Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.108 n.3-4 Pretoria Jan. 2012
RESEARCH ARTICLES
The evolution of cranial form in mid-Pleistocene Homo
G. Philip Rightmire
Department of Human Evolutionary Biology, Harvard University, Cambridge, USA
ABSTRACT
Interactions of the brain and cranium in archaic populations remain poorly understood. Hominin fossils from Middle Pleistocene localities in Africa and Europe have been allocated to one or more species distinct from Homo erectus, the Neanderthals and modern humans, based on the assumption that characters of the vault and face are developmentally independent. However, it is possible that increased frontal width, parietal lengthening, midvault expansion and occipital rounding all reflect encephalisation occurring within the H. erectus lineage. If specimens from Broken Hill and Elandsfontein (in southern Africa) and Sima de los Huesos and Petralona (in Europe) differ from H. erectus only in brain volume, then it will be difficult to distinguish and diagnose Homo rhodesiensis or Homo heidelbergensis adequately. In this study, correlation analysis showed that the brain fails to influence vault breadth within either H. erectus or the mid-Pleistocene sample. Instead, the (large) cranial base has a major effect on width. Variation in brain volume is not associated with frontal flattening. In H. erectus and in individuals such as Bodo and Petralona, the massive face seems to override the brain as a determinant of frontal curvature. Small H. erectus crania have rounded occipitals, whilst larger individuals show greater flexion. Later hominins do not follow this trend, and encephalisation cannot explain the occipital rounding that is present. Few of the vault characters considered diagnostic for the mid-Pleistocene fossils can be attributed to increasing brain volume. The situation is complex, as of course the brain must influence some traits indirectly. The cranial base is also an instrument of change. Discriminant analysis identified basicranial breadth as critical to distinguishing individuals such as Broken Hill, Sima de los Huesos and Petralona from H. erectus.
Introduction
Beginning with the Broken Hill discoveries in 1921, many hominin fossils have been found at mid-Pleistocene localities in Africa. At Broken Hill in Zambia, miners quarrying for lead and zinc broke into the lower part of an extensive cavern. This section of ore deposits contained mineralised bones and stone artefacts. Along with postcranial elements, a nearly complete human cranium was recovered. This well-preserved specimen remains one of the treasures of prehistory (Figure 1). In 1953, pieces of a similar skull were picked up at Elandsfontein in South Africa. In blowouts amongst the dunes at this locality, animal bones and artefacts are exposed on a calcareous duricrust known as Elandsfontein Main, probably representing an ancient land surface. Thousands of bones, later Acheulean bifaces, cores and flakes have been collected, mostly in surface scatters. Only at 'Cutting 10' was such an assemblage uncovered in situ.1 The fauna from Cutting 10 may not be associated directly with the artefacts, but in general the contemporaneity of many of the Elandsfontein fossils with a later Acheulean industry is not in doubt. The diverse fauna includes numerous large herbivores, and there are archaic elements such as a dirk-toothed cat, a sivathere, a giant gelada baboon and at least four archaic hartebeest-like or wildebeestlike antelope species. In all, some 15 of 48 mammalian species occurring at Elandsfontein have no historic descendants. An analysis conducted by Klein et al.2 suggests that this assemblage is between 600 kyr and 1 Myr old.
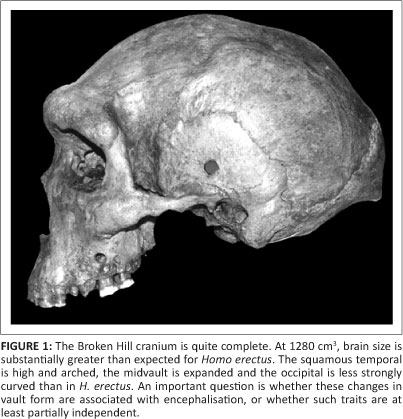
At the time of these discoveries, there were suggestions that the hominins might resemble the Neanderthals then known from Europe. It is now recognised that this comparison was inappropriate. The crania lack the specialised characters of later Neanderthals but are similar to other African individuals such as Bodo from the Middle Awash of Ethiopia. Like the Broken Hill specimen, Bodo resembles Homo erectus in that it possesses a low braincase and a massive face. The flattened frontal with midline keeling, parietal angular torus and thick vault bones give the specimen a pronounced archaic appearance. In other respects, the Bodo cranium is more derived in its morphology. Brain size is close to 1250 cm3 and thus is greater than expected for H. erectus. Frontal breadth proportions, the high arched shape of the squamous temporal, and some (although not all) traits of the cranial base are like those of more modern humans. Although the face is very broad with a prominent glabellar region, the supraorbital tori are divided into medial and lateral segments, the nasal border is vertical rather than forward sloping, and the incisive canal opens into the front of the hard palate. These are apomorphic conditions expressed also in the face of recent Homo.3 The Bodo hominins were found in conglomerates and sands containing later Acheulean tools. Fauna from the Bodo site has been compared to that from Bed IV at Olduvai Gorge in Tanzania and Olorgesailie in Kenya, and an early Middle Pleistocene date is indicated. 40Ar/39Ar measurements support this biochronology, and the evidence points to an age of about 600 kyr for the Bodo assemblage.4
The African crania resemble earlier Middle Pleistocene hominins from Europe. Excavations in the Sima de los Huesos (Atapuerca) in northern Spain have produced remains representing virtually all parts of the skeleton. Two of the Sima adult specimens provide estimates for brain size. At close to 1100 cm3, SH 5 is rather small, but SH 4, with a capacity of 1390 cm3, is one of the larger mid-Quaternary braincases. The skulls are primitive in some respects, and the face of SH 5 is surmounted by a prominent browridge. The Sima fossils also share a number of derived traits with their African contemporaries. First applications of U-series dating to a speleothem present in the lower part of the Sima stratigraphic sequence suggested a date of >350 ka.5 More recent sampling from the same speleothem has produced high resolution U-series dates averaging 600 ka, and a conservative minimum estimate for the age of the fossils is now said to be 530 ka.6 Another hominin from Petralona in Greece is like the Broken Hill specimen in many aspects of vault shape - in height, breadth and massive construction of the upper face and cheek; orientation of the infraorbital region; and several measures of facial projection.7,8,9 The same is true for the less complete cranium from Arago Cave in France. Arago 21 dated to circa 450 ka has a face that is largely intact but damaged as a result of its long interment in compacted cave sediments. The discoverers have been able to correct some of this distortion in a reconstruction, and it is evident that Arago 21 is slightly smaller than the Petralona or Broken Hill specimens in brow thickness, upper facial width and facial height. Overall, the specimens are similar in shape.8,9
These mid-Pleistocene hominins are distinct morphologically from H. erectus. Brain size is increased, the frontal is less constricted, the squamous temporal is arched, and there is more rounding of the parietal vault and occiput. The occipital upper scale exceeds the nuchal plane in length. The temporomandibular joint generally resembles that of Homo sapiens, as is the case for the tympanic and petrous portions of the temporal bone. Nevertheless, the frontal profile is flattened, and the vault is less globular relative to that of recent people. In the Broken Hill, Elandsfontein and Petralona specimens, the supraorbital torus is very massive. There is no reduction in overall face size, and the facial skeleton seems to be 'hafted' to the braincase in much the same way as in H. erectus.
The evolutionary and developmental determinants of skull form in earlier Homo remain poorly understood. Growth and development of anatomical modules within the cranium, and their integration, have been much studied for diverse groups of primates and in extant humans.10,11,12,13,14,15,16,17 But the process whereby a comparatively small, wide and flattened braincase is transformed during hominin evolution into a more voluminous and vaulted shape, and the accompanying constraints on facial morphology, are known only in broad outline. A key question is whether changes occurring in the mid-Pleistocene cranium are coupled solely to increasing brain size, or whether traits such as a longer, 'bossed' parietal and rounding of the occipital are partially independent from encephalisation. Either conclusion has important systematic implications. On the strength of craniofacial attributes presumed to be genetically and functionally independent, the Broken Hill and penecontemporary fossils from Europe are routinely classified as one or more species separate from H. erectus, the Neanderthals and recent humans.18,19,20,21,22 It is appropriate to ask whether the morphological evidence, examined critically, justifies the recognition of so many taxa within Homo.23 Framed in a more formal way, the null hypothesis of interest states that the mid-Pleistocene hominins do not differ at the species level from H. erectus. Acceptance of this hypothesis implies that populations of H. erectus did not change appreciably prior to the evolution of Neanderthals in Europe and modern humans in Africa, probably after 300 ka.
Materials and methods
Fossil specimens
A total of 11 of the more complete crania from mid-Pleistocene localities in Africa and western Eurasia were used in this study (Table 1). The comparative sample comprised 34 H. erectus specimens (Table 2). For the archaic hominins, sample sizes remain small. In a number of cases, the more fragile facial structures are not preserved, and the basioccipital, sphenoid body and ethmoid are either missing or deformed by crushing. Often, key landmarks such as basion and sella cannot be located. Despite these obstacles, brain volume, along with the major external dimensions of the basicranium and vault, can be treated in correlation studies and multivariate analysis.
Measurements
The variables utilised were endocranial volume (VOL), glabella-occipital length (GOL), porion-vertex height (PVH), basion-nasion length (BNL), glabella-bregma arc (GBR), frontal angle (FRA),24 bregma-lambda arc (BLR), lambda-inion chord (LIC), inion-opisthion chord (IOC), lambda-opisthion arc (LOR), occipital angle (OCA),24 maximum cranial breadth (XCB), biparietal breadth (XPB), biauricular breadth (AUB), biasterionic breadth (ASB) and vertical thickness of the supraorbital torus (TOR). Note that VOL is converted to the cube root of measured capacity. An additional variable (GMN) was used to estimate overall cranial size. GMN was calculated as the geometric mean of eight linear measurements (GOL, PVH, GBR, BLR, LOR, XCB, ASB and TOR). Volume was omitted, so as to make GMN at least partially independent from brain size.
Cranial capacities are from the literature.25 Measurements of PVH and GBR were provided by F. Spoor (2008, personal communication, May 10). Measurements of the Sima crania are those of Arsuaga et al.26 KNM-ER 42700 is described by Spoor et al.27, and dimensions for Daka are those of Gilbert and Asfaw28. Selected dimensions for the Bukuran, Sambungmacan and Ngandong fossils are from Kaifu et al.29 Data for Ngawi are listed by Widianto and Zeitoun30, but measurements of sagittal chords and arcs were taken from a cast held at the American Museum of Natural History. Sangiran IX is documented by Arif et al.31 Measurements of the Zhoukoudian crania are those of Weidenreich32, supplemented by observations from casts at the Peabody Museum at Harvard University. Other data are my own, taken from the original fossils.
Methods of analysis
Correlation techniques were used to find associations amongst measurements for the H. erectus assemblage and within the mid-Pleistocene sample. Brain volume and other cranial dimensions were included, in an effort to find patterns of covariation and assess the extent to which modules making up the skull are integrated. Pearson correlation coefficients were used to assess the strength of association between continuous variables that are normally distributed. Partial correlation is a method for disentangling relationships involving multiple variables, by computing 'corrected' correlations for any given pair. For example, in a situation where linear measurements are influenced by one another and also by brain volume or overall cranial size, it is possible to keep volume and/or size constant. The strength of the correlation remaining between the linear dimensions can then be determined, with the effects of other variables controlled.
Discriminant analysis facilitates the comparison of two or more groups, the composition of which is established in advance. Individual crania of unknown affiliation may also be included, with the objective of assigning them to one of the groups analysed. Measurements are used to construct linear functions that will maximise differences between these populations relative to variation within each sample. With a stepwise entry procedure, it is possible to ascertain how each of the original variables contributes to the analysis and whether any can be dropped without loss of substantial information. The discriminant axes are mutually orthogonal. Interpretation of the functions is facilitated by correlating the original measurements with the discriminant scores. Variables with high correlations can be identified as contributing importantly to variation on a given function, and this information helps to reveal the nature of essential population differences.
For each analysis, details concerning the measurements selected, the size adjustments utilised, the composition of groups, and problems with missing variables are given in the sections below. Correlations, partial correlations and discriminant computations were performed using JMP.33
Brain volume, cranial arc lengths and sagittal curvature
The influence of brain size on craniofacial variation was investigated first. For H. erectus, average endocranial capacity is 952 cm3 (Table 3). In the mid-Pleistocene group, brain size is expanded by 277 cm3, and this difference is significant (t = -4.77,p < 0.0001). Indeed, the mid-Quaternary hominins fall within the range documented for diverse modern humans. Arcs measured from glabella-bregma, bregmalambda and lambda-opisthion are greater in the mid-Pleistocene crania than in H. erectus, and increases in the parietal and occipital lengths are pronounced. Nevertheless, proportions of the frontal, parietal and occipital segments relative to the total sagittal profile remain comparable in the archaic populations. Only in recent H. sapiens is the occipital contribution substantially reduced.
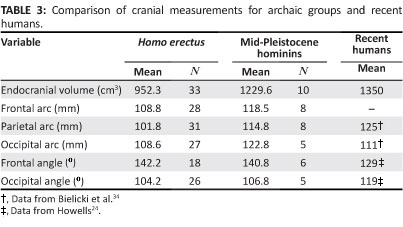
For the 18 H. erectus crania in which the necessary landmarks are preserved, the frontal angle averages 142.2°. The frontal squama is inclined posteriorly and flattened. Similar morphology characterises the mid-Pleistocene specimens, but in the modern populations studied by Howells24, means for FRA are lower for both male specimens (130.2°) and female specimens (128.2°), indicating that the frontal surface is more convex. The occipital angle can be measured for 26 H. erectus, and the average is 104.2°. The back of the braincase is more tightly flexed in H. erectus than in H. sapiens, where OCA is substantially greater for both male and female individuals drawn from 17 regional populations.24 The mid-Pleistocene group is intermediate in occipital form. With just one exception (Petralona), these specimens have OCA values of 106° or greater, and this finding is consistent with the observation that the rear of the vault is more rounded than in H. erectus.
Homo erectus almost certainly differs from modern humans in having a primitive, more rapid life history schedule,35,36,37,38 which implies alterations in the growth process. The H. erectus brain may have matured over a relatively short time period, as is true for apes,39,40 although this view has been challenged.41,42,43 Supraorbital tori, which are so prominent in H. erectus, must have begun to enlarge early in ontogeny, before the cessation of brain growth. Even in the Mojokerto infant from Indonesia, a small but clearly defined browridge is developed, along with an incipient supratoral sulcus and constriction of the frontal bone.44 Also, the H. erectus vault must have expanded posteriorly to a degree not seen in recent humans. This extra growth would have helped to produce the low neurocranial profile and elongated occiput that are characteristic of the taxon.17
Despite such differences, it can be assumed that for H. erectus, as in recent humans, the basicranium was the first part of the skull to reach adult size, and this provided the platform on which the brain and neurocranium could expand. Facial growth probably continued for at least a moderate interval after completion of the neuro-basicranial complex. Because of integration, changes in one region are reflected in the morphology of other structures. If the developmental model tested by Lieberman et al.45 in a diverse, pooled-sex sample of modern adult crania applies to closely related species, it can be hypothesised that the brain constrained earlier Pleistocene vault form in predictable ways.
Sagittal arcs should increase in skulls with larger volumes. This is the case for the H. erectus sample, where the frontal arc is highly correlated with brain size (Table 4). Pearson correlations for the parietal and occipital arcs are also significant, but particularly the association of BLR with VOL is weakened. Within H. erectus, changes in parietal length are linked less closely with variations in brain volume than is true for other segments of the vault. If parietal form is not greatly influenced by VOL, then it must be questioned whether the increase in BLR that characterises the Broken Hill, Elandsfontein, Omo 2, SH and Petralona specimens can be the outcome of encephalisation coupled with phyletic (within-lineage) evolution.
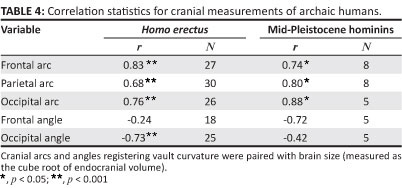
Male individuals of recent humans often have sloping frontal contours, whilst female individuals exhibit more 'bulbous' profiles. This difference offers a basis for predicting that in the H. erectus sample, larger crania will have more open frontal angles than less voluminous ones. However, the findings shown in Table 4 do not support this hypothesis. The correlation of FRA with VOL is low and negative (r = -0.24). Although this association is not significant, it is evident that several of the smaller H. erectus fossils display frontal profiles that are especially flattened. For the mid-Quaternary hominins, FRA again covaries negatively with VOL, whilst r does not reach significance. Discounting SH 5, where brain size is relatively small but there is little frontal curvature, this correlation would be close to zero. Neither for H. erectus nor within the mid-Pleistocene assemblage is brain size an important determinant of frontal flattening.
Given the findings27 pointing to a negative allometric relationship between occipital chord and arc lengths in H. erectus, it is expected that OCA will covary negatively with VOL. This covariance is confirmed, and it is evident that larger skulls show greater flexion of the occipital, whilst smaller crania tend to be more rounded. This trend continues in both early (African) and late (Asian) H. erectus populations. OCA is generally >106° for the mid-Pleistocene crania. Here, OCA is not significantly correlated with brain size, and the structural relationship apparent during the long evolutionary span of H. erectus is not maintained. Thus, the occipital rounding apparent in the later populations cannot readily be interpreted as a consequence of brain expansion within a vault that was otherwise erectus-like.
Interactions of the skull base and neurocranium
Vault form should be influenced by variations in the cranial base. In recent H. sapiens, maximum cranial breadth is strongly correlated with base breadth as well as endocranial capacity. These associations occur independently, as brain volume does not covary with dimensions of the base when overall size is held constant using partial correlation.45 The H. erectus basicranium is appreciably longer and wider than that of modern humans (Table 5). Given its relatively large area, this structure should have a primary influence on neurocranial size and shape. Indeed, maximum breadth of the vault covaries positively with base breadth measured at the auricular points (Table 6). Paired comparisons show that cranial breadth is also correlated with the external length of the base (BNL) and VOL. However, only the association of XCB with AUB remains strong, when the effects of other variables are controlled in partial correlation analysis. The influence of VOL is much reduced, and it is evident that the base, rather than the brain, has a major effect on the width of the H. erectus neurocranium. In mid-Pleistocene hominins, the cranial base is again (very) large relative to the modern condition (Table 5). Within this sample, XCB is highly correlated with AUB but is not significantly influenced by BNL or VOL (Table 7). This result parallels the findings for H. erectus.
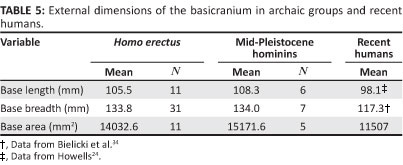
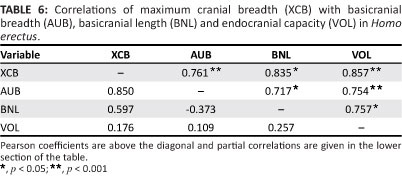
The length of the modern human cranium is dependent on the growing brain. Given their model of developmental interactions occurring during ontogeny, Lieberman et al.45 hypothesise that maximum length will have little association with variations in the adult basicranium, and this expectation is supported. A conspicuous feature of the H. erectus skull is its uniquely low, elongated, and hence poorly filled appearance, arising from the relationship of the (small) brain to the (massive) cranial base. Cranial length is influenced by the brain, as in extant humans (Table 8). Length is also strongly correlated with variations in the basicranium. There is a significant association of GOL with BNL, and the positive correlation of GOL with AUB indicates a tendency for length to increase in braincases with wider bases. Partial correlation analysis leads to the same conclusion. The correlation of GOL with BNL remains strong when 'corrected' for the potential effects of AUB and VOL, and GOL continues to covary positively with AUB when the other measurements are held constant. Here, H. erectus differs from recent humans, where such associations should be insignificant or negative.
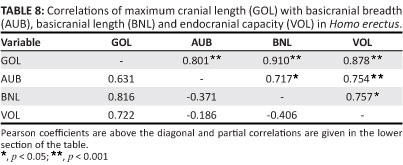
An important question is whether cranial length measured for the mid-Quaternary group is influenced only by brain size, as in modern populations, or whether this dimension is linked also to the basicranium, as in H. erectus. As expected, GOL covaries strongly (and significantly) with volume (Table 7). But the correlations of GOL with BNL and AUB are of about the same magnitude, and it is primarily because sample sizes are small that r does not quite reach the p< 0.05 level of significance. Although the data are limited, it can be inferred that the mid-Pleistocene population exhibits the H. erectus pattern. A possible explanation for this finding is the low level of encephalisation attributed to all of the archaic hominins.17,46,47,48 Neither absolute brain size nor the ratio of volume to base width (VOL:AUB) reaches recent population averages. For the large H. erectus sample, there is no significant covariation of VOL:AUB with any of the principal neurocranial diameters (Rightmire, unpublished data). Within the mid-Pleistocene group, VOL:AUB covaries only weakly with GOL and vertex height (PVH) (Table 7). The relationship of VOL:AUB with XCB is stronger but negative (a likely response to the high correlation of base breadth with vault breadth). Only in later H. sapiens does encephalisation drive values of the VOL:AUB ratio upwards, and such increases are linked with longer, higher braincases.45 Apparently the incremental changes in brain size that characterise mid-Pleistocene hominins do not result in a modern pattern of cranial development. Refining and testing this hypothesis may be difficult, given the present limits of the fossil record.
Overall cranial form
Individual arcs, angles and other dimensions provide important information, but it is helpful to assess skull shape from a multivariate perspective. As a species, H. erectus presents much variation. One component of this variation must represent sex dimorphism within populations, but there are regional differences as well as time-related trends. The Middle Pleistocene fossils also vary in their morphology. Discriminant analysis employs measurements as a basis for contrasting such groups, seeking to maximise differences between populations relative to variation within each sample. Discriminant analysis was utilised here to explore the extent to which morphological trends begun with H. erectus are continued, altered, or cut short, following the appearance of mid-Pleistocene hominins.
The eight variables selected for this exploration were VOL, vertex height : cranial length (PVH:GOL), maximum breadth : vertex height (XCB:PVH), biparietal breadth : maximum breadth (XPB:XCB), AUB, BLR, LIC and TOR. AUB, BLR, LIC and TOR were adjusted for size using the geometric mean. The three cranial indices represent proportions. In certain cases, where proportions are of some specified whole (e.g. the geochemical compositions of rocks), the observations must sum to unity, which can result in analytical difficulties. The indices utilised here are not subject to such constraints.
The analysis was performed on 25 H. erectus and 9 mid-Pleistocene crania, which were complete enough to be included. These individuals were assigned a priori to groups of approximately equal size. Partly on the basis of chronology, H. erectus specimens from East Africa were placed with the Dmanisi specimens (from the Republic of Georgia). The Sangiran and Sambungmacan specimens from Indonesia comprised a second sample, and the relatively large-brained hominins from Ngandong, Indonesia were allocated to a third sample. The skulls from Zhoukoudian in China constituted an additional grouping within H. erectus, whilst the mid-Pleistocene hominins were treated as a separate assemblage.
Four functions were obtained. The first is the most important, accounting for 69.9% of the total discrimination. The second and subsequent axes contributed 15.6%, 11.7% and 2.7% of the discrimination, respectively. These axes are mutually orthogonal, and together they describe a multivariate space within which the several groups are separated, although there is overlap in the distributions of scores. A plot of function 1 versus function 2 is provided (Figure 2). It can be seen that African and earlier Asian H. erectus crania receive low scores on function 1, whilst the mid-Pleistocene group lies at the opposite pole. The Ngandong hominins are intermediate. On function 2, there is less discrimination overall, but H. erectus at Sangiran and Sambungmacan is contrasted to the Zhoukoudian population. Axis 3 (not illustrated) provides further but limited separation, particularly between H. erectus from Africa and/or the Caucasus and specimens from eastern Asia. On axis 4, accounting for only a fraction of the total discrimination, the mid-Pleistocene fossils are driven positively, whilst most (but not all) of the H. erectus scores are lower.
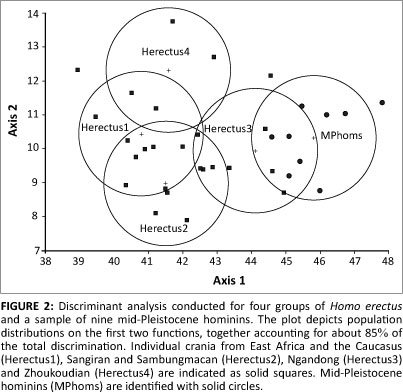
Correlations of the discriminant scores with the original variables indicate that function 1 is associated most strongly with VOL (r = 0.86), AUB (r = -0.72), XCB:PVH (r = -0.64) and XPB:XCB (r = 0.64). The latter indices register vault breadth relative to height, and coronal expansion of the parietals. Brain size contributing to a higher vertex and parietal bossing must underlie a part of the variation on this axis, but the basicranium is also important. Broad bases are correlated (negatively) with low scores for earlier H. erectus, whilst narrower bases contribute to the higher scores of mid-Pleistocene individuals. In terms of base width as well as brain size, the skulls from Ngandong occupy a sort of middle ground. Function 2 is again strongly correlated with AUB (r= 0.51) and with breadths of the vault. The Zhoukoudian crania present broad bases coupled with vaults that are less expanded laterally in comparison to the condition at Sangiran and Sambungmacan. Most notably, the posterior vault is narrowed in the Chinese fossils.32,49 Function 3 is associated with BLR (r = 0.62) and LIC (r = 0.57). Differences in these sagittal lengths set African H. erectus apart from populations in the Far East. Function 4, correlated principally with TOR (r = 0.73), seems to sort the crania on the basis of supraorbital development, giving high scores to such heavy-browed individuals as Broken Hill, Petralona and SH 5, as well as Steinheim (from Germany) and Ndutu (from Tanzania).
Together, the functions effectively discriminate amongst the populations. Within the constellation of all 25 H. erectus, two crania are 'misclassified' (assigned to inappropriate regional samples), but no H. erectus is mistaken as a mid-Pleistocene individual, and none of the latter specimens is misclassified. Some of this discrimination is related to brain size. However, the analysis does not suggest that a simple trend toward encephalisation, rooted in the ancient demes of East Africa or Java and encompassing later groups from China, can 'explain' the emergence of hominins in the Middle Pleistocene. The Zhoukoudian skulls themselves should not be viewed as transitional in form, as they seem to constitute an outlying population, distinct from other Asian H. erectus crania and from the mid-Pleistocene hominins. Ngandong does occupy an intermediate position in multivariate space (Figure 2). The systematic significance of Ngandong remains to be fully clarified. In relation to Trinil and Sangiran, average brain size is increased, and frontal constriction is reduced. A unique lengthening of the basioccipital and structures lying adjacent to the foramen magnum has been noted, along with marked upward inclination of the posterior aspect of the supramastoid crest.29 Apart from the presence of these traits reflecting local evolution, the fossils are quite similar to other Javanese H. erectus.50,51 It must be questioned whether the Ngandong assemblage exhibits many (if any) elements of neurocranial form that would be anticipated in antecedents to Broken Hill, the Sima, Petralona and more modern populations.
Discussion and conclusions
An increase in brain size is one of several keys to the transformation of the skull occurring in Middle and Late Pleistocene Homo. Endocranial volume is significantly greater in the mid-Pleistocene group than in H. erectus, and this change influences vault length, vertex height and sagittal arc lengths. Encephalisation is probably also linked to frontal constriction, parietal bossing and temporal squama height. The brain fails to influence vault breadth within either H. erectus or the mid-Pleistocene sample (Table 9). Conformity across the archaic populations suggests that a low correlation of volume with breadth may be counted as the primitive state. Apparently as a consequence of changes to the shape of the brain or its interactions (evolutionary 'tinkering'52), there is a positive association of brain volume with cranial breadth in H. sapiens. This condition must be recently derived. Flattening of the frontal is pronounced for H. erectus and in the mid-Pleistocene crania. In the Broken Hill, Bodo, SH 5 and Petralona specimens, the face is large, forward projecting and surmounted by heavy brows. As with H. erectus, facial morphology seems to override brain size as a determinant of frontal curvature and the entire cranium retains an elongated form. In anatomically modern H. sapiens, the face is shorter and retracted, and the large brain contributes to a more convex forehead and a globe-like vault.53,54,55
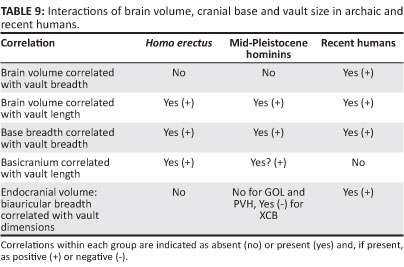
Developmental and functional interactions of the brain with components of the skull may be quite complex. Such interactions result in morphological integration.56,57,58 At this point, it is useful to ask whether H. erectus and the mid-Pleistocene hominins differ in their integration. Clues are provided by changes in character covariation registered during the course of evolution, along with the inferred polarity of these adjustments. In the examples of modular interaction listed in Table 9, there are few differences between the archaic taxa. This is the case for the relationship of VOL and GOL, and the covariation of AUB with XCB. These interactions can be interpreted as symplesiomorphic. For VOL and XCB, the primitive pattern of no association prevails prior to the emergence of H. sapiens. The VOL:AUB ratio has little effect on neurocranial dimensions of H. erectus, and for this 'trait,' the mid-Pleistocene hominins again possess the primitive condition. However, strong and positive interactions occur in the skull of recent H. sapiens. The lack of association between AUB and GOL represents an apomorphy accompanying the advent of modern humans. Overall, there is consistency in the interactions documented for H. erectus and its mid-Pleistocene descendants, and no secure case for differences in integration can be made. Where there are changes in the covariation of the brain or basicranium with the vault and face, these are manifest most frequently with the appearance of H. sapiens. The efficacy of such comparisons is limited by small sample size.59 Nevertheless, such findings might be expected, as previous studies have shown patterns of cranial integration to be similar (but not identical) in humans, African apes and Neanderthals.60,61
It is evident that variation in the skull base is an important instrument of craniofacial change. As noted above, the basicranium, independently from the brain, covaries with dimensions of the vault. Structural adjustments to the base itself separate the mid-Pleistocene sample from H. erectus. Diagnostic characters include a larger petrotympanic angle associated with coronal alignment of the petrous and tympanic axes, 'erosion' of the pyramid apex leading to enlargement of the foramen lacerum, reduction of the spine of the crista petrosa, decreased robusticity of the tympanic plate, development of a projecting sphenoid spine and greater definition of an articular tubercle at the anterior margin of the mandibular fossa. Some of these derived states may be linked to shortening of the basioccipital and sphenoid body, or to midline flexion as measured by the cranial base angle. Other changes are likely influenced by the masticatory apparatus. During development, both the cranial base angle and facial form must respond to encephalisation, and integration is expected. But the basicranium is clearly critical to the architecturally complex 'remodelling' of the skull that occurs in later Homo.
Given the influences on cranial form that have been identified, the null hypothesis posed earlier can be reconsidered. Whilst the situation poses undeniable challenges relating to both structure and function, there is evidence to support the view that some changes in the Broken Hill, Elandsfontein, Bodo and European populations are dependent on encephalisation. In other instances where the brain influences vault shape, the mid-Pleistocene specimens follow the H. erectus pattern, and there is no difference in the interaction(s) observed. Traits that are symplesiomorphic offer little insight into evolutionary history. Additional characters of the mid-Pleistocene neurocranium are derived but not appreciably influenced by variations in brain size. The parietals are lengthened, and the occipital profile is rounded in the mid-Pleistocene hominins, but these changes cannot result primarily from encephalisation within populations of H. erectus. It is clear that the base is essential to the alteration of cranial form in mid-Pleistocene Homo. Discrete morphology of the temporal bone and mandibular fossa is derived in the Broken Hill, Ndutu and, in part, the Bodo specimens, and similar determinations can be made for several of the European individuals where the basicranium is preserved.
Additional information was provided by discriminant analysis. Along with endocranial volume, basicranial width contributes importantly to the separation of mid-Pleistocene hominins from H. erectus on function 1. Cranial base breadth is also the variable most highly correlated with function 2, on which specimens from Sangiran and Zhoukoudian occupy opposite poles. Axis 2 does not by itself distinguish the mid-Pleistocene crania from H. erectus but acts jointly with other functions to classify all of the former hominins 'correctly.' The analysis demonstrates that mid-Quaternary Homo is significantly distant from African and Asian H. erectus. Not surprisingly, this multivariate distance incorporates brain size, but changes to the form of the neurocranium and face cannot be described as driven solely by encephalisation.
In sum, results suggest rejection of the initial null hypothesis. If the mid-Pleistocene assemblage can be diagnosed on the basis of characters that vary independently from brain volume, then Broken Hill, Elandsfontein, Bodo, Ndutu, and the European fossils should be recognised as distinct taxonomically from H. erectus. H. heidelbergensis is the nomen that has been proposed for the European specimens.18,26,62 On the strength of shared morphology, it can be argued that the African group belongs within this taxon.18,19,20,22 Alternatively, the African fossils may be attributed to Homo rhodesiensis.21 Just how the individuals are to be sorted will likely remain controversial. But it is evident that the Homo family tree can accommodate at least one mid-Pleistocene species in addition to Neanderthals and H. sapiens.
Acknowledgements
I thank F. Spoor, Y. Kaifu, R. Quam and A. Nevgloski for assistance with gathering data central to this research. Ian Tattersall gave me access to materials held in the American Museum of Natural History. Anne Hull executed the figure. Dan Lieberman and Rebecca Ackermann provided insightful comments on the manuscript. An early version of this paper was presented at the Langebaanweg 2010 conference held in Cape Town. For the invitation to participate in this conference and for local support, I am grateful to the LBW 2010 organising committee (T. Matthews, D. Roberts, R. Smith, D. Stynder and J. Ackerman). Additional funding was provided by the American School of Prehistoric Research at Harvard University.
Competing interests
I declare that I have no financial or personal relationships which may have inappropriately influenced me in writing this article.
References
1. Singer R, Wymer J. Archaeological investigations at the Saldanha skull site in South Africa. S Afr Archaeol Bull. 1968;25:63-74. http://dx.doi.org/10.2307/3888485 [ Links ]
2. Klein RG, Avery G, Cruz-Uribe K, Steele TE. The mammalian fauna associated with an archaic hominin skullcap and later Acheulean artifacts at Elandsfontein, Western Cape Province, South Africa. J Hum Evol. 2007;52:164-186. http://dx.doi.org/10.1016/j.jhevol.2006.08.006, PMid:17030056 [ Links ]
3. Rightmire GP. The human cranium from Bodo, Ethiopia: Evidence for speciation in the Middle Pleistocene? J Hum Evol. 1996;31:21-39. http://dx.doi.org/10.1006/jhev.1996.0046 [ Links ]
4. Clark JD, De Heinzelin J, Schick KD, et al. African Homo erectus: Old radiometric ages and young Oldowan assemblages in the Middle Awash Valley, Ethiopia. Science. 1994;1907-1910. http://dx.doi.org/10.1126/science.8009220, PMid:8009220 [ Links ]
5. Bischoff JL, Shamp DD, Aramburu A, Arsuaga JL, Carbonell E, Bermúdez de Castro JM. The Sima de los Huesos hominids date to beyond U/Th equilibrium (>350 kyr) and perhaps to 400-500 kyr: New radiometric dates. J Archaeol Sci. 2003;30:275-280. http://dx.doi.org/10.1006/jasc.2002.0834 [ Links ]
6. Bischoff JL, Williams RW, Rosenbauer RJ, et al. High-resolution U-series dates from the Sima de los Huesos hominids yields 600 + ∞ - 66 kyrs: Implications for the evolution of the early Neanderthal lineage. J Archaeol Sci. 2007;34:763-770. http://dx.doi.org/10.1016/j.jas.2006.08.003 [ Links ]
7. Rightmire GP. Human evolution in the Middle Pleistocene: The role of Homo heidelbergensis. Evol Anthropol. 1998;6:218-227. http://dx.doi.org/10.1002/(SICI)1520-6505(1998)6:6<218::AID-EVAN4>3.0.CO;2-6 [ Links ]
8. Harvati K, Hublin JJ, Gunz P. Evolution of middle-late Pleistocene human cranio-facial form: A 3-D approach. J Hum Evol. 2010;59:445-464. http://dx.doi.org/10.1016/j.jhevol.2010.06.005, PMid:20708775 [ Links ]
9. Harvati K, Hublin JJ, Gunz P. Three dimensional evaluation of Neanderthal craniofacial features in the European and African Middle Pleistocene human fossil record (abstract). Am J Phys Anthropol. 2011;52:157. [ Links ]
10. Moore WJ, Lavelle CJB. Growth of the facial skeleton in the Hominoidea. London: Academic Press; 1974. [ Links ]
11. Sirianni JE, Swindler DR. Growth and development of the pigtailed macaque. Boca Raton, FL: CRC Press; 1985. [ Links ]
12. Sperber GH. Craniofacial embryology. 4th ed. London: Wright; 1989. [ Links ]
13. Enlow DH. Facial growth. 3rd ed. Philadelphia: WB Saunders; 1990. [ Links ]
14. Ross CF, Ravosa MJ. Basicranial flexion, relative brain size and facial kyphosis in nonhuman primates. Am J Phys Anthropol. 1993;91:305-324. http://dx.doi.org/10.1002/ajpa.1330910306, PMid:8333488 [ Links ]
15. Lieberman DE, Ross CF, Ravosa MJ. The primate cranial base: Ontogeny, function, and integration. Yearb Phys Anthropol. 2000;43:117-169. http://dx.doi.org/10.1002/1096-8644(2000)43:31+<117::AID-AJPA5>3.3.CO;2-9 [ Links ]
16. Bastir M, Rosas A, Stringer C, et al. Effects of brain and facial size on basicranial form in human and primate evolution. J Hum Evol. 2010;58:424-431. http://dx.doi.org/10.1016/j.jhevol.2010.03.001, PMid:20378153 [ Links ]
17. Lieberman DE. The evolution of the human head. Cambridge: Belknap (Harvard University) Press; 2011. [ Links ]
18. Stringer CB. New views on modern human origins. In: Rasmussen DT, editor. The origin and evolution of humans and humanness. Boston, MA: Jones and Bartlett, 1993; p. 75-94. [ Links ]
19. Rightmire GP. Human evolution in the Middle Pleistocene: The role of Homo heidelbergensis. Evol Anthropol. 1998;6:218-227. http://dx.doi.org/10.1002/(SICI)1520-6505(1998)6:6<218::AID-EVAN4>3.0.CO;2-6 [ Links ]
20. Rightmire GP. Homo in the Middle Pleistocene: Hypodigms, variation, and species recognition. Evol Anthropol. 2008;17:8-21. http://dx.doi.org/10.1002/evan.20160 [ Links ]
21. Hublin JJ. The origin of Neandertals. Proc Natl Acad Sci USA. 2009;106:16022-16027. http://dx.doi.org/10.1073/pnas.0904119106, PMid:19805257 [ Links ]
22. Mounier A, Marchal F, Condemi S. Is Homo heidelbergensis a distinct species? New insight on the Mauer mandible. J Hum Evol. 2009;56:219-246. http://dx.doi.org/10.1016/j.jhevol.2008.12.006, PMid:19249816 [ Links ]
23. Lieberman DE, Bar-Yosef O. Apples and oranges: Morphological versus behavioral transitions in the Pleistocene. In: Lieberman DE, Smith RJ, Kelley J, editors. Interpreting the past: Essays on human, primate and mammal evolution. Boston, MA: Brill Academic Publishers, 2005; p. 275296. [ Links ]
24. Howells WW. Cranial variation in man: A study by multivariate analysis of patterns of difference among recent human populations. Pap Peabody Mus. 1973;67:1-259. [ Links ]
25. Holloway RL, Broadfield DC, Yuan MS, Schwartz JH, Tattersall I. Brain endocasts: The paleoneurological evidence. The human fossil record 3. Hoboken, NJ: Wiley-Liss; 2004. http://dx.doi.org/10.1002/0471663573 [ Links ]
26. Arsuaga JL, Martinez I, Gracia A, Lorenzo C. The Sima de los Huesos crania (Sierra de Atapuerca, Spain). A comparative study. J Hum Evol. 1997;33:219-281. http://dx.doi.org/10.1006/jhev.1997.0133, PMid:9300343 [ Links ]
27. Spoor F, Leakey MG, Gathogo PN, et al. Implications of new early Homo fossils from Ileret, east of Lake Turkana, Kenya. Nature. 2007;448:688-691. http://dx.doi.org/10.1038/nature05986 [ Links ]
28. Gilbert WH, Asfaw B. Homo erectus. Pleistocene evidence from the Middle Awash, Ethiopia. Berkeley, CA: University of California Press; 2008. [ Links ]
29. Kaifu Y, Aziz F, Indriati E, Jacob T, Kurniawan I, Baba H. Cranial morphology of Javanese Homo erectus: New evidence for continuous evolution, specialization, and terminal extinction. J Hum Evol. 2008;55:551-580. http://dx.doi.org/10.1016/jJhevol.2008.05.002, PMid:18635247 [ Links ]
30. Widianto H, Zeitoun V. Morphological description, biometry and phylogenetic position of the skull of Ngawi 1 (East Java, Indonesia). Int J Osteoarchaeol. 2003;13:339-351. http://dx.doi.org/10.1002/oa.694 [ Links ]
31. Arif J, Baba H, Suparka ME, Zaim Y, Setoguchi T. Preliminary study of Homo erectus skull IX (Tig-1993.05) from Sangiran, central Java, Indonesia. Bull Nat Sci Mus Tokyo D. 2001;27:1-17. [ Links ]
32. Weidenreich F. The skull of Sinanthropus pekinensis: A comparative study of a primitive hominid skull. Palaeontol Sin D. 1943;10:1-484. [ Links ]
33. JMP. Version 8.0. Cary, NC: SAS Institute Inc; 2009. [ Links ]
34. Bielicki T, Miszkiewicz B, Hauser G. Archaic and modern craniometric variants in contemporary man. Monographs of the Institute of Anthropology. Wroclaw: Polish Academy of Sciences; 1990. [ Links ]
35. Dean MC, Leakey MG, Reid D, et al. Growth processes in teeth distinguish modern humans from Homo erectus and earlier hominins. Nature. 2001;414:628-631. http://dx.doi.org/10.1038/414628a, PMid:11740557 [ Links ]
36. Dean MC. Tooth microstructure tracks the pace of human life-history evolution. Proc R Soc B. 2006;273:2799-2808. http://dx.doi.org/10.1098/rspb.2006.3583, PMid:17015331 [ Links ]
37. Dean MC, Smith BH. Growth and development of the Nariokotome youth, KNM-WT 15000. In: Grine FE, Fleagle JG, Leakey RE, editors. The first humans: Origin and early evolution of the genus Homo. New York: Springer, 2009; p. 101-120. http://dx.doi.org/10.1007/978-1-4020-9980-9_10 [ Links ]
38. Graves RR, Lupo AC, McCarthy RC, Wescott DJ, Cunningham DL. Just how strapping was KNM-WT 15000? J Hum Evol. 2010;59:542-554. http://dx.doi.org/10.1016/j.jhevol.2010.06.007, PMid:20846707 [ Links ]
39. Coqueugniot H, Hublin JJ, Veillon F, Houet F, Jacob T. Early brain growth in Homo erectus and implications for cognitive ability. Nature. 2004;431:299-302. http://dx.doi.org/10.1038/nature02852, PMid:15372030 [ Links ]
40. Hublin JJ, Coqueugniot H. Absolute or proportional brain size: That is the question. J Hum Evol. 2006;50:109-113. http://dx.doi.org/10.1016/j.jhevol.2005.08.009 [ Links ]
41. Leigh, SR. Brain ontogeny and life history in Homo erectus. J Hum Evol. 2006;50:104-108. http://dx.doi.org/10.1016/jJhevol.2005.02.008, PMid:16226296 [ Links ]
42. DeSilva J, Lesnik J. Chimpanzee neonatal brain size: Implications for brain growth in Homo erectus. J Hum Evol. 2006;51:207-212. http://dx.doi.org/10.1016/j.jhevol.2006.05.006, PMid:16824583 [ Links ]
43. Zollikofer CPE, Ponce de León MS. The evolution of hominin ontogenies. Sem Cell Dev Biol. 2010;21:441-452. http://dx.doi.org/10.1016/j.semcdb.2009.10.012 [ Links ]
44. Antón SC. Developmental age and taxonomic affinity of the Mojokerto child, Java, Indonesia. Am J Phys Anthropol. 1997;102:497-504. http://dx.doi.org/10.1002/(SICI)1096-8644(199704)102:4<497::AID-AJPA6>3.0.CO;2-P [ Links ]
45. Lieberman DE, Pearson OM, Mowbray KM. Basicranial influence on overall cranial shape. J Hum Evol. 2000;38:291-315. http://dx.doi.org/10.1006/jhev.1999.0335, PMid:10656780 [ Links ]
46. Ruff CB, Trinkaus E, Holliday TW. Body mass and encephalization in Pleistocene Homo. Nature. 1997;387:173-176. http://dx.doi.org/10.1038/387173a0, PMid:9144286 [ Links ]
47. Rosenberg KR, Lu Z, Ruff CB. Body size, body proportions and encephalization in the Jinniushan specimen (abstract). Am J Phys Anthropol. 1999;28:235. [ Links ]
48. Rightmire GP. Brain size and encephalization in early to mid-Pleistocene Homo. Am J Phys Anthropol. 2004;124:109-123. http://dx.doi.org/10.1002/ajpa.10346, PMid:15160365 [ Links ]
49. Antón S. Evolutionary significance of cranial variation in Asian Homo erectus. Am J Phys Anthropol. 2002;118:301-323. http://dx.doi.org/10.1002/ajpa.10091, PMid:12124912 [ Links ]
50. Rightmire GP. The relationship of Homo erectus to later Middle Pleistocene hominids. Cour Forsch-Inst Senckenberg. 1994;171:319-326. [ Links ]
51. Antón SC. Natural history of Homo erectus. Yearb Phys Anthropol. 2003;46:126-169. [ Links ]
52. Lieberman DL, Krovitz GE, McBratney-Owen B. Testing hypotheses about tinkering in the fossil record: The case of the human skull. J Exp Zool (Mol Dev Evol). 2004;302B:284-301. http://dx.doi.org/10.1002/jez.b.21004, PMid:15211687 [ Links ]
53. Day MH, Stringer CB. A reconsideration of the Omo Kibish remains and the erectus-sapiens transition. In: De Lumley MA, editor. [Homo erectus and the place of Tautavel man among the fossil hominids]. Nice: CNRS, 1982; p. 814-846. French. [ Links ]
54. Lieberman DE, McBratney BM, Krovitz G. The evolution and development of cranial form in Homo sapiens. Proc Natl Acad Sci USA. 2002;99:1134-1139. http://dx.doi.org/10.1073/pnas.022440799, PMid:11805284 [ Links ]
55. Pearson OM. Statistical and biological definitions of 'anatomically modern' humans: Suggestions for a unified approach to modern morphology. Evol Anthropol. 2008;17:38-48. http://dx.doi.org/10.1002/evan.20155 [ Links ]
56. Cheverud JM. Morphological integration in the saddle-back tamarin (Saguinus fuscicollis) cranium. Am Nat. 1995;145:63-89. http://dx.doi.org/10.1086/285728 [ Links ]
57. Lieberman DE, Ross CF, Ravosa MJ. The primate cranial base: Ontogeny, function and integration. Yearb Phys Anthropol. 2000;43:117-169. http://dx.doi.org/10.1002/1096-8644(2000)43:31+<117::AID-AJPA5>3.3.CO;2-9 [ Links ]
58. Rolian C, Willmore KE. Morphological integration at 50: patterns and processes of integration in biological anthropology. Evol Biol. 2009;39:1-4. http://dx.doi.org/10.1007/s11692-009-9052-0 [ Links ]
59. Ackermann RR. Morphological integration and the interpretation of fossil hominin diversity. Evol Biol. 2009;36:149-156. http://dx.doi.org/10.1007/s11692-009-9050-2 [ Links ]
60. Ackermann RR. Ontogenetic integration of the hominoid face. J Hum Evol. 2005;48:175-197. http://dx.doi.org/10.1016/j.jhevol.2004.11.001, PMid:15701530 [ Links ]
61. Roseman CC, Weaver TD, Stringer CB. Do modern humans and Neandertals have different patterns of cranial integration? J Hum Evol. 2011;60:684-693. http://dx.doi.org/10.1016/j.jhevol.2010.04.010, PMid:21463884 [ Links ]
62. Rosas A, Bermúdez de Castro JM. The Mauer mandible and the evolutionary significance of Homo heidelbergensis. Geobios. 1998;31:687-697. http://dx.doi.org/10.1016/S0016-6995(98)80055-7 [ Links ]
 Correspondence to:
Correspondence to:
Philip Rightmire
Postal address: HEB, Peabody Museum
11 Divinity Avenue
Cambridge, MA 02138, USA
Email: gprightm@fas.harvard.edu
Received: 20 Apr. 2011
Accepted: 26 Sept. 2011
Published: 09 Mar. 2012














