Thermal and thermomechanical properties of poly(butylene succinate) nanocomposites. J Nanosci Nanotechnol. 2008;8(4):1679-1689. [ Links ]
53. Vatta LL, Kramer J, Koch KR. Diethylenetriamine functionalized silica coated magnetite nanoparticles for selective palladium ion extraction from aqueous solutions separation. Sep Sci Technol. 2007;42(9):1985-2002. http://dx.doi.org/10.1080/01496390701401402 [ Links ]
54. OECD. Comparison of guidance on selection of skin protective equipment and respirators for use in the workplace: Manufactured nanomaterials. ENV/JM/MONO(2009)17 [document on the Internet]. c2009 [cited 2012 June 06]. Available from: http://www.oecd.org/dataoecd/15/56/43289781.pdf [ Links ]
55. Linkov I, Satterstrom FK, Steevens J, et al. Multi-criteria decision analysis and environmental risk assessment for nanomaterials. J Nanopart Res. 2007;9(4):543-554. [ Links ]
56. Savolainen K, Alenius H, Norppa H, Pylkkanen L, Tuomi T, Kasper G. Risk assessment of engineered nanomaterials and nanotechnologies - A review. Toxicology. 2010;269(2-3):92-104. http://dx.doi.org/10.1016/j.tox.2010.01.013 [ Links ]
57. Elder A, Lynch I, Grieger K, et al. Human health risks of engineered nanomaterials: Critical knowledge gaps in nanomaterials risk assessment. In: Linkov I, Steevens J, editors. Nanomaterials: Risks and benefits. Dordrecht: Springer, 2009; p. 3-29. http://dx.doi.org/10.1007/978-1-4020-9491-0_1 [ Links ]
58. Guzelian PS, Victoroff MS, Halmes C, et al. Clear path: Towards an evidence-based toxicology (EBT). Hum Exp Toxicol. 2009;28(2-3):71-79. http://dx.doi.org/10.1177/0960327109102802 [ Links ]
59. Takagi A, Hirose A, Nishimura T, et al. Induction of mesothelioma in p53+/- mouse by intraperitoneal application of multi-wall carbon nanotube. J Toxicol Sci. 2008;33(1):105-116. http://dx.doi.org/10.2131/jts.33.105 [ Links ]
60. Rushton EK, Jiang J, Leonard SS, et al. Concept of assessing nanoparticle hazards considering nanoparticle dosemetric and chemical/biological response metrics. J Toxicol Environ Health A. 2010;73(5):445-461. http://dx.doi.org/10.1080/15287390903489422 [ Links ]
 Correspondence to:
Correspondence to:
Mary Gulumian
PO Box 4788
Johannesburg 2000, South Africa
Email: mary.gulumian@nioh.nhls.ac.za
© 2012. The Authors. Licensee: AOSIS OpenJournals. This work is licensed under the Creative Commons Attribution License.














