Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.108 n.7-8 Pretoria Jan. 2012
RESEARCH ARTICLE
Citrus black spot is absent in the Western Cape, Northern Cape and Free State Provinces
Elma CarstensI, II; Hendrik F. le RouxII; Michael A. HoltzhausenIII; Liezl van RooyenIII; Joey CoetzeeIII; Ria WentzelIII; Wilhelm LaubscherIII; Zorina DawoodIII; Elrita VenterIV; Gerhardus C. SchutteI, II; Paul H. FourieI, II; Vaughan HattinghII, V
IDepartment of Plant Pathology, Stellenbosch University, Stellenbosch, South Africa
IICitrus Research International, Nelspruit, South Africa
IIIDepartment of Agriculture, Forestry and Fisheries, Pretoria, South Africa
IVAlternafruit, Paarl, South Africa
VDepartment of Conservation Ecology and Entomology, Stellenbosch University, Stellenbosch, South Africa
ABSTRACT
The South African citrus industry is strongly focused on exports and South Africa is a signatory member of both the World Trade Organisation Agreement on the application of Sanitary and Phytosanitary Measures and the International Plant Protection Convention. Citrus black spot, caused by Guignardia citricarpa, does not occur in all the South African citrus production areas and, therefore, South Africa has a responsibility to provide those trading partners that have identified G. citricarpa as a regulated pest with reliable information about the distribution of citrus black spot within South Africa. Detection surveys were conducted in citrus production areas in the Western Cape, Northern Cape and Free State Provinces and appropriate diagnostic protocols were used to ensure reliable detection of G. citricarpa. Trees in commercial orchards and home gardens on farms and in towns of 17, 9 and 5 magisterial districts in the Western Cape, Northern Cape and Free State Provinces, respectively, were sampled between 1995 and 2010. Fruit samples were taken during June and July, and leaf samples from November to January. None of the 3060 fruit and leaf samples collected during these surveys tested positive for G. citricarpa. Phyllosticta capitalensis, a non-pathogenic, ubiquitous, endophytic species was, however, detected during these surveys. In compliance with relevant International Standards for Phytosanitary Measures and based on the outcome of these official surveys, these three provinces in South Africa can be recognised as citrus black spot pest free areas.
Introduction
The South African citrus industry, currently the second largest exporter of fresh citrus fruit in the world,1 was initiated in 1654 when Commander Jan van Riebeeck planted the first citrus trees (oranges) on his farm, Bosheuvel, and in the Company's Garden near Table Mountain.2 These trees planted by van Riebeeck were brought from the Island of St. Helena to the Cape of Good Hope by a ship named Tulp.2,3 During that time, the Island of St. Helena was known as an important stopover for ships on their voyages from Asia to Europe and all the fruit trees planted on the island came with the tradesmen from the East.4 Although the possibility exists that not all the routes along which citrus material first entered South Africa are known, three records exist of the importation of citrus material into South Africa prior to 1895, when the first trained horticulturists were brought to the Cape to develop the fruit industries in South Africa.5 The other two records are that of a shipment of orange trees that were directly imported from India in 1656 and that of a shipment of grafted (budded) trees that were imported in 1850 from Brazil.5 Although there is not much horticultural information available about these first citrus plantings, they were the ancestors of the citrus trees that moved inland in South Africa from the Cape with the pioneer settlers.6
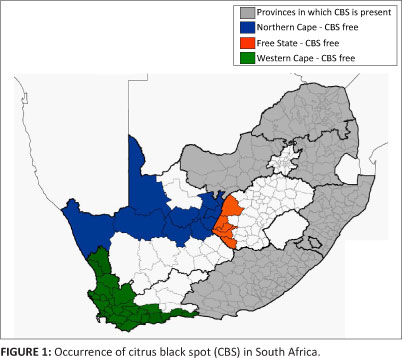
Citrus black spot (CBS) is caused by Guignardia citricarpa Kiely [anamorph Phyllosticta citricarpa (McAlpine) Aa]. Although the disease is found on several continents, it is known to not occur in Europe, Central America and the Caribbean Region.7,8 The disease is found predominantly in areas with warm, wet or humid climates with summer rainfall.7 In South Africa, the disease is known to occur in the citrus producing provinces of KwaZulu-Natal, Mpumalanga, Limpopo, North West and the Eastern Cape (Figure 1).9 However, no records exist of the presence or absence of this disease in the citrus producing provinces of the Western Cape and Northern Cape,9,10 although history indicates that the first citrus propagating material introduced into South Africa and planted in the Western Cape Province was from countries in which CBS is known to be present.7
G. citricarpa can infect the fruit, leaves and twigs of citrus trees. The epidemiology of CBS in South Africa has been unravelled by Kotzé11,12,13, McOnie14,15,16,17 and Truter18. The disease economically affects all species of citrus that are commercially grown, except for sour orange and its hybrids and Tahiti limes. Fruit symptoms are more distinctive and common than leaf symptoms. Two types of spores can be produced, namely waterborne conidia (asexual spores) and windborne ascospores (sexual spores). Ascospores, considered to be the main source of infection,12,15,16,17 are released from perithecia that have developed on fallen, infected leaves, lying on the orchard floor (leaf litter). Perithecia occur only on leaf litter and not on fruit or on leaves that are still on the tree.11,14
Studies to date have shown that more than one distinct species of Guignardia are associated with citrus.14,18,19,20 In South Africa, the presence of two species, G. citricarpa and G. mangiferae, have been reported on citrus.21 G. mangiferae is an omnipresent, non-pathogenic, endophyte species that occurs on many plants.19 The name Phyllosticta capitalensis was recently proposed instead of G. mangiferae for the endophytic species in Citrus.22
South Africa, being a signatory member of both the World Trade Organisation Agreement on the application of Sanitary and Phytosanitary Measures and the International Plant Protection Convention has certain associated responsibilities. Some of these responsibilities are that phytosanitary measures to protect South Africa must be based on scientific data and international standards and that reliable information must be provided to trading partners about the presence and distribution of plant pests within South Africa.23 South Africa is currently free from a number of devastating exotic citrus pests; however, certain pests present in South Africa, such as the fungus that causes CBS, are of quarantine importance (as regulated pests)24 to some of South Africa's trading partners. The USA only permits import of fresh citrus fruit from CBS-free areas, while Japan and India allow the import of consignments of fresh citrus fruit that are free from visible symptoms of CBS. The European Union and Iran allow the import of fresh citrus fruit that has been produced in CBS-free areas or from production sites where no CBS infected fruit have been detected in official inspections. Consequently, the reliable identification of CBS-free citrus production areas in South Africa is of great importance for the South African citrus industry because of its heavy reliance on exports.
The aim of this study was to determine the presence or absence of the CBS pathogen, G. citricarpa, in the Western Cape, Northern Cape and Free State Provinces. This aim was achieved through various surveys spanning a period of 15 years and by using available and accepted identification protocols.
Materials and methods
Detection surveys
South Africa is divided into nine provinces, which in turn are divided into magisterial districts. In order to assess the status of Guignardia species in three provinces (Western Cape, Northern Cape and Free State), magisterial districts with commercial citrus production or with the potential for future plantings were included in the surveys (Figure 1).
The surveys were conducted in a manner that is relevant to the biology of G. citricarpa, and in accordance with relevant In ternational Standards for Phytosanitary Measures (ISPM). The survey procedures were determined by diagnostic protocols available at the time. Lemons and Valencia oranges are the most susceptible to CBS and, according to Kotzé12, in an area with no history of CBS, the disease appears first on lemons. Trees were therefore sampled in commercial orchards and in home gardens on farms and towns to preferentially include older orchards (at least 15 years old and neglected) of the sensitive citrus types (lemons and Valencia oranges). However, in areas with no lemon or Valencia trees, samples were taken from Navel oranges, grapefruit and soft citrus trees.
The first surveys in the Western Cape were conducted during 1995 in 11 magisterial districts and a sample site was regarded as a single selected tree. In this survey, samples were only collected from trees in commercial orchards. Samples (four mature fruits and nine leaves) were collected between May and June from the 860 sample sites (Table 1).
In the Northern Cape, the first survey was conducted in 1998 in the magisterial district of Hartswater (the only district with commercial citrus production at that time) and a sample site was regarded as a randomly selected tree. Samples were taken from trees in commercial orchards and home gardens. Fruit samples (four mature fruits) were collected in August. Leaf samples (12 dry abscised leaves from underneath the tree) were collected in November from the 300 sample sites previously visited to collect fruit samples (Table 1).
As citrus production has expanded in South Africa, more plantings have been established in the Western Cape and Northern Cape and therefore further detection surveys were conducted in these two provinces from 2000 onwards. Surveys were also extended to the Free State Province, which borders the Northern Cape. Preliminary surveys were followed by more comprehensive confirmatory surveys. For these surveys, a sample site in orchards was regarded as 25 trees per 2 ha, and in home gardens as a single selected tree. Fruit samples were collected in June and July and leaf samples between October and January from the sites previously visited to collect fruit samples.
Preliminary surveys were conducted in the Northern Cape and Free State in 2002 and 2004, respectively. In 2002, 31 sites were sampled in six magisterial districts of the Northern Cape and in 2004, 20 sites were sampled in four magisterial districts of the Free State. In 2005 and 2006, a comprehensive survey was conducted which included three additional magisterial districts in the Northern Cape and one in the Free State. A total of 350 fruit and leaf samples were taken (Table 1).
Preliminary and comprehensive detection surveys were also conducted between 2002 and 2010 in six additional magisterial districts of the Western Cape. In 2002, 2004 and 2005, preliminary detection surveys were conducted in the magisterial districts of Mossel Bay and Knysna. Only leaf samples were taken from these sites (Table 1). In 2007 and 2008, a comprehensive survey was conducted in these two magisterial districts and also in the magisterial districts of Vredendal, Van Rhynsdorp and George. A total of 347 sites were sampled to collect fruit samples. Leaf samples were taken from 325 of these sites. At 24 sites, no leaves were available for sampling because trees or orchards had been removed (Table 1). Follow up surveys were conducted in 2010 in the magisterial districts of Mossel Bay and Knysna. As part of this survey, the neighbouring Western Cape magisterial district of Riversdale was included where leaf samples were taken from 35 sites (Table 1).
In the surveys from 2000 onward, fruit samples consisting of three to five mature fruits were collected from each sample site. In commercial orchards, 300-500 fallen leaves were collected per site; in home gardens all fallen leaves were collected per site.
Laboratory procedures for fruit samples
In 1995 and 1998, three fruits per sample were incubated in plastic buckets lined with wet paper towels. Buckets were sealed with plastic wrap and placed in a growth chamber at 29 °C and high humidity for 2 weeks to stimulate the development of CBS symptoms. In 1995, fruit was inspected for CBS symptoms on arrival and after the incubation period of 14 days. In 1998, the samples were inspected on arrival, after incubation for 2 weeks and again after 6 weeks. If suspected lesions were found, direct isolations were made onto potato dextrose agar (PDA) plates (Biolab, Johannesburg, South Africa). Direct plating of small pieces of the rind from one fruit per sample was also done onto PDA plates in 1995 and 1998. These plates were incubated at 22 °C for 13 days under a white fluorescent light before being examined for presumptive positive G. citricarpa isolates based on general colony characteristics. Pure cultures of presumptive positive G. citricarpa isolates were subjected to a random amplified polymorphic DNA (RAPD) analysis technique25 for identification.
From 2000 onward, all the fruit per sample was incubated at temperatures from 23 °C to 25 °C under a high light intensity for 6 weeks. The fruit was inspected for the development of symptoms on arrival and weekly thereafter. If suspected lesions were found, the sample was subjected to a molecular diagnostic protocol with species-specific primers for G. citricarpa.26 Direct isolations of the suspected lesions were also done onto oatmeal agar (OMA) plates (Biolab).19 OMA plates were incubated at 25 °C for 7 days and monitored for presumptive positive G. citricarpa isolates based on general colony characteristics.
Laboratory procedures for leaf samples
In 1995, seven leaves from each sampling site (including two of the leaves sampled from underneath the trees) and, in 1998, ten leaves per site were incubated in plastic buckets and monitored under the same conditions as described for fruit samples. Direct plating of small pieces of the remaining two leaves per sampling site was done onto PDA plates and incubated and monitored as described for fruit samples.
From 2000 onwards, detection and presumptive identification of Guignardia ascospores were done according to the method described by Truter et al.27,28 If any ascospores resembling the morphology of G. citricarpa were found, the sample was subjected to a molecular diagnostic protocol with species-specific primers for G. citricarpa.26 Direct isolation of these samples was also done onto OMA plates. OMA plates were incubated and monitored as described for fruit samples.
Identification
Identification of presumptive positive isolates in 1995 and 1998 was performed using light microscopy and taxonomic keys for the genus,29 as well as RAPD analysis.25 From 2000, identification was performed using colony characteristics on OMA (where isolates producing a distinct diffuse yellow pigment were regarded as G. citricarpa19) and species-specific primers for G. citricarpa.26
For the molecular-based identifications, leaves or fruit lesions of the presumptive positive Guignardia samples were ground to a fine powder in liquid nitrogen. DNA extraction was performed using the Qiagen DNEasy Extraction Kit (Qiagen, Hilden, Germany) as per the manufacturer's instructions. The species-specific primer pair 5'-GAA AGG TGA TGG AAG GGA G-3' (CITRIC1) and 5'-AGT ATA CAA AAC TCA AGA ATT C-3' (CAMEL2), developed by Meyer et al.26, and the ITS4 primer were used in the amplification reaction. Amplification with these primers was performed using the polymerase chain reaction (PCR) conditions recommended by Meyer et al.26 Positive and negative controls (using water instead of template DNA) were included. PCR products were analysed in a 1.5% (wt/vol) agarose gel and visualised under UV light after ethidium bromide (10 mg/mL) staining. An amplicon of approximately 580 bp was recorded as positive for G. citricarpa and an amplicon of 430 bp was recorded as positive for P. capitalensis, but negative for G. citricarpa.
Results
During the surveys conducted in 1995 and 1998, no typical CBS symptoms were observed either during field inspections or by visual examination of the samples after incubation of fruit and leaves. Furthermore, no G. citricarpa isolates were found in 1995 from a total of 17 200 isolations made from fruit and leaves on PDA plates. Likewise there were no positive G. citricarpa identifications from the 4000 isolates obtained in 1998. In the surveys conducted from 2000 onward, no symptoms of CBS were observed during field inspections or by visual examination of the fruit and leaf samples and incubated fruit.
During the preliminary detection surveys in Mossel Bay and Knysna in 2002, 2004 and 2005, 14 leaf samples tested positive for Guignardia spores using the inoculum monitor. Four samples tested positive for Guignardia spores during the comprehensive survey conducted in 2005 and 2006 in the Northern Cape. These samples were collected in the magisterial districts of Herbert, Gordonia and Hopetown. Guignardia spores were also found in four samples collected during the follow-up survey in 2010 in the magisterial district of Knysna. However, none of these 22 presumptive positive samples yielded a 580-bp amplicon following PCR amplification with the species-specific primer pairs. Only DNA from the CBS positive control yielded an amplicon of 580 bp. All of the 22 samples yielded an amplicon of 430 bp with the species-specific primer pairs. None of the 22 isolates produced the distinct diffuse yellow pigment on OMA plates (Table 1). These samples were, therefore, recorded as positive for P. capitalensis, and negative for G. citricarpa.
Discussion
Plant health has always influenced international trade.30 A key element in market access negotiations is the provision of lists of all pests associated with a specific crop within the exporting country to its trading partners. Long delays can occur when this information is not readily available. South Africa is an export-driven country and the citrus industry is continuously seeking access to new markets, and at the same time, the expansion and maintenance of existing markets.
In order for the South African government to take part in such negotiations, they must have access to accurate and scientifically peer-reviewed information on the distribution and occurrence of plant pests identified by trading partners to be of phytosanitary importance.
In South Africa, CBS has never been observed or detected in certain citrus production areas (the Western Cape and Northern Cape), although history indicates that orchards in the Western Cape were established with propagation material which originated from countries with CBS. Subsequently, orchards were also established in these areas with propagation material from the northern parts of the country where CBS occurs, before legislation was implemented to control the movement of citrus propagation material. Infected propagation material is regarded as the most prominent means of distribution of CBS to new areas,12 and according to Wager31 there is evidence in South Africa of CBS being introduced into CBS-free areas by means of latently infected trees. However, climatic conditions play an important role in the successful establishment of this disease.32 Climate modelling studies9,18 have indicated that the Western Cape and Northern Cape Provinces are unsuitable for CBS, which therefore explains why these areas have remained CBS free despite repeated challenges.
Whereas there was strong circumstantial evidence that CBS is not present in the provinces surveyed,10,12,31 scientific evidence was required by trading partners to support the declaration of such areas as pest free.33 Our detection surveys conducted over a period of 15 years (1995 to 2010) provide such evidence. In accordance with the relevant ISPM (ISPM 4 - Requirements for the establishment of pest free areas)33 and based on the outcome of these official surveys from 1995 to 2010, the Western Cape, Northern Cape and Free State Provinces can be recognised as CBS pest free areas.
The status of CBS pest free areas is continuously monitored, through the routine official quality and phytosanitary inspections conducted on fruit for export. The pest free status of these provinces is also protected by government legislation.34 This legislation prohibits the movement of propagation material of species of Citrus and related plants such as Fortunella, Limonia, Poncirus, Swinglea and crosses thereof from provinces where CBS is present to these CBS-free provinces. Nonetheless, periodic follow-up surveys should be undertaken to ensure compliance with the relevant International Standards for Phytosanitary Measures.
Acknowledgements
The Department of Agriculture, Forestry and Fisheries, Citrus Research International, Citrus Growers Association of Southern Africa, University of Pretoria, Agricultural Research Council, QMS Agri Science, Hans la Grange, Ballie Wahl and Marelize de Villiers are thanked for financial, technical and administrative support. Prof. J.M. Kotzé is gratefully acknowledged for his most valuable support, guidance and advice on these surveys.
Competing interests
We declare that we have no financial or personal relationships which may have inappropriately influenced us in writing this paper.
Authors' contributions
H.l.R., V.H. and M.H. were the project leaders. H.l.R., P.F., R.W., E.V. and E.C. were responsible for the experimental and project design. Most of the field work and experiments were performed by H.l.R., E.C., J.C., R.W., L.v.R. and W.L. Some of the experiments and field work were performed by E.V., Z.D. and G.S. The manuscript was written by E.C.
References
1. Key industry statistics. Durban: Citrus Growers' Association of Southern Africa; 2010." [ Links ]
2. Chapot H. The citrus plant. In: Citrus. Basle: Ciba-Geigy Agrochemicals, Ciba-Geigy Ltd., 1975; p. 6-13. [ Links ]
3. Powell HC. The culture of the orange and allied fruits. South African Agricultural series no. 8. Pretoria: Central News Agency Ltd.; 1930. [ Links ]
4. Allwright WJ. The controlled marketing of citrus fruit in South Africa. PhD thesis, Pretoria, University of Pretoria, 1945. [ Links ]
5. Allwright WJ. South African Co-operative Citrus Exchange Limited. Pretoria: Electric Printing Works (Pty) Ltd; 1957. [ Links ]
6. Oberholzer PCJ. Citrus culture in Africa south of the Sahara. Proc Int Soc Citric. 1969;1:111-120. [ Links ]
7. CAB International Crop Protection Compendium Globule Module. Wallingford, UK: Commonwealth Agricultural Bureau International; 2011. [ Links ]
8. Phytosanitary alert system. Ottawa: North American Plant Protection Organization; 2010. Available from: http://www.pestalert.org/oprDetail.cfm?oprID=421. [ Links ]
9. Paul I, Van Jaarsveld AS, Korsten L, Hattingh V. The potential global geographical distribution of citrus black spot caused by Guignardia citricarpa Kiely: Likelihood of disease establishment in the European Union. Crop Prot. 2005;24:297-308. http://dx.doi.org/10.1016/j.cropro.2004.08.003 [ Links ]
10. McOnie KC. Apparent absence of Guignardia citricarpa Kiely from localities where citrus black is absent. S Afr J Agri Science. 1964;7:347-354. [ Links ]
11. Kotzé JM. Studies on the black spot disease of citrus caused by Guignardia citricarpa Kiely, with particular reference to its epiphytology and control at Letaba. DSc(Agric) thesis, Pretoria, University of Pretoria, 1963. [ Links ]
12. Kotzé JM. Epidemiology and control of citrus black spot in South Africa. Plant Dis. 1981;65:945-950. http://dx.doi.org/10.1094/PD-65-945 [ Links ]
13. Kotzé JM. History and epidemiology of citrus black spot in South Africa. Proc Int Soc Citric. 1996;2:1296-1299. [ Links ]
14. McOnie KC. The latent occurrence in citrus and other hosts of a Guignardia easily confused with G. citricarpa, the citrus black spot pathogen. Phytopathology. 1964;54:40-43. [ Links ]
15. McOnie KC. Source of inoculum of Guignardia citricarpa, the citrus black spot pathogen. Phytopathology. 1964;54:64-67. [ Links ]
16. McOnie KC. Orchard development and discharge of ascospores of Guignardia citricarpa and the onset of infection in relation to the control of citrus black spot. Phytopathology. 1964;54:1148-1453. [ Links ]
17. McOnie KC. Germination and infection of citrus by ascospores of Guignardia citricarpa in relation to control of black spot. Phytopathology. 1967;57:743-746. [ Links ]
18. Truter M. Epidemiology of citrus black spot disease in South Africa and its impact on phytosanitary trade restrictions. PhD thesis, Pretoria, University of Pretoria, 2010. [ Links ]
19. Baayen RP, Bonants PJM, Verkley G, et al. Nonpathogenic isolates of the citrus black spot fungus, Guignardia citricarpa, identified as a cosmopolitan endophyte of woody plants, Guignardia mangiferae (Phyllosticta capitalensis). Phytopathology. 2002;92:464-477. http://dx.doi.org/10.1094/PHYTO.2002.92.5.464, PMid:18943020 [ Links ]
20. Wulandari NF, To-anun C, Hyde KD, et al. Phyllosticta citriasiana sp. nov., the cause of citrus tan spot of Citrus maxima in Asia. Fungal Divers. 2009;34:23-39. [ Links ]
21. Meyer L, Slippers B, Korsten L, Kotzé JM, Wingfield MJ. Two distinct Guignardia species associated with citrus in South Africa. S Afr J Science. 2001;97:191-194. [ Links ]
22. Glienke C, Pereira OL, Stringari D, et al. Endophytic and pathogenic Phyllosticta species, with reference to those associated with citrus black spot. Persoonia. 2011;26:47-56. http://dx.doi.org/10.3767/003158511X569169, PMid:22025803, PMCid:3160796 [ Links ]
23. Van Halteren P. Diagnostics and National Plant Protection Organizations. EPPO Bulletin. 2000;30:357-359. http://dx.doi.org/10.1111/j.1365-2338.2000.tb00910.x [ Links ]
24. Food and Agriculture Organization of the United Nations. Secretariat of the International Plant Protection, International Standards for Phytosanitary Measures, Publication No 5: ISPM no 5 Glossary of phytosanitary terms. Rome: FAO; 2007. [ Links ]
25. Schutte GC, Visser AA, Oosthuizen MC, Kotzé JM. The use of random amplified polymorphic DNA markers for the detection of genetic diversity in Phyllosticta citricarpa. Proc Int Soc Citric. 1996;1:373-378. [ Links ]
26. Meyer L, Sanders GM, Jacobs R, Korsten L. A one day sensitive method to detect and distinguish between the citrus black spot pathogen Guignardia citricarpa and endophyte Guignardia mangiferae. Plant Dis. 2006;90:97-101. http://dx.doi.org/10.1094/PD-90-0097 [ Links ]
27. Truter M, Kotzé JM, Janse van Rensburg TN, Korsten L. A sampler to determine available Guignardia citricarpa inoculum on citrus leaf litter. Biosyst Eng. 2004;89:515-519. http://dx.doi.org/10.1016/j.biosystemseng.2004.08.018 [ Links ]
28. Truter MP, Labuschagne M, Kotzé JM, Meyer L, Korsten L. Failure of Phyllosticta citricarpa pycnidiospores to infect Eureka lemon leaf litter. Australas Plant Path. 2007;36:87-93. http://dx.doi.org/10.1071/AP06087 [ Links ]
29. Sutton BC, Waterston JM. Guignardia citricarpa. CMI descriptions of pathogenic fungi and bacteria no. 85. Wallingford, UK: CAB International; 1966. [ Links ]
30. McRae CF, Wilson D. Plant health as a trade policy issue. Australas Plant Path. 2002;31:103-105. http://dx.doi.org/10.1071/AP02011 [ Links ]
31. Wager VA. The black spot disease of citrus in South Africa. Sci B Dep Agric Union S Afr. 1952;303:1-52. [ Links ]
32. Guignardia citricarpa. Quarantine pests for Europe. 2nd ed. Wallingford, UK: CAB International, 1997; p. 773-778. [ Links ]
33. Food and Agriculture Organization of the United Nations. Secretariat of the International Plant Protection, International Standards for Phytosanitary Measures, Publication No 4: ISPM no 4 Requirements for the establishment of pest free areas. Rome: FAO; 1996. [ Links ]
34. Agricultural Pests Act 36 of 1983, South Africa. Available from: www.daff.gov.za. [ Links ]
 Correspondence to:
Correspondence to:
Elma Carstens
Private Bag X1, Stellenbosch University,
Matieland 7601, South Africa
Email:ec@cri.co.za
Received: 08 Aug. 2011
Accepted: 24 Feb. 2012
Published: 06 Jul. 2012
© 2012. The Authors. Licensee: AOSIS OpenJournals. This work is licensed under the Creative Commons Attribution License.














